Abstract
Sweet pepper crops suffer major losses from anthracnose caused by Colletotrichum spp. In the present work we describe the combined effects of cultivation in the dry and rainy seasons, of nitrogen fertilization doses (0-450 Kg/ha), the use of organic (grass) and plastic soil mulches, as well as the application of a synthetic protectant fungicide (chlorothalonil) for disease control. Overall results indicate that choice of planting season is the most decisive factor for sweet pepper anthracnose management. Use of fungicide is a less decisive but still important factor, followed by choice of soil mulch substrate and nitrogen fertilization. The combined effects of all cultural practices were demonstrated: for instance, in the rainy season, fungicide alone was not as efficient as the combined use of fungicide and grass mulch. Conversely, in the dry season fungicide applications may be discarded, when organic soil mulch is adopted. The effects of the type of soil cover on the splash dispersal of Colletotrichum conidia are discussed. Management practices described here are of practical value for either organic or conventional growers.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Anthracnose is an important Capsicum disease, affecting green or ripe fruits of hot and sweet peppers, both in the field and at the postharvest stage (Ali et al. 2016; Ivey et al. 2004; Silva et al. 2019). Worldwide, and mostly in chilli peppers, several Colletotrichum species are known as causal agents, mostly in the C. truncatum (Schwein.) Andrus & W.D. Moore, C. acutatum J.H. Simmonds and C. gloeosporioides (Penz.) Penz. & Sacc. species complexes (Mongkolporn et al. 2010; Silva et al. 2019). In Brazil, sweet pepper (C. annuum L.) anthracnose has been related only to C. gloeosporioides and C. acutatum (Tozze-Júnior et al. 2006), but recently C. brevisporum Noireung, Phouliv., L. Cai & K.D. Hyde was found in C. chinensis Jacq. (hot pepper) in the Brazilian Amazon (Almeida et al. 2017).
Because the disease directly affects the produce, it frequently has a major economic impact. In environments of high relative humidity and frequent rains, such as in the Brazilian summer cropping season, losses up to 100% may occur in individual fields.
Colletotrichum produces splash-dispersed conidia, which are spread by the impact of water drops onto the fungus acervulae and go through several spore transport stages in secondary droplets, until conidia are deposited onto new infection sites (Madden 1992; Madden et al. 1993; Yang et al. 1990). Thus, anthracnose is a polycyclic disease (Vanderplank 1963) of potentially high epidemic rates. Moreover, due to its dispersal mechanism, it may be either constrained or strongly favoured by the conditions prevailing during any particular planting season. In other words, the rate of the epidemics may be inhibited by cultural practices that hamper spore splash dispersal.
Chemical control of the disease is not wholly efficient, cannot be used by organic farmers and is increasingly scrutinized for environmental and market impacts, even among conventional farmers. Therefore, alternative, integrated methods to help manage the disease are needed. One alternative is choice of a planting date to avoid conducive environmental conditions. Another factor that affects disease progress is the nature of the soil cover. Coelho et al. (2008) found that organic mulches, as opposed to plastic, had a strong positive impact on the control of strawberry (Fragaria X ananassa Duch.) anthracnose, caused by C. acutatum. Others (Ferraz et al. 1999; Napoleão et al. 2005) showed that apothecia formation by Sclerotinia sclerotiorum (Lib.) de Bary, and bean (Phaseolus vulgaris L.) white mould development were reduced by the use of grass mulch. Freitas et al. (2002) showed that no-till practices (keeping grass mulch on the soil) greatly reduced soybean (Glycine max (L.) Merril) stem canker, caused by Diaporthe phaseolorum (Cke. & Ell.) Sacc. f.sp. meridionalis. Recently, Costa-Coelho et al. (2016) showed that no-till reduced bean web blight (Thanatephorus cucumeris (A.B. Frank) Donk), and Likins et al. (2019) reported on the effect of cypress or pine bark mulching to prevent soil inoculum of Calonectria pseudonaviculata (Crous, J.Z. Groenew. & C.F. Hill) L. Lombard, M.J. Wingf. & Crous from reaching boxwood (Buxus sempervirens L.) foliage. Additionally, Nitrogen, which is a limiting macronutrient for sweet pepper fruit yields, is known to affect the severity of many plant diseases (Datnoff et al. 2007).
The effects of joint methods for control of Capsicum anthracnose have not been studied in commercial field conditions. Here, we present results of field studies on the combined effect of soil mulch materials, N fertilization, and a protectant fungicide, for management of sweet pepper anthracnose in two planting seasons with contrasting environmental settings.
Materials and methods
Field trials
Field trials were installed at the Biological Experimental Station of the Universidade de Brasília, Brazil (15°44’13"S and 47°52’56"W. 1010 m a.s.l.) in a dystrophic red latosol whose chemical analysis is presented in Table 1, following standard cropping practices. The first series of trials was conducted in the dry planting season (March to July). A second series of trials was carried out during the rainy season (September to December). Fields had no history of Capsicum cultivation. Crops were cultivated on 1.0 m-wide raised beds with two plant rows, spaced 0.5 m apart. Fertilization followed standard practices: 4.0 ton.ha− 1 of NPK (4-14-8) and 8.6 ton.ha− 1 of poultry manure. A supplement of 15 g/plant of NPK 20-05-20 was applied as side dressing in the soil mulch and fungicide trials, 30 and 60 days after planting (DAT). Forty-five-day-old sweet pepper plantlets (cv. Maximos F1, Clause Seed®) were grown in Styrofoam trays at the Experimental Station greenhouses. Overhead supplemental irrigation was provided in the first 10 DAT in the rainy season and twice a day in the dry season for the duration of the crop.
Soil mulch and fungicide experiments
Experiments followed a split-plot design, with subplots distributed in a 2 × 2 factorial, with three replicates. The main factors were chemical treatments and sub-factors corresponded to soil mulch materials. The two main factors were weekly applications, starting at fructification, of the synthetic protectant fungicide chlorothalonil (75% of active principle as wettable powder, Iharabrás Chem®) at 20 g of commercial product/10L, or control plots, where dipotassium phosphate (K2HPO4, Sigma®, 50 g/10L) was applied following the same schedule. Phosphate salts have been reported to have an effect on eliciting plant defences in other pathosystems (e.g.. Pagani et al. 2014), including cucumber anthracnose (Gottstein and Kuc 1989), and many Brazilian vegetable growers routinely apply phosphate salts as a general preventative treatment for overall crop health. The two sub-factors were plastic mulch and organic (grass) mulch, the latter represented by cut and dried leaves of Andropogon sp. Experimental plots were 5.0 m long with 20 plants in each.
Nitrogen fertilization experiments
Two nitrogen fertilization field assays were carried out in the same dry and rainy seasons described above. Experiments followed a randomized complete block with three replicates, with experimental units made up of 20 plants, planted in beds and cultivated as above. The effect of four N levels was studied: 0, 12, 35 and 102 g/plant in the form of CO(NH2)2 (urea, 45–46% N), divided into three applications at 30, 60 and 90 DAT, which corresponded to 0, 50, 150 and 450 Kg.ha− 1 respectively. Ten g / plant of potassium chloride (KCl) was applied to all treatments as dressing at 30 DAT.
Inoculum and inoculation procedures
Inoculum was prepared with a mixture of equal parts of three monosporic isolates of C. gloeosporioides s.l. (Embrapa codes CNPH 1, CNPH 6 and CNPH 56), the aggressiveness of which had previously been established in preliminary trials. Individual cultures were grown in PDA at 25ºC (± 1ºC) and a photoperiod of 12 h, for 30 days. Conidia were removed from Petri dishes, suspended in water and filtered in gauze, and the suspension was adjusted to 3 × 105 conidia/ml by use of a Neubauer Chamber. This suspension was used to inoculate green indene fruits of sweet pepper by injecting 100 ml into the interior of the fruits. Inoculated fruits were then kept in humid chambers for eight days, when external lesions were abundant. These fruits were in turn used as field inoculum, by distributing five halves of the inoculated fruit onto the soil of each experimental plot. Only the experiments conducted in the dry season were inoculated, at 85 DAT. The summer experiment, which was conducted in the same field, was not inoculated but relied on the residual inoculum of the previous dry season.
Environmental and variable data collection and statistical analyses
Meteorological data were obtained from the Brazilian National Institute of Meteorology (INMET). Incidence and severity of disease data were collected at 90, 93, 96, 99 and 102 DAT in the dry season and at 60, 63, 66, 69 and 72 DAT in the rainy season. Disease incidence was estimated by the percentage of fruits bearing anthracnose symptoms. Disease severity was assessed by a standard area diagram with nine classes proposed by Pedroso et al. (2011), with the following grades: 0%, 1%, 2%, 5%, 10%, 20%, 40%, 60% and 80%. The scale was modified to adapt its use to the Leaf Doctor software (Pethybridge and Nelson 2015), and allow the calculation of anthracnose disease severity index, DSI = ∑((n + nc)100)/tf, where: n = number of diseased fruits in each disease class; nc = value of class; tf = total number of fruits. Box Plots were generated for data interpretation (McGill et al. 1978). Values outside the upper and lower limits of the Box Plot were considered outliers. The areas under disease progress curves (AUDPCs) for incidence and severity were calculated according to Shaner and Finney (1977). Disease progress curves’ best fits were based on the lowest coefficient of determination and other parameters, as in Campbell and Madden (1990). Statistical analyses were performed with the software Assistat 7.7 (Silva and Azevedo 2016) and Genes (Cruz 2006). Treatment means were compared by Tukey´s when data were normally distributed, and the Friedman test was applied for non-normally-distributed data (Friedman 1937).
Results
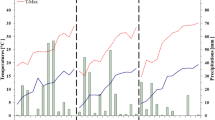
Whereas other environmental parameters were similar, precipitation differed sharply between planting seasons. Despite the fact that some rainfall was initially recorded in the first 30 days, the dry season was mostly arid afterwards. On the other hand, rains were frequent throughout in the wet season, especially in the last 60 days, when fruits form and mature (Fig. 1).
Effect of planting season, soil mulch material and synthetic fungicide
The disease severity in relation to the effects of soil mulch material or fungicide use points to three general trends (Fig. 2): (i) Overall, anthracnose intensities were much lower in the dry season; (ii) much lower disease severity was recorded in the synthetic fungicide treatments; and (iii) somewhat less anthracnose developed in the organic (grass) soil cover, as compared to the plastic mulch plots. Combined effects of each cultural factor were also noticeable. The Mann-Whitney non-parametric test (MacFarland et al. 2016) indicated lower severity levels in the dry season (U = 5.59; p-value 5% = 1.96). Application of the synthetic fungicide reduced anthracnose both in the dry (F = 48.5048, p-value = 30.1435, p < 0.01) and the rainy seasons (F = 49.0647, p-value = 36.1908, p < 0.01). As for the effect of the soil mulches, only in one instance (rainy season control plots) were disease levels significantly lower with the adoption of organic soil cover (p < 0.01).
Box Plots of anthracnose severity indexes recorded in the dry and rainy growing seasons in plots treated or not treated with fungicide, and in soil under organic (grass) or plastic mulch. Median followed by the same capital letter did not differ between soil cover materials (Mann-Whitney test, p < 0.05). Median values followed by the same lower-case letter did not differ between fungicide treatments (Friedman test, p < 0.01). Org. Mulch: Organic mulch; Plast. Mulch: Plastic mulch; Synthetic Fungicide: Chlorothalonil
In the dry season (Fig. 2, upper graph), anthracnose levels were lower in plots treated with chlorothalonil, as compared to control plots (where only K2HPO4 was applied), irrespective of the nature of soil cover. In fact, median disease indexes were four to five times lower with chlorothalonil applications (DSI = 55–65) than in control plots (DSI = 250–300). In the dry season, the effect of the organic soil cover was less pronounced than in the wet season, but still discernible: in the absence of the synthetic fungicide grass mulch plots had a median DSI of c. 250 vs. c. DSI of 300 in the plastic mulch plots (Fig. 2).
In the rainy season (Fig. 2, lower graph), the same trends were repeated, although with much higher disease intensities. Again, the synthetic fungicide gave the best anthracnose control (DSIs = 200–250), still very high when compared to the disease levels in the dry season. Control treatments in the wet season had DSIs of 420 and 580, for the grass mulch plots and plastic mulch plots, respectively. The benefit of the organic soil cover is discernible in the absence of chlorothalonil, but not when the synthetic fungicide was applied.
The cumulative temporal effects of the environment (dry vs. rainy season plantings), of the fungicide and of the soil mulches on the epidemiology of sweet pepper anthracnose are presented in Fig. 3. In the dry season (upper graph), following inoculation at 85 DAT, disease progressed rapidly when no fungicide was applied, but was kept uniformly low when chlorothalonil was used. In addition, progress with or without fungicide is consistently slower under organic mulch, in contrast with the respective treatment under plastic mulch. On the other hand, extremely conducive conditions in the rainy season (Fig. 3, lower graph) lead to much more severe epidemics, especially in the absence of the fungicide (compare y-axis scales of upper and lower graphs). Due to early disease onset, and frequent rains, the crop cycle was shorter (see x-axis scale). In all, the high disease levels during the wet season (even with no additional inoculum added) led to an inadequate disease control level. However, the hindering effect of grass mulch on the disease progress curves is observable in all treatment combinations (Fig. 3).
Anthracnose temporal progress curves in experimental sweet pepper field plots grown in the dry and rainy growing seasons, in plots treated or not treated with the fungicide chlorothalonil and in soil under organic (grass) or plastic mulch. Note the differences in the temporal and disease scales on the x and y-axis between seasons.
Effects of treatment combinations and planting season were significant for AUDPC, but no interaction between treatment combinations and planting season was found (Table 2). Lack of interactions between treatment combinations and planting season indicates that treatment effects were consistent, irrespective of disease pressure levels. AUDPC is a disease variable that integrates the cumulative effects over time and shows that the choice of planting season is the most determining factor in management of sweet pepper anthracnose, followed by the mulch-fungicide combination.
The AUDPC values were significant (p < 0.01), ranging from 3062 to 3466 to 5191–6276 in the control treatments, and from 650 to 827 to 2724–2976, in the fungicide treatments. Grass mulch was associated with a consistent decrease in disease incidence (Table 3).
Nitrogen fertilization effects
Heavy nitrogen fertilization (450 Kg/ha), both in the dry and wet seasons, increased anthracnose severity. Conversely, up to 150 Kg/ha did not cause significant differences in severity, as measured by AUDPC (Table 4). The effect of excessive N level was compounded by the more conducive environment in the rainy season (Table 4).
Discussion
Abundance of rains was the most significant factor for difference between cropping seasons, and it accounts for the vast difference in anthracnose intensities detected. The climatic data for each epidemic period (Fig. 1) strongly support the hypothesis that environmental conditions, especially rains, are the key determinants of the rate of disease progress. Indeed, the risk of loss incurred by planting in the rainy season is very high, as reflected by all disease parameters measured. The synthetic protectant fungicide was the second most important factor for controlling sweet pepper anthracnose. Chlorothalonil-sprayed plots consistently presented lower disease severities, but fungicide application is likely not sufficient as the sole method of disease control in the rainy season. Ivey et al. (2004) also found that this active principle was partially effective for chemical management of bell pepper anthracnose, caused by C. acutatum in the US. Other protectant fungicides, such as the ones based on copper (La Torre et al. 2018), which are often used by organic growers, have a similar protectant effect. They would probably yield analogous results, and their effect should be examined. Conversely, the choice of a systemic fungicide, which moves into the plant tissue, would likely have enhanced disease control.
Yang et al. (1990), Madden et al. (1993) and Ntahimpera et al. (1997) illustrated the effect of rains on the spread of Colletotrichum conidia and infection of strawberry fruit. Ntahimpera et al. (1998) and Coelho et al. (2008) showed that organic soil mulches hindered spore dispersal and led to lower anthracnose levels on strawberry. These authors discussed the role of splash dispersal, which is a function of surface topography as provided by the substrate of each mulch. We contend that a similar restraining process is in effect for the dispersal of sweet pepper anthracnose conidia, even though the sweet pepper fruits are placed considerably higher above the soil level than the strawberry fruits. While we did not check for presence of Colletotrichum propagules in the plant residues in the soil, these probably provided the inoculum sources for the epidemic, as indicated by the persistence of inoculum between growing seasons. The importance of the nature of the surface onto which rains falls has also been demonstrated in other pathosystems (Costa-Coelho et al. 2016; Freitas et al. 2002; Likins et al. 2019).
Excess nitrogen fertilization has been associated with an increase in severity of many plant diseases, especially for pathogens that attack succulent tissue, such as fruit organs (Agrios 2005). In hot pepper, greater Cercospora leaf spot and anthracnose severities have been associated with high doses of N (up to 500 Kg/ha) cultivated in tropical lowland conditions (Vos and Frinking 1997). Our results point to the same conclusion for sweet pepper anthracnose. Therefore, high levels of nitrogen fertilization, which are commonly found in vegetable growers’ fields in Brazil, must be avoided, and especially so in the rainy season, in order to reduce the risk of anthracnose epidemics.
Several previous works have indicated that the combination of different management methods is needed for the successful control of important plant diseases (e.g.. Pagani et al. 2014). The synergistic effect of integrating different management strategies, including use of pine or grass soil mulch for the control of strawberry anthracnose, has also been presented (Coelho et al. 2008). This paper demonstrates that for the successful management of sweet pepper anthracnose, choice of planting season is the most determining factor. Use of synthetic fungicide is a less impacting but still important factor (chiefly in the rainy season), followed by choice of an organic mulch material and moderate Nitrogen fertilization. Furthermore, the additive effects of these practices are evident: in the rainy season, use of fungicide alone is not as efficient as the combined use of fungicide and grass mulch. This practice is applicable to all growers, conventional or otherwise. On the other hand, in the dry season, fungicide applications may even be abandoned when growing sweet pepper over grass mulch, which is a particularly valuable piece of information for organic growers.
References
Agrios, G. N. 2005. Plant Pathology. 5th Edition. Elsevier Academic Press.
Ali, A., Bordoh, P. A., Singh, A., Siddiqui, Y., & Droby, S. (2016). Post-harvest development of anthracnose in pepper (Capsicum spp.): Etiology and management strategies. Crop Protection, 90, 132–141.
Almeida, L. B., Matos, K. S., Assis, L. A. G., Hanada, R. E., & Silva, G. F. (2017). First report of anthracnose of Capsicum chinense in Brazil caused by Colletotrichum brevisporum. Plant Disease, 101, 1035.
Campbell, C. L., & Madden, L. V. (1990). Introduction to Plant Disease Epidemiology. New York: Wiley.
Coelho, M. V., Palma, F. R., & Café-Filho, A. C. (2008). Management of strawberry anthracnose by choice of irrigation system, mulching material and host resistance. International Journal of Pest Management, 54, 347–354.
Costa-Coelho, G. R., Toledo-Souza, E. D., Café-Filho, A. C., & Lobo Jr, M. (2016). Dynamics of common bean web blight epidemics and grain yields in different tillage systems. Tropical Plant Pathology, 41, 306–311.
Cruz, C. D. (2006). Programa Genes - Estatística Experimental e Matrizes. Viçosa: Editora UFV.
Datnoff, L. E., Elmer, W. H., & Huber, D. M. (2007). Mineral Nutrition and Plant Disease. St. Paul: APS Press.
Ferraz, L. C. L., Café-Filho, A. C., Nasser, L. C. B., & Azevedo, J. (1999). Effects of soil moisture, organic matter and grass mulching on the carpogenic germination of sclerotia and infection of bean by Sclerotinia sclerotiorum. Plant Pathology, 48, 77–82.
Freitas, M. A., Café-Filho, A. C., & Nasser, L. C. B. (2002). Cultural practices and genetic resistance as factors affecting soybean stem canker and plant yield in the cerrado. Fitopatologia Brasileira, 27, 5–11.
Friedman, M. (1937). The use of ranks to avoid the assumption of normality implicit in the analysis of variance. Journal of the American Statistical Association, 32, 675–701.
Gottstein, H. D., & Kuc, J. (1989). Induction of systemic resistance to anthracnose in cucumber by phosphates. Phytopathology, 79, 176–179.
Ivey, M. L. L., Nava-Diaz, C., & Miller, S. A. (2004). Identification and management of Colletotrichum acutatum on immature bell peppers. Plant Disease, 88, 1198–1204.
La Torre, A., Iovino, V., & Caradonia, F. (2018). Copper in plant protection: current situation and prospects. Phytopathologia Mediterranea, 57, 201–236.
Likins, T. M., Kong, P., Avenot, H. F., Marine, S. C., Baudoin, A., & Hong, C. X. (2019). Preventing soil inoculum of Calonectria pseudonaviculata from splashing onto healthy boxwood foliage by mulching. Plant Disease, 103, 357–363.
MacFarland, T. W., Yates, J. M., & Mann–Whitney, U. 2016. Introduction to Nonparametric Statistics for the Biological Sciences Using R. Springer International Publishing.
Madden, L. (1992). Rainfall and the dispersal of fungal spores. Advances in Plant Pathology, 8, 39–79.
Madden, L., Wilson, L., & Ellis, M. (1993). Field spread of anthracnose fruit rot of strawberry in relation to ground cover and ambient weather conditions. Plant Disease, 77, 861–866.
McGill, R., Tukey, J. W., & Larsen, W. A. (1978). Variations of box plots. The American Statistician, 32, 12–16.
Mongkolporn, O., Montri, P., Supakaew, T., & Taylor, P. W. J. (2010). Differential reactions on mature green and ripe chilli pepper fruit infected by three Colletotrichum species. Plant Disease, 94, 306–310.
Napoleão, R., Café-Filho, A. C., Nasser, L. C. B., Lopes, C. A., & Silva, H. R. (2005). Intensidade do mofo-branco do feijoeiro em plantio convencional e direto sob diferentes lâminas d’água. Fitopatologia Brasileira, 30, 374–379.
Ntahimpera, N., Madden, L., & Wilson, L. (1997). Effect of rain distribution alteration on splash dispersal of Colletotrichum acutatum. Phytopathology, 87, 649–655.
Ntahimpera, N., Ellis, M., Wilson, L., & Madden, L. (1998). Effects of a cover crop on splash dispersal of Colletotrichum acutatum conidia. Phytopathology, 88, 536–543.
Pagani, A. P., Dianese, A. C., & Café-Filho, A. C. (2014). Management of wheat blast with synthetic fungicides, partial resistance and silicate and phosphite minerals. Phytoparasitica, 42, 609–617.
Pedroso, C., Lage, D. A. C., Henz, G. P., & Café-Filho, A. C. (2011). Development and validation of a diagrammatic scale for estimation of anthracnose on sweet pepper fruits for epidemiological studies. Journal of Plant Patholology, 93, 219–225.
Pethybridge, S. J., & Nelson, S. C. (2015). Leaf doctor: a new portable application for quantifying plant disease severity. Plant Disease, 99, 1310–1316.
Shaner, G., & Finney, R. (1977). The effect of nitrogen fertilization on the expression of slow-mildewing resistance in Knox wheat. Phytopathology, 67, 1051–1056.
Silva, F. A. S., & Azevedo, C. A. V. (2016). The Assistat software version 7.7 and its use in the analysis of experimental data. African Journal of Agricultural Research, 11, 3733–3740.
Silva, D. D., Groenewald, J. Z., Crous, P. W., Ades, P. K., Nasruddin, A., Mongkolporn, O., & Taylor, P. W. J. (2019). Identification, prevalence and pathogenicity of Colletotrichum species causing anthracnose of Capsicum annuum in Asia. IMA Fungus, 10, 8. https://doi.org/10.1186/s43008-019-0001-y.
Tozze-Júnior, H. J., Mello, M. B. A., & Massola-Júnior, N. S. (2006). Caracterização morfológica e fisiológica de isolados de Colletotrichum sp. causadores de antracnose em solanáceas. Summa Phytopathologica, 32, 71–79.
Vanderplank, J. E. (1963). Plant Diseases: Epidemics and Control. New York: Academic Press.
Vos, J. G. M., & Frinking, H. D. (1997). Nitrogen fertilization as a component of integrated crop management of hot pepper (Capsicum sp.) under tropical lowland conditions. International Journal of Pest Management, 43, 1–10.
Yang, X., Madden, L., Wilson, L., & Ellis, M. (1990). Effects of surface topography and rain intensity on splash dispersal of Colletotrichum acutatum. Phytopathology, 80, 1115–1120.
Acknowledgements
C Pedroso and LM Anjos were supported by graduate student scholarships from the Brazilian National Research Council (CNPq). AC Café-Filho is a recipient of a CNPq Research Fellowship (grant no. 301.758/2017-4). JJ Dias-Neto received a postdoctoral fellowship from the Brazilian Ministry of Education (CAPES – PNPD grant no. 20133002–53001010021P0).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pedroso, C., Dias-Neto, J.J., Rossato, M. et al. Managing sweet pepper anthracnose by choice of planting season, organic soil mulch, fungicide and moderate nitrogen fertilization. Phytoparasitica 48, 525–533 (2020). https://doi.org/10.1007/s12600-020-00819-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12600-020-00819-2