Abstract
Background
Anemia and malnutrition are highly prevalent, frequently concomitant and associated with negative outcomes and mortality in the elderly.
Objectives
To evaluate the association between these two entities, and test the hypothesis that protein-energy deficit could be etiology of anemia.
Design
Prospective case-control study. Setting: Geriatric and Rehabilitation Hospital, Geneva University Hospitals, Switzerland.
Participants
392 patients (mean age 84.8 years old, 68.6% female).
Main outcome measures
Hematological (hemoglobin (Hb)), chemical (iron work up, cyanocobalamin, folates, renal function, C-Reactive Protein (CRP)) and nutrition (albumin, prealbumin) parameters, and mini nutritional assessment short form (MNA-SF).
Results
The prevalence of anemia (defined as Hb<120 g/l) was 39.3%. Anemic patients were more frequently malnourished or at risk of malnutrition according to the MNA-SF (p=0.047), with lower serum albumin (p <0.001) and prealbumin (p <0.001) levels. Thirty-eight percent of these patients had multiple causes and 14.3% had no cause found for anemia. Among the latter 90.9% of patients with unexplained anemia had albumin levels lower than 35g/l. After exclusion of iron,vitamin B12 and folic acid deficits, anemic patients had lower albumin (p<0.001) and prealbumin (p 0.007) levels. Albumin level explained 84.5% of the variance in anemia. In multivariate analysis albumin levels remain associated with Hb only in anemic patients, explaining 6.4% of Hb variance (adj R2) and 14.7% (adj R2) after excluding inflammatory parameters (CRP>10).
Conclusions
Albumin levels are strongly associated with anemia in the elderly. Screening for undernutrition should be included in anemia assessment in those patients. Further prospective studies are warranted in order to explore the effect of protein and energy supplementation on hemoglobin level.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Anemia is very frequent in elderly and associated with negative clinical outcomes (1) as functional impairment, falls, multiple co-morbid conditions, hospitalization rate, longer hospital length of stay and mortality (2-4).
It may be part of the frailty process of ageing and its prevalence in over 65 year-old community-dwelling people is estimated at 11% in men, and 10.2% in women. It rises up to more than 20% in 85 years and older (5, 6). In oldest old hospitalized patients the prevalence is even higher (40%) (7, 8). Even so, anemia is often undertreated and simply considered as age-related (4). Concerning the etiologies of anemia, it is considered that one third of cases are due to nutritional deficits (iron, folic acid and/or vitamin B12). Half of them are due to an iron deficit (combined or not with vitamin deficiencies), which could be the sole cause of anemia in 25% of cases (9). Another third is associated with chronic diseases or chronic renal insufficiency, together qualified as anemia of chronic disease (ACD) (5, 10, 11). ACD is frequent in elderly, directly related to the various inflammation pathways, independently from the underlying cause of inflammation. Anemia in elderly is often multifactorial and ACD may coexist with nutritional deficits. Even after large work-ups including the above-mentioned causes, one third of anemia cases remain of unknown origin (5).
Associated with negative outcomes and long-term mortality in community-dwelling populations and older inpatients, malnutrition is another important and frequent concern in elderly (12-16). Defined as a state of insufficient dietary intakes concerning proteins, calories and micronutrients, its prevalence is higher than 40-50% in hospital and nursing home settings. Thirty to 60% of elderly patients admitted to a hospital are either malnourished or at risk of malnutrition and 23% and 51% respectively in geriatric in-patients (15, 16). Malnutrition may overlap with cachexia, which corresponds to a reaction to a disease state or to co-morbidities, usually associated with some degree of inflammation, and anemia related to cachexia is probably caused by the underlying chronic condition and inflammatory reaction (12, 13). Nevertheless, except cases of cachexia, it would be interesting to assess whether malnutrition may be associated with anemia, independently of an underlying chronic inflammation or an underlying nutritional deficit in iron/vitamin B12 or folic acid. An association between anemia, hypo-albuminemia and other biological and clinical nutritional parameters has already been demonstrated in hospitalized elderly patients (17), in long-term care structures (18) and community-dwelling populations (19). However, association between anemia and malnutrition has never been studied after exclusion of potential confounding factors as co-existing nutritional deficits, such as iron, vitamin B12 and folic acid, or underlying inflammatory state.
The aim of the study is to describe the epidemiology of anemia in a very old hospitalised group and assess the association between hemoglobin and nutritional status, in patients without iron and vitamin deficiencies and taking into account inflammation and chronic kidney disease. If this association can be detected, we could raise the hypothesis as to whether malnutrition could contribute to the pathogenesis of anemia of unknown etiology.
Methods
The study took place between February 2010 and November 2012 in the Geriatrics and Rehabilitation Hospital of the Geneva University Hospitals, Switzerland. This hospital is composed of general geriatric, internal medicine units and one geriatric-orthopaedics unit. In its prospective case-control design, the study was sequentially proposed to all patients hospitalized in the general geriatric and internal medicine units. All included patients signed a written consent form. Exclusion criteria for the study were: refusal, Mini-Mental State Evaluation (MMSE) <18/30, ongoing hemodialysis, palliative care, secondary diabetes (for exemple after pancreatectomy), corticosteroid treatments, known myelodysplastic disease and post-operative state.
Primary outcomes were the hematological parameters allowing definition and categorization of anemia and the biological, clinical and anthropometric parameters of nutrition. Secondary measurements were general informations as age and provenance, treatments and cognitive status. The inclusion was performed within 7 days after hospital admission. Between day 1 and 4 after inclusion, blood tests were done and the patients were evaluated during a clinical consultation done by the same medical and nurse team for the clinical parameters.
Data collected for every included patient were: date of birth, sex, provenance (other medical unit or home) and medications. The biological parameters studied on a blood sample were hemoglobin, mean corpuscular volume, hematocrit, red blood cells, reticulocytes, white blood cell count, thrombocytes, iron parameters (iron, transferrin, transferrin soluble receptor (sTfR) and ferritin), cyanocobalamin, folates, renal function (urea, creatinine, estimated creatinine clearance with Cockroft-Gault formula (eGFR)), albumin, prealbumin and CRP. Reference values used by the hospital laboratory are detailed in Table 1. Clinical nutritional parameters studied were: weight (kg), height (m), BMI (kg/m2) and heel-knee distance (cm). Clinical scales used were MNA-SF for malnutrition screening (20) and MMSE for cognitive impairment evaluation (21).
We compared all these parameters in the cohort, according to anemia status. Thresholds used in the literature to define anemia vary greatly (22). Mindell’s work supported the use of the WHO thresholds (130g/L in men and 120 g/L in women) (23) but most definitions are based on data from populations less than 65 years of age (24). The threshold used in our study was hemoglobin <120 g/l for both sexes, based on existing clinical studies about the link between anemia and nutrition including patients with median age of 83 to 85 and above (17, 25) For the MNA-SF, patients with 0-7 points were considered as malnourished, 8-11 points as at risk of malnutrition and 12-14 points as having a normal nutritional status (26). The definitions used for causes of anemia were: ferritin <30 ng/ml or transferrin saturation <15% or sTfR >2 for iron deficit and levels under 150 pmol/l and 5.9 nmol/l for cyanocobalamin and folic acid deficits respectively (5, 27-29).
Inflammatory anemia was defined by CRP>10 or sTfR <1.5 and renal insufficiency as an estimated creatinine clearance under 30ml/min/1.73m2.
Statistics
Patients’ characteristics were compared using chi-square tests or unpaired t tests as appropriate. Due to 5 (1.3%) missing height measurements, the corresponding BMI scores had to be imputed using a regression model adjusted for age, sex and anemia status. For the other parameters, with less than 5% of missing data (mostly for iron work-up), imputation was also performed.
To assess the association between hemoglobin, the dependent variable, and each demographics, nutritional, inflammatory and anemia markers listed in tables 3 and 4, we applied univariate linear regression models. Then, hemoglobin levels were associated simultaneously with all the demographics, nutritional, inflammatory and anemia markers variables using a multiple linear regression.
First, these two sets of regression models were performed in patients without iron and vitamin deficiencies, in those with and without anemia (Table 3). Second, the same two sets of regression models were applied to patients with CRP levels ≤ 10 [mg/l], according to their anemia status (Table 4) to exclude a potential confounding effect of inflammation.
The proportions of the variance of hemoglobin level accounted for by the different regression models were estimated by the coefficient of determination (r-square or R2) and adjusted according to the number of variables included in the models (adjusted R2) and expressed in percent. P-values <0.05 were considered statistically significant. Stata Statistical Software release 12.1 was used to compute all statistics.
Results
Patients’ flow diagram according to Consort is presented in Figure 1. Four hundred and eleven patients were initially included in the study. After exclusion of patients with missing data on iron status, MNA-SF score and hemoglobin, the study population counted 392 patients. Characteristics of this study population will be first presented according to anemia status, with further details for the anemic subgroup. Then, results of a cohort of 261 patients obtained after exclusion of patients with iron, vitamin B12 and folic acid deficits will be presented, also according to anemia status.
Characteristics of the included patients (N=392) after exclusion of missing data are presented in Table 1 (categorical and continuous data) according to anemia status (Hb<120 g/l). Mean age in the cohort (N=392) was 84.8 ±6.3 years, with a majority of women. Overall mean hemoglobin level was 121.3 +/-16.2 g/l. One hundred fifty four patients (39.3%) were anemic. Considering the MNA-SF categories, 57.7% of all patients were malnourished or at risk of malnutrition and 12% of patients had a BMI less than 20 kg/m2. Mean levels of albumin and prealbumin were below normal ranges. Mean eGFR was 54.2 ± 20.6 ml/minute. Mean CRP levels were 28.4 ±40.4mg/l. There were no significant age differences between non-anemic and anemic patients. Part from significantly lower hemoglobin, hematocrit and erythrocyte count, anemic patients had a higher thrombocytes count but no differences in reticulocytes and leucocytes. Anemic patients had mean hemoglobin of 105.9 +/-12.2 g/l. Considering the nutritional status, anemic patients were significantly more frequently malnourished or at risk for malnutrition (p=0.047), had a significantly lower serum albumin and prealbumin level (p <0.001) but with no difference regarding BMI, iron and folic acid levels. Ferritin, soluble transferrin receptor, B12 vitamin and CRP were higher in the anemic group and transferrin was lower.
In the anemic group (N=154), 30 patients (19.5%) had iron deficiency, 32 (20.8%) vitamin B12 deficiency, 7 (4.6%) folic acid deficiency, and 11 (7.14%) suffered from renal failure. In 117 patients (75.9%), chronic inflammation was considered as one of the anemia causes. In only a few patients a unique anemia cause was discovered. More precisely, 6 patients (3.9%) had sole iron deficit, 4 (2.6%) vitamin B12 deficit, 1 (0.7%) had a pure renal failure anemia and 63 (40.9%) had chronic inflammation related anemia. None had a sole folate deficiency. Thirty-eight patients (37.7%) had multiple anemia causes and 22 (14.3%) had no specific cause found. Among the latter, albumin levels were lower than 35 g/l in 20 patients (90.9%).
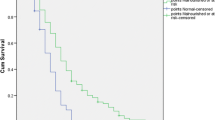
After further excluding patients with iron, vitamin B12 and folic acid deficits in the whole cohort, we studied a subgroup of 261 patients, of which 106 were anemic (41%) and 155 non-anemic (59%)(Table 2). Anemic patients were older, with a significant lower mean level of albumin (p<0.001) and prealbumin (p<0.007). CRP and creatinine levels were higher in anemic patients, whereas eGFR was lower. In this subgroup of anemic patients without iron, vitamin B12 and/or folic acid deficits, an univariate and multiple linear regression model was applied in order to predict hemoglobin according to albumin (Table 3). After adjustment for several variables (age, sex, creatinine, eGFR, ferritin, albumin, CRP, folic acid, vitamin B12 levels) albumin was significantly associated with hemoglobin in patients with anemia, but not in non-anemics. Albumin was the best predictor of anemia (adj. R2), and explained 84.5% of the anemia proportion (Figure 2), as well as 6.4 % (p = 0.005) of hemoglobin’s variance (Figure 3). In order to exclude a potential confounding effect of inflammation, we repeated the analysis according to CRP levels. Thus, after excluding patients with CRP>10mg/l, albumin was an even stronger statistical predictor of anemia and explained 14.7% (p = 0.016) of the variance of hemoglobin in anemic patients (Figure 4) (Table 4).
Discussion
Our study confirmed that the prevalence of anemia is very high in hospitalized elderly populations (almost 40%) as was the prevalence of malnutrition (57.7% of all patients were malnourished or at risk of malnutrition). In the whole cohort, there was a significant association between nutritional status and anemia, considering clinical and biological criteria (MNA-SF and albumin/prealbumin). There was no association with BMI. Nevertheless BMI is a very general and imprecise nutritional index and may be greatly influenced by parameters such as fluid retention. Iron and folic acid levels were not significantly different between the two groups. Ferritin was higher in the anemic group probably because of inflammation related cases and higher vitamin B12 levels due to higher rates of substitution in older patients when anemia was present.
Regarding anemia causes, transferrin saturation and sTfR were added to low ferritin levels to define iron deficiency with more accuracy (30-32). Despite epidemiological definition used for iron deficiency (ferritin level under 12 ng/ml), (5) we chose a higher ferritin level (30 ng/ml) to define iron deficiency, several studies having suggested higher thresholds in the elderly (33, 34). Levels used for cyanocobalamin and folic acid deficits were those described in epidemiological studies (5). We associated sTfR to CRP to define inflammatory anemia, since sTfR helps differential diagnosis between ACD and iron deficiency-dependent anemia (32). The prevalence of iron deficiency, vitamin B12 and folic acid deficiencies was high (almost 20%, 20.8% and 4.6% respectively), in agreement with the literature (5, 35)
After considering other frequent aetiologies (renal failure and inflammation), it was clear that the co-existence of multiple anemia causes was very frequent in our study, with a single cause confirmed in only 14.3%. Amongst patients with no specific anemia cause (simple or multiple), low albumin was extremely frequent (prevalence: 90.9%). This association has never been described so far.
In the subgroup without iron-B12-folic acid deficiency, 41% of patients were anemic. Patients with anemia were older and had lower mean levels of albumin and prealbumin thus supporting our hypothesis of a potential link between anemia and nutrition. CRP and creatinine levels were higher in anemic patients, with lower creatinin clearance levels. A possible explanation would be the inclusion in this group of patients with anemia potentially related to chronic inflammation and chronic renal failure.
A multivariate analysis was carried out in order to better explore the association between nutrition and anemia. So, the model was adjusted for age and sex, but also for creatinine, eGFR, ferritin and CRP in order to exclude any influence of chronic inflammation and renal failure in the hemoglobin/albumin-prealbumin association. After adjustment albumin remained significantly associated with hemoglobin in patients with anemia but not in non-anemics. In this analysis, albumin was the best predictive variable of anemia (adjusted R-square) and explained 6.4 % of its variance. This link between hemoglobin and albumin persisted and became even stronger after excluding patients with CRP over 10 mg/l. In this subgroup albumin was even stronger associated with anemia, explaining 14.7% of the hemoglobin variance. The relation between hemoglobin and prealbumin was not studied since prealbumin values could reflect the hospital nutrition care.
Several studies have previously explored the link between anemia and nutrition and a potential link has already been suggested. Ramel et al. described nutritional status as a predictor of hemoglobin in geriatric inpatients (83 ±7.9 years old) where the prevalence of anemia was 36.7% (25). Hemoglobin was correlated with biological (albumin and prealbumin, lymphocyte count) and clinical (body mass index (BMI), loss of weight, triceps skinfold thickness, mid arm muscle circumference) parameters of nutrition (25). The same association between anemia and albumin, was shown by Mitrache and Silva in hospitalized and community dwelling geriatric populations (17, 19). Nakashima also described hypoalbuminemia as a risk factor of anemia in long term care institutions (36). Nevertheless, our study is the only to consider the relation between nutrition and anemia after taking into account the bias of iron, B12 vitamin and folic acid deficits, as well as inflammation as a cause. Results showed that low albumin, a marker for nutritional status amongst other disorders, is associated to anemia, independently from iron, vitamin deficiencies and inflammatory status. One third of anemia cases in the elderly are of unknown origin. Our findings provide a potential cause for a number of these cases. In fact, hemoglobin is a protein containing four chains of amino acids with an iron ion in the middle, which has bonds with the essential amino acid of histidine. Insufficient dietary intakes of proteins as occurring in undernutrition, could potentially explain anemia in these situations. Alternatively, proteinenergy malnutrition may impair haematopoiesis. Although there is no available study in the elderly, an abnormal erythroid progenitor cell pool has been described in children with protein-energy malnutrition (37) and in animal models, with ineffective erythropoiesis in malnourished mice (independently of any iron or erythropoietin deficiency) and reduction of bone marrow myeloid compartment resulting in anemia and leukopenia (38, 39). Decrease in the percentage of proliferating bone marrow cells has also been shown after short periods of dietary deficiency in rats, with a recovery after nutritional replenishment (40). Protein malnutrition results in pathological changes in mice associated with peripheral leukopenia, bone marrow hypoplasia and alterations in the bone marrow microenvironment leading to hematopoietic failure (41).
The strengths of the study are the prospective design, the large number of patients and the fact that a great part of anemia causes (vitamin and iron deficits, inflammation) were considered in the analysis. Also, the relation between hemoglobin and albumin was studied after exclusion of nutritional deficits. The limitations are the missing data, due to the prescription of laboratory tests by different wards. Also, the number of patients with anemia after excluding various deficits was rather small. We had no indications of possible current substitutions in vitamins/iron or other micronutritional parameters that could have interfered with biological values, and some other causes of anemia in the old has not been excluded, as small intestinal bacterial overgrowth, celiac disease, malabsorption syndromes, late onset hypogonadism, medications with influence on haematological parameters or inflammatory conditions, as not all inflammatory conditions show elevated CRP. In particular, for example, obesity or excess of fat deposition induce inflammation without CRP elevation (33% of the study population has BMI>25 kg/m2).
Conclusion
Anemia and malnutrition are very frequent in elderly hospitalized populations and associated with negative clinical outcomes and with high mortality. Anemia may be caused by one, but more frequently, a multitude of factors. Nevertheless, one third of anemia cases in elderly populations have no identified cause. We found that protein and energy deficit could contribute to the pathogenesis of anemia of unknown origin given the association found between hemoglobin and albumin, independently from iron or B12/folate deficiencies. Further prospective studies should be carried out in order to explore the potential improvement or correction of hemoglobin values by protein and energy supplements. Considering the prognostic value of albumin and hemoglobin on outcomes in elderly, these associations clearly deserve further studies.
Acknowledgements: The authors wish to acknowledge the contribution of Chantal Genet for her valuable help for data entry.
Funding: This work was supported by a grant from the Swiss National Science Foundation (320030-134973/1 to F.R.H., R. R. and U.M.V.)
Conflicts of interest: None.
Sponsor’s Role: None.
Author’s contribution: Study conception and design: E Frangos, UM Vischer, FR Herrmann. Acquisition of subjects and data: E Frangos, CE Graf, UM Vischer, V Lachat. Analysis and interpretation of data: all authors. Preparation of manuscript: E Frangos, UM Vischer, FR Herrmann, N Samaras. Critical revision: all authors. All persons who contributed significantly to the work have been listed.
Ethical Standards: This work was aproved by the local ethical committee.
References
Steensma DP, Tefferi A. Anemia in the elderly: how should we define it, when does it matter, and what can be done? Mayo Clin Proc 2007;82(8):958–66.
Moore KL, Boscardin WJ, Steinman MA, et al. Patterns of chronic co-morbid medical conditions in older residents of U.S. nursing homes: differences between the sexes and across the agespan. J Nutr Health Aging 2014;18(4):429–36.
Bien B, Bien-Barkowska K, Wojskowicz A, et al. Prognostic factors of long-term survival in geriatric inpatients. Should we change the recommendations for the oldest people? J Nutr Health Aging 2015;19(4):481–8.
Frangos E, Samii K, Perrenoud JJ, et al. [Anemia in the elderly: a frequent condition that should not be overlooked]. Rev Med Suisse 2010;6(270):2125–6, 28-9.
Guralnik JM, Eisenstaedt RS, Ferrucci L, et al. Prevalence of anemia in persons 65 years and older in the United States: evidence for a high rate of unexplained anemia. Blood 2004;104(8):2263–8.
Patel KV. Epidemiology of anemia in older adults. Semin Hematol 2008;45(4):210–7.
Smieja MJ, Cook DJ, Hunt DL, et al. Recognizing and investigating iron-deficiency anemia in hospitalized elderly people. CMAJ 1996;155(6):691–6.
Gaskell H, Derry S, Andrew Moore R, et al. Prevalence of anaemia in older persons: systematic review. BMC Geriatr 2008;8:1.
Artz AS, Thirman MJ. Unexplained anemia predominates despite an intensive evaluation in a racially diverse cohort of older adults from a referral anemia clinic. J Gerontol A Biol Sci Med Sci 2011;66(8):925–32.
Ble A, Fink JC, Woodman RC, et al. Renal function, erythropoietin, and anemia of older persons: the InCHIANTI study. Arch Intern Med 2005;165(19):2222–7.
Adamson JW. Renal disease and anemia in the elderly. Semin Hematol 2008;45(4):235–41.
Vischer UM, Frangos E, Graf C, et al. The prognostic significance of malnutrition as assessed by the Mini Nutritional Assessment (MNA) in older hospitalized patients with a heavy disease burden. Clin Nutr, 2012;31(1):113–7.
Wilson MM, Vaswani S, Liu D, et al. Prevalence and causes of undernutrition in medical outpatients. Am J Med 1998;104(1):56–63.
Herrmann FR, Safran C, Levkoff SE, et al. Serum albumin level on admission as a predictor of death, length of stay, and readmission. Arch Intern Med 1992;152(1):125–30.
Trombetti A, Cheseaux J, Herrmann FR, et al. A critical pathway for the management of elderly inpatients with malnutrition: effects on serum insulin-like growth factor-I. Eur J Clin Nutr 2013;67(11):1175–81.
Sullivan DH, Bopp MM, Roberson PK. Protein-energy undernutrition and lifethreatening complications among the hospitalized elderly. J Gen Intern Med 2002;17(12):923–32.
Mitrache C, Passweg JR, Libura J, et al. Anemia: an indicator for malnutrition in the elderly. Ann Hematol 2001;80(5):295–8.
Nakashima AT, de Moraes AC, Auler F, et al. Anemia prevalence and its determinants in Brazilian institutionalized elderly. Nutrition 2012;28(6):640–3.
Silva CL, Lima-Costa MF, Firmo JO, et al. [Hemoglobin level in older adults and the association with nutritional status and use of health services: the Bambui Project]. Cad Saude Publica 2012;28(11):2085–94.
Bauer JM, Kaiser MJ, Anthony P, et al. The Mini Nutritional Assessment—its history, today’s practice, and future perspectives. Nutr Clin Pract 2008;23(4):388–96.
Folstein MF, Folstein SE, McHugh PR. «Mini-mental state». A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 1975;12(3):189–98.
Beghe C, Wilson A, Ershler WB. Prevalence and outcomes of anemia in geriatrics: a systematic review of the literature. Am J Med 2004;116 Suppl 7A:3S–10S.
Mindell J, Moody A, Ali A, et al. Using longitudinal data from the Health Survey for England to resolve discrepancies in thresholds for haemoglobin in older adults. Br J Haematol 2013;160(3):368–76.
Kilpatrick GS, Hardisty RM. The prevalence of anaemia in the community. A survey of a random sample of the population. Br Med J 1961;1(5228):778–82.
Ramel A, Jonsson PV, Bjornsson S, et al. Anemia, nutritional status, and inflammation in hospitalized elderly. Nutrition 2008;24(11-12):1116–22.
Guigoz Y, Vellas B. The Mini Nutritional Assessment (MNA) for grading the nutritional state of elderly patients: presentation of the MNA, history and validation. Nestle Nutr Workshop Ser Clin Perform Programme, 1999;1:3–11; discussion 11-2.
Andres E, Loukili NH, Noel E, et al. Vitamin B12 (cobalamin) deficiency in elderly patients. CMAJ, 2004;171(3):251–9.
Clark SF. Iron deficiency anemia: diagnosis and management. Curr Opin Gastroenterol 2009;25(2):122–8.
Johnson-Wimbley TD, Graham DY. Diagnosis and management of iron deficiency anemia in the 21st century. Therap Adv Gastroenterol 2011;4(3):177–84.
Koulaouzidis A, Saeed AA, Abdallah M, et al. Transferrin receptor level as surrogate peripheral blood marker of iron deficiency states. Scand J Gastroenterol 2009;44(1):126–7.
Koulaouzidis A, Said E, Cottier R, et al. Soluble transferrin receptors and iron deficiency, a step beyond ferritin. A systematic review. J Gastrointestin Liver Dis 2009;18(3):345–52.
Skikne BS, Punnonen K, Caldron PH, et al. Improved differential diagnosis of anemia of chronic disease and iron deficiency anemia: a prospective multicenter evaluation of soluble transferrin receptor and the sTfR/log ferritin index. Am J Hematol 2011;86(11):923–7.
Guyatt GH, Oxman AD, Ali M, et al. Laboratory diagnosis of iron-deficiency anemia: an overview. J Gen Intern Med 1992;7(2):145–53.
Guyatt GH, Patterson C, Ali M, et al. Diagnosis of iron-deficiency anemia in the elderly. Am J Med 1990;88(3):205–9.
Clarke R, Grimley Evans J, Schneede J, et al. Vitamin B12 and folate deficiency in later life. Age Ageing 2004;33(1):34–41.
Nakashima AT, Ferreira de Moraes AC, Auler F, et al. Anemia prevalence and its determinants in Brazilian institutionalized elderly. Nutrition 2011.
Wickramasinghe SN, Akinyanju OO, Grange A. Ultrastructure and cell cycle distribution of bone marrow cells in protein-energy malnutrition. Clin Lab Haematol 1988;10(2):135–47.
Borelli P, Blatt S, Pereira J, et al. Reduction of erythroid progenitors in proteinenergy malnutrition. Br J Nutr 2007;97(2):307–14.
Nakajima K, Crisma AR, Silva GB, et al. Malnutrition suppresses cell cycle progression of hematopoietic progenitor cells in mice via cyclin D1 down-regulation. Nutrition 2014;30(1):82–9.
Dunki Jacobs PB, Ruevekamp M, Hart GA, et al. Dietary influences on cell proliferation in bone marrow. Eur J Cancer Clin Oncol 1989;25(6):953–7.
Cunha MC, Lima Fda S, Vinolo MA, et al. Protein malnutrition induces bone marrow mesenchymal stem cells commitment to adipogenic differentiation leading to hematopoietic failure. PLoS One 2013;8(3):e58872.
Author information
Authors and Affiliations
Corresponding authors
Additional information
The author passed away on March 19, 2012.
Rights and permissions
About this article
Cite this article
Frangos, E., Trombetti, A., Graf, C.E. et al. Malnutrition in very old hospitalized patients: A new etiologic factor of anemia?. J Nutr Health Aging 20, 705–713 (2016). https://doi.org/10.1007/s12603-015-0641-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12603-015-0641-6