Abstract
Background
Respiratory motion due to breathing during cardiac positron emission tomography (PET) results in spatial blurring and erroneous tracer quantification. Respiratory gating might represent a solution by dividing the PET coincidence dataset into smaller respiratory phase subsets. The aim of our study was to compare the resulting imaging quality by the use of a time-based respiratory gating system in two groups administered either adenosine or dipyridamole as the pharmacological stress agent.
Methods and Results
Forty-eight patients were randomized to adenosine or dipyridamole cardiac stress 82RB-PET. Respiratory rates and depths were measured by a respiratory gating system in addition to registering actual respiratory rates. Patients undergoing adenosine stress showed a decrease in measured respiratory rate from initial to later scan phase measurements [12.4 (±5.7) vs 5.6 (±4.7) min−1, P < .001] and tended to have a lower frequency of successful respiratory gating compared to dipyridamole (47% vs 71%, P = .12). As a result, imaging quality was superior in the dipyridamole group compared to adenosine.
Conclusions
If respiratory gating is considered for use in cardiac PET, a dipyridamole stress protocol is recommended as it, compared to adenosine, causes a more uniform respiration and results in a higher frequency of successful respiratory gating and thereby superior imaging quality.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In cardiac perfusion positron emission tomography (PET), static and dynamic images are obtained at rest and during pharmacological stress. While motion of the heart due to respiration can be up to 18 mm in mainly the caudocranial direction and due to the fact that patients are unable to hold their breath during the entire PET acquisition, respiratory motion will result in spatial blurring and motion artifacts, which ultimately may result in incorrect data quantification.1,2 A reduction in motion-induced artifacts in PET images is therefore desirable.3-5 Respiratory gating could represent a solution to avoid motion blurring by dividing the PET coincidence dataset into N respiratory bins, where N represents the desired number of respiratory phases.6 Crucial to this technique is the recording of respiratory motion as a function of scan time.
Two different respiratory gating methods have been suggested: a time-based gating method and an amplitude-based gating method.7,8 In the present study, a time-based gating method was used. In brief, the time-based respiratory gating method divides the respiratory signal into N time-equal bins (8 bins in our study), determined by either the inspiratory or the expiratory peak. The amplitude-based respiratory gating method divides the respiratory signal in each respiratory cycle with reference to the magnitude of the respiratory signal. The time-based binning is based on assumptions of a steady, nonchanging respiratory rate and depth throughout the PET acquisition. This is often the case during resting conditions, but not during stress image acquisition since pharmacologic stress agents alter the respiration pattern. The most commonly used pharmacological stress agents for cardiac PET are adenosine and dipyridamole. Pharmacodynamics and pharmacokinetics including half-lives differ between the two agents, which is why they, according to recent guidelines, are administered using different protocols.8-12 Adenosine is administered as a continuous intravenous infusion during the stress scan simultaneously with the administration of the PET tracer, whereas dipyridamole is administered as a bolus injection prior to the PET scan. The different stress protocols could affect breathing differently and thus influence the likelihood of success of the use of a respiratory gating system.
The aim of our study was to compare the resulting imaging quality by the use of a time-based respiratory gating system in two groups administered either adenosine or dipyridamole as the pharmacological stress agent.
Methods
Study Population
The study group consisted of forty-eight patients referred for cardiac rest and stress Rubidium-82 (82Rb) PET scanning at our department on the suspicion of ischemic heart disease. The only inclusion criteria were suspected ischemic heart disease as the indication to perform 82Rb-PET and a successful calibration of the respiratory belt prior to the PET acquisition.
PET/CT Scan and Respiratory Gating System
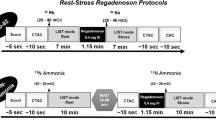
The patients underwent 82Rb-PET computed tomography (CT) using the same 64-slice PET/CT scanner (Siemens mCT, Siemens, Knoxville, USA). Two dynamic PET perfusion scans were obtained, initially under resting conditions and subsequently under pharmacologically induced stress (please refer to stress protocols below). Each PET acquisition took 7 minutes and was performed under the administration of 1000-1200 MBq 82Rb.
A respiratory gating system (Anzai 733v – Anzai Medical Co., Ltd., Tokyo, Japan), consisting of a strain gage strapped around the chest of the patients, was used to continuously monitor respiratory depths/amplitude and rate during scan. Respiratory rate and amplitude were analyzed within the first 60 seconds of the scan, and in the 6th minute of the scan (300-360 seconds) after PET tracer infusion start. Both actual/observed respiratory rates and measured/registered respiratory rates were registered; actual/observed respiratory rates were the actual respiratory rate derived by manually analyzing the respiratory curve and measured/registered respiratory rates were derived from automatic computerized respiratory rate registration. This allowed the detection of changes in respiration during the two different stress protocols as well as a comparison of the concordance between the actual and measured respiratory rates.
A standardization of the respiratory cycles in the Anzai program (Anzai Medical Co., Ltd., Tokyo, Japan) was necessary to obtain homogenous respiratory depths for all patients due to the individual calibration settings of the system. These standardizations were performed in a custom-made program (MatLab R2013b, Mathworks, USA).
The program used the maximum amplitude given by the strain gage (10 mA) and the maximum respiratory depth (150 arbitrary units) to normalize the respiratory cycles. The normalization was computed using the following equation:
where PatientAmplitude is the individual patient amplitude measured by the gage and Average depth is the average depth of breath cycles measured in the program within the analyzed time frames.
Successful respiratory gating was defined based upon image quality under the discretion of the observer as a respiratory gating of ≥60%.
82Rubidium and Stress Protocols
82Rb myocardial perfusion PET-CT was performed as a 7-minute dynamic PET myocardial perfusion rest scan under the administration of 1000-1200 MBq 82Rb followed by a 7-minute dynamic stress PET scan. PET images were analyzed using Cedars-Sinai Cardiac Suite (Cedars-Sinai Medical Center, Los Angeles, USA) for Syngo.Via (Siemens, Knoxville, USA).
Two different stress protocols were used: one using adenosine and the other using dipyridamole as the pharmacologic stress agent in accordance with the existing guidelines10.
Adenosine was given as a continuous infusion of 840 µg/kg initiated 2.5 minutes before scan start and infused over 6 minutes. Dipyridamole was given as a slowly injected bolus of 560 µg/kg 6 minutes before scan start.
Rate Pressure Product
Blood pressure and heart rate were measured at rest and during stress. Rate pressure products (RPPs) were calculated at rest and stress as the product of systolic blood pressure and heart rate at rest and stress, respectively. Hemodynamic responses to adenosine and dipyridamole were evaluated by comparing differences between RPP at rest and at stress between groups.
Statistical Analysis
Categorical variables were expressed as percentages and continuous variables were reported as means and standard deviations (±SD) or standard error of the mean (±SEM). Differences in baseline characteristics between adenosine and dipyridamole groups were assessed using Chi-square test for discrete variables and Student’s t test for continuous variables. Differences between the initial and late respiratory rates and amplitudes as well as RPP at rest and stress were assessed with a paired t test. Frequencies of successful respiratory gating in adenosine and dipyridamole groups were tested using Chi-square test. A two-tailed P value < 0.05 was considered statistically significant. Statistical analyses were performed using SAS® for Windows, version 9.2 (SAS institute, Cary, North Carolina, USA).
Results
Baseline Characteristics
Baseline characteristics are given in Table 1. The dipyridamole group consisted of nonsmoking women without a family history of cardiovascular disease and a mean age of 64 years. The adenosine group consisted primarily of men (86%) at a mean age of 57 years, of whom 39% were current smokers and 48% had a family history of cardiovascular disease. There were no differences in the frequencies of hypertension, diabetes, and cholesterol level between the two groups.
Respiratory Gating Data
Examples of typical respiratory gating schemes for patients undergoing pharmacological stress with adenosine or dipyridamole are shown in Figure 1. Respiratory rates and amplitudes were fairly constant during rest in both groups and under dipyridamole stress. Patients, given adenosine, on the other hand showed a decrease in respiratory rate during the late phase of stress compared to the initial phase. Furthermore, the baseline of the respiratory amplitude tended to drift thus exceeding the minimum requirement of 50 units in order to register breaths under stress, which resulted in a loss of trigger signals (Figure 1).
Respiratory response of two different patients undergoing dipyridamole to the left (rest study: A + C; stress study: B + D) and adenosine stress to the right (rest study: E + G; stress study: F + H). Trigger signals for registered breath cycles are shown with red vertical lines at the time of the respiratory peak (blue curves). Clinical characteristics of the two patients: Dipyridamole (A-D)—a 68-year-old female without known hypertension, diabetes, or family history of ischemic heart disease; smoking status unknown. Adenosine (E-H)—a 59-year-old male known with hypertension and a family history of ischemic heart disease, no diabetes, and previous smoker
Respiratory Amplitudes
Respiratory amplitudes in dipyridamole and adenosine groups are given in Table 2. There were no differences between the initial and late respiratory amplitudes in either the adenosine or the dipyridamole group under rest or stress. On the other hand, respiratory amplitudes were significantly lower in the adenosine group under stress in both the initial and late phases compared to the dipyridamole group [0.10 vs 0.21 (P < .05) and 0.08 vs 0.20 (P < .01), respectively].
Respiratory Rates
The observed and registered respiratory rates in the dipyridamole and adenosine groups are shown in Table 3 and Figure 2. In the dipyridamole group, no differences between the initial and late phases were noted in either the observed or the registered respiratory rates under rest or stress. Similarly, no differences between the initial and late phases were found in either the observed or the registered respiratory rates in the adenosine group under rest. On the other hand, both the observed and measured respiratory rates were significantly lower in the later phase of adenosine stress compared to the initial phase [observed: 11.1 vs 14.2 min−1 (P < .01) and measured: 5.6 vs 12.4 min−1 (P < .001)]. Additionally, no differences in the observed respiratory rates between the dipyridamole and adenosine groups were found at rest (data not shown). Under stress, the observed respiratory rates tended to be higher initially and lower in the late phase in the adenosine group compared to the dipyridamole group, although being nonsignificant [initial phase: 14.2 vs 12.9 min−1 (P = .29) and late phase: 11.1 vs 13.2 min−1 (P = .15)]. Furthermore, the measured respiratory rates were slightly but significantly lower than the observed respiratory rates in both groups under rest as well as stress, most notable in the late phase of adenosine stress [5.6 vs 11.1 min−1 (P < .0001) for the late phase of adenosine stress].
Frequencies of successful respiratory gating tended to be higher in the dipyridamole group compared to the adenosine group (71% vs 47%, P = .12) (Table 4). Furthermore, lower resting respiratory rates were associated with successful respiratory gating.
The effect of successful versus failed respiratory gating in image quality is illustrated in Figure 3.
Hemodynamic Response
Hemodynamic responses in dipyridamole and adenosine groups are shown in Table 5. Under stress, RPP was found to be significantly higher than that at rest in both the adenosine and dipyridamole groups [8925 vs 10,501 mmHg/minute (P < .01) and 8601 vs 10,246 mmHg/minute (P < .01), respectively]. There were no differences in RPP at rest or stress nor were there any differences in RPP at rest or stress between the groups (data not shown).
Discussion
The major finding of our study was that adenosine and dipyridamole influenced the breathing pattern differently which could potentially influence the choice of stress agent if respiratory gating is intended to be used as part of a cardiac PET protocol. While dipyridamole did not change the respiratory rate during the cardiac stress PET acquisition, adenosine induced a nonsignificant increase in respiratory rate in the initial phase of the study and a significant decrease in the later phase. Registered respiratory rates were slightly lower than the actual respiratory rates in both groups during both rest and stress.
There were no differences in the respiratory amplitude between the initial and late phases at rest in either the dipyridamole or the adenosine group. This led to the conclusion that the respiratory belt was able to obtain the correct gating sequences and that calibration of the respiratory belt was equal in both groups. Under stress, respiratory amplitudes were constant in both groups, but there seemed to be a drift of respiratory baseline in the adenosine group leading to a loss of trigger signals.
The drift in respiratory baseline in conjunction with the change in respiratory rate from the initial to the late phase under adenosine stress resulted in a loss of respiratory trigger signals and thus a difference between the registered and actual respiratory rates. Dipyridamole, on the other hand, not influencing the breathing pattern, showed a better correlation between the registered and actual respiratory rates.
There were no difference in RPP increase from rest to stress between the dipyridamole and adenosine group, indicating an equal hemodynamic response in the two groups.
The changes in respiratory cycles are anticipated to be a result of the pharmacokinetics and pharmacodynamics of the two stress agents, with adenosine having a half-life of about 10 seconds and dipyridamole having 62 minutes. Furthermore, the initial increase of respiratory rate due to adenosine is well known, while the drop in respiratory rate afterwards might be a compensatory mechanism.11,12 Additionally, the administration of adenosine and dipyridamole is initiated at different time points with regard to the stress examination, potentially contributing to the observed breath changes.
In the present study, we used the Anzai AZ-733V belt, but other commercially and noncommercially available systems have been suggested. Many of these methods rely on optical triggering signals, by use of either infrared tracking or normal video-cameras.3,13-15 Others systems are based on using respiratory elastic straps for clinical SPECT/CT analysis, which operates in the same way as the respiratory belt used in this study.16 The systems using the optical monitoring of the respiratory motions all requires both a correct positioning of the fiducial markers and that the patient is positioned correct in order to obtain the triggering signals. The fiducial markers are often created as a disk, which are used to track the respiratory cycle. These systems often require a standard breath pattern in order to obtain the triggering signals, which can only be achieved by training the patients. Training sessions like these are not feasible in cardiac PET centers with a high throughput of patients. Other methods for respiratory gating include a pressure-based air bag system working like a spirometer,17 electrical impedance plethysmography,18 a thermal sensor in a mask or a nasal cannula,6 a respiratory flowmeter in a mouthpiece,19 a displacement transducer in a belt around the upper abdomen,20,21 the Polaris system15, and an electrocardiogram-derived respiratory motion tracking system.5
A time-based gating was chosen for this study due to our current scanner setup and the post-processing of the images. Time-based gating provides a static image quality for each gate, due to the definition of the binning procedure.7 The amplitude-based binning setup has different noise characteristics as the majority of respiratory cycles would have been assigned to a single bin due to the change of perceived respiratory depth obtained by the respiratory belt when using adenosine. Due to these characteristics, we do not recommend to use amplitude-based binning unless a full motion compensation is performed. This is, however, not possible in our current scanner setup where only gated images can be provided.
The newer data-driven methods include list-mode-based motion detection approaches, which use inhomogenous sensitivity profile of the scanner systems to detect the respiratory cycle directly from the count rate.3,22,23 This method has proven efficient and could potentially eliminate the use of external markers and the accompanied potential loss of trigger signals due to erroneously calibrated systems.3,22,23 Furthermore, data-driven methods have the potential to use amplitude-based gating which is superior to time-based gating.7
Quantitative gated PET imaging also requires gated attenuation CT images for an accurate estimation of radiotracer uptake but at the sacrifice of a more complex CT acquisition protocol and increased radiation exposure. Still, nongated CT images can be used for attenuation correction of the PET emission data and are far superior to PET transmission scans and reduce scan times as well as provide noiseless attenuation correction.
Existing respiratory gating systems require a uniform breath pattern throughout the examination in order to obtain correct gating. In this study, dipyridamole showed the most constant respiratory rate throughout the stress examination and the best correspondence between the observed and registered respiratory rates compared to adenosine. Therefore, a dipyridamole stress protocol is recommended if respiratory gating is intended to be used in cardiac PET.
New Knowledge Gained
This is to our knowledge the first time the administration of adenosine and dipyridamole in myocardial perfusion PET has been compared with regard to the benefits of the use of a respiratory gating system. A dipyridamole stress protocol is superior to an adenosine one if respiratory gating is considered in myocardial perfusion PET.
Limitations
A potential limitation is the heterogeneity of the two groups. Most striking perhaps are the differences in gender; the adenosine group constituted mainly of men and the dipyridamole group is exclusively represented by women. However, we have no reason to believe that these differences in groups should have an impact on the conclusion of our manuscript. In respiratory gating, we are measuring the respiratory rate and no differences have ever been reported in respiratory rates regarding gender.24 Respiratory depth/tidal volume is correlated to body surface area and while women are smaller than men, their tidal volume due to lower body surface area is lower compared to men. Thus, if a difference in respiratory gating would exist, expectedly the drift of the respiratory baseline amplitude would more often exceed the minimum for respiratory registration, and erroneous respiratory gating, in women; actually opposite to the findings of this manuscript. Another concern could be that the respiratory responses to adenosine and dipyridamole could be different in men compared to women. While the response in myocardial blood flow during adenosine and dipyridamole stress is exclusively dependent on body weight, and not gender, stress protocols never take gender into account. Accordingly, we have no reason to believe that the respiratory responses to adenosine and dipyridamole should be different in men compared to women.
Another limitation is the smaller size of the dipyridamole group.
Conclusion
If respiratory gating is considered for use in cardiac PET, a dipyridamole stress protocol is recommended as it, compared to adenosine, causes a more uniform respiration and results in a higher frequency of successful respiratory gating and thereby superior imaging quality.
Abbreviations
- CT:
-
Computed tomography
- PET:
-
Positron emission tomography
- 82Rb:
-
Rubidium-82
- RPP:
-
Rate pressure product
References
Livieratos L, Rajappan K, Stegger L, Schafers K, Bailey DL, Camici PG. Respiratory gating of cardiac PET data in list-mode acquisition. Eur J Nucl Med Mol Imaging 2006;33:584-8.
Wang Y, Riederer SJ, Ehman RL. Respiratory motion of the heart: Kinematics and the implications for the spatial resolution in coronary imaging. Magn Reson Med 1995;33:713-9.
Buther F, Dawood M, Stegger L, Wubbeling F, Schafers M, Schober O, et al. List mode-driven cardiac and respiratory gating in PET. J Nucl Med 2009;50:674-81.
Schafers KP, Stegger L. Combined imaging of molecular function and morphology with PET/CT and SPECT/CT: Image fusion and motion correction. Basic Res Cardiol 2008;103:191-9.
Klein R. Editorial: Derivation of respiratory gating signals from ECG signals. J Nucl Cardiol 2016;23:84-6.
Boucher L, Rodrigue S, Lecomte R, Benard F. Respiratory gating for 3-dimensional PET of the thorax: Feasibility and initial results. J Nucl Med 2004;45:214-9.
Dawood M, Buther F, Lang N, Schober O, Schafers KP. Respiratory gating in positron emission tomography: A quantitative comparison of different gating schemes. Med Phys 2007;34:3067-76.
Dawood M, Buther F, Stegger L, Jiang X, Schober O, Schafers M, et al. Optimal number of respiratory gates in positron emission tomography: A cardiac patient study. Med Phys 2009;36:1775-84.
Lerman BB, Belardinelli L. Cardiac electrophysiology of adenosine. Basic and clinical concepts. Circulation 1991;83:1499-509.
Henzlova MJ, Cerqueira MD, Mahmarian JJ, Yao SS. Stress protocols and tracers. J Nucl Cardiol 2006;13:e80-90.
Jonzon B, Sylven C, Beermann B, Brandt R. Adenosine receptor mediated stimulation of ventilation in man. Eur J Clin Invest 1989;19:65-71.
Mahony C, Wolfram KM, Cocchetto DM, Bjornsson TD. Dipyridamol kinetics. Clin Pharmacol Ther 1982;31:330-8.
Nehmeh SA, Erdi YE, Ling CC, Rosenzweig KE, Schoder H, Larson SM, et al. Effect of respiratory gating on quantifying PET images of lung cancer. J Nucl Med 2002;43:876-81.
Nehmeh SA, Haj-Ali AA, Qing C, Stearns C, Kalaigian H, Kohlmyer S, et al. A novel respiratory tracking system for smart-gated PET acquisition. Med Phys 2011;38:531-8.
Beach RD, Pretorius PH, Boening G, Bruyant PP, Feng B, Fulton RR, et al. Feasibility of stereo-infrared tracking to monitor patient motion during cardiac SPECT imaging. IEEE Trans Nucl Sci 2004;51:2693-8.
Kovalski G, Israel O, Keidar Z, Frenkel A, Sachs J, Azhari H. Correction of heart motion due to respiration in clinical myocardial perfusion SPECT scans using respiratory gating. J Nucl Med 2007;48:630-6.
Ohara K, Okumura T, Akisada M, Inada T, Mori T, Yokota H, et al. Irradiation synchronized with respiration gate. Int J Radiat Oncol Biol Phys 1989;17:853-7.
Cho K, Kumiata S, Okada S, Kumazaki T. Development of respiratory gated myocardial SPECT system. J Nucl Cardiol 1999;6:20-8.
Kim BH, Ishida Y, Tsuneoka Y, Matsubara N, Hiraoka T, Takeda H, et al. Effects of spontaneous respiration on right and left ventricular function: Evaluation by respiratory and ECG gated radionuclide ventriculography. J Nucl Med 1987;28:173-7.
Ehman RL, McNamara MT, Pallack M, Hricak H, Higgins CB. Magnetic resonance imaging with respiratory gating: Techniques and advantages. AJR Am J Roentgenol 1984;143:1175-82.
Tada T, Hosono M, Fujioka T, Fukuda H, Nakajima T, Inoue Y. Monitoring of respiratory movement of the diaphragm for gated radiotherapy. Radiat Med 2005;23:10-3.
Bundschuh RA, Martinez-Moeller A, Essler M, Martinez MJ, Nekolla SG, Ziegler SI, et al. Postacquisition detection of tumor motion in the lung and upper abdomen using list-mode PET data: A feasibility study. J Nucl Med 2007;48:758-63.
He J, O’Keefe GJ, Gong SJ, Jones G, Saunder T, Scott AM, et al. A novel method for respiratory motion gated with geometric sensitivity of the scanner in 3D PET. IEEE Trans Nucl Sci 2008;55:2557-65.
Barrett KE, Barman SM, Boitano S, Brooks H. Ganong’s review of medical physiology. 24th ed. New York: McGraw-Hill Medical; 2012. p. 619.
Disclosures
MLL, TR, TEC, AK, and PH have nothing to disclose.
Author information
Authors and Affiliations
Corresponding author
Additional information
See related editorial, doi:10.1007/s12350-016-0666-1.
The authors of this article have provided a PowerPoint file, available for download at SpringerLink, which summarises the contents of the paper and is free for re-use at meetings and presentations. Search for the article DOI on SpringerLink.com.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lassen, M.L., Rasmussen, T., Christensen, T.E. et al. Respiratory gating in cardiac PET: Effects of adenosine and dipyridamole. J. Nucl. Cardiol. 24, 1941–1949 (2017). https://doi.org/10.1007/s12350-016-0631-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12350-016-0631-z