Abstract
The present study aimed to clarify the current status, therapeutic strategy, and 1-year outcome in acute limb ischemia (ALI) patients in Japan. The EnDOvascular treatment (Edo) registry database includes 324 patients from 10 institutes who were registered between November 2011 and October 2013. A total of 70 ALI patients (mean age 74.0 years) from the Edo registry database were enrolled in this study. Of the 70 included patients, 72.9% were male and 35.7% had embolism. Of patients, 38.6%, 42.9%, and 18.6% underwent EVT, surgery, and hybrid thrombectomy, respectively, in primary revascularization strategy. Limb ischemia was categorized into four classes at initial evaluation: SVS/ISCVS class I (n = 13, 18.6%), SVS/ISCVS class IIa (n = 36, 51.4%), SVS/ISCVS class IIb (n = 21, 30%), and SVS/ISCVS class III (n = 0, 0%). Three patients with SVS/ISCVS class IIb limb ischemia developed myonephropathic metabolic syndrome. No catheter-directed thrombolysis was employed as a primary revascularization strategy. The 1-year rates of all-cause death, major amputation, and a composite of perioperative death or major adverse limb event were 28.6%, 5.7%, and 40.0%, respectively. Lower age, male sex, dyslipidemia, high estimated glomerular filtration rate, high albumin level, and low C-reactive protein level were independent positive predictors of all-cause death. In this registry, SVS/ISCVS class IIa ALI was predominant. Approximately 40% of primary revascularization strategy was surgery and EVT, followed by hybrid therapy. All-cause death and major amputation rates at 1 year were less than 30% and 6%, respectively.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Peripheral artery disease (PAD) is one of the representative chronic non-communicable diseases affecting an increasing number of individuals in recent decades [1]. Therefore, PAD has been extensively discussed as a chronic disease worldwide [2,3,4,5,6]. PAD patients are at high risk of developing cardiovascular diseases; in particular, patients with critical limb ischemia have poorer prognosis than those with claudication or asymptomatic PAD. Improved prognosis following revascularization performed with consideration of lesion location has been indicated. A cohort study conducted in Southwestern France reported that PAD patients with acute limb ischemia (ALI) had poorer prognosis than patients with intermittent claudication [7]. A recent cohort study performed in Japan similarly indicated poorer prognosis for ALI than for intermittent claudication [8]. Furthermore, a retrospective study from the United States provided details on revascularization strategy and reported the efficacy of surgical embolectomy and risk factors for amputation and in-hospital mortality [9], and a previous study from Europe showed the efficacy of the combination of surgical embolectomy and endovascular techniques (hybrid procedure) for ALI [10]. In a multicenter cohort of patients receiving endovascular treatment (EVT), ALI patients were observed to experience more short-term adverse events than critical limb ischemia patients [11]. No sufficient evidence in favor of thrombolysis as treatment currently exists, and a meta-analysis suggested that thrombolysis might be associated with higher risk of hemorrhagic complications [12]. Moreover, data from a multicenter cohort regarding the current therapeutic strategy for ALI patients and their outcome are lacking, particularly in Japan. The present study aimed to clarify therapeutic strategy, and 1-year outcome in ALI patients in Japan.
Methods
Study design
The present study was a multicenter cohort study. The EnDOvascular treatment (EDO) registry was established to determine the real-world clinical practice of catheter therapy for specific situations (subclavian artery stenosis/occlusion, common femoral artery stenosis/occlusion, popliteal artery stenosis/occlusion, deep vein thrombosis, and ALI) in the Tokyo area. Our group had already reported EVT for deep vein thrombosis from the EDO registry [13]. Using data from the EDO registry database, we analyzed the real-world clinical practice of revascularization for ALI. The study protocol was approved by the ethics committee of Toho University Ohashi Medical Center (reference no. 23–48) and the ethics committees of each participating facility. Furthermore, this study was conducted in accordance with the Declaration of Helsinki, and all patients provided written informed consent prior to their study participation.
Study population
A total of 324 patients from 10 institutes were registered between November 2011 and October 2013 in the Edo registry database. The present study enrolled 70 consecutive ALI patients from the Edo registry database. Patients aged < 20 years and those who refused to provide informed consent were excluded from the analysis.
Data collection and follow-up protocol
Demographic, laboratory, angiographic, and procedural data were collected by independent researchers from hospital charts or database according to predetermined definitions. The study office collectively managed all data. Follow-up data were obtained either from hospital charts or from patients or their family members who were contacted by telephone. Patients were followed up at 1, 6, and 12 months after EVT.
Endpoints
The primary endpoints were all-cause death and major adverse limb event (MALE) + perioperative death (POD), which was a composite of perioperative all-cause death, unscheduled ipsilateral lower limb revascularization, ALI event, and major amputation (defined as amputation above the ankle). Major adverse cardiovascular event (MACE) was a composite of all-cause death, acute myocardial infarction (MI), and stroke.
Definitions
ALI was defined as a sudden decrease in limb perfusion that causes potential threat to limb viability, with its acute clinical presentation occurring within 2 weeks after symptom onset [14]. Bleeding event was defined as life-threatening intracerebral bleeding or substantial hemodynamic collapse requiring hospitalization and transfusion. Smoking was defined as current smoking. Heart failure was defined as a history of hospitalization for heart failure or the presence of New York Heart Association class III or IV symptoms [15] Atrial fibrillation was defined as the presence of atrial fibrillation on electrocardiogram upon admission. MI was defined as a significant increase in serum biomarker level (troponin T level > 0.1 ng/mL or creatine phosphokinase level twice the normal value) or new Q waves on electrocardiogram. Coronary artery disease was defined as stable angina with > 50% stenosis, a history of percutaneous coronary intervention or coronary artery bypass grafting, or previous MI. Stroke was defined as cerebral stroke persisting for ≥ 24 h that indicated the presence of a neurological deficit. Diabetes mellitus was defined as a hemoglobin A1c level ≥ 6.5%, casual plasma glucose level > 200 mg/dL, fasting glucose level > 126 mg/dL, or treatment with oral hypoglycemic agents or insulin injection [16]. Hypertension was defined as systolic blood pressure ≥ 140 mmHg and/or diastolic blood pressure ≥ 90 mmHg or ongoing therapy for hypertension [17]. Dyslipidemia was defined as low-density lipoprotein cholesterol level ≥ 140 mg/dL or ongoing medical treatment [18].
Statistical analysis
Continuous variables are expressed as mean ± standard deviation, whereas categorical data are presented as numbers with percentages. Student’s t test and Fisher’s exact test were used for continuous and categorical variables, respectively. The difference in primary procedure, proximal lesion location, or in-hospital limb event was assessed using analysis of variance and Fisher’s exact test for continuous and categorical variables, respectively. Odds ratios and 95% confidence intervals were calculated using univariate logistic analysis. The cut-off value was determined using receiver-operating characteristic curve analysis for independent predictors of all-cause death among patients. This cut-off value was adapted to all-cause death and MALE. We removed administration at discharge in the analysis of predictive factors for 1-year events, because 1-year events included events during hospitalization. Multivariate analysis was not performed because of insufficient number of events. Statistical significance was set at P < 0.05. All analyses were performed using SPSS version 19 (IBM Corporation, Armonk, NY, USA).
Results
Etiology
Table 1 presents the details on etiology. Overall, 35.7% of the 70 included patients had embolism, which was primarily caused by atrial fibrillation. In comparison, 64.3% of the patients had thrombosis, which was mainly due to in situ thrombosis, graft failure, and stent failure. The rates of atrial fibrillation, in situ thrombosis, graft failure, and stent failure were 28.1%, 31.4%, 12.6%, and 8.6%, respectively.
Patients’ demographics, characteristics, and admission status
Table 1 also summarizes the patients’ demographics, characteristics, and admission status. The mean age of the 70 included patients was 74.0 years, of whom 72.9% were males. The mean body mass index (BMI) was 21.8, with 7% of the patients having a BMI below 18. The rates of hypertension, dyslipidemia, diabetes mellitus, coronary artery disease, cerebrovascular disease, and heart failure were 72.9%, 44.3%, 31.4%, 24.3%, 12.9%, and 11.4%, respectively. Of patients, 28.6% were current smokers, 10.0% were on regular hemodialysis, and 4.3% had a history of ALI. Furthermore, 40.0% and 2.9% underwent revascularization for PAD and prior valvular surgery, respectively.
On admission, low-flow status (defined as shock, dehydration, or congestive heart failure) and atrial fibrillation were detected in 4.3% and 41.4% of patients, respectively. In addition, the mean systolic blood pressure and heart rate on admission were 145 mmHg and 83 beats/min, respectively.
Proximal lesion location and limb ischemia SVS/ISCVS classification at initial evaluation
Table 1 shows proximal lesion location. In 24.3%, 14.3%, 55.7%, and 5.7% of patients, the proximal lesion was detected in the aortoiliac artery, common femoral artery, femoropopliteal artery, and below-the-knee artery, respectively. Limb ischemia was categorized at initial evaluation into four classes: SVS/ISCVS class I (n = 13, 18.6%), SVS/ISCVS class IIa (n = 36, 51.4%), SVS/ISCVS class IIb (n = 21, 30%), and SVS/ISCVS class III (n = 0, 0%). Three patients with SVS/ISCVS class IIb limb ischemia developed myonephropathic metabolic syndrome (MNMS), leading to a rate of 25.7% and 4.3% for SVS/ISCVS classes IIb and III.
Onset-to-intervention time and laboratory data
The mean onset-to-intervention time was 60 ± 73 h. The distribution of onset-to-intervention time was various, as shown in Table 1. Laboratory data on admission and peak creatine phosphokinase during hospitalization are presented in Table 1.
Primary revascularization strategy
Details on primary revascularization strategy are summarized in Table 2. Of patients, 38.6%, 24.3%, 10.0%, 5.7%, and 18.6% underwent EVT, angiography-guided Fogarty catheter thrombectomy, blind Fogarty catheter thrombectomy, bypass surgery, and hybrid thrombectomy procedure, respectively. Among primary revascularization strategies, no thrombolysis was performed, although systemic thrombolysis and catheter-directed thrombolysis (CDT) were used as adjunctive therapy for a few cases.
Analysis of variance showed no difference between proximal lesion location and primary procedure (Fig. 1). However, the rate of EVT procedure performed in the aortoiliac artery (11.8%, 2/17) was significantly lower than the rate of EVT procedure performed in the femoropopliteal artery (53.8%, 21/39).
EVT only and hybrid EVT procedure in detail
Details on EVT only and hybrid thrombectomy procedure are summarized in Table 3. Mechanical thrombectomy with Fogarty catheter thrombectomy was performed in all patients belonging to the hybrid EVT procedure group. Contrast volume, procedure time, and the rates of balloon procedure and intravascular ultrasound use were almost equal in both the EVT only group and hybrid thrombectomy procedure group. However, the EVT only group required stent and distal protection device use compared with the hybrid thrombectomy procedure group. Table 4 shows the final angiography findings and limb event. The final angiography findings indicated that the cases of 87.5% could get the flow to ankle at least. However, the final angiography findings did not linked to an in-hospital limb event.
Primary procedure and in-hospital limb event
Figure 2 shows the relationship between primary procedure and in-hospital limb event. In-hospital limb event included major amputation and unscheduled limb revascularization. Blind mechanical thrombectomy using a Fogarty catheter without angiography tended to have poorer limb prognosis than EVT or angiography-guided mechanical thrombectomy.
Oral administration at discharge (N = 61, with nine patients who died during hospitalization)
Table 5 summarizes the patients’ drug treatments at the time of discharge. Overall, 65.6%, 27.9%, 21.3%, 60.7%, 6.6%, 39.3%, and 60.7% of patients took aspirin, thienopyridine, cilostazol, warfarin, direct oral anticoagulants, statin, and antihypertensive drugs at the time of discharge, respectively.
All-cause death, major amputation, bleeding event, MALE + POD, and major amputation + all-cause death
Table 6 presents details on all-cause death, major amputation, bleeding event, and MALE + POD. The all-cause death rates at 1 month, 6 months, and 1 year were 12.9%, 25.7%, and 28.6%, respectively. The major amputation rates at 1 month, 6 months, and 1 year were 5.7%, 5.7%, and 5.7%, respectively. The MACE rates at 1 month, 6 months, and 1 year were 14.3%, 25.7%, and 30.0%, respectively. The MALE + POD rates at 1 month, 6 months, and 1 year were 28.6%, 34.3%, and 40.0%, respectively. The major bleeding rates were 2.9%, 5.7%, and 5.7%, respectively. Major amputation + all-cause death rates at 1 month, 6 months, and 1 year were 15.7%, 27.1%, and 30.0%, respectively.
Causes of death are presented in Table 7.
Predicted risk for all-cause death and MALE in univariate logistic analysis with proportional hazard models
The results of univariate logistic analysis using proportional hazards models of all-cause death and MALE + POD at 1 year are shown in Table 8. Lower age, male sex, dyslipidemia, current smoking, high estimated glomerular filtration rate, high albumin level, and low C-reactive protein (CRP) level were independent positive predictors of all-cause death. Albumin and hemoglobin levels were independent predictors of MALE + POD.
The detail of three patients with MNMS
In all three cases with MNMS, proximal lesion location was femoropopliteal artery. Two cases were treated with surgical embolectomy with Fogarty catheter; the rest case was treated with hybrid thrombectomy. Two cases with surgical embolectomy had blood pressure dropped after getting blood flow to lower limb during revascularization procedure and died due to shock. In the rest case with hybrid thrombectomy, respiratory condition deteriorated after revascularization and acute respiratory distress syndrome developed on the next day and died. Onset-to-intervention times were 6 h., 9 h., and 72 h., respectively. The etiology of two cases was in situ thrombosis and the rest case was embolism.
Discussion
This multicenter prospective cohort study included not only ALI patients managed with EVT but also those who underwent surgery. This is the first prospective multicenter study that analyzes these patients simultaneously.
Etiology
The etiology of ALI can be broadly categorized into thrombosis and embolism. A previous study conducted from 1987 to 1995 reported that the cause of ALI had shifted from embolism to thrombosis [19]. It became clear that the rate of thrombosis had exceeded that of embolism in ALI patients treated from 2005 to 2011 [20]. In this study, the frequency of thrombosis was higher than that of embolism, which is similar to the finding of the previous report. To the best of our knowledge, this study is the first one to clarify the details about etiology. Cardiac thrombosis due to atrial fibrillation accounts for approximately 75% of embolism etiologies. The most frequent thrombosis etiology was in situ thrombosis, which represented approximately 50% of all thrombosis etiologies. Although the rate of popliteal aneurysm as a cause of thrombosis is approximately 6–7%, we cannot ignore popliteal aneurysms because of their severity [21,22,23]. The rate of stent thrombosis and bypass graft occlusion was approximately 20% in this study and approximately 50% in another previous study [20]. We should expend effort in selecting the appropriate method for revascularization or administration of antithrombotic therapy after revascularization to avoid stent thrombosis and graft occlusion. However, the data to analyze this remain insufficient, especially in Japan.
The rate of low-flow status (defined as shock, dehydration, or congestive heart failure) was 4.3% in this study. Low-flow status can lead to vascular flow limitation. The rate of ALI involvement with low-flow status was clarified in this study.
Armand Trousseau reported “Trousseau syndrome” as cancer-associated thrombosis [24]. The rate of ALI complicated with malignancy in this study was lower than that in a previous study (4.3% vs. 23%, respectively) [20]. Insufficient examination might be considered a possible reason for this lower rate.
Proximal lesion location and primary revascularization strategy
As shown in Fig. 1, surgical revascularization tended to be more selected for aortoiliac artery lesion than for femoropopliteal artery lesion. This selection bias could possibly be attributed to thrombus volume. In other words, surgical revascularization is preferred for aortoiliac artery lesions with considerably greater thrombus volume than for infrainguinal artery lesions. Conversely, EVT was actively selected as the revascularization strategy for femoropopliteal artery lesions, with a relatively small amount of thrombus. Furthermore, a common femoral artery lesion features a non-stenting zone in the EVT procedure and is widely used in surgery as an approach site for revascularization for ALI, as surgical revascularization is actively selected irrespective of the amount of thrombus in this lesion [10].
Limb ischemia classification at initial evaluation
Approximately 14% of patients deemed to have SVS/ISCVS class IIb limb ischemia before revascularization were revealed to have SVS/ISCVS class III limb ischemia instead, suggesting the difficulty of evaluating SVS/ISCVS classes IIb and III. The clinical category of ALI is definite [25]. However, in real-world clinical practice, there exist cases of ALI in which the iliac artery lesion causes large-area ischemia and leg paralysis despite the viability of the leg. Although these cases are actually cases of SVS/ISCVS class IIb limb ischemia, they are diagnosed as SVS/ISCVS class III limb ischemia regardless of the definite clinical category. In contrast, MNMS can be developed on the basis that it is judged to be milder as SVS/ISCVS class IIb in SVS/ISCVS class III. In this study, all three patients with MNMS died owing to shock or acute respiratory distress syndrome. No clear indicator of the onset of MNMS exists in current practice. Hence, SVS/ISCVS class IIb needs to be more clearly and objectively distinguished from SVS/ISCVS class III, and predictive factors for the occurrence of MNMS in daily practice should be identified.
Onset-to-intervention time
In complete arterial obstruction, irreversible changes occur in the nerve, muscles, and skin at 4–6 h, 6–8 h, and 8–12 h, respectively [23]. Therefore, the onset-to-intervention time for suitable revascularization is considered to be within 6 h (otherwise known as the “golden time”). However, in this study, only 25% of cases were within the golden time, and more than 1 day had passed in about half of cases. This phenomenon can be explained by prolonged thrombotic disease. ALI patients with thrombosis etiologies can develop collateral circulation and getting therapeutic marginal times because of underlying chronic occlusive lesions and ischemia. We should not evaluate severity based on onset time alone because of the increasing rate of thrombosis etiologies in current practice.
Primary procedure
In this study, no CDT was employed as a primary revascularization strategy. Unlike surgical embolectomy, gradual reperfusion by CDT could possibly reduce the occurrence of MNMS; moreover, CDT is effective for lesions with a large amount of thrombus. It can be presumed that we can avoid the performance of CDT, considering the risk of bleeding in Japan. Revascularization was preferably completed in only one EVT procedure. Instead of CDT, techniques using various devises for ALI are employed in Japan [26]. Nevertheless, new devices for EVT procedure have not been approved in Japan, and percutaneous mechanical thrombectomy for ALI is considered more effective, particularly in western countries [27]. If these devices can be used in Japan, treatment for ALI with a large amount of thrombus can be possibly completed using mechanical thrombectomy with a single EVT procedure. If possible, we should learn the basics of CDT for ALI at the present stage without new devices.
As shown in Fig. 2, mechanical thrombectomy without angiography tended to have poorer limb prognosis than EVT or angiography-guided mechanical thrombectomy. A previous study reported the effectiveness of routine angiography during mechanical thrombectomy [28]. As the condition of the vessel (e.g., dissection, residual thrombosis, and degree of vessel stenosis after thrombectomy using a Fogarty catheter) could not be checked, routine angiography should thus be performed during surgical thrombectomy.
Prognosis
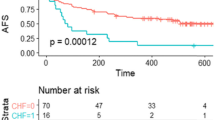
A previous study showed that ALI patients had poorer life prognosis than claudication patients [11]. In this study, the mortality rate at 1 year in ALI patients was 28.6%, which is fairly similar to that in previous Japanese and European cohort studies (24.5% and 23.0%, respectively) [7, 8]. However, a noteworthy difference between this study and previous studies was the cause of death. The rate of cardiovascular death was approximately 35% in this study and 64% in a previous European study [7], which might have been due to a race difference in the risk of atherosclerotic disease onset. Furthermore, the rate of major amputation was approximately 6% in this study, which is lower than that in a previous European cohort study [7].
Event onset time
The results of this study seem to indicate that most events occurred within 6 months. The short-term results for a Japanese prospective cohort suggested that all-cause death and cardiovascular events tended to occur more often within 5 months than over 5 months [8]. Furthermore, a previous multicenter cohort study showed that atherothrombosis patients with a prior history of ischemic events in the past year had significantly higher rate of cardiovascular death, myocardial infarction, or stroke than those without [29]. Atherothrombosis might require a certain period of time to stabilize, and this period has been suggested to be approximately 6 months for ALI. Therefore, we should pay more attention to the prevention of complications or life-threatening events within 6 months.
Limitations
The present study has three main limitations. First, patients who were treated using medical therapy alone were not included in this study. Second, the criteria for the selection of revascularization strategy for ALI patients varied among facilities in this study. This selection bias might have affected prognosis. Finally, the follow-up period was short. Further studies enrolling a large number of patients are required to verify which therapy can improve life and limb prognoses in ALI patients, and the efficacy of hybrid thrombectomy procedure or CDT revascularization for ALI patients.
Conclusion
In this registry, SVS/ISCVS class IIa ALI was predominant. Approximately 40% of primary revascularization strategy were surgery and EVT, followed by hybrid therapy. All-cause death and major amputation rates at 1 year were less than 30% and 6%, respectively.
References
Fowkes FG, Rudan D, Rudan I, et al. Comparison of global estimates of prevalence and risk factors for peripheral artery disease in 2000 and 2010: a systematic review and analysis. Lancet. 2013;382:1329–40.
Diehm C, Allenberg JR, Pittrow D, et al. Mortality and vascular morbidity in older adults with asymptomatic versus symptomatic peripheral artery disease. Circulation. 2009;120:2053–61.
Kullo IJ, Rooke TW. Clinical practice. Peripheral artery disease. N Engl J Med. 2016;374:861–71.
Utsunomiya M, Takahara M, Iida O, et al. Wound blush obtainment is the most important angiographic endpoint for wound healing. JACC Cardiovasc Interv. 2017;10:188–94.
Yamauchi Y, Takahara M, Shintani Y, et al. One-year outcomes of endovascular therapy for aortoiliac lesions. Circ Cardiovasc Interv. 2019;12:e007441.
Kawarada O, Yasuda S, Huang J, et al. Contemporary infrapopliteal intervention for limb salvage and wound healing: harmonization of revascularization and wound management. Circ J. 2014;78:1540–9.
Cambou JP, Aboyans V, Constans J, Lacroix P, Dentans C, Bura A. Characteristics and outcome of patients hospitalised for lower extremity peripheral artery disease in France: the COPART Registry. Eur J Vasc Endovasc Surg. 2010;39:577–85.
Higashitani M, Uemura Y, Mizuno A, et al. Cardiovascular outcome and mortality in patients undergoing endovascular treatment for symptomatic peripheral artery disease—short-term results of the Toma-Code Registry. Circ J. 2018;82:1917–25.
Eliason JL, Wainess RM, Proctor MC, et al. A national and single institutional experience in the contemporary treatment of acute lower extremity ischemia. Ann Surg. 2003;238:382–9.
de Donato G, Setacci F, Sirignano P, Galzerano G, Massaroni R, Setacci C. The combination of surgical embolectomy and endovascular techniques may improve outcomes of patients with acute lower limb ischemia. J Vasc Surg. 2014;59:729–36.
Darwood R, Berridge DC, Kessel DO, Robertson I, Forster R. Surgery versus thrombolysis for initial management of acute limb ischaemia. Cochrane Database Syst Rev. 2018;8:CD002784.
Inagaki E, Farber A, Kalish JA, et al. Outcomes of peripheral vascular interventions in select patients with lower extremity acute limb ischemia. J Am Heart Assoc. 2018;7:e004782.
Mizuno A, Anzai H, Utsunomiya M, et al. Real clinical practice of catheter therapy for deep venous thrombosis: periprocedural and 6-month outcomes from the EDO registry. Cardiovasc Interv Ther. 2015;30:251–9.
Creager MA, Kaufman JA, Conte MS. Clinical practice. Acute limb ischemia. N Engl J Med. 2012;366:2198–206.
Hunt SA, Baker DW, Chin MH, et al. ACC/AHA guidelines for the evaluation and management of chronic heart failure in the adult: executive summary. A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to revise the 1995 Guidelines for the Evaluation and Management of Heart Failure). J Am Coll Cardiol. 2001;38:2101–13.
The International Expert Committee. International Expert Committee report on the role of the A1c assay in the diagnosis of diabetes. Diabetes Care. 2009;32:1327–34.
Japanese Society of Hypertension Guidelines Subcommittee for the Management of Hypertension. Guidelines for the management of hypertension for general practitioners. Hypertens Res. 2001;24:613–34.
Teramoto T, Sasaki J, Ueshima H, et al. Diagnostic criteria for dyslipidemia. Executive summary of Japan Atherosclerosis Society (JAS) guideline for diagnosis and prevention of atherosclerotic cardiovascular diseases for Japanese. J Atheroscler Thromb. 2007;14:155–8.
Dormandy J, Heeck L, Vig S. Acute limb ischemia. Semin Vasc Surg. 1999;12:148–53.
Byrne RM, Taha AG, Avgerinos E, Marone LK, Makaroun MS, Chaer RA. Contemporary outcomes of endovascular interventions for acute limb ischemia. J Vasc Surg. 2014;59:988–95.
Ravn H, Björck M. Popliteal artery aneurysm with acute ischemia in 229 patients. Outcome after thrombolytic and surgical therapy. Eur J Vasc Endovasc Surg. 2007;33:690–5.
Cervin A, Tjärnström J, Ravn H, et al. Treatment of popliteal aneurysm by open and endovascular surgery: a contemporary study of 592 procedures in Sweden. Eur J Vasc Endovasc Surg. 2015;50:342–50.
Obara H, Matsubara K, Kitagawa Y. Acute limb ischemia. Ann Vasc Dis. 2018;11:443–8.
Trousseau A. Phlegmasia alba dolens. In: Cormack JR, editor. Clinique médicale de l’Hôtel-Dieu de Paris [Lectures on clinical medicine at Hôtel-Dieu de Paris]. London: New Sydenham Society; 1872. p. 281–332.
Rutherford RB, Baker JD, Ernst C, et al. Recommended standards for reports dealing with lower extremity ischemia: revised version. J Vasc Surg. 1997;26:517–38.
Honma Y, Urasawa K, Sato K, Koshida R. A new device for percutaneous mechanical thrombectomy in acute limb ischemia. Cardiovasc Interv Ther. 2011;26:166–71.
Leung DA, Blitz LR, Nelson T, et al. Rheolytic pharmacomechanical thrombectomy for the management of acute limb ischemia: results from the PEARL Registry. J Endovasc Ther. 2015;22:546–57.
Zaraca F, Stringari C, Ebner JA, Ebner H. Routine versus selective use of intraoperative angiography during thromboembolectomy for acute lower limb ischemia: analysis of outcomes. Ann Vasc Surg. 2010;24:621–7.
Bhatt DL, Eagle KA, Ohman EM, et al. Comparative determinants of 4-year cardiovascular event rates in stable outpatients at risk of or with atherothrombosis. JAMA. 2010;304:1350–7.
Acknowledgements
The authors thank Yuichi Suzuki, Yoshinori Nagashima, and Junji Yajima for collecting data and improving our discussion. The authors also express great gratitude for the cooperation of the hospitals involved in the Edo registry.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
All authors have no conflicts of interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Higashitani, M., Anzai, H., Mizuno, A. et al. One-year limb outcome and mortality in patients undergoing revascularization therapy for acute limb ischemia: short-term results of the Edo registry. Cardiovasc Interv and Ther 36, 226–236 (2021). https://doi.org/10.1007/s12928-020-00662-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12928-020-00662-6