Abstract
Objective
Caloric restriction (CR) is proven to be effective in increasing life span and it is well known that, nutritional habits, sleeping pattern and meal frequency have profound effects on human health. In Ramadan some Muslims fast during the day-light hours for a month, providing us a unique model of intermittent fasting (IF) in humans. In the present study, we have investigated the effects of IF versus CR on the same non-diabetic obese subjects who were followed for two years according to the growth hormone (GH)/Insulin like growth factor (IGF)-1 axis and insulin resistance.
Design
Single-arm Interventional Human Study.
Participants
23 female subjects (Body Mass Index (BMI) 29-39, aged between 28-42years).
Setting
Follow-up is designed as 12 months of CR, after which there was a month of IF and 11 months of CR again, to be totally 24 months. Subjects’ daily diets were aligned as low calorie diet during CR and during the IF period, the same subjects fasted for 15 hours in a day for a month and there was no daily calorie restriction. Nutritional pattern was changed as 1 meal in the evening and a late supper before sleeping and no eating and drinking during the day light hours in the IF model. Subjects made brisk walking twice a day during the whole follow-up including both CR and IF periods. BMI, Blood glucose, insulin, TSH, GH, HbA1c, IGF-1, Homa-IR and urinary acetoacetate levels were monitored once in three months and twice in the fasting month.
Measurements and Results
While subjects lost 1250 ± 372g monthly during the CR, in the IF period, weight loss was decreased to 473 ±146 g. BMI of all subjects decreased gradually and as the BMI decreased, glucose, HbA1c, insulin, Homa-IR and TSH levels were decreased. GH levels were at baseline at the beginning, increased in the first six months and stayed steady during the CR and IF period than began decreasing after the IF period, while IGF-I increased gradually during the CR period and beginning with the 7th day of IF period, it decreased and kept on decreasing till the end of the follow-up. Urinary acetoacetate levels were higher during the IF period suggesting a constant lipid catabolism.
Conclusion
Our results suggest that, CR affects metabolic parameters positively which will help especially pre-diabetic and insulin resistant patients without any pharmacological approach. In addition IF without calorie restriction can enhance health and cellular resistance to disease without losing weight and those effects may be attributed to different signalling pathways and circulating ketones during IF. Changes observed during IF are probably due to the changes in eating and sleeping pattern and thus changes in metabolic rhythm.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
High caloric intake and a sedentary life style are well known environmental risk factors having an impact on life-span. Caloric restriction (CR) is proven to be effective in increasing life-span and disease resistance in rats, mice, and Rhesus monkeys in recent studies (1-3). Caloric restriction models generally refer to a 30-40% decrease in daily caloric intake (3, 4). On the other hand, intermittent fasting (IF) is also shown to have positive impact on life span (4). IF model in mice is defined as every other day feeding without caloric restriction (4). Although there is no consensus and defined IF model in humans, a prolonged intermittent fasting time for a month during Ramadan provides a good IF model. In Ramadan every year, depending on the time of year, Muslims fast for approximately 15 hours during the day and eat at night without caloric restriction for a month. Many physiological changes are observed during Ramadan fasting that are likely due to the changes in eating and sleeping patterns. Many societies have recognized the beneficial effects on health and longevity of limited food intake for certain periods of time, either for religious reasons or when the food supply was insufficient (5). In the present study, we have investigated the effects of IF versus CR (40%) on obese non-diabetic subjects regarding the GH/IGF axis, insulin resistance.
Methods
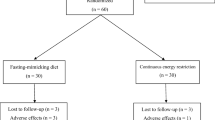
Twenty-three female subjects without diabetes whose body mass indices (BMI) were 29-37 (mean ± SD, 34.15 ± 2.15 and aged 28-42 (mean ± SD, 36 ± 3.12) years were followed for 24 months. The study design and plan were thoroughly explained to the patients orally and in writing, and written informed consent was obtained from all patients and the study was approved by the ethics committee of Maltepe University, School of Medicine. The study has begun with 30 patients, 7 of whom, were excluded since they were not adherent to the diet regimens.
Follow-up is designed as 12 months of CR, after which there was a month of IF (Ramadan) and 11 months of CR again, to be totally 24 months. Patients’ routine daily diets were followed for a month with a questionnaire and daily caloric intake was calculated (mean ± SD, 2610 ± 409 calories) before the study began. Mean caloric intake of all the subjects was aligned to be 40% less during the study, which was followed and recorded by weekly interviews. The main restriction was from carbohydrates (CH); daily CH intake was restricted to 120g and total caloric intake was aligned to be nearly (mean ± SD) 1518 ± 212 calories during the study. Subjects made brisk walking twice a day (am and pm) for an hour in total. This was the CR model. The same subjects fasted for 15 hours/day for 30 days during the month of Ramadan. This model was used as the IF model, which is also called “Prolonged intermittent fasting”. During IF, there was no daily calorie restriction. The nutritional pattern was changed to one meal in the evening and a late supper one hour before sleeping, with no eating or drinking during the day. Daily brisk walking sessions were continued and subjects continued their daily activities during the fasting days.
The follow-up began 12 months before the fasting month in 2014 and ended at June 2015. Nine fasting blood and urine samples were taken during the follow-up (once in three months before Ramadan, at the end of the first week (7th day), and the last week (21st day) of Ramadan Month and three more samplings after Ramadan), sampling design and frequency are summarized in Table 1. Totally 9 samplings were performed. Glucose, insulin, thyroid stimulating hormone (TSH), growth hormone (GH), hemoglobin (Hb)A1c, insulin like growth factor (IGF)-1, Homa-IR, and urinary acetoacetate levels were monitored. Twenty-four hour urine samples were collected for six times during the follow-up and 24 hour urinary ketones were determined semi-quantitatively. Additionally, 24 hour urinary volumes were monitored in order to determine any kind of dehydration during IF. Glucose was analyzed by hexokinase/ G6PDH method in RxLMax (Siemens/Germany) autoanalyzer. Insulin, TSH, GH were measured by electrochemiluminescence immunoassay (ECLIA) method in Roche/ Cobas (Germany) systems, and the analytic sensitivity of GH reagent was 0.002 ng/mL. Homa-IR was calculated as multiplying fasting serum insulin (FPI) by fasting serum glucose (FPG), then dividing by the constant 22.5, i.e. HOMA-IR = (FPI×FPG)/22.5 (6), IGF-1 was measured by chemiluminescence immunoassay method (CLIA) in IDS-ISYS (Immunodiagnostic Systems-France) and finally HbA1c was measured by HPLC method (BioRad- Variant-England/USA) and only five samplings were made for HbA1c during the entire follow-up (Table 2). All blood samples were taken after 12 hour fasting, and during Ramadan, great attention was paid to wait for 12 hours after the last meal.
Statistical analysis was performed by using repeatedmeasures ANOVA since each subject served as her own control. Post hoc testing was performed with Dunn’s multiple comparison test and p values <0.05 were considered to be statistically significant.
Results
While subjects lost 1250 ± 372g on average in a month during the CR follow-up, in the IF period, weight loss decreased to 473 ± 146g. Twenty-four hour urinary volumes did not change significantly when the IF period was compared to CR period (data not shown).

As the BMI decreased, glucose, insulin, and Homa-IR levels decreased significantly with CR. During the IF period while glucose levels continued to decrease, insulin levels remained at the same level (Fig and 2). TSH levels were 3.15 ± 0.85 uIU/ mL on average at the beginning, and decreased gradually with decreasing BMI in all patients (Table 2). HbA1c percentage begun the study with 6.46 ± 1.26% on average and after 24 months it was 4.85 ± 1.20 % (Table 2). GH levels were at baseline at the beginning, increased in the first six months, stayed steady during the follow-up and IF period then decreased after the IF period (Fig 3), while IGF-I increased gradually during the follow-up and beginning at the 7th day of IF period, it decreased and kept on decreasing till the end of the follow-up (Fig 3). Urinary acetoacetate levels were monitored six times, which were higher during the IF period, suggesting an increased lipid catabolism during the fasting days (Table 2).

Discussion
There are great struggles to extend healthy lifespan in humans. Longevity-extending studies have focused on GH, IGF-1, and insulin axis, and their effects on survival and healthy aging. It has been thoroughly demonstrated that nutritional habits, sleeping patterns, and meal frequency have profound effects on human health (7, 8). Caloric restriction and IF are both shown to be potential diets for healthy body and brain aging (5). A reduction in food intake without malnutrition, as in the CR model, may decrease the activity of nutrientsignaling pathways that mimic the periods of food shortage in nature (1), which in turn may activate cellular stressresistance pathways. Furthermore, in a study by Anson et al., it is shown that without caloric restriction, intermittent fasting (IF) had beneficial effects on neuronal resistance and aging, showing the same effects as the CR model without a significant weight loss in mice (4).

In the present study, after the 24 month follow up, BMI, fasting glucose, insulin, HbA1C, and HOMA-IR levels of all subjects decreased gradually overtime, suggesting long term CR and physical exercise have great beneficial effects on the health of obese patients. Decreasing HOMA-IR and insulin levels together with decreasing HbA1C concentrations show that the risk of type 2 diabetes and metabolic syndrome is significantly reduced by long term CR. This beneficial effect has been well reported by previous studies and already applied to therapy (9, 10). The unknown is, whether prolonged IF has similar influences on the health and risk of metabolic diseases. To date, there have been limited comparison studies on humans to understand the effects of long term CR and IF. The present study showed both long term caloric restriction and a short term IF model have beneficial effects on human health while they have different influences on the IGF-GH-insulin axis. During CR, IGF-1 levels increased gradually, while during IF, with the 7th day of fasting period there was a decline which continued till the end of the follow-up (Fig. 3). Although IF lasted only for one month and again a CR period began, its effects seem to continue for at least 11 months till the end of the study. This decline was not clear in GH levels, it began to decrease after the IF period. These findings suggest that there may be a difference in the signaling pathways of IF and CR according to the GH-IGF-1and insulin axis.
GH secretion is regulated by a complex network of neurotransmitters and neuropeptides. It is mainly under control of GH-releasing hormone and somatostatin besides other hormones, including sex steroids, glucocorticoids, gastrointestinal hormones and metabolic factors. Nutrition and metabolism play a key role in the regulation the GH-IGF-1 axis. It is shown that in obesity, stimulated GH peak is negatively correlated with BMI (11).
IGF-1 mediates many of the somatic effects of GH and GH concentrations are controlled by a negative feedback due to free IGF-1 levels. Recent data have shown that IGF-1 has insulinlike effects on the peripheral uptake of glucose and fatty acids (12). Most cells that express the IGF-1 receptor (IGF-IR) and IGF-1 in circulation are mostly produced by the liver, and its levels largely regulated by GH. However, other factors may also affect hepatic IGF-1 synthesis, including nutritional status, such as caloric intake and protein consumption, insulin, and inflammatory cytokines (13-15). There is growing evidence that shows increased IGF-1 levels have been associated with reduced insulin resistance (16). In the present study increase in IGF-I levels and the decrease in HOMA-IR values during CR are consistent with these findings. Administration of recombinant IGF-1 was found to reduce serum glucose levels and improve insulin sensitivity in healthy adults as well as in insulin resistant subjects and type 2 diabetics (17). This insulin sensitizing effect of IGF-1 may not only be due to its GH suppressing effect, but also due to independent IGF-1 effects. It has been reported that insulin like growth factor binding protein (IGFBP)-3, the most abundant IGFBP in circulation (among the known six of them), may play a role in glucose homeostasis by binding to a nuclear receptor that interacts with peroxisome proliferator activated receptor-gamma (PPAR-γ), a nuclear protein involved in the regulation of glucose and lipid metabolism (18, 19). The metabolic effects of IGFBP-3 are largely opposite of those of IGF-1 (20). Transgenic animal data demonstrate that over expression of IGFBP-3 is associated with fasting hyperglycemia and impaired glucose tolerance in mice (21). In the current study, the increase in IGF-1 during CR may result in decreased levels of IGFBP-3 due to binding, resulting in increased insulin sensitivity. We have shown in this particular study that GH and IGF-I concentrations were down regulated during and after IF period. GH/IGF-I signaling with nutrition has different aspects. A decreased GH/IGF-I signaling is a common characteristic of aging where as a constitutively decreased pathway extends longevity (2). GH/ IGF-I pathway down modulation may reflect a defensive response for minimizing cell growth and metabolism during systemic damage trying to escape from death. It is shown that organisms with a constitutively decreased GH/IGF-I pathway can survive longer (2). This GH/IGF-I axis down regulation during and after the IF period may be due to low energy intake and adaptation of the metabolism.
During IF, BMI did not change significantly, but after the IF period, it again began to drop with ongoing CR, suggesting day-long fasting model (IF) cannot be assumed as a weight loss program, but can be thought of as a detoxification/regeneration or a longevity-extending process since IGF-1 levels were lower during IF and also circulating ketones were elevated which stayed high during nine months after IF period. During IF, increased reliance on β-oxidation of fatty acids for meeting energy needs as a response to reduced availability of nutrients, may be associated with improved metabolic homeostasis. A shift from carbohydrate to fatty acid utilization in response to reduced nutrients is supposed to be an important metabolic adaptation of mitochondrial function and one of the key mechanisms of extended longevity (22). Mice on an IF regimen have been shown to develop a two-fold increase in the fasting serum concentrations of ketones compared with mice on CR (5). This shift to ketogenesis may play a direct role in the cytoprotective effects of IF, since it has been reported that rats that were fed a ketogenic diet exhibited increased resistance to seizures and β-hydroxybutirate itself can protect neurons in models of Alzheimer’s and Parkinson’s disease (23, 24). Changes in the IF period may be due to omitting at least two meals when the body is metabolically active. Moreover, glycogen stores of all cells must be regenerated every day since there was absolute fasting during the day for 15 hours. Regarding IGF-1, recent data have shown an inverse correlation between levels of IGF-1 with CRP and other cytokines, indicating IGF-1 may be involved in the altering inflammatory response to insulin resistance and its progression to type-2 diabetes (25). Furthermore, it is shown that circulating levels of adiponectin, an important anti-inflammatory adipokine, are elevated in long-lived, calorie restricted mice, whereas the expression of proinflammatory cytokines, IL-6, and TNF-α are reduced (26). In the current study, although concentration of serum adiponectin was not determined in the subjects, one of the previous studies of the researchers indicated that IL-6 and CRP levels significantly decreased in an IF model (7). Adiponectin, in addition to its anti-inflammatory properties, also promotes B-oxidation of fatty acids and enhances insulin sensitivity by activating adenosine monophosphate-activated protein kinase (27). Moreover, cellular stress-resistance pathways may be activated by IF; It has been shown that levels of chaperones and neurotropic factors increased in rats maintained on an IF regimen (4). In our IF model, during the fasting day light hours, catabolism is active and on gorging, anabolism is active. Alternating periods of anabolism and catabolism may play a mechanistic role in triggering cellular stress-resistance pathways and repair of damaged cells. Hence the beneficial effects of this IF model may be due to the stress associated with fasting rather than the caloric intake. Since it is shown that IGF-1 signaling is neuroprotective (28, 29), it is important to determine the mechanisms by which CR and IF differentially affect IGF-1 levels and insulin signaling and their influence on energy metabolism, disease resistance, and longevity. Therefore, it must be well investigated in future studies whether a reduction in calorie intake is the only dietary method by which to increase healthy longevity or whether IF without calorie restriction might have beneficial effects similar to CR.
This study had certain limitations one of which is we did not have a different control group, we thought it is important to see the changes particularly on the same subjects, since they served as their own controls and by this way we had avoided the inter-individual variation in the monitored parameters. Second limitation is about the sampling; We had no sampling just before the IF period, the last sampling was three months ago. Since CR had began 12 months ago before the IF period, which means subjects had already reached the basal levels of all metabolic parameters, we assessed the results comparing to these measurements. Finally the last limitation is about the sample size of the study which seems to be small, we had begun the study with 30 subjects but as stated before 7 of them had to be excluded because of the lack of adherence to the diet regimen since it is a really challenging task for the individuals for 24 months. But still 23 subjects seem to be proper enough for reflecting the differences between CR and IF model. There is limited data on humans in IF model and in our opinion the present study adds valuable data to this area.
In conclusion, this study demonstrated, constantly losing weight with CR and physical exercise affects metabolic parameters positively, which will particularly aid pre-diabetic and insulin resistant patients leading to non-pharmacologic approaches. Moreover one of the most striking result of the study is that IF can enhance health and cellular resistance to disease by different mechanisms, even if the fasting period is followed by a period of overeating such that overall caloric intake is not decreased. Further human studies on a molecular basis are needed in order to reveal the signaling mechanisms that differentially occur during intermittent fasting and CR to support these results.
Conflict of Interest: None
Acknowledgements: We want to thank to our subjects who made this work come to life with their adherence to our programme for two years.
Ethical Standards: Experiments comply with the current laws of country in which they were performed.
References
Fontana L, Patridge L, Longo V. Dietary restriction, growth factors and aging. From yeast to humans. Science April 2010;16; 328(5976):321–326
Lopez-Otin C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell 2013;53: 1194–1217
Colman RJ, Anderson RM, Johnson SC, Kastman EK, Kosmatka KJ, Beasly TM, et al. Caloric restriction delays disease onset and mortality in rhesus monkeys. Science 2009;10;325 (5937):201–204
Anson RM, Guo Z, Cabo R, Lyun T, Rios M, Hagepanos A, et al. Intermittent fasting dissociates beneficial effects of dietary restriction on glucose metabolism and neuronal resistance to injury from calorie intake. PNAS 2003;100: 6216–6220
Martin B, Mattson M, Maudsley S. Caloric restriction and intermittent fasting: two potential diets for successful brain aging. Aging Research Reviews 2006;58(3): 332–353
Ascaso JF, Pardo S, Real JT, Lorente RI, Priego A, et al. Diagnosing Insulin Resistance by Simple Quantitative Methods in Subjects With Normal Glucose Metabolism. Diabetes Care 2003;26: 3320–3325.
Aksungar FB, Topkaya AE, Akyildiz M. Interleukin-6, C-reactive protein and biochemical parameters during prolonged intermittent fasting. Ann Nutr Metab 2007;51: 88–95
Bogdan A, Bouchareb B, Touitou Y. Ramadan fasting alters endocrine and neuroendocrine circadian patterns. Meal-time as a synchronizer in humans? Life Sci 2001;68: 1607–1615
Beavers KM, Gordon MM, Easter L, Beavers DP, Hairston KG, Nicklas BJ, Vitolins MZ. Effect of protein source during weight loss on body composition, cardiometabolic risk and physical performance in abdominally obese, older adults: a pilot feeding study. J Nutr Health Aging. Jan;2015;19(1): 87–95. doi: 10.1007/s12603-015-0438-7.
Normandin E, Senecal M, Prudhomme R, Rabasa-Lhorat R, Brochu M. Effects of Caloric Restriction with or without Resistance Training in Dynapenic-Overweight and Obese Menopausal Women: A Monet Study. J Frailty Aging 2015;4(3): 155–162
Vottero A, Guzetti C, Loche S. New Aspects of The Physiology of The GH-IGF-I Axis. Endocr Dev 2013;24: 96–105
Rajpathak SN, Gunter MJ, Wylie-Rosett J, Ho GYF, Kaplan RC, Muzumdar R, et al. The role of insulin-like growth factor-I and its binding proteins in glucose homeostasis and type 2 diabetes. Diabetes Metab Res Rev 2009;25: 3–12
Underwood LE, Thissen JP, Lemozy S, Ketelslegers JM, Clemmons DR. Hormonal and nutritional regulation of IGF-I and its binding proteins. Horm Res 2004,42: 145–151.
Thissen JP, Ketelslegers JM, Underwood LE. Nutritional regulation of the insulinlike growth factors. Endocr Rev 1994,15: 80–101.
Kaushal K, Heald AH, Siddals KW, et al. The impact of abnormalities in IGF and inflammatory systems on the metabolic syndrome. Diabetes Care 2004,27: 2682–2688.
Woods KA, Camacho-Hubner C, Bergman RN, Barter D, Clark AJ, Savage MO. Effects of insulin-like growth factor I (IGF-I) therapy on body composition and insulin resistance in IGF-I gene deletion. J Clin Endocrinol Metab 2000,85:1407–1411.
Moses AC, Young SC, Morrow LA, O’Brien M, Clemmons DR. Recombinant human insulin-like growth factor I increases insulin sensitivity and improves glycemic control in type II diabetes. Diabetes 1996;45: 91–100.
Yamanaka Y, Fowlkes JL, Wilson EM, Rosenfeld RG, Oh Y. Characterization of insulin-like growth factor binding protein-3 (IGFBP-3) binding to human breast cancer cells: kinetics of IGFBP-3 binding and identification of receptor binding domain on the IGFBP-3 molecule. Endocrinology 1999;140: 1319–1328.
Schedlich LJ, Le Page SL, Firth SM, Briggs LJ, Jans DA, Baxter RC. Nuclear import of insulin-like growth factor-binding protein-3 and -5 is mediated by the importin beta subunit. J Biol Chem 2000;275: 23462–23470.
Muzumdar RH, Ma X, Fishman S, et al. Central and opposing effects of IGF-I and IGF-binding protein-3 on systemic insulin action. Diabetes 2006;55: 2788–2796.
Silha JV, Gui Y, Murphy LJ. Impaired glucose home2006;ostasis in insulin-like growth factor-binding protein-3-transgenic mice. Am J Physiol Endocrinol Metab 2002;283: E937–E945.
Poplawski MM, Mastaitis JW, Yang XJ, Mobbs CV. Hypothalamic responses to fasting indicate metabolic reprogramming away from glycolysis toward lipid oxidation. Endocrinol 2008;295: 101–105.
Bough KJ, Valiyil R, Han FT, Eagles DA. Seizure resistance is dependent upon age and calorie restriction in rats fed a ketogenic diet. Epilepsy Res 1999;35: 21–28
Kashiwaya Y, Takeshima T, Mori N, Nakashima K, Clarke K, Veech R. D-betahydroxybutyrate protects neurons in models of Alzheimer’s and Parkinson’s disease. Proc. Natl. Acad. Sci. 2000;97: 5440–5444.
Rajpathak SN, McGinn AP, Strickler HD, et al. Insulin-like growth factor (IGF)-axis, inflammation, and glucose intolerance among older adults. Growth Horm IGF Res 2008;18(2): 166–173.
Wang Z, Al-Regaiey KA, Masternak MM, Bartke A. Adipocytokines and lipid levels in Ames dwarf and caloric restricted mice. J Gerontol A Biol Sci Med Sci 2006;61A: 323–331
Brown-Borg HM, Bartke A. GH and IGF1: Roles in energy metabolism of longliving GH mutant mice. J Gerontol A Biol Sci Med Sci. June; 2012;67A(6): 652–660
Cheng, B. & Mattson, M.P. IGF-I and IGF-II protect cultured hippocampal and septal neurons against calcium-mediated hypoglycemic damage. J. Neurosci 1992;12: 1558–1566.
Hsieh, C.C., DeFord, J.H., Flurkey, K., Harrison, D.E., Papaconstantinou, J. Implications for the insulin signaling pathway in Snell dwarf mouse longevity: a similarity with the C. elegans longevity paradigm. Mech. Ageing Dev 2002;123: 1229–1244.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aksungar, F.B., Sarikaya, M., Coskun, A. et al. Comparison of intermittent fasting versus caloric restriction in obese subjects: A two year follow-up. J Nutr Health Aging 21, 681–685 (2017). https://doi.org/10.1007/s12603-016-0786-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12603-016-0786-y