Abstract
Purpose
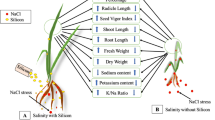
Salt stress poses a threat to wheat performance that can be managed by silicon application. Therefore, the current short term experiment was aimed to learn the effect of silicon on the defense system, ionic composition and growth of wheat in the presence and absence of salt stress.
Methods
In the current experiment, the influence of silicon (6 mM) on anti-oxidative potential and dry matter yield of wheat genotypes (salt sensitive; Fareed-06 and salt tolerant; Inqlab-91) was studied under saline (10 dS/m) and control (2 dS/m) hydroponic solutions. The whole Si was applied to the pots of the Si+ treatment (6 mM) using calcium silicate solution. CaCl2 solution was applied to the pots of the Si-deficient treatment to balance the same total of Ca as in the Si+ treatment to obtain the sole effect of Si. Various ionic, biochemical and growth parameters of wheat plants were recorded after 34 days of transplanting according to standard procedures.
Results
Silicon addition to the hydroponic solution increased wheat seedling weight, K:Na with limited Na and enhanced K uptake. Moreover, Si increased chlorophyll chl a/b ratio and promoted the actions of superoxide dismutase (SOD) and catalase (CAT).
Conclusions
It can be concluded that silicon supplementation improved the wheat growth and plant defense suppressed by the salt applied. Further, salt tolerant variety gave better performance than the salt sensitive variety.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Abbreviations
- Abbreviation :
-
Explanation
- Si:
-
Silicon
- SOD:
-
Superoxide dismutase
- CAT:
-
Catalase
- Chl :
-
Chlorophyll
References
Wang W, Vinocur B, Shoseyov O (2000) Altman A Biotechnology of plant osmotic stress tolerance physiological and molecular considerations. In: IV International Symposium on In Vitro Culture and Horticultural Breeding 560, pp 285–292
Ashraf M (2009) Biotechnological approach of improving plant salt tolerance using antioxidants as markers. Biotechnol Adv 27(1):84–93
Garg BK, Burman U, Kathju S (2004) The influence of phosphorus nutrition on the physiological response of moth bean genotypes to drought. J Plant Nutr Soil Sci 167(4):503–508
Raza SH, Athar H-U-R, Ashraf M (2006) Influence of exogenously applied glycinebetaine on the photosynthetic capacity of two differently adapted wheat cultivars under salt stress. Pak J Bot 38(2):341–351
Tester M, Davenport R (2003) Na + tolerance and Na + transport in higher plants. Ann Bot 91(5):503–527
Weisany W, Sohrabi Y, Heidari G, Siosemardeh A, Ghassemi-Golezani K (2012) Changes in antioxidant enzymes activity and plant performance by salinity stress and zinc application in soybean (’Glycine max’L.)
Gunes A, Inal A, Bagci EG, Pilbeam DJ (2007) Silicon-mediated changes of some physiological and enzymatic parameters symptomatic for oxidative stress in spinach and tomato grown in sodic-B toxic soil. Plant Soil 290(1–2):103–114
Zhu Z, Wei G, Li J, Qian Q, Yu J (2004) Silicon alleviates salt stress and increases antioxidant enzymes activity in leaves of salt-stressed cucumber (Cucumis sativus L.) Plant Sci 167(3):527– 533
JohnsonCM SP, Broyer T, Carlton A (1957) Comparative chlorine requirements of different species. Plant Soil 8:337–353
Wolf B (1982) A comprehensive system of leaf analyses and its use for diagnosing crop nutrient status. Commun Soil Sci Plant Anal 13(12):1035–1059
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1):248–254
Lutts S, Kinet J, Bouharmont J (1996) NaCl-induced senescence in leaves of rice (Oryza sativa L.) cultivars differing in salinity resistance. Ann Bot 78(3):389–398
Giannopolitis CN, Ries SK (1977) Superoxide dismutases I. Occurrence in higher plants. Plant Physiol 59 (2):309–314
Chance B, Maehly A (1955) [136] Assay of catalases and peroxidases. Methods Enzymol 2:764–775
Scholander PF, Hammel H, Hemmingsen E, Bradstreet E (1964) Hydrostatic pressure and osmotic potential in leaves of mangroves and some other plants. Proc Natl Acad Sci U S A 52(1):119
Yildirim B, Yaser F, Ozpay T, Ozpay D, Turkozu D, Terziodlu O, Tamkoc A (2008) Variations in response to salt stress among field pea genotypes (Pisum sativum sp. arvense L.) J Anim Veter Adv 7:907–910
Tabaei-Aghdaei S, Harrison P (2000) Expression of dehydration-stress related genes in crown of wheat grass species having contrasting acclimation to salt, cold and drought. Plant Cell Environ 23:561–571
Chen CT, Li CC, Kao CH (1991) Senescence of rice leaves. xxxi. Changes of chlorophyll, protein and polyamine contents and ethylene production during senescence of a chlorophyll-deficient mutant. J Plant Growth Reg 10:201–205
Munns R (2005) Genes and salt tolerance: bringing them together. New Phytol 167(3):645–663
Ahmad R, Zaheer SH, Ismail S (1992) Role of silicon in salt tolerance of wheat (Triticum aestivum L.) Plant Sci 85(1):43–50
Yeo A, Flowers S, Rao G, Welfare K, Senanayake N, Flowers T (1999) Silicon reduces sodium uptake in rice (Oryza sativa L.) in saline conditions and this is accounted for by a reduction in the transpirational bypass flow. Plant, Cell & Environment 22(5):559–565
Gong Hj, Chen Km, Chen Gc, Wang Sm, Zhang Cl (2003) Effects of silicon on growth of wheat under drought. J Plant Nutr 26(5):1055–1063
Gong H, Zhu X, Chen K, Wang S, Zhang C (2005) Silicon alleviates oxidative damage of wheat plants in pots under drought. Plant Sci 169(2):313–321
Moussa HR (2006) Influence of exogenous application of silicon on physiological response of salt-stressed maize (Zea mays L.) Int J Agric Biol 8(3):293–297
Liang Y, Shen Q, Shen Z, Ma T (1996) Effects of silicon on salinity tolerance of two barley cultivars. J Plant Nutr 19(1):173–183
Romero-Aranda MR, Jurado O, Cuartero J (2006) Silicon alleviates the deleterious salt effect on tomato plant growth by improving plant water status. J Plant Physiol 163(8):847– 855
Dionisio S, Tobita S (1999) Antioxidative responses of shoots and roots of wheat to increasing NaCl concentration. J Plant Physiol 155:274–280
Ali A, Basra SMA, Iqbal J, Hussain S, Subhani MN, Sarwar M, Ahmad M (2012) Si-mediated biochemical changes in wheat under salinized and non-salinized solution culture. African J Biotech 11(3):606–615
Epstein E (2001) Silicon in plants: facts vs. concepts. In: Datnoff LE, Snyder GH, Korndorfer GH (eds) Silicon in agriculture. Elsevier, Amsterdam, pp 1–16
Elliot CL, Snyder GH (1991) Autoclave-induced digestion for the calorimetric determination of silicon in rice straw. J Agri Food Chem 39:1118–1119
Arnon DT (1949) Copper enzyme in isolated chloroplasts polyphenoloxidase in Beta vulgaris. Plant Physiol 24:1–15
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ali, A., Haq, T.u., Mahmood, R. et al. Stimulating the Anti-Oxidative Role and Wheat Growth Improvement Through Silicon Under Salt Stress. Silicon 11, 2403–2406 (2019). https://doi.org/10.1007/s12633-015-9378-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12633-015-9378-4