Abstract
The polychaete assemblages at Dakhla Bay (Atlantic coast of South Morocco) were studied during spring 2013 and winter 2014 to analyze the spatial-temporal variability in diversity and community structure, as well as their relationships with the main environmental variables. Forty-two stations were sampled all over the bay, yielding a total of 22 species (21 in spring, 22 in winter) belonging to 16 families. The three most abundant species were Maldane sarsi (25.1%) Eunice vittata (11.1%), and Nainereis laevigata (10%) in spring and Ophelia rathkei (43%), M. sarsi (21.5%), and N. laevigata (5.9%) in winter. The main drivers of the polychaete community structure at Dakhla Bay were the hydrographic characteristics of the bay as well as the type sediment (in spring and winter) and food availability, organic matter, and chl-a content (in spring). Accordingly, the cluster analysis identified three assemblages in spring and winter, named according to the dominant species. The M. sarsi assemblage occurred in inner bay fine sediments and was replaced by the E. vittata (spring) and N. laevigata (winter) assemblages in mid-bay medium grain-sized sediments, and by the C. tentaculata one in sandy sediments closer to the outer inlet. This represents a shift from a typical brackish, lacunar assemblage to two different, temporal aspects of a marine assemblage, with a transitional one in between. Our study confirms the singularity of the macrofaunal pool at Dakhla Bay and provides a fundamental baseline for future monitoring of an endangered southern Moroccan wetland that will contribute to facilitate its management and protection.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Benthic organisms play a key role in marine ecosystem functioning. Among other relevant processes, they contribute to nutrient cycle regulation, pollutants’ burying, and dispersal and secondary production (Snelgrove 1998), particularly in semi-enclosed areas such as estuaries (e.g., Kristensen et al. 2014; Magallaes et al. 2011), bays, and gulfs (e.g., Belan 2003; Junoy et al. 2013; Girbert et al. 2015; Sardá et al. 2000). The knowledge on the spatial-temporal structure of the benthic assemblages is a key issue allowing environmental health assessments (Borja et al. 2000; Aubry and Elliott 2006; Arbi et al. 2017).
Among benthic marine macroinvertebrates, polychaetes, crustaceans, molluscs, and echinoderms are usually the dominant and most diverse taxa (Snelgrove 1998; Byers and Grabowski 2014). Particularly, the polychaetes are a basal group in any study on marine benthic communities. They dominate the macrofaunal assemblages in terms of abundance, biomass, and diversity at practically all depths and benthic habitats (Jumars et al. 2015). Since classical studies on responses of benthic organisms to environmental gradients (e.g., Perason and Rosenberg 1978), the polychaetes are well known by their adaptability to a wide variety of environmental conditions, thus playing a key role in ecosystem functioning. Furthermore, they have been used successfully as surrogates to estimate the diversity, spatial-temporal dynamics, and functional roles of benthic communities (Olsgard et al. 2003; Papageorgiou et al. 2006; Dixon-Bridges et al. 2014). In fact, analyzing the polychaete assemblage structure proved to be an efficient tool in environmental health assessment. Thus, it is commonly used as a biological criterion for water quality in biomonitoring studies (Pocklington and Wells 1992; Olsgard et al. 2003; Samuelson 2001; Giangrande et al. 2005) and as a relevant component in ecological indexes (see Dauvin et al. 2016, and references herein).
Dakhla Bay, located in the Atlantic coasts of southern Morocco, is one of the most productive natural systems of the country, whose remarkable ecological, biological, and socio-economic services have been recently recognized (Zidane et al. 2008). The bay harbors a significant amount of exploitable native populations of bivalves, such as the grooved carpet shell (Ruditapes decussatus (Linnaeus 1758)), the edible cockle (Cerastoderma edule (Linnaeus 1758)), the grooved razor shell (Solen marginatus Pulteney, 1799), and the mussel (Perna perna (Linnaeus 1758)) (Zidane et al. 2008). The bay is also well known for its ornithological importance, constituting a stopover migratory and a winter refuge appreciated for numerous species of birds (Qninba et al. 2003). Thus, it has been classified as a site of biological and ecological interest (SIBE), through the Protected Areas Master Plan of Morocco, and as a RAMSAR site since 2005. Overall, Morocco is a very rich country in terms of wetlands, including bays, estuaries, lagoons, lakes, and rivers, among others. However, many of them need to be well managed and better exploited, thus requiring baseline descriptive studies to assess faunal patterns and trends. Dakhla Bay is not an exception. In fact, the studies conducted to date in the bay mainly focused on its oceanographic features and aquaculture capacities (Guelorget et al. 1996; Dafir et al. 1997; Saad et al. 2013; Zidane et al. 2008, 2017), while there is no information on the characteristics of its benthic macroinvertebrate assemblages.
In this context, our study represents the first baseline study focusing on the patterns and trends of the benthic organisms from Dakhla Bay, using the polychaete assemblages as surrogates. We analyze the spatial-temporal variability of both diversity and community structure descriptors, as well as their relationships with the main environmental variables. As a result, we described for the first time the polychaete assemblages inhabiting Dakhla Bay. We also intend to reveal its singularity, which acquires a particular relevancy taking into account the worsening anthropogenic-driven conditions in the bay.
Materials and methods
Study area
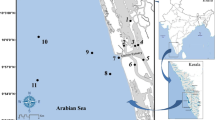
Dakhla Bay is located at 23° 35′ N and 16° W, along the Atlantic coast of south Morocco (Fig. 1). This region shows a typically desert climate and receives an oceanic influence, particularly as a result of the interaction between the cold Canary current and the subtropical ridge currents (Orbi et al. 1999). Dakhla Bay is 37 km long and ranges from 10 to 12 km wide. It has a NE-SW orientation and is separated from the Atlantic Ocean by the peninsula of Oued Ed-Dahab. The bay is divided into two different areas according to its bathymetry and distance to the opening: (1) the inner, southern zone, which has a series of channels oriented in the same direction as the bay and (2) the outer northern zone, which has a more regular morphology. Bathymetry increases from both shores toward the middle of the bay, where it reaches a maximum depth of 20 m (Orbi et al. 1995).
Sampling and data analysis
Forty-two stations (Fig. 1) were sampled both in spring (May 2013) and in winter (February 2014). All samples (two replicates) were collected using a Van Veen grab (0.0625 m2 in surface area). Despite samples were collected at approximately the same location in each station, the surface area at each selected site was large enough, compared to sample surface, to prevent interferences between the successive seasonal sampling. The samples were sieved in situ through a 1-mm pore size mesh. The material retained on the mesh was transferred to containers and fixed in a 10% formalin/seawater solution.
At each station, water salinity (‰) and temperature (°C) and distance from the bay opening (i.e., distance) were recorded. An additional sediment sample was collected to analyze grain size, organic matter, and chlorophyll-a contents. Grain size was measured with a laser granulometer (Malvern, Mastersizer) at the LETG (Littoral, Environnement, Geomatique, Teledetection) (UMR 6554, University of Nantes) and expressed as mean grain size, in micrometers. Organic matter contents (OM, %) were obtained as weight losses of dried samples (24 h, 60 °C) after ignition (4 h, 450 °C). The chlorophyll-a content (chl-a, mg/m2) was determined according to the Lorenzen method (Holm-Hansen et al. 1965).
All macroinvertebrates were sorted under a binocular microscope. The polychaetes were identified at the lowest taxonomic level possible and counted. Selected specimens of the most relevant species have been deposited in the collections of the Centre d’Estudis Avançats de Blanes (CEAB) (Table 1). To analyze the assemblage structure, the following indices were calculated: (1) species richness (as number of species per sample); (2) density (as ind./m2); (3) Shannon diversity (log2 basis) (Shannon 1948); and evenness (Pielou 1966).
A hierarchical ascending classification analysis (HAC), based on Euclidean distance, the Wards method, and log10 (x + 1) transformed data to limit the influence of the most dominant taxa (Vakharia and Wemmerlöv 1995; Cao et al. 1997), was used to analyze the spatial structure of the populations, both in spring and in winter. The most representative species of each community were identified by the IndVal index (Dufrêne and Legendre 1997). Wilcoxon tests were used to determine the significance (P < 0.05) of the difference between seasons. Canonical correspondence analyses (CCA) based on log10 (x + 1) transformed data were performed to analyze the relationships between environmental variables and the polychaete assemblages, both in spring and in winter. One-way permutational multivariate analysis of variance (PERMANOVA) based on Euclidean distances and 9999 permutations (Anderson 2001) was used to test for differences between the assemblages identified in the hierarchical ascending classification analysis in both seasons. All analyses were carried out using the PAST software package (Hammer et al. 2001), Statistical Version 2.17 for Windows software.
Results
Environmental variables
Salinity in Dakhla Bay ranged from 36.2 to 39.9‰ and progressively increased downstream-upstream both in winter and spring (Fig. 2a). The temperature ranged from 15.8 to 19.5 °C in spring and from 16.3 to 19.5 °C in winter, following also the same increasing gradient as in the case of salinity (Fig. 2b). Chl-a showed a very heterogeneous distribution and ranged from 0.4 to 24.9 mg m−2 in spring and from 0.2 to 34.6 mg m−2 in winter (Fig. 2c). Organic matter contents ranged from 0.3 to 9.6% in spring and from 0.2 to 7.5% in winter, showing maxima in the center and in the north of the bay at both seasons (Fig. 2d). The granulometry varied from sandy (upstream) to sandy-silt or silt (downstream) (Fig. 2e). No significant seasonal variations were detected for the analyzed environmental variables.
Polychaete assemblage descriptors
Four thousand three hundred twenty-four polychaete individuals belonging to 22 species and 16 genera were collected during this study (Table 1). Among them, 2144 individuals from 21 species occurred in spring and 2160 from 22 species in winter.
The most abundant species in spring were Maldane sarsi (25.1%), Eunice vittata (11.1%), Naineris laevigata (10%), Platynereis dumerilii (7.4%), and Arabella iricolor (6.3%), while in winter these were Ophelia rathkei (43%), M. sarsi (21 .5%), N. laevigata (5.9%), and Cirriformia tentaculata (5.2%).
Polychaete densities did not differ significantly between winter and spring, although they tended to be higher in the former (0 to 856 ind./m2) than in the latter (0 to 176 ind./m2). The number of species and the Shannon diversity also did not differ significantly between winter and spring, being overall similar at both seasons (ranging from 0 to 7 species and from 0 to 1.9 bits, respectively). Evenness was significantly lower (P < 0.05) in winter than in spring (Fig. 3).
Three station clusters were obtained both during spring and winter, which differed significantly (PERMANOVA, Table 3). In spring (Fig. 4a), cluster 1 included 12 stations located mainly in the inner region of the bay. They were characterized by sediments mostly composed by different proportions of silt and clay, with a 4.3 ± 3.8% OM and 7 ± 6.6 of chl-a, on average. The polychaete assemblages showed relatively low average density (60 ± 35.9 ind./m2), species richness (2.7 ± 1.4), and diversity (0.8 ± 0.4), while the average evenness (c.a. 0.8) was similar in the three clusters. The dominant species were Maldane sarsi and Paraprionospio pinnata (Table 2). Cluster 2 consisted of 7 stations occupying mostly the inner-central region of the bay. They had mainly silty and clayed sands, as well as high chl-a (11.6 ± 4.2 mg/m2) and moderately high OM (2 ± 1.3%). They show high average density (94.9 ± 28.3 ind./m2), species richness (5.1 ± 1.6), and diversity (1.4 ± 0.4 bits). The dominant species was Eunice vittata (Table 2). Cluster 3 consisted of 12 stations mainly located close to the bay opening. Accordingly, they had predominantly sandy sediments, low OM (1.4 ± 1.2%), and moderate chl-a (8.6 ± 4.9 mg/m2). Polychaete assemblages showed relatively low average density (63.3 ± 50.3 ind./m2), species richness (3.7 ± 1.9), and diversity (1.0 ± 0.5 bits). The dominant species was Cirriformia tentaculata (Table 2).
During winter (Fig. 4b), Cluster 1 included 10 stations, mainly located in the inner-central region of the bay, which were characterized by having relatively high OM (3.2 ± 2.7%), low chl-a (4.9 ± 4.4 mg/m2), clay, and silty clay, together with a moderate average density (56 ± 38.1 ind./m2) and low species richness (2.5 ± 1.9). Diversity (0.5 ± 0.7 bits) and evenness (0.4 ± 0.4). The dominant species was Maldane sarsi (Table 2). Cluster 2 included 13 stations located all along the bay, but mostly in the central part. They were characterized by having high chl-a (12.2 ± 9.2), moderately high OM (2.8 ± 2.5%), and silty and clayed sands, while there was low average density (14.2 ± 12.3ind./m2), species richness (1.2 ± 0.4), diversity (0.1 ± 0.3 bits), and evenness (0.2 ± 0.4). The dominant polychaete was Naineris laevigata (Table 2). Cluster 3 included 11 stations located mostly near the bay opening. They were characterized by having low OM (1.3 ± 0.7%), moderately high chl-a (11.3 ± 5.9 mg/m2), and a high percentage of sand. Moreover, this cluster showed the highest recorded average density (136 ± 239.5 ind./m2), species richness (4.8 ± 1.5), diversity (1.4 ± 0.5 bits), and evenness (0.9 ± 0.3), while the dominant species were Cirriformia tentaculata and Ophelia rathkei (Table 2).
Relationships between polychaete assemblage descriptors and environmental variables
In spring, the first two CCA axes accounted for 61.88% of the observed variance. The species composition was mainly related to granulometry and distance (Fig. 5a). In winter, the first two CCA axes accounted for 59.28% of the relationships, with the most influencing environmental variables being chl-a, granulometry, and OM (Fig. 5b).
In spring, the projections of the species on the environmental parameter vectors (Fig. 5a) showed positive correlations of Panousea Africana, Cirriformia tentaculata, Platynereis dumerilii, Schistomeringos neglecta, Maldane sarsi, Glycera alba, Glycera cf. tridactyla, Paraprionospio pinnata, and Pistella lornensis with distance, OM, salinity, and temperature. In turn, Eunice vittata, Eumida sanguinea, Axiothella constricta, and Glycinde nordmanni were positively correlated with granulometry and chl-a. As for the stations, they are left-right oriented for axis 1, which corresponds to a downstream-upstream gradient. Accordingly, downstream stations (e.g., 31, 33, 34, 35, 36, 38, 39) were characterized by larger grain sizes and high chl-a, whereas the upstream stations (e.g., 1, 2, 3, 4, 5, 8, 9, 10, 11, 19) were characterized by high distance, OM, salinity, and temperature.
In winter, the projections of the species on the environmental parameters vectors (Fig. 5b) showed the positive correlations of Pistella lornensis, Schistomeringos neglecta, Naineris laevigata, Eunice vittata, Glycinde nordmanni, Phyllodoce sp., Cirriformia tentaculata, and Ophelia rathkei with granulometry, chl-a and temperature, while Maldane sarsi, Panousea africana, Sthenelais boa, Platynereis dumerilii, and Glycera alba were positively correlated with distance, OM, and salinity. As for the stations, they are oriented toward the right of axis 1, which also corresponds to a downstream-upstream gradient, with the downstream stations (e.g., 28, 32, 33, 34, 35, 37, 38, 39) being characterized by larger grain sizes and high chl-a, whereas the upstream stations (e.g., 1, 4, 3, 6, 8, 10, 11, 12, 15) were characterized by having high distance, salinity, and OM.
Discussion
Dakhla Bay is one of the most important wetlands along the Moroccan Atlantic coastline. Situated in a Saharan area, this ecosystem is highly diverse both in terms of fauna and flora. Curiously enough, it has not been extensively studied, and the present work contributes to this knowledge by providing the first report describing the temporal and spatial patterns of the diversity and structure of the polychaete assemblages and their relationships with the main environmental parameters. The novelty of this study for the southern Atlantic coast of Morocco leads us to consider it as a good baseline for future ecological research.
The lower temperature at the entrance of the bay reflects the influence of the cold ocean waters Dafir (1997). The other stations tended to be easily warmed because of their shallow depth and the loss of momentum. However, in our study, this trend was only evident in spring, as reflected by the positive correlation between temperature and the distance, while it does not occur in winter. Similarly, and for the same reason, downstream salinity tended to be lower, reaching almost the oceanic 35‰ (Aminot and Chaussepied, 1983). This finding has also been reported by Saad et al. (2013) and coincided with our own observations in which there was a positive correlation between salinity and the distance both in spring and in winter. On the other hand, the overall distribution of the granulometry and organic matter follows the hydrodynamic state (Dafir 1997; Zidane 2009; Zidane et al. 2017), with the upstream area tending to be relatively calm and to have more fine sediments and organic matter, although the relationships of both parameters and the distance were non-significant.
The presence of 12 families and 22 species of polychaetes in Dakhla Bay (Table 1) represents a moderately high diversity. It was certainly higher than in Camamu Bay, Brazil (17 species) (Paixão et al. 2010), and Chabahar Bay, Iran (16 species) (Taheri et al. 2010), but was lower than in Veys Bay, France (27 species) (Timsit et al. 2004), Tijucas Bay, Brazil (47 species) (de Almeida and Vivan 2011), Admiralty Bay, New Zealand (76 species) (Pabis and Siciński, 2012), Blanes Bay, Spain (ca. 80 species) (Pinedo et al. 1996), and Els Alfacs Bay, Spain (101 species) (Martin et al. 2000). Among other reasons, differences in sampling devices, sampling effort, and area covered may contribute to explain the variability in species diversity, but also intrinsic environmental factors such as the depth range, sedimentary heterogeneity, and hydrodynamic regimes have been argued to explain the local diversity patterns. Despite the temporal environmental differences found in Dakhla Bay, and, to some extent, the differences in composition, the structure of the polychaete assemblages was almost the same during the two study periods (Fig. 4a, b). The environmental descriptors, including the granulometry and, particularly, the salinity and temperature (the latter only in spring), conformed a regular gradient, which was mirrored by the polychaete assemblages. In fact, the Maldane sarsi assemblage found in the fine sediments of the inner bay was replaced by the E. vittata (spring) N. laevigata (winter) ones in the medium grain-sized sediment of the mid-bay, and by the C. tentaculata one in the sandy sediments closer to the outer inlet. The shift was, in fact, from a classical, brackish, lacunar assemblage to two different, temporal aspects of a marine assemblage (close to the outer inlet), with a transition assemblage in between.
The diversity, distribution, and structure of the polychaete assemblages are closely driven by environmental parameters (Carrasco and Carbajal 1998; Simboura et al. 2000; Labrune et al. 2007; El Asri et al. 2017), with a special relevance in the case of sediment characteristics and organic matter (Glémarec 1973; Martin et al. 2000; Barbosa et al. 2010; Martins et al. 2013). In Dakhla Bay, sediments on one hand (in spring and winter) and the parameters related with the food availability, organic matter, and chl-a content (in spring) highly influenced the polychaete density distribution (Fig. 5a, b), while they show a range of different degrees of influence at the species level (in most cases resulting in positive relationships) (Table 3).
The environmental factors most often reported as being responsible for the observed patterns of benthic assemblages in marine bays tend to be rather similar. Among them, gradients in sediment composition, organic matter content, hydrodynamics and, occasionally, salinity are suggested as the main driving factors. However, the dominant species are clearly different, suggesting that other factors may be more strongly influent. Among them, the assemblages inhabiting a given bay may be dependent on but also on biogeographic and, to some extent, historical reasons, but also on the regional organisms’ pool, which led different species to respond to equivalent sets of environmental conditions in bays with markedly different biogeographical locations. Accordingly, the local environmental stressors (i.e., mainly particle size, chl.-a, salinity, OM, and temperature) combined with regional species pool lead the following species to be dominant in Dakhla Bay: M. sarsi (25.1%), E. vittata (11.1%), and N. laevigata (10%) in spring and O. rathkei (43%), M. sarsi (21.5%), and N. laevigata (5.9%) in winter. This pattern of dominant species seems to be highly characteristic for a given bay, and may vary depending on the local environmental conditions, and the regional species pool. For instance, in Bou Ismail Bay the dominant species were Aponuphis bilineata (Baird 1870) and Hialinoecia brementi Fauvel, 1916 (Bakalem 2008). Algiers Bay was dominated by Owenia fusiformis Delle Chiaje, 1844 (Bakalem 2008). Admiralty Bay was dominated by Capitella sp. (as C. capitata) and Leodamas marginatus (Ehlers, 1897) (Sicinski and Janowska 1993). The most abundant species in Terra Nova Bay were Tharyx cincinnatus (Ehlers, 1908) and Spiophanes tcherniai Fauvel, 1950 (Gambi et al. 1997). In Chile Bay, M. sarsi and T. cincinnatus were the dominant species (Gallardo et al. 1988), while in Todos os Santos Bay (Brazil), the most abundant species was Armandia polyophthalma Kükenthal, 1887 (Pires-Vanin et al. 2011).
Moreover, all the above-mentioned bays differ considerably from one another in size, morphology, water depth, salinity, etc. This may certainly contribute to explain the differences in composition and dominance of the respective polychaete assemblages. The strong dominance in abundance by a few number of species can be explained by the existence of large fluctuations in the environmental conditions, which ensure that only species tolerant to changes in the driving abiotic factors (e.g., salinity, temperature, oxygen availability, and type of substrate) turned to be adapted to survive and proliferate in this kind of coastal ecosystem (Guelorget and Michel 1979; Martin et al. 2000; El Asri et al. 2015). Moreover, drastic alterations in species composition through time may also occur, particularly if there are associated changes in anthropogenic pressure (Hernández-Guevara et al. 2008). Finally, inter-specific interactions among polychaetes, but also with other benthic invertebrates, including predation, competition etc., may also contribute to explain the changes in species dominance, but also in the structure of the whole polychaete assemblage (Gambi et al. 1995; Artemis et al. 2006; Schückel et al. 2014).
Despite the unavoidable usefulness of baseline studies like the present one at Dakhla Bay to assess future changes in benthic environments and the associated assemblages, more specific studies are required to fully understand the particular functioning of the bay. Nonetheless, it is necessary to emphasize that the benthic communities in the bay are severely threatened by the worsening conditions mainly in relation with the spread of human activities. The growing relevance of the commercial exploitation of benthic species and the touristic developments certainly increases their influence on the bay that, as a semi-enclosed, transition ecosystem, is particularly vulnerable to threats derived from human activities (Newton et al. 2014).
The polychaetes are well known as relevant contributors to the biodiversity of marine sediments since the earliest reviews by Snelgrove (1998, 1999). Despite their relevance, however, they are the single taxa being described from Dakhla Bay. Therefore, there is a high risk that many other not yet studied taxa would disappear, even before their presence in the Bay will be registered. We are aware that the present characterization of the polychaete assemblages from Dakhla Bay may be considered as incomplete (particularly in terms of seasonal and inter-annual variability). However, the information generated by the present biotic inventory, including spatial and, in part, seasonal variability, arose as a key dataset to allow future monitoring of the functioning of these communities and their responses to the threats to which they are submitted. Hopefully, it may also stimulate further studies of the endangered wetland environments in southern Moroccan coast, which would be a major contribution to facilitate the management and protection of these natural resources.
References
Aminot A, Chaussepied M (1983) Manuel des analyses chimiques en milieu marin. Ed. CNEXO, BNDO / Documentation, BREST
Anderson MJ (2001) A new method for non-parametric multivariate analysis of variance. Austral Ecol 26:32–46
Arbi I, Zhang J, Liu S, Wu Y, Huang X (2017) Benthic habitat health assessment using macrofauna communities of a sub-tropical semi-enclosed bay under excess nutrients. Mar Poll Bull 119(2):39–49
Artemis N, Petrou K, Kormas KA, Reizopoulou S (2006) Inter-annual variability of soft bottom macrofaunal communities in two Ionian Sea lagoons. Hydrobiologia 555:89–98
Aubry A, Elliott M (2006) The use of environmental integrative indicators to assess seabed disturbance in estuaries and coasts: application to the Humber Estuary, UK. Mar Poll Bull 53:175–185
Bakalem A (2008) Contribution à l’étude des peuplements benthiques du plateau continental algérien. Doctorat d’état, Université des sciences et de la technologie Houari Boumediene, Faculté Des Sciences Biologiques. Algeria
Barbosa LDS, Soares-Gomes A, Paiva PC (2010) Distribution of polychaetes in the shallow, sublittoral zone of Admiralty Bay, King George Island, Antarctica in the early and late austral summer. Nat Sci 2(10):1155–1163
Belan TA (2003) Benthos abundance pattern and species composition in conditions of pollution in Amursky Bay (the Peter the Great Bay, the Sea of Japan). Mar Poll Bull 46:1111–1119
Borja A, Franco J, Pérez V (2000) A marine biotic index to establish the ecological quality of soft-bottom benthos within European estuarine and coastal environments. Mar Poll Bull 40(12):1100–1114
Byers JE, Grabowski JH (2014) Soft-sediment communities. In: Bertness MD, Bruno JF, Silliman BR, Stachowicz JJ (eds) Marine community ecology and conservation. Sinauer Associates, Sunderland, pp 227–249
Cao Y, Bark AW, Williams WP (1997) A comparison of clustering methods for river benthic community analysis. Hydrobiologia 347:24–40
Carrasco F, Carbajal W (1998) The distribution of polychaete feeding guilds in organic enriched sediments of San Vicente Bay, Central Chile. Int Rev Hydrobiol 83:233–249
Dafir JE (1997) Application de la dynamique du phosphore à l’étude de l’organisation et du fonctionnement de certains écosystèmes aquatiques (lagune Nador, baie Dakhla, barrage Al Massira) (gestion et préservation). Doctorat d’état, Université Hassan II, Faculté des Sciences Ain Chock, Casablanca (Maroc)
Dauvin, JC, Andrade H, de-la-Ossa-Carretero JA, Del-Pilar-Ruso Y, Riera R (2016) Polychaete/amphipod ratios: an approach to validating simple benthic indicators. Ecol Indic 63: 89–99
De Almeida TCM, Vivan JM (2011) Macrobenthic associations in a South Atlantic Brazilian enclosed bay: the historical influence of shrimp trawling. Mar Poll Bull 62(10):2190–2198
Dixon-Bridges K, Hutchings P, Gladstone W (2014) Effectiveness of habitat classes as surrogates for biodiversity in marine reserve planning. Aquat Conserv Mar Fresh Ecosyst 24(4):463–477
Dufrêne M, Legendre P (1997) Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecol Soc Am 67:345–366
El Asri F, Zidane H, Maanan M, Tamsouri M, Errhif A (2015) Taxonomic diversity and structure of the molluscan fauna in Oualidia lagoon (Moroccan Atlantic coast). Environ Monit Assess 187:1–10
El Asri F, Zidane H, Errhif A, Tamsouri M, Maanan M, Malouli Idrissi M, Martin D (2017) Polychaete diversity and assemblage structure in the Oualidia Lagoon, Moroccan Atlantic coast. J Mar Biol Assoc U K:1–10
Gallardo VA, Medrano SA, Carrasco FD (1988) Taxonomic composition of the sublittoral soft-bottom Polychaeta of Chile Bay (Greenwich Island, South Shetland Islands, Antarctica). Ser Cient INACH 37:49–67
Gambi MC, Giangrande A, Martinelli M, Chessa LA (1995) Polychaetes of a Posidonia oceanica bed off Sardinia (Italy)-spatiotemporal distribution and feeding guild analysis. Sci Mar 59:129–141
Gambi MC, Castelli A, Guizzardim M (1997) Polychaete populations of the shallow bottoms off Terra Nova Bay (Ross Sea, Antarctica): distribution, diversity and biomass. Polar Biol 17:199–210
Giangrande A, Licciano M, Musco L (2005) Polychaetes as environmental indicators revisited. Mar Poll Bull 50:1153–1162
Gilbert F, Stora G, Cuny P (2015) Functional response of an adapted subtidal macrobenthic community to an oil spill: macrobenthic structure and bioturbation activity over time throughout an 18-month field experiment. Environ Sci Poll Res 22(20):15285–15293
Glémarec M (1973) The benthic communities of the European North Atlantic continental shelf. Oceanogr Mar Biol An Annu Rev 11:263–289
Guelorget O, Michel P (1979) Les peuplements benthiques d’un étang littoral languedocien, l’étang du Prévost (Hérault). 2. Etude quantitative de la macrofaune des sables. Téthys 9:65–77
Guelorget O, Orbi A, Lefebvre A (1996) La baie de Dakhla: Organisation biologique et fonctionnement. Institut National de Recherche Halieutique, Maroc
Hammer O, Harper DAT, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis. Palaeontol Electron 4:1–9
Hernández-Guevara NA, Pech D, Ardisson P (2008) Temporal trends in benthic macrofauna composition in response to seasonal variation in a tropical coastal lagoon, Celestun, Gulf of Mexico. Mar Fresh Res 59:772–779
Holm-Hansen O, Lorenzen CJ, Holmes RW, Strickland JDH (1965) Fluorometric determination of chlorophyll. J Mar Sci 30:3–15
Jumars PA, Dorgan KM, Lindsay SM (2015) Diet of worms emended: an update of polychaete feeding guilds. Annu Rev Mar Sci 7:497–520
Junoy J, Castellanos C, Vieitez JM, Riera R (2013) Seven years of macroinfauna monitoring at Ladeira Beach (Corrubedo Bay, NW Spain) after the Prestige oil spill. Oceanologia 55:393–407
Kristensen E, Delefosse M, Quintana CO, Flindt MR, Valdemarsen T (2014) Influence of benthic macrofauna community shifts on ecosystem functioning in shallow estuaries. Front Mar Sci 1(41):1–14
Labrune C, Grèmare A, Amouroux JM, Sardá R, Gil J, Taboada S (2007) Assessment of soft-bottom polychaete assemblages in the Gulf of Lions (NW Mediterranean) based on a mesoscale survey. Estuar Coast Shelf Sci 71(1–2):133–147
Magalhaes WF, Barros F (2011) Structural and functional approaches to describe polychaete assemblages: ecological implications for estuarine ecosystems. Mar Fresh Res 62(8):918–926
Martin D, Pinedo S, Sardá R (2000) Distribution patterns and trophic structure of soft-bottom Polychaete assemblages in a North-Western Mediterranean shallow-water Bay. Ophelia 53:1–17
Martins R, Sampai L, Rodrigues AM, Quintino V (2013) Soft-bottom Portuguese continental shelf polychaetes: diversity and distribution. J Mar Syst 123-124:41–54
Newton A, Icely J, Cristina S, Brito A, Cardoso AC, Colijn F, Riva SD, Gertz F, Hansen JW, Holmer M, Ivanova K, Leppäkoski E, Canu DM, Mocenni C, Mudge S, Murray N, Pejrup M, Razinkovas A, Reizopoulou S, Pérez-Ruzafa A, Schernewski G, Schubert H, Carr L, Solidoro C, Pierluigi V, Zaldívar J-M (2014) An overview of ecological status, vulnerability and future perspectives of European large shallow, semi-enclosed coastal systems, lagoons and transitional waters. Estuar Coast Shelf Sci 140:95–122
Olsgard F, Brattegard T, Holthe T (2003) Polychaetes as surrogates for marine biodiversity: lower taxonomic resolution and indicator groups. Biodivers Conserv 12:1033–1049
Orbi A, Dafir JM, Berraho A (1995) Etude pluridisciplinaire de la baie de Dakhla. Travaux et document. INRH. In: N°86
Orbi A, Dafir JE, Berraho A, Sarf F (1999) Etude Pluridisciplinaire de la Baie de Dakhla. Note d’information (interne) sur la planification de l’aquaculture dans la baie de Dakhla. Maroc
Pabis K, Siciński J (2012) Is polychaete diversity in the deep sublittoral of an Antarctic fiord related to habitat complexity? Polish Polar Res 33:181–197
Paixão JF, de Oliveira OMC, Domingueza JML, Coelho ACD, Garcia KS, Carvalho GC, Magalhães WF (2010) Relationship of metal content and bioavailability with benthic macrofauna in Camamu Bay (Bahia, Brazil). Mar Poll Bull 60(3):474–481
Papageorgiou N, Arvanitidis C, Eleftheriou A (2006) Multicausal environmental severity: a flexible framework for microtidal sandy beaches and the role of polychaetes as an indicator taxon. Estuar Coast Shelf Sci 70:643–653
Pearson TH, Rosenberg R (1978) Macrobenthic succession in relation to organic enrichment and pollution in marine environment. Oceanogr Mar Biol An Annu Rev 16:229–311
Pielou EC (1966) Shannon’s formula as a measure of specific diversity: its use and measure. Am Nat 100:463–465
Pinedo S, Sardá R, Martin D (1996) Seasonal dynamics and structure of soft-bottom assemblages in the Bay of Blanes (Western Mediterranean Sea). Pub Esp InstEsp Oceanogr 22:61–70
Pires-Vanin AMS, Muniz P, de Léo FC (2011) Benthic macrofauna structure in the northeast area of Todos os Santos Bay, Bahia state, Brazil: patterns of spatial and seasonal distribution. Braz J Oceanogr 59(1):27–42
Pocklington P, Wells PG (1992) Polychaetes: key taxa for marine environmental quality monitoring. Mar Pollut Bull 24:593–598
Qninba A, Radi M, Benhoussa A, Bazairi H, Menioui M (2003) Fiche descriptive sur les zones humides Ramsar (FDR) : Baie d’Ad-Dakhla (Maroc)
Saad Z, Orbi A, Abouabdellah R, Saad A, Oudra B (2013) Impact of economic development on the dynamics of phytoplankton and physic-chemical quality of Dakhla Bay (south of Morocco). South Asian J Exp Biol 3(5):274–287
Samuelson GM (2001) Polychaetes as indicators of environmental disturbance on subartic tidal flats, Iqaluit, Baffin Island, Nunavut territory. Mar Pollut Bull 42(9):733–741
Sardá R, Pinedo S, Dueso A (2000) Estimating secondary production in natural populations of polychaetes: some general constraints. In: Proceedings of the VI International Polychaete Conference. Bull Mar Sci 67:433–447
Schückel U, Beck M, Kröncke I (2014) Macrofauna communities of tidal channels in Jade Bay (German Wadden Sea): spatial patterns, relationships with environmental characteristics, and comparative aspects. Mar Biodivers 45:841–855
Shannon CE (1948) A mathematical theory of communication. Bell Syst Tech J 27:379–423
Sicinski J, Janowska E (1993) Polychaetes of the shallow sublittoral of Admiralty Bay, King George Island, South Shetland Islands. Antarct Sci 5:161–167
Simboura N, Nicolaidou A, Thessalou-Legaki M (2000) Polychaete communities of Greece: an ecological overview. Mar Ecol 21:129–144
Snelgrove PVR (1998) The biodiversity of macrofaunal organisms in the marine sediment. Biodivers Conserv 7:1123–1132
Snelgrove PVR (1999) Getting to the bottom of marine biodiversity: sedimentary habitats. BioScience 49(2):129–138
Taheri M, Yazdani FM, Bagheri H (2010) Community structure and biodiversity of intertidal sandy beach macrofauna in Chabahar Bay, northeast of Oman Gulf, IR Iran. J Persian Gulf 1:17–25
Timsit O, Bernard S, Lefeuvre JD (2004) Evolution du macrozoobenthos intertidal de la baie de Veys de 1985 à 2000. Comptes Rendus – Biol 327(1):51–64
Vakharia AJ, Wemmerlöv U (1995) A comparative investigation of hierarchical clustering techniques and dissimilarity measures applied to the cell formation problem. J Oper Manag 13:117–138
Zidane H (2009) Biologie et potentialités aquacoles de l’huître creuse Crassostrea gigas (Thunberg, 1793) en élevage dans la baie de Dakhla (sud du Maroc). Thèse de Doctorat, Université Ibn Tofail, Kenitra (Maroc)
Zidane H, Orbi A, Mouradi A, Zidane F, Blais JF (2008) Structure hydrologique et édaphique d’un site ostréicole: Duna Blanca (La Baie De Dakhla Sud Du Maroc). Environ Technol 29:1031–1042
Zidane H, Maanan M, Mouradi A, Maanan M, El Barjy M, Zourarah B, Blais JF (2017) Environmental and ecological risk of heavy metals in the marine sediment from Dakhla Bay, Morocco. Environ Sci Poll Res 24:7970–7981
Acknowledgements
This paper is part of a research program on coastal resources of the LERL/DRH-Casablanca (National Fisheries Research Institute) and of the laboratory Geolittomer, UMR 6554 LETG University of Nantes, France, as well as a contribution of DM to the research project PopCOmics (CTM2017-88080), funded by the Agencia Estatal de Investigación and the European Funds for Regional Development and to the Consolidated Research Group on Marine Benthic Ecology of the Generalitat de Catalunya (2017SGR378). We are deeply grateful for all the INRH-Dakhla staff for their help during sampling and human quality and would like to address special thanks to Mr. Mohmoud El khattat for their help during the field samplings (INRH Dakhla). We are also grateful to the reviewers for their critical and constructive comments, which highly contributed to improve the quality of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with animals performed by any of the authors.
Sampling and field studies
All necessary permits for sampling and observational field studies have been obtained by the authors from the competent authorities and are mentioned in the acknowledgements, if applicable.
Additional information
Communicated by P. Lana
Rights and permissions
About this article
Cite this article
El Asri, F., Martin, D., Tamsouri, MN. et al. Spatial and temporal variability in distribution, diversity, and structure of the polychaete assemblages from Dakhla Bay (Atlantic coast of South Morocco). Mar Biodiv 49, 1271–1281 (2019). https://doi.org/10.1007/s12526-018-0910-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12526-018-0910-9