Abstract
The Sugarbeet is one of the main sugar crops in the world. In the search for sustainability and economic value, the complete utilization of the crop is necessary. In addition to sugar and animal feed, sugarbeets can provide many value-added co-products for biofuels, human nutrition, plastics, and pharmaceuticals. Current research efforts are reviewed.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Economic Impact and Value
Sugarbeet (Beta vulgaris) provides about 20 % of the global demand for sugar. The European Union is one of the global leaders in sugar production along with the United States. The United States provides about 11 % of the world’s supply of beet sugar (Biancardi et al. 2010). Sugarbeet and sugarcane dominate the U.S. domestic market; mainly due to preferential international trade and agriculture agreements. In 2012, over 1.2 million acres were planted in the United States yielding over 35 million tons of Sugarbeets (National Agricultural Statistics Service 2013a). Primary growing regions are California, the Upper and Central Great Plains, the Upper Midwest and the Northwest. The value of the U.S. Sugarbeet crop in 2011 was in excess of $2.0 trillion with an average price of $69.50 per ton (National Agricultural Statistics Service 2013b). In other markets, the value of Sugarbeet to sugar production and its co-products is more limited. For example, in China, Sugarbeets only account for about 6 % of the total sugar output in 2006 (Wei and Li 2006).
Brief History of the Sugarbeet
Before 1750, all sugar production came from sugar cane grown in the tropics and shipped around the world (Alamzan et al. 1998). Around this time, a German chemist extracted the sugar from beets (B. vulgaris) grown primarily as animal fodder, and discovered beet sugar was exactly the same as cane sugar: sucrose (Cooke and Scott 1993; Jodidi 1911). It took 50 years for a large scale production method to be developed. Economic embargoes of sugar during decades of war made it necessary for continental Europe to develop its own sugar industry using sugarbeets (Fig. 1a). In the late 19th century and the beginning of the 20th century, Sugarbeets were introduced into the United States and facilitated by the U.S. Department of Agriculture through its network of agricultural colleges, experiment stations and cooperative extensions (Fig. 1b) (Alamzan et al. 1998; Cattanach et al. 1991). Sugarbeets are very tolerant of different climates and soils and could be grown on marginal land. Plant breeding programs have selected for nutrition, sugar content, yield, and disease/pest resistance (Stevanato and Panella 2013). In addition, the whole beet with its coproducts of greens, molasses, and pulp residue could be utilized as an animal feed or a feedstock for alcohol production. Detailed information on breeding, cultivation, protection, harvest and storage of Sugarbeets may be found in Draycott’s 2006 work, Sugar Beet (Draycott 2006).
Historical posters from United States National Archives and Records Administration. a Source Office for Emergency Management. Office of War Information. Domestic operations branch. Bureau of special services. 1943–1945. b Source United States Department of Agriculture. Production and Marketing Administration, 1946
Sugar Production and Its Co-products
Sugar (sucrose) is extracted from beets using hot water in a multi-step step process: the initial extraction of a syrup then concentration followed by cyclic washing and finally drying the Sugar Association (2013). In general, modern sugarbeets have around 20 % sugar by weight. The non-crystallized syrup is called Beet molasses which has upwards of 50 % by weight of sugar. Beet molasses is usually fermented into alcohol. The leftover molasses is rich in nitrogen and is used either as animal feed or as a fertilizer. Once the sugar is extracted, the “pressed” and dehydrated beet pulp is normally fed to animals. Sugarbeet pulp (SBP) also contains a significant fraction of cell wall polysaccharides including pectin and dietary fiber. Utilization of the co-products reduce waste and add value to the crop (Broughton et al. 1975).
Livestock Feed
The primary market for Sugarbeets as fodder is ruminants such as cows and pigs (Teimouri Yansari 2013; Zijlstra and Beltranena 2013). Beet tops with or without the crowns can be left in the field or ensilaged for animal feed. Generally, the crude protein of beet tops is around 15 % (dry weight basis) and has a D-value (digestibility) of 55. They are considered slightly inferior or equivalent to alfalfa in feeding cattle and sheep. In comparison, sugar cane tops and bagasse has a D-value of 57 with 6–8 % crude protein (Steg and Van Der Meer 1985). If harvest is not possible, the entire beet can be left in the field as forage. Beet pulp, leftover from sugar production, may also be used as animal feed and contains about 9 % crude protein and a D-value of 75 (Hartnell et al. 2005). There are research efforts to increase the protein content by fermentation (Iconomou et al. 1998). Because specific nutritional requirements of ruminants and their feed composition are beyond the scope of this paper, the reader is invited to explore numerous articles in both scientific literature and trade publications; two reviews may be of particular interest (Fadel et al. 2000; Kelly 1983).
Current Research Efforts on the Utilization of Sugarbeet
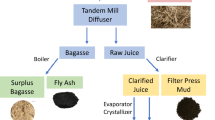
In the search for sustainability and profitability of a crop, the utilization of the entire plant is mandatory beyond the traditional roles of harvesting for food or feed and then directly or indirectly returning the residue to the soil, burning for fuel or non-environmental disposal. The composition of SBP suggests that it could be used to produce several value-added products (Table 1).
Food Ingredients
Fiber products from SBP have been Generally Recognized as Safe (GRAS) since 1991 (Nordic Sugar 2012) and are produced with a relatively simple process. Nutritional data from commercial products show that beet fiber contains around 8 % protein (by weight) and 67 % carbohydrates such as hemicelluloses (28 %), cellulose (19 %), and pectin (18 %) (Michel et al. 1988). Dietary fiber available for human digestion is generally more than 20 % (Cho 2001). Fiber products from Sugarbeets can be either the whole pulp or a purified pectic substance like arabinan (Goodban and Owens 1956) and have a wide range of beneficial effects on human health (Ralet et al. 2009). Excellent reviews on dietary fiber in food and its effects on human nutrition/health can be found in the Handbook for Dietary Fiber (Cho 2001) and Fiber Ingredients (Cho and Samuel 2009). For example, the effect of SBP on cholesterol levels was investigated with positive results (Leontowicz et al. 2001). The use of Sugarbeet fiber in processed foods is limited by its texture and taste. It is generally used in meat patties, bakery products, cereals and assorted products that need thickening or bulking agents (Dhingra et al. 2012).
In the 1990s, some effort was made to evaluate Sugarbeet leaf protein as a food component in comparison to other leafy green matter (Fantozzi 1990; Jwanny et al. 1993). Beet tops are considered a healthy choice in both salads and juices.
One recent health claim is that the pectic oligosaccharides in SBP, such as arabinan, may function as a prebiotic in the human gut (Tamimi et al. 2006). Prebiotics affect the microbial population in the human gut and generally favor bacteria beneficial to human health.
The phenolic compounds may be extracted from SBP and used as antioxidants in food (Mohdaly et al. 2010).
Sugarbeet molasses can be used as the sugar/carbon source for Xanthan fermentation by micro-organisms (Moosavi and Karbassi 2010). Xanthan is used extensively as a thickener in the food industry.
Pectin
Pectin is a cell-wall polysaccharide consisting of galacturonic acid with ramnose sidechains in varying proportions. Pectin is best known for its gelling properties in fruit products (Norsker et al. 2000). Beet pectin exhibits better emulsifying properties than other sources of pectin (Ma et al. 2013). Pectin can be obtained in good yields (+95 %) from SBP by pectinolytic enzymes and mild, organic acids (Concha-Olmos and Zuniga-Hansen 2012). The effects of extraction temperature, time and pH on the yield, color, rheological and emulsifying properties of sugarbeet pectin has been reported (Lv et al. 2013). The structure of pectin includes functional groups such as acetic and ferulic acids. These functional groups provide pectin with the ability to form networks through Calcium bridges, acid-sugar bridges and oxidative crosslinks (Oosterveld et al. 2000). There is some evidence that the excellent stabilization properties of sugar beet pectin in emulsions is due to the attachment of a protein (Fishman et al. 2013; Kirby et al. 2006).
Araban (or Arabinan), a low molecular weight colloid, was isolated from SBP and could be used as an adhesive, emulsion stabilizer (Fishman et al. 2009) and suspension agent in cosmetics or pharmaceuticals (Goodban and Owens 1956). For example, Sugarbeet pectin can be used to suspend colorants such as anthocyanins from berries (Buchweitz et al. 2012).
There has been recent interest in isolating cellulose microfibrils (Fishman et al. 2011) while degrading the pectin into its constituent monomers (Leijdekkers et al. 2013). The cellulose would be used in polymer composites while the monomers would be used in other high-value ways (Dufresne et al. 1997; Leitner et al. 2007).
Plastics and Composites
Plastics are prevalent in the global market. Most are derived from petroleum, and there are many research efforts to replace petro-plastics with bioplastics derived from renewable resources. In some cases, plant polymers only need be extracted before use while others need to be synthesized from small molecules also derived from plants.
Sugar beet pulp was processed in a twin-screw extruder using plasticizers to obtain thermoplastic films (Liu et al. 2011b; Rouilly et al. 2006, 2009). The resulting composite could be characterized as cellulose microfibrils suspended in a pectin matrix. SBP was also used as a polyol source for the production of urethanes (Pavier and Gandini 2000). Sugarbeet pulp was combined with a biobased polymer, polylactic acid, to form polymer composites that had similar tensile properties to commodity plastics (Chen et al. 2008; Finkenstadt et al. 2008). The SBP could be plasticized and used as a co-polymer rather than as a filler in both PLA and poly(butylene adipate-co-terepthalate) (Liu et al. 2011a).
Microbial and plant sourced polyesters such as polyhydroxyalkanoates (PHA) are making inroads into the plastics market. Sugarbeet juice was used as a sugar feedstock for the production of PHAs such as poly(hydroxybutyrate) (Wang et al. 2013). PHB is an important biobased polymer with plastic properties similar to synthetics such as polypropylene. It is also compostable and environmentally friendly. The “carbon source” cost of PHA production, calculated based on Sugarbeet molasses as a sole feedstock, was approximately $1.40/kg (Castilho et al. 2009).
Pectin, extracted from SBP, is also used in plastic packaging materials (Li et al. 2012; Liu et al. 2012). In some cases, pectin can be used to protect active ingredients from thermal shock during processing into thin films for food packaging (Liu et al. 2007).
Conversion to Platform Chemicals
The search for renewable and sustainable fuels has driven the development of technology for complete utilization of biomass (Hood et al. 2013). In general, lignocellulosic grasses or woody plants garner most of the attention of technology developments to harvest fermentable sugars for conversion to a wide range of platform chemicals; ethanol being the current favorite. The low lignin content and the high digestibility of its carbohydrates (Table 1) make SBP a feedstock candidate for biorefineries. Fermentation requires the breakdown of cell wall networks and their components like, in the case of SBP, pectin and cellulose. The severity of the pre-treatment and the use of different enzyme treatments affect the fermentable sugar yield from SBP (Kuhnel et al. 2011; Martinez et al. 2009; Micard et al. 1997).
Both the small amounts of ferulic acid and large amounts of cellobiose (glucose) extracted from SBP could be used to produce “natural” vanillin in a bioconversion process using fungal enzymes (Bonnin et al. 2000). Ferulic acid is used as a pre-cursor in the food and drug industries (Kroon et al. 1996).
Sugar beet vinasse, leftover from ethanol production, contains Betaine (15 %) which is used as amphoteric surfactants in personal care products. Betaine, and other polyphenols, can be recovered using ion-exchange resins (Caqueret et al. 2008; Soto et al. 2011).
Galactinol dehydrate and myo-inositol were first isolated from Sugarbeet syrup in 1965 and are used in proteomics and pharmaceutical applications (McCready et al. 1965).
Oxalic acid can be formed from Sugarbeet molasses using a vanadium catalyst with a 75 % yield (Guru et al. 2001).
In order to design an efficient and (relatively) inexpensive conversion of any vegetal matter, one needs accurate knowledge of the structure and composition of the components, the interaction and structure between the components, and the synergistic effects of thermochemical and enzymatic treatments to separate and purify the constituents (Van Dyk et al. 2013). This is not a trivial matter when designing a production facility to process different crops all with different compositions.
Energy Production
Energy production from renewable resources is increasingly in demand. Using Sugarbeets as the feedstock, the main target for biofuel production is ethanol. Once extracted, sucrose can be directly fermented into ethanol using any number of traditional, industrial-scale methods. In contrast, starchy crops need additional processing steps to obtain fermentable sugars. Advances in lignocellulosic bioconversion will allow the use of the beet tops and SBP for bioenergy production. For example, ethanol production was demonstrated using SBP and a mixed enzymatic culture to solubilize pectin and cellulose and then the sugars were converted via fungal enzymes (Sutton and Doran-Peterson 2001). In this case, the Sugarbeet would become a dual purpose crop: sugar and energy (Panella 2010). A combined sugar-ethanol plant was studied while introducing beet co-products in various stages (Krajnc et al. 2007) including the immobilization of a prevalent yeast, Saccharomyces cerevisiae (Vucuroviç and Razmovski 2012), in a single-tank hydrolysis and fermentation process (Reziç et al. 2013). A study on the integration of the storage, hydrolysis and fermentation of sugar beet pulp has shown that ethanol production can be increased by 50 % over current methods (Zheng et al. 2012). Optimization of the biofuel production process is ongoing; for example, there have been recent developments into membrane ultafiltration for increasing the ethanol yields of Sugarbeet feedstocks (Kawa-Rygielska et al. 2013). Of course, environmental impacts must be considered; for example, water usage is one of the more important factors imported into any model (Gerbens-Leenes and Hoekstra 2012). To be viable as a biofuel, ethanol must have high net energy gain, be competitive in price, provide ecological benefits, and have the ability to be produced on a large scale. The design and introduction of new technologies (Santek et al. 2010) and the assessment of environmental advantages (Salazar-Ordonez et al. 2013) will increase the sustainability and profitability of the Sugarbeet. Regional efforts are aimed at designing ethanol plants with minimal waste generation using locally grown energy crops such as Sugarbeet (Vucuroviç et al. 2012).
Ethanol may be driving biofuel technology right now, but second generation biofuels are being studied such as hydrogen, methane, methanol and butanol. The wet storage difficulty could be leveraged by capturing the biogas produced during ensilage by anaerobic digestion (Panella 2010). Biogasification (anaerobic digestion) of the molasses co-product by mixed cultures of microorganisms produces methane and carbon dioxide which can be captured and used as a fuel for electricity generation (300 kWh/tonne raffinate) onsite (Polematidis et al. 2010). Work is continuing to produce methane gas as a biofuel using Sugarbeet residues (Tian et al. 2013). In addition, there are a variety of pre-treatments that improve the formation of biogases from SBP (Zieminski et al. 2012).
In addition to biochemical conversion, there are thermochemical conversion techniques for biomass. Torrefaction, pyrolysis, and gasification are three high-temperature processes to produce biofuels (Basu 2013). Pyrolysis (450 °C) and gasification converts solid biomass into liquid or gaseous fuel. Torrefaction (250–400 °C) is used to produce value-added products such as biochar and carbon fibers.
Carbon Materials for Removal of Contaminants
Renewable, sustainable energy research and concerns regarding the production of greenhouse gases has driven the need for the utilization of biomass in so-called carbon–neutral processes. Contamination of water sources by toxic substances is an ongoing environmental and health concern. Using agricultural materials have some advantages over conventional processes including low cost, regeneration of biosorbents, and potential recovery of heavy metals (Kolodynska et al. 2012; Sud et al. 2008). The binding capacity of SBP as an ion-exchanger can be improved (Dronnet et al. 1998) making it more valuable in the market.
Biochar is produced using high temperatures to burn biomass leaving only carbon (carbonization). Biochar has been shown to enhance soil fertility and water holding capability and sequesters carbon. Biochar also has potential as a low-cost absorbent as it shows high affinity for heavy metals. While any biomass can be carbonized, economics suggest that agricultural wastes would be more suitable. Using slow pyrolysis (600 °C), Sugarbeet residue was carbonized and was shown to capture phosphates (Yao et al. 2011). Using high temperature pyrolysis (500–700 °C), activated carbon was produced using ZnCl2-activated Sugarbeet bagasse (Demiral and Gunduzoglu 2010), and it was successful at removing nitrates from water. An alternate, low-energy technique converts lignocelluloses to carbon by sulfuric acid dehydration and has been shown to absorb heavy metals especially Cr(VI) (Altundogan et al. 2007). Sugar beet pulp was carbonized and used to decolor sugar syrups (Mudoga et al. 2008).
Cellulose
Cellulose has been isolated from SBP (Togrul and Arslan 2003), modified, and used to preserve fresh fruit during transportation and storage (Togrul and Arslan 2004). In paper products, treated and untreated SBP has been used to increase the internal bond strength of paper products mainly through the suspension of cellulose microfibrils in a gelatinous matrix of pectin (Dinand et al. 1999; Fiserova et al. 2007; Gigac et al. 2008).
Conclusions
Utilization of agricultural commodities in sustainable, economic, and ethical ways is essential in the competitive global marketplace.
The value-added coproducts of sugarbeet are summarized in Fig. 2. The complete utilization of the sugarbeet should be examined on the basis of a biobased economy in order to select the optimal parameters for the industry (Langeveld 2010; Liang et al. 2012). One must also consider the future impact of global climate change on the sugarbeet crop (Angulo et al. 2013; Reineke et al. 2013). Life cycle models will give researchers and leaders insight into the environmental, economic, political, and social value of sugarbeets.
References
Alamzan, O., L. Gonzalez, L. Galvez. 1998. The sugar cane: Its by-products and co-products. In Réduit, Mauritius: Food and Agricultural Research Council, xiii–xxv.
Altundogan, H.S., N. Bahar, B. Mujde, and F. Tumen. 2007. The use of sulphuric acid-carbonization products of sugar beet pulp in Cr(VI) removal. Journal of Hazardous Materials 144: 255–264.
Angulo, C., R. Rotter, R. Lock, A. Enders, S. Fronzek, and F. Ewert. 2013. Implication of crop model calibration strategies for assessing regional impacts of climate change in Europe. Agricultural and Forest Meteorology 170: 32–46.
Basu, P. 2013. Biomass gasification, pyrolysis and torrefaction, 2nd ed. Boston: Academic Press.
Biancardi, E., J.M. McGrath, L.W. Panella, R.T. Lewellen, and P. Stevanato. 2010. Sugar Beet. In Root and tuber crops, ed. J.E. Bradshaw, 173–219. New York: Springer.
Bonnin, E., H. Grange, L. Lesage-Meessen, M. Asther, and J.F. Thibault. 2000. Enzymic release of cellobiose from sugar beet pulp, and its use to favour vanillin production in Pycnoporus cinnabarinus from vanillic acid. Carbohydrate Polymers 41: 143–151.
Broughton, N.W., C.C. Dalton, G.C. Jones, and E.L. Williams. 1975. Adding value to sugar beet pulp. International Sugar Journal 97: 57–60.
Buchweitz, M., A. Nagel, R. Carle, and D.R. Kammerer. 2012. Characterisation of sugar beet pectin fractions providing enhanced stability of anthocyanin-based natural blue food colourants. Food Chemistry 132: 1971–1979.
Caqueret, V., S. Bostyn, B. Cagnon, and H. Fauduet. 2008. Purification of sugar beet vinasse—Adsorption of polyphenolic and dark colored compounds on different commercial activated carbons. Bioresource Technology 99: 5814–5821.
Castilho, L.R., D.A. Mitchell, and D.M.G. Freire. 2009. Production of polyhydroxyalkanoates (PHAs) from waste materials and by-products by submerged and solid-state fermentation. Bioresource Technology 100: 5996–6009.
Cattanach, A.W., A.G. Dexter, and E.S. Oplinger. 1991. Sugarbeets. In Alternative field crops manual, ed. Alternative Field Crops Manual. Madison: University of Wisconsin Cooperative Extension.
Chen, F., L. Liu, P.H. Cooke, K.B. Hicks, and J. Zhang. 2008. Performance enhancement of poly(lactic acid) and sugar beet pulp composites by improving interfacial adhesion and penetration. Industrial and Engineering Chemistry Research 47: 8667–8675.
Cho, S.S., and P. Samuel. 2009. Fiber ingredients: Food applications and health benefits. Florence: Taylor & Francis.
Cho, S.S. 2001. Handbook of dietary fiber. New York: CRC Press.
Concha-Olmos, J., and M.E. Zuniga-Hansen. 2012. Enzymatic depolymerization of sugar beet pulp: Production and characterization of pectin and pectic-oligosaccharides as a potential source for functional carbohydrates. Chemical Engineering Journal 192: 29–36.
Cooke, D.A., and R.K. Scott. 1993. The sugar beet crop: Science into practice. New York: Chapman & Hall.
Demiral, H., and G. Gunduzoglu. 2010. Removal of nitrate from aqueous solutions by activated carbon prepared from sugar beet bagasse. Bioresource Technology 101: 1675–1680.
Dhingra, D., M. Michael, R. Hradesh, and R.P. Patil. 2012. Dietary fibre in foods: a review. Journal of Food Science Technology 49: 255–266.
Dinand, E., H. Chanzy, and R.M. Vignon. 1999. Suspensions of cellulose microfibrils from sugar beet pulp. Food Hydrocolloids 13: 275–283.
Draycott, A.P. 2006. Sugar Beet. London: Blackwell (Wiley).
Dronnet, V.M., M.A.V. Axelos, C.M.G.C. Renard, and J.F. Thibault. 1998. Improvement of the binding capacity of metal cations by sugar-beet pulp. 1. Impact of cross-linking treatments on composition, hydration and binding properties. Carbohydrate Polymers 35: 29–37.
Dufresne, A., J.Y. Cavaille, and M.R. Vignon. 1997. Mechanical behavior of sheets prepared from sugar beet cellulose microfibrils. Journal of Applied Polymer Science 64: 1185–1194.
Fadel, J.G., E.J. Depeters, and A. Arosemena. 2000. Composition and digestibility of beet pulp with and without molasses and dried using three methods. Animal Feed Science and Technology 85: 121–129.
Fantozzi, P. 1990. What is the future of leaf-protein in human nutrition? Italian Journal of Food Science 11: 3–8.
Finkenstadt, V.L., C.K. Liu, P.H. Cooke, L.S. Liu, and J.L. Willett. 2008. Mechanical property characterization of plasticized sugar beet pulp and poly(lactic acid) green composites using acoustic emission and confocal microscopy. Journal of Polymers and the Environment 16: 19–26.
Fiserova, M., J. Gigac, and R. Butas. 2007. Influence of sugar beet pulp on bond strength and structure of paper. Wood Research 52: 59–74.
Fishman, M.L., H.K. Chau, D.R. Coffin, P.H. Cooke, P. Qi, M.P. Yadav, and J. Hotchkiss. 2011. Physico-chemical characterization of a cellulosic fraction from sugar beet pulp. Cellulose 18: 787–801.
Fishman, M.L., H.K. Chau, P.H. Cooke, M.P. Yadav, and A.T. Hotchkiss. 2009. Physico-chemical characterization of alkaline soluble polysaccharides from sugar beet pulp. Food Hydrocolloids 23: 1554–1562.
Fishman, M.L., H.K. Chau, P.X. Qi, J. Hotchkiss, and M.P. Yadav. 2013. Physico-chemical characterization of protein-associated polysaccharides extracted from sugar beet pulp. Carbohydrate Polymers 92: 2257–2266.
Gerbens-Leenes, W., and A.Y. Hoekstra. 2012. The water footprint of sweeteners and bio-ethanol. Environment International 40: 202–211.
Gigac, J., M. Fsíerova, and M. Rosenberg. 2008. Improvement of paper strength via surface application of sugar beet pectin. Chemical Papers 62: 509–515.
Goodban, A.F., and H.S. Owens. 1956. Isolation and properties of sugar beet araban. Journal of Sugar Beet Research IX: 129–132.
Guru, M., A.Y. Bilgesu, and V. Pamuk. 2001. Production of oxalic acid from sugar beet molasses by formed nitrogen oxides. Bioresource Technology 77: 81–86.
Hartnell, G.F., T. Hvelplund, and M.R. Weisbjerg. 2005. Nutrient digestibility in sheep fed diets containing Roundup Ready or conventional fodder beet, sugar beet, and beet pulp. Journal of Animal Science 83: 400–407.
Hood, E., Teoh, K., Devaiah, S., and Vicuna Requesens, D. 2013. Biomass crops for biofuels and bio-based products. In Sustainable food production (eds. P. Christou, R. Savin, B. Costa-Pierce, I. Misztal, and C. B. Whitelaw), 250–279. New York: Springer.
Iconomou, D., K. Kandylis, C. Israilides, and P. Nikokyris. 1998. Protein enhancement of sugar beet pulp by fermentation and estimation of protein degradability in the rumen of sheep. Small Ruminant Research 27: 55–61.
Jodidi, S.L. 1911. The Sugar beet and beet sugar. Chicago: Beet Sugar Gazette Company.
Jwanny, E.W., L. Montanari, and P. Fantozzi. 1993. Protein production for human use from Sugarbeet: Byproducts. Bioresource Technology 43: 67–70.
Kawa-Rygielska, J., W. Pietrzak, P. Regiec, and P. Stencel. 2013. Utilization of concentrate after membrane filtration of sugar beet thin juice for ethanol production. Bioresource Technology 133: 134–141.
Kelly, P. 1983. Sugar beet pulp—a review. Animal Feed Science and Technology 8: 1–18.
Kirby, A.R., A.J. MacDougall, and V.J. Morris. 2006. Sugar beet pectin-protein complexes. Food Biophysics 1: 51–56.
Kolodynska, D., R. Wnetrzak, J.J. Leahy, M.H.B. Hayes, W. Kwapinski, and Z. Hubicki. 2012. Kinetic and adsorptive characterization of biochar in metal ions removal. Chemical Engineering Journal 197: 295–305.
Krajnc, D., M. Mele, and P. Glavic. 2007. Improving the economic and environmental performances of the beet sugar industry in Slovenia: increasing fuel efficiency and using by-products for ethanol. Journal of Cleaner Production 15: 1240–1252.
Kroon, P. A., Faulds, C. B., Brezillon, C., and Williamson, G. 1996. Enzymic release of ferulic acid from sugar beet pulp using a specific esterase from Aspergillus niger. In Progress in Biotechnology Pectins and Pectinases Proceedings of an International Symposium (ed. J. Visser), 761–768. Amsterdam: Elsevier.
Kuhnel, S., H.A. Schols, and H. Gruppen. 2011. Aiming for the complete utilization of sugar-beet pulp: Examination of the effects of mild acid and hydrothermal pretreatment followed by enzymatic digestion. Biotechnology for Biofuels 4: 14.
Langeveld, H. 2010. The Biobased Economy: Biofuels, Materials and Chemicals in the Post-oil Era. London: Earthscan Ltd.
Leijdekkers, A.G.M., J.P.M. Bink, S. Geutjes, H.A. Schols, and H. Gruppen. 2013. Enzymatic saccharification of sugar beet pulp for the production of galacturonic acid and arabinose; a study on the impact of the formation of recalcitrant oligosaccharides. Bioresource Technology 128: 518–525.
Leitner, J., B. Hinterstoisser, M. Wastyn, J. Keckes, and W. Gindl. 2007. Sugar beet cellulose nanofibril-reinforced composites. Cellulose 14: 419–425.
Leontowicz, M., S. Gorinstein, E. Bartnikowska, H. Leontowicz, G. Kulasek, and S. Trakhtenberg. 2001. Sugar beet pulp and apple pomace dietary fibers improve lipid metabolism in rats fed cholesterol. Food Chemistry 72: 73–78.
Li, W., D.R. Coffin, T.Z. Jin, N. Latona, C.K. Liu, B. Liu, J. Zhang, and L. Liu. 2012. Biodegradable composites from polyester and sugar beet pulp with antimicrobial coating for food packaging. Journal of Applied Polymer Science 126: E361–E372.
Liang, S., M. Xu, and T. Zhang. 2012. Unintended consequences of bioethanol feedstock choice in China. Bioresource Technology 125: 312–317.
Liu, B., S. Bhaladhare, P. Zhan, L. Jiang, J. Zhang, L. Liu, and A.T. Hotchkiss. 2011a. Morphology and properties of thermoplastic sugar beet pulp and poly(butylene adipate-co-terepthalate) blends. Industrial and Engineering Chemistry Research 50: 13859–13865.
Liu, B., J. Zhang, L. Liu, and A.T. Hotchkiss. 2011b. Preparation and properties of water and glycerol-plasticized sugar beet pulp plastics. Journal of Polymers and the Environment 19: 559–567.
Liu, B., J. Zhang, L. Liu, and A.T. Hotchkiss. 2012. Utilization of pectin extracted sugar beet pulp for composite application. Journal of Biobased Materials and Bioenergy 6: 185–192.
Liu, L.S., V.L. Finkenstadt, C. Liu, T. Jin, M.L. Fishman, and K.B. Hicks. 2007. Preparation of poly (lactic acid) and pectin composite films intended for applications in antimicrobial packaging. Journal of Applied Polymer Science 106: 801–810.
Lv, C., Y. Wang, Lj Wang, D. Li, and B. Adhikari. 2013. Optimization of production yield and functional properties of pectin extracted from sugar beet pulp. Carbohydrate Polymers 95: 233–240.
Ma, S., Sj Yu, Xl Zheng, Xx Wang, Qd Bao, and Xm Guo. 2013. Extraction, characterization and spontaneous emulsifying properties of pectin from sugar beet pulp. Carbohydrate Polymers 98: 750–753.
Martinez, M., B. Gullon, H.A. Schols, J.L. Alonso, and J.C. Parajo. 2009. Assessment of the production of oligomeric compounds from sugar beet pulp. Industrial and Engineering Chemistry Research 48: 4681–4687.
McCready, R.M., J.B. Stark, and A.E. Goodban. 1965. Preparation of galactinol and myoinositol from sugar beet sirup by chromatography on a cation exchange resin. Journal of Sugar Beet Research 14: 127–132.
Micard, V., C.M.G.C. Renard, and J.F. Thibault. 1997. Influence of pretreatments on enzymic degradation of a cellulose-rich residue from sugar-beet pulp. LWT—Food Science and Technology 30: 284–291.
Michel, F., J.F. Thibault, J.L. Barry, and R. de Baynast. 1988. Preparation and characterisation of dietary fibre from sugar beet pulp. Journal of the Science of Food and Agriculture 42: 77–85.
Mohdaly, A.A., M.A. Sarhan, I. Smetanska, and A. Mahmoud. 2010. Antioxidant properties of various solvent extracts of potato peel, sugar beet pulp and sesame cake. Journal of the Science of Food and Agriculture 90: 218–226.
Moosavi, A., and A. Karbassi. 2010. Bioconversion of sugar-beet molasses into xanthan gum. Journal of Food Processing and Preservation 34: 316–322.
Mudoga, H.L., H. Yucel, and N.S. Kincal. 2008. Decolorization of sugar syrups using commercial and sugar beet pulp based activated carbons. Bioresource Technology 99: 3528–3533.
National Agricultural Statistics Service. 2013a. Crop production 2012 summary. Washington, DC: United States Department of Agriculture.
National Agricultural Statistics Service. 2013b. Crop value 2012 summary. Washington, DC: United States Department of Agriculture.
Nordic Sugar. 2012. GRAS notification for sugar beet fiber. http://www.fda.gov/Food/IngredientsPackagingLabeling/GRAS/NoticeInventory/ucm347789.htm. Accessed 20 Oct 2013.
Norsker, M., M. Jensen, and J. Adler-Nissen. 2000. Enzymatic gelation of sugar beet pectin in food products. Food Hydrocolloids 14: 237–243.
Oosterveld, A., G. Beldman, and A.G.J. Voragen. 2000. Oxidative cross-linking of pectic polysaccharides from sugar beet pulp. Carbohydrate Research 328: 199–207.
Panella, L. 2010. Sugar beet as an energy crop. Sugar Tech 12: 288–293.
Pavier, C., and A. Gandini. 2000. Oxypropylation of sugar beet pulp. 1. Optimisation of the reaction. Industrial Crops and Products 12: 1–8.
Polematidis, I., A. Koppar, and P. Pullammanappallil. 2010. Biogasification potential of desugarized molasses from Sugarbeet processing plants. Journal of Sugar Beet Research 47: 89–104.
Ralet, M.-C., Guillon, F., Renard, C., and Thibault, J. F. 2009. Sugar beet fiber: Production, characteristics, food applications and physiological benefits. In Fiber ingredients: Food applications and health Benefits (eds. S. S. Cho and P. Samuel). Florence: Taylor & Francis.
Reineke, H., N. Stockfisch, and B. Marlander. 2013. Analysing the energy balances of sugar beet cultivation in commercial farms in Germany. European Journal of Agronomy 45: 27–38.
Reziç, T., D. Oros, I. Markoviç, D. Kracher, R. Ludwig, and B. Santek. 2013. Integrated hydrolysation and fermentation of the sugar beet pulp to bioethanol. Journal of Microbiology and Biotechnology 23: 1244–1252.
Rouilly, A., C. Geneau-Sbarta, and L. Rigal. 2009. Thermo-mechanical processing of sugar beet pulp. III. Study of extruded films improvement with various plasticizers and cross-linkers. Bioresource Technology 100: 3076–3081.
Rouilly, A., J. Jorda, and L. Rigal. 2006. Thermo-mechanical processing of sugar beet pulp. I. Twin-screw extrusion process. Carbohydrate Polymers 66: 81–87.
Salazar-Ordonez, M., P.P. Perez-Hernandez, and J.M. Martin-Lozano. 2013. Sugar beet for bioethanol production: An approach based on environmental agricultural outputs. Energy Policy 55: 662–668.
Santek, B., G. Gwehenberger, M.I. Santek, M. Narodoslawsky, and P. Horvat. 2010. Evaluation of energy demand and the sustainability of different bioethanol production processes from sugar beet. Resources, Conservation and Recycling 54: 872–877.
Soto, M.L., A. Moure, H. Dominguez, and J.C. Paraja. 2011. Recovery, concentration and purification of phenolic compounds by adsorption: A review. Journal of Food Engineering 105: 1–27.
Steg, A., and J.M. Van Der Meer. 1985. Differences in chemical composition and digestibility of beet and cane molasses. Animal Feed Science and Technology 13: 83–91.
Stevanato, P., and L.W. Panella. 2013. History of Sugarbeets, 17–21. Idaho Falls: Sugar Producer Magazine.
Sud, D., G. Mahajan, and M.P. Kaur. 2008. Agricultural waste material as potential adsorbent for sequestering heavy metal ions from aqueous solutions—A review. Bioresource Technology 99: 6017–6027.
Sutton, M.D., and J.B. Doran-Peterson. 2001. Fermentation of sugarbeet pulp for ethanol production using bioengineered Klebsiella oxytoca strain P2. Journal of Sugar Beet Research 38: 19–34.
Tamimi, M.A., R.J. Palframan, J.M. Cooper, G.R. Gibson, and R.A. Rastall. 2006. In vitro fermentation of sugar beet arabinan and arabino-oligosaccharides by the human gut microflora. Journal of Applied Microbiology 100: 407–414.
Teimouri Yansari, A. 2013 Physically effectiveness of beet pulp-based diets in dairy cows as assessed by responses of feed intake, digestibility, chewing activity and milk production. Journal of Animal Physiology and Animal Nutrition (in press).
The Sugar Association. 2013. Refining and processing sugar. http://www.sugar.org/images/docs/refining-and-processing-sugar.pdf Accessed 20 Aug 2013.
Tian, Z., D. Chauliac, and P. Pullammanappallil. 2013. Comparison of non-agitated and agitated batch, thermophilic anaerobic digestion of Sugarbeet tailings. Bioresource Technology 129: 411–420.
Togrul, H., and N. Arslan. 2003. Flow properties of sugar beet pulp cellulose and intrinsic viscosity—molecular weight relationship. Carbohydrate Polymers 54: 63–71.
Togrul, H., and N. Arslan. 2004. Extending shelf-life of peach and pear by using CMC from sugar beet pulp cellulose as a hydrophilic polymer in emulsions. Food Hydrocolloids 18: 215–226.
Van Dyk, J.S., R. Gama, D. Morrison, S. Swart, and B.I. Pletschke. 2013. Food processing waste: Problems, current management and prospects for utilisation of the lignocellulose component through enzyme synergistic degradation. Renewable and Sustainable Energy Reviews 26: 521–531.
Vucuroviç, D.G., S.N. Dodiç, S.D. Popov, J.M. Dodiç, and J.A. Grahovac. 2012. Process model and economic analysis of ethanol production from sugar beet raw juice as part of the cleaner production concept. Bioresource Technology 104: 367–372.
Vucuroviç, V.M., and R.N. Razmovski. 2012. Sugar beet pulp as support for Saccharomyces cerivisiae immobilization in bioethanol production. Industrial Crops and Products 39: 128–134.
Wang, B., R.R. Sharma-Shivappa, J.W. Olson, and S.A. Khan. 2013. Production of polyhydroxybutyrate (PHB) by Alcaligenes latus using Sugarbeet juice. Industrial Crops and Products 43: 802–811.
Wei, Y.A., and Y.R. Li. 2006. Status and trends of sugar industry in China. Sugar Tech 8: 10–15.
Yao, Y., B. Gao, M. Inyang, A.R. Zimmerman, X. Cao, P. Pullammanappallil, and L. Yang. 2011. Biochar derived from anaerobically digested sugar beet tailings: Characterization and phosphate removal potential. Bioresource Technology 102: 6273–6278.
Zheng, Y., C. Yu, Y.S. Cheng, C. Lee, C.W. Simmons, T.M. Dooley, R. Zhang, B.M. Jenkins, and J.S. VanderGheynst. 2012. Integrating sugar beet pulp storage, hydrolysis and fermentation for fuel ethanol production. Applied Energy 93: 168–175.
Zieminski, K., I. Romanowska, and M. Kowalska. 2012. Enzymatic pretreatment of lignocellulosic wastes to improve biogas production. Waste Management 32: 1131–1137.
Zijlstra, R. T. and Beltranena, E. 2013. Alternative feedstuffs in swine diets. In Sustainable swine nutrition, 229–253. Oxford: Blackwell.
Acknowledgments
Ms. Sara Lee, Special Collections Librarian, at the National Agricultural Library (Beltsville, MD), was invaluable in tracking down historical documents from the National Archives. The author would like to thank Ms. Marcia Wood, Public Affairs Specialist, on the ARS Information Staff (Beltsville, MD), for the inspiration to write this review.
Disclosure
Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Finkenstadt, V.L. A Review on the Complete Utilization of the Sugarbeet. Sugar Tech 16, 339–346 (2014). https://doi.org/10.1007/s12355-013-0285-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12355-013-0285-y