Abstract
Essential tremor (ET) might be a family of diseases unified by the presence of kinetic tremor, but also showing etiological, pathological, and clinical heterogeneity. In this review, we will describe the most significant clinical evidence, which suggests that ET is linked to the cerebellum. Data for this review were identified by searching PUBMED (January 1966 to May 2015) crossing the terms “essential tremor” (ET) and “cerebellum,” which yielded 201 entries, 11 of which included the term “cerebellum” in the article title. This was supplemented by articles in the author’s files that pertained to this topic. The wide spectrum of clinical features of ET that suggest that it originates as a cerebellar or cerebellar outflow problem include the presence of intentional tremor, gait and balance abnormalities, subtle features of dysarthria, and oculomotor abnormalities, as well as deficits in eye-hand coordination, motor learning deficits, incoordination during spiral drawing task, abnormalities in motor timing and visual reaction time, impairment of social abilities, improvement in tremor after cerebellar stroke, efficacy of deep brain stimulation (which blocks cerebellar outflow), and cognitive dysfunction. It is unlikely, however, that cerebellar dysfunction, per se, fully explains ET-associated dementia, because the cognitive deficits that have been described in patients with cerebellar lesions are generally mild. Overall, a variety of clinical findings suggest that in at least a sizable proportion of patients with ET, there is an underlying abnormality of the cerebellum and/or its pathways.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Essential tremor (ET) is the most common of the 20 known tremor disorders and is also one of the most common neurological disorders among adults [1–3]. ET is a widespread condition, affecting people of all races and cultures, from the remote Araihazar, Bangladesh, to the urban area of Madrid, Spain (NEDICES study) [1, 4]. Its most recognizable clinical feature is a 4–12-Hz kinetic tremor of the arms (i.e., tremor during voluntary movement), which often is later accompanied by head and voice tremors [5–8]. The traditional view of ET as a monosymptomatic condition is being replaced, as a spectrum of clinical features is increasingly being documented. These features comprise both motor and non-motor elements, including gait abnormalities [9], parkinsonism [10], cognitive impairment [11–13], dementia [14, 15], personality disturbances [16], depressive symptoms [17], sensory abnormalities (e.g., mild olfactory dysfunction and hearing impairment) [18–20], voice disturbances [21], and sleep disorders [22]. The emerging view is that ET might be a family of disease unified by the presence of kinetic tremor, but also showing etiological, pathological, and clinical heterogeneity [5–8].
The idea that ET is linked to the cerebellum is not novel. In the landmark paper by Critchley and Greenfield, published in 1949, the authors wrote: “Although anatomical proof is as yet lacking, there are at least a number of clinical points to make question whether ‘essential tremor’ may not, at times any rate, represent an incomplete or a premature variant of one of the cerebellar atrophies” [23]. Although not further elaborated on by those authors, these clinical characteristics include its insidious onset, association with advanced aging (i.e., both prevalence and incidence rates increase with age), gradually progressive nature, and the presence of “cerebellar” features (e.g., intention tremor and ataxia) on examination [23]. Indeed, these are many of the clinical features typical of neurodegenerative diseases of the cerebellum [24].
In this review, we will describe the most significant clinical evidence, which suggests that ET is linked to the cerebellum.
Methods
Data for this review were identified by searching PUBMED (January 1966 to May 2015) crossing the terms “essential tremor” (ET) and “cerebellum,” which yielded 201 entries, 11 of which included the term “cerebellum” in the article title. This was supplemented by articles in the author’s files that pertained to this topic. Of note is that a recent review of the 100 most cited papers on ET revealed that only one was related to the cerebellum [25, 26].
Clinical Evidence That Links Essential Tremor to the Cerebellum
Intention Tremor
Tremor is an involuntary, rhythmic, muscle movement involving oscillations of one or more parts of the body, resulting from repeated contraction and relaxation of opposing muscle groups [5–7]. Tremor can be classified according to the consensus statement of the Movement Disorder Society [27]. Tremor at rest is separated from action tremor, which is produced by voluntary muscle contraction. Action tremor includes postural, isometric, and kinetic tremors, and the latter form including intention tremor [27].
Canonically, the dominant clinical feature of ET was considered to be a “postural tremor not made strikingly worse during action” (i.e., tremor while the arms are outstretched against gravity) [28]. However, growing evidence point to a kinetic tremor (i.e., tremor during voluntary motion), affecting mainly the arms and hands, which often is later accompanied by head and voice tremors, as the primary type of tremor in ET [29–31]. Kinetic arm tremor occurs during any voluntary movement, including pouring water into a cup, drinking from a cup, eating with utensils, or drawing Archimedes’ spirals [5–8]. The kinetic tremor in ET often has an intentional component; for instance, during visually guided movements, such as the finger-to-nose maneuver, the amplitude of the tremor increases as the target is approached [5–8]. This characteristic provides further support for an abnormality of cerebellar function in ET. Kinetic tremor leads to difficulties with eating, drinking, writing, dressing, and various other activities of daily living [5–8]. The intentional component of kinetic tremor in ET is usually more evident in more advanced ET cases and may occur in approximately one in three ET cases [32, 33]. In a quantitative analysis of a grasping movement, involving two subgroups of ET patients (one with predominant postural tremor and another with intention tremor), and two control groups (one with cerebellar disease and another of normal subjects), the amplitude measurements of intention tremor were clearly abnormal and of comparable magnitude for ET patients with intention tremor and those with cerebellar disease [32]. The occurrence of intention tremor therefore suggests abnormalities of cerebellar functions in ET [32].
Gait and Balance Abnormalities
Gait and balance abnormalities in ET patients have been described in a number of studies; these abnormalities include slower gait speed [34–36], impaired dynamic balance (indicated by greater number missteps) [36–39], gait asymmetry [36], and balance impairment during tandem walk [36]. Unlike controls, significant correlations between older age and quantitative gait measures have been described [36, 39, 40], suggesting that the effect of age on ET-related gait abnormalities is more obvious than in a healthy population, even in advanced ages. Moreover, ET patients report greater subjective functional gait, experiencing more fear of falls and near falls than controls [41], which indicates that gait and balance problems in ET are not merely a subclinical phenomenon. However, no increased stride-to-stride variability—considered an important hallmark of cerebellar pathology—has been found in ET patients [34, 36, 42], suggesting that the changes in ET are not as advanced as those seen in the cerebellar ataxias.
Although early studies failed to find an association between impairments of balance and gait and tremor severity in ET patients [37, 43, 44], more recent studies have consistently revealed significant correlations between tandem gait abnormalities and the severity of intention tremor of the hands [42], hands and legs [35], and cranial tremors involving the neck, jaw, and voice [9, 39, 41]. The finding that the subgroup of ET patients with head tremor is the most functionally impaired [9, 41] is consistent with a voxel-based morphometry magnetic resonance imaging (MRI) study [45], which demonstrated atrophy of the cerebellar vermis, especially in ET patients with head tremor, and one postmortem analysis that revealed more vermal Purkinje cell axonal swellings in ET patients than in controls, with the highest vermal torpedo counts in ET cases with voice, jaw, and neck tremors [46]. Taken together, these results suggest that ET-related abnormalities of balance and gait found may correspond to lesions of the medial regions of the cerebellum that play a primary role in the control of balance and locomotion [47].
Dysarthria
Subclinical signs of dysarthria (increased syllable duration) may be seen in a subset of patients, which would suggest that ET is associated with an impairment of the cerebellum [48].
Oculomotor Abnormalities
Deficits of pursuit initiation that correlate with the intensity of intention tremor, and pathological suppression of the vestibulo-ocular reflex have been reported in ET patients [49]. These oculomotor deficits may indicate an impairment of the caudal vermis in ET. [49]
Eye–Hand Incoordination
In a study on eye–hand coordination, abnormal kinematic changes in the early phase of pointing movements have been reported in ET patients [50]. In contrast to normal subjects, ET patients’ saccadic latency did not decrease during combined eye–hand movements compared with saccades performed in isolation [50]. In addition, hand movements had a longer duration in ET patients, with decreased peak acceleration, an increased latency of the peak velocity, and peak deceleration [50]. Altogether, these changes suggest an underlying abnormality of the cerebellum and/or its pathways [50].
Motor Learning Deficits
Disordered acquisition and retention of the classically conditioned eyeblink response—a well-established model to study motor learning—has been described in ET patients, providing further evidence that ET is caused by a functional disturbance of olivo-cerebellar circuits, which may cause cerebellar dysfunction [51].
Incoordination During Spiral Drawing Task
The Spiral Width Variability Index (SWVI) is a graphonomic measure of the variability around an ideal trajectory, which may be increased in patients with cerebellar pathology [52, 53]. The finding that ET cases exhibit higher SWVI than controls and that higher SWVI scores correlated not only with more severe kinetic tremor but also with the presence and severity of other cerebellar signs in ET (e.g., intention tremor and tandem gait mis-steps) provides additional support for the notion that the underlying pathology in ET is at least in part cerebellar [54].
Motor Timing and Visual Reaction Time
ET patients present significantly impaired motor performance, at least in some tasks, such as rapid repetitive finger movements (finger tapping and frequency) and visual reaction time than normal controls, suggesting a severe deficit of event-based rhythm generation on both sides in ET [55, 56]. The finding that both spinocerebellar ataxia and ET patients are significantly worse at intercepting moving target than are patients with early Parkinson’s disease and normal controls suggests that the cerebellum may play an essential role in integrating incoming visual information with motor output in a timely manner [57].
Impairment of Social Abilities
Cognitive aspects of the theory of mind have been reported to be selectively impaired in ET patients [58]. Theory of mind is the ability to attribute mental states (beliefs, intents, desires, pretending, and knowledge) to oneself and others, and to understand that others have beliefs, desires, and intentions different from one’s own, and thus, it may be regarded as an essential prerequisite for successful human social interaction [59]. The dorsolateral prefrontal cortex is anatomically and functionally connected with the posterior lobule of neocerebellum [60], and this neural circuit is thought to be involved in several cognitive functions [61, 62], including social cognition [63]. Based on these facts, the selective impairment of cognitive theory of mind found in ET patients might be conceived of as a further consequence of dysfunctional dorsolateral prefrontal cortex–cerebellum circuit [58].
Improvement in Tremor After Cerebellar Stroke
Vascular lesions of cerebellar–thalamic–cortical pathway have been reported to improve the tremor of ET patients [64–66]. There are anectodical cases of ET patients in whom tremor improved or disappeared after stroke. There are several described cases in the literature. For example, an ET patient in whom tremor disappeared on the right side after a homolateral cerebellar infarct has been reported [64]. In another case, an ET patient improved after a sensorimotor stroke related to a small cortical infarct near by the left precentral region of the brain [66]. Finally, the unilateral tremor of an ET patient with a large area of damage in the right hemisphere, as a result from two strokes, disappeared in the left arm after the second stroke [65]. Contrary to the previous observations, lesions of the cerebellum may also trigger ET. For example, a patient who developed ET ipsilateral to cerebellar hemispherectomy has been described [67].
Efficacy of Stereotactic Thalamotomy and Thalamic Ventralis Intermedius Nucleus Deep Brain Stimulation
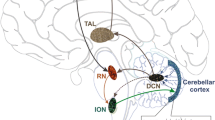
Surgery has been a treatment for ET since the early 1950s [8]. Initially, a number of different brain regions were targeted for tremor control. However, the optimal target was eventually determined to be the ventralis intermedius (VIM) nucleus of the thalamus, since tremor is thought to be mediated by a neuronal loop involving cerebello-thalamo-cortical pathways [5–8]. Stereotactic thalamotomy and thalamic VIM nucleus deep brain stimulation (DBS) offer high rates of tremor reduction in the contralateral arm [5–8]. The effects of thalamic DBS on the cerebellothalamocortical pathway have been studied [68]. With DBS turned off, excitability of the cerebello-thalamo-cortical pathways was reduced [68]. Turning DBS on resulted in facilitation of the cerebello-thalamo-cortical pathways, which suggests that thalamic DBS appears to activate rather than inhibit [68].
In a recent study, using diffusion tensor imaging, a group of ET patients that had undergone unilateral, left, thalamotomy, and ET patients that did not undergo thalamotomy (control group) were compared [69]. Fractional anisotropy and increased mean diffusivity values in the right superior cerebellar peduncle leading to the left, lesioned thalamus, only in the thalamotomy group, were observed, which suggests that long-term structural changes in the cerebellorubrothalamic tract after thalamotomy may occur [69].
Neuropsychological Deficits
Although the cerebellum has been linked historically to the control of posture, gait, and skilled voluntary movement, converging clinical and neuroimaging evidence has implicated the cerebellum in many different neuropsychological non-motor functions, thereby defining a novel vision of the cerebellar contribution to cognitive processes and emotional behavior [70]. This view gives rise to the dysmetria of thought theory, which holds that cerebellar modulation of movement, cognition, and emotion is a manifestation of the same neurological process [71, 72]. Schmahmann and Sherman [73] provided clinical evidence for this theory based on a constellation of cognitive, affective, and behavioral manifestations in patients whose lesions were confined to the cerebellum. The authors argued that those non-motor dysfunctions were related to the cerebellar disorder itself, particularly when the posterior lobe was involved, which led them to propose the cerebellar cognitive affective syndrome, characterized by the following features: (1) disturbances of executive function (this includes deficient planning, set-shifting, abstract reasoning, working memory, and decreased verbal fluency), (2) impaired spatial cognition, (3) personality changes, and (4) linguistic difficulties [73]. All these deficits have been attributed to the disruption of the neural circuits linking prefrontal, temporal, posterior parietal, and limbic cortices with the cerebellum [73]. The subsequent description that patients with cerebellar stroke in the posterior lobe had no or minimal cerebellar motor signs provides further indirect support to the notion that the “motor cerebellum” is situated in the anterior lobe (lobules I–V), and the “cognitive cerebellum” within the posterior lobe (predominantly lobule VII) [74].
As in other neurodegenerative cerebellar diseases [75, 76], mild cognitive deficits, mainly in attention and frontal executive functions, verbal memory, and visuospatial processes, have been reported to occur in ET patients in a series of independent studies [11, 13, 77–86]. Taken together, these studies indicate that a disorder of the frontal cortical or frontal cortical–cerebellar pathways might occur in some patients with ET [11, 13, 77–86]. Overall, the degree of cognitive impairment in ET is mild and is thought to be due to a dysfunction in the dorsolateral prefrontal cortex and in the inferior parietal cortex, including the cerebral–cerebellar loop [87].
It is worth noting how the observed ET-related neuropsychological deficits resemble those reported in the literature to occur after isolated cerebellar lesions:
-
1.
ET patients experience cognitive difficulties impacting executive functions and frontal lobe tasks [11, 13, 77–86], and similarly, several reports of cerebellar patients have also noted impaired executive function [88–90]. For example, working memory, a cognitive process, which can be considered an example of a test of executive function, has been consistently shown to strongly engage cerebellar circuits [91, 92], suggesting that the role of the cerebellum in working memory includes aspects of encoding, maintenance, and retrieval [93]. Overactivation (abnormally enhanced cerebellar response) of posterior lobules of the cerebellum (crus I/lobule VI) during verbal working memory represents increased effort to subvocally refresh stimuli during attention-demanding conditions, such as high-load working memory trials and, hence, it may represent a brain compensatory mechanism that maintains the behavioral performances within a normal range [94]. Noteworthy, this abnormally enhanced cerebellar response has been reported in both individuals suffering from chronic alcoholism, a toxic condition associated with neurodegeneration of the cerebellum [94], and patients with ET during a verbal working memory task [95], providing evidence that disrupted functional interactions within distinct cortical–cerebellar circuits responsible for verbal working memory are an important mechanism underlying cognitive dysfunction in ET.
-
2.
The observed fluency deficits among ET patients (more impaired on lexical than semantic verbal fluency) [77, 96] also parallel those reported after cerebellar lesions [73, 97].
-
3.
Given that the posterior parietal cortex receives afferent connections from cerebellum via the pons and thalamus, the poor performance in visuospatial functions among ET patients may reflect the involvement of the cerebello-ponto-thalamo-parietal pathways [81].
There are also observations which suggest alternative explanations for the ET-related cognitive deficits. First, cognitive deficits in motor and premotor ET (that is, ET cases who underwent neuropsychological evaluations at a baseline visit prior to the onset of their tremor) are not static, and they appear to be progressing at a faster rate than in elders who do not develop this disease [13, 86]. In addition, an association between elderly-onset ET and prevalent mild cognitive impairment was demonstrated in the NEDICES study [12]. In line with this, in the same population, an association between elderly-onset ET and prevalent dementia was also evident; ET cases with tremor onset after 65 years of age were 70 % more likely to have dementia than were similarly aged controls [14]. Further, in an incidence cohort from the same population, ET cases with tremor onset after 65 years of age were 89 % more likely to develop incident dementia than were controls [15]. It is unlikely, however, that cerebellar dysfunctions per se fully explain dementia in ET, because the cognitive deficits that have been described in patients with cerebellar lesions are generally mild [74, 98]. Second, there is preliminary evidence to suggest that there are gray matter changes in ET patients with ET [99], the significance of which in relation to cognitive deficits requires further study. Moreover, the link between elderly patients with ET and dementia would suggest that this subgroup of patients with ET had involvement of the cerebral cortex during the course of their illness [14, 15], which raise the question whether elderly patients with ET are suffering from the same illness as younger patients with ET or whether there is a difference in pathophysiology based on age of onset. However, a recent neuroimaging study has reported that ET patients display, relative to controls, disrupted functional coupling between lobule VI and the executive control circuit (dorsolateral prefrontal cortex; parietal lobules) as well as the default mode network (precuneus cortex, ventromedial prefrontal cortex, and hippocampus) [95]. This functional imbalance can be viewed as a dysfunction of switching from default mode network to executive control circuit [95]. This “switcher role” of the cerebellum would be essential to guarantee a correct optimization of cognitive resources in accordance to ongoing needs [95]. Thus, given that diffuse abnormalities within the executive control circuit and the default mode network are the hallmarks of Alzheimer’s disease [100], it could be that the fundamental brain mechanisms underlying cognitive deficits in ET resemble those implicated in Alzheimer’s disease.
Table 1 summarizes the main studies that link ET to cerebellum.
Conclusion
ET may be a family of disease unified by the presence of kinetic tremor, but also showing etiological, pathological, and clinical heterogeneity. The wide spectrum of clinical features of ET that suggest that it originates as a cerebellar or cerebellar outflow problem include the presence of intentional tremor, gait and balance abnormalities, dysarthria, and oculomotor abnormalities, as well as deficits in eye-hand coordination, motor learning deficits, incoordination during spiral drawing task, motor timing and visual reaction time, impairment of social abilities, improvement in tremor after cerebellar stroke, efficacy of deep brain stimulation (which blocks cerebellar outflow), and cognitive dysfunction. It is unlikely, however, that cerebellar dysfunctions per se fully explain ET-associated dementia, because the cognitive deficits that have been described in patients with cerebellar lesions are generally mild. Overall, a variety of clinical findings suggest that in at least a sizable proportion of patients with ET, there is an underlying abnormality of the cerebellum and/or its pathways.
References
Benito-León J, Bermejo-Pareja F, Morales JM, Vega S, Molina JA. Prevalence of essential tremor in three elderly populations of central Spain. Mov Dis: Off J Mov Dis Soc. 2003;18:389–94. doi:10.1002/mds.10376.
Benito-León J, Bermejo-Pareja F. Louis ED and Neurological Disorders in Central Spain Study G. Incidence of essential tremor in three elderly populations of central Spain. Neurology. 2005;64:1721–5. doi:10.1212/01.wnl.0000161852.70374.01.
Benito-León J. How common is essential tremor? Neuroepidemiology. 2009;32:215–6. doi:10.1159/000195692.
Louis ED, Hafeman D, Parvez F, Alcalay RN, Islam T, Siddique AB, et al. Prevalence of essential tremor in Araihazar, Bangladesh: a population-based study. Neuroepidemiology. 2011;36:71–6. doi:10.1159/000323389.
Benito-León J, Louis ED. Essential tremor: emerging views of a common disorder. Nat Clin Pract Neurol. 2006;2:666–78. doi:10.1038/ncpneuro0347. quiz 2p following 91.
Benito-León J, Louis ED. Clinical update: diagnosis and treatment of essential tremor. Lancet. 2007;369:1152–4. doi:10.1016/S0140-6736(07)60544-3.
Benito-León J, Louis ED. Update on essential tremor. Minerva Med. 2011;102:417–39.
Benito-León J and Louis ED. Management of essential tremor, including medical and surgical approaches. Handbook of clinical neurology / edited by PJ Vinken and GW Bruyn 2011; 100:449-56. doi 10.1016/B978-0-444-52014-2.00034-3.
Louis ED, Rios E, Rao AK. Tandem gait performance in essential tremor: clinical correlates and association with midline tremors. Mov Dis: Off J Mov Dis Soc. 2010;25:1633–8. doi:10.1002/mds.23144.
Benito-León J, Louis ED, Bermejo-Pareja F, Neurological Disorders in Central Spain Study G. Risk of incident Parkinson’s disease and parkinsonism in essential tremor: a population based study. J Neurol Neurosurg Psychiatry. 2009;80:423–5. doi:10.1136/jnnp.2008.147223.
Benito-León J, Louis ED. Bermejo-Pareja F and Neurological Disorders in Central Spain Study G. Population-based case-control study of cognitive function in essential tremor. Neurology. 2006;66:69–74. doi:10.1212/01.wnl.0000192393.05850.ec.
Benito-León J, Louis ED, Mitchell AJ, Bermejo-Pareja F. Elderly-onset essential tremor and mild cognitive impairment: a population-based study (NEDICES). J Alzheimer’s Dis: JAD. 2011;23:727–35. doi:10.3233/JAD-2011-101572.
Benito-León J, Louis ED, Sánchez-Ferro A, Bermejo-Pareja F. Rate of cognitive decline during the premotor phase of essential tremor: a prospective study. Neurology. 2013;81:60–6. doi:10.1212/WNL.0b013e318297ef2b.
Benito-León J, Louis ED. Bermejo-Pareja F and Neurological Disorders in Central Spain Study G. Elderly-onset essential tremor is associated with dementia. Neurology. 2006;66:1500–5. doi:10.1212/01.wnl.0000216134.88617.de.
Bermejo-Pareja F, Louis ED, Benito-León J, Neurological Disorders in Central Spain Study G. Risk of incident dementia in essential tremor: a population-based study. Mov Disord: Off J Mov Disord Soc. 2007;22:1573–80. doi:10.1002/mds.21553.
Chatterjee A, Jurewicz EC, Applegate LM, Louis ED. Personality in essential tremor: further evidence of non-motor manifestations of the disease. J Neurol Neurosurg Psychiatry. 2004;75:958–61.
Louis ED, Benito-León J, Bermejo-Pareja F, Neurological Disorders in Central Spain Study G. Self-reported depression and anti-depressant medication use in essential tremor: cross-sectional and prospective analyses in a population-based study. Eur J Neurol: Off J Eur Fed Neurol Soc. 2007;14:1138–46. doi:10.1111/j.1468-1331.2007.01923.x.
Louis ED, Bromley SM, Jurewicz EC, Watner D. Olfactory dysfunction in essential tremor: a deficit unrelated to disease duration or severity. Neurology. 2002;59:1631–3.
Ondo WG, Sutton L, Dat Vuong K, Lai D, Jankovic J. Hearing impairment in essential tremor. Neurology. 2003;61:1093–7.
Benito-León J, Louis ED, Bermejo-Pareja F, Neurological Disorders in Central Spain Study G. Reported hearing impairment in essential tremor: a population-based case-control study. Neuroepidemiology. 2007;29:213–7. doi:10.1159/000112463.
Gamboa J, Jiménez-Jiménez FJ, Nieto A, Cobeta I, Vegas A, Ortí-Pareja M, et al. Acoustic voice analysis in patients with essential tremor. J Voice: Off J Voice Found. 1998;12:444–52.
Benito-León J, Louis ED, Bermejo-Pareja F. Short sleep duration heralds essential tremor: a prospective, population-based study. Mov Disord : off J Mov Disord Society. 2013;28:1700–7. doi:10.1002/mds.25590.
Critchley M. Observations on essential (heredofamial) tremor. Brain: J Neurol. 1949;72:113–39.
Benito-León J. Essential tremor: a neurodegenerative disease? Tremor Other Hyperkinet Mov (N Y). 2014; 4:252. doi 10.7916/D8765CG0.
Jenkins IH, Bain PG, Colebatch JG, Thompson PD, Findley LJ, Frackowiak RS, et al. A positron emission tomography study of essential tremor: evidence for overactivity of cerebellar connections. Ann Neurol. 1993;34:82–90. doi:10.1002/ana.410340115.
Benito-León J and Louis ED. The top 100 cited articles in essential tremor. Tremor Other Hyperkinet Mov (N Y). 2013; 3.
Deuschl G, Bain P, Brin M. Consensus statement of the Movement Disorder Society on Tremor. Ad Hoc Scientific Committee. Mov Disord: Off J Mov Disord Soc. 1998;13 Suppl 3:2–23.
Findley LJ, Koller WC. Essential tremor: a review. Neurology. 1987;37:1194–7.
Biary N, Koller W. Kinetic predominant essential tremor: successful treatment with clonazepam. Neurology. 1987;37:471–4.
Brennan KC, Jurewicz EC, Ford B, Pullman SL, Louis ED. Is essential tremor predominantly a kinetic or a postural tremor? A clinical and electrophysiological study. Mov Disord: Off J Mov Disord Soc. 2002;17:313–6.
Louis ED. The primary type of tremor in essential tremor is kinetic rather than postural: cross-sectional observation of tremor phenomenology in 369 cases. European journal of neurology : the official journal of the European Federation of Neurological Societies. 2013;20:725–7. doi:10.1111/j.1468-1331.2012.03855.x.
Deuschl G, Wenzelburger R, Loffler K, Raethjen J, Stolze H. Essential tremor and cerebellar dysfunction clinical and kinematic analysis of intention tremor. Brain: Journal Neurol. 2000;123(Pt 8):1568–80.
Louis ED, Frucht SJ, Rios E. Intention tremor in essential tremor: prevalence and association with disease duration. Mov Disord: Off J Mov Disord Soc. 2009;24:626–7. doi:10.1002/mds.22370.
Earhart GM, Clark BR, Tabbal SD, Perlmutter JS. Gait and balance in essential tremor: variable effects of bilateral thalamic stimulation. Mov Disord: Off J Mov Disord Soc. 2009;24:386–91. doi:10.1002/mds.22356.
Fasano A, Herzog J, Raethjen J, Rose FE, Muthuraman M, Volkmann J, et al. Gait ataxia in essential tremor is differentially modulated by thalamic stimulation. Brain: J Neurol. 2010;133:3635–48. doi:10.1093/brain/awq267.
Rao AK, Gillman A, Louis ED. Quantitative gait analysis in essential tremor reveals impairments that are maintained into advanced age. Gait Posture. 2011;34:65–70. doi:10.1016/j.gaitpost.2011.03.013.
Singer C, Sanchez-Ramos J, Weiner WJ. Gait abnormality in essential tremor. Mov Disord: Off J Mov Disord Soc. 1994;9:193–6. doi:10.1002/mds.870090212.
Hubble JP, Busenbark KL, Pahwa R, Lyons K, Koller WC. Clinical expression of essential tremor: effects of gender and age. Mov Disord: Off J Mov Disord Soc. 1997;12:969–72. doi:10.1002/mds.870120620.
Hoskovcova M, Ulmanova O, Sprdlik O, Sieger T, Novakova J, Jech R, et al. Disorders of balance and gait in essential tremor are associated with midline tremor and age. Cerebellum. 2013;12:27–34. doi:10.1007/s12311-012-0384-4.
Cinar N, Sahin S, Okluoglu Onay T, Karsidag S. Balance in essential tremor during tandem gait: is the first mis-step an important finding? JClin Neurosci: Off J Neuro Soc Australasia. 2013;20:1433–7. doi:10.1016/j.jocn.2013.01.013.
Louis ED, Rao AK, Gerbin M. Functional correlates of gait and balance difficulty in essential tremor: balance confidence, near misses and falls. Gait Posture. 2012;35:43–7. doi:10.1016/j.gaitpost.2011.08.002.
Stolze H, Petersen G, Raethjen J, Wenzelburger R, Deuschl G. The gait disorder of advanced essential tremor. Brain: J Neurol. 2001;124:2278–86.
Bove M, Marinelli L, Avanzino L, Marchese R, Abbruzzese G. Posturographic analysis of balance control in patients with essential tremor. Mov Disord: Off J Mov Disord Soc. 2006;21:192–8. doi:10.1002/mds.20696.
Parisi SL, Heroux ME, Culham EG, Norman KE. Functional mobility and postural control in essential tremor. Arch Phys Med Rehabil. 2006;87:1357–64. doi:10.1016/j.apmr.2006.07.255.
Quattrone A, Cerasa A, Messina D, Nicoletti G, Hagberg GE, Lemieux L, et al. Essential head tremor is associated with cerebellar vermis atrophy: a volumetric and voxel-based morphometry MR imaging study. AJNR Am J Neuroradiol. 2008;29:1692–7. doi:10.3174/ajnr.A1190.
Louis ED, Faust PL, Ma KJ, Yu M, Cortes E, Vonsattel JP. Torpedoes in the cerebellar vermis in essential tremor cases vs. controls. Cerebellum. 2011;10:812–9. doi:10.1007/s12311-011-0291-0.
Morton SM, Bastian AJ. Mechanisms of cerebellar gait ataxia. Cerebellum. 2007;6:79–86. doi:10.1080/14734220601187741.
Kronenbuerger M, Konczak J, Ziegler W, Buderath P, Frank B, Coenen VA, et al. Balance and motor speech impairment in essential tremor. Cerebellum. 2009;8:389–98. doi:10.1007/s12311-009-0111-y.
Helmchen C, Hagenow A, Miesner J, Sprenger A, Rambold H, Wenzelburger R, et al. Eye movement abnormalities in essential tremor may indicate cerebellar dysfunction. Brain: J Neurol. 2003;126:1319–32.
Trillenberg P, Fuhrer J, Sprenger A, Hagenow A, Kompf D, Wenzelburger R, et al. Eye-hand coordination in essential tremor. Mov Disord: Off J Mov Disord Soc. 2006;21:373–9. doi:10.1002/mds.20729.
Kronenbuerger M, Gerwig M, Brol B, Block F, Timmann D. Eyeblink conditioning is impaired in subjects with essential tremor. Brain: J Neurol. 2007;130:1538–51. doi:10.1093/brain/awm081.
Longstaff MG, Heath RA. Spiral drawing performance as an indicator of fine motor function in people with multiple sclerosis. Hum Mov Sci. 2006;25:474–91. doi:10.1016/j.humov.2006.05.005.
Hsu AW, Piboolnurak PA, Floyd AG, Yu QP, Wraith JE, Patterson MC, et al. Spiral analysis in Niemann-Pick disease type C. Mov Disord: Off J Mov Disord Soc. 2009;24:1984–90. doi:10.1002/mds.22744.
Louis ED, Gillman A, Boschung S, Hess CW, Yu Q, Pullman SL. High width variability during spiral drawing: further evidence of cerebellar dysfunction in essential tremor. Cerebellum. 2012;11:872–9. doi:10.1007/s12311-011-0352-4.
Farkas Z, Szirmai I, Kamondi A. Impaired rhythm generation in essential tremor. Mov Disord: Off J Mov Disord Soc. 2006;21:1196–9. doi:10.1002/mds.20934.
Jiménez-Jiménez FJ, Rubio L, Alonso-Navarro H, Calleja M, Pilo-de-la-Fuente B, Plaza-Nieto JF, et al. Impairment of rapid repetitive finger movements and visual reaction time in patients with essential tremor. Eur J Neurol: Off J Eur Fed Neurol Soc. 2010;17:152–9. doi:10.1111/j.1468-1331.2009.02784.x.
Bares M, Lungu OV, Husarova I, Gescheidt T. Predictive motor timing performance dissociates between early diseases of the cerebellum and Parkinson’s disease. Cerebellum. 2010;9:124–35. doi:10.1007/s12311-009-0133-5.
Santangelo G, Trojano L, Barone P, Errico D, Improta I, Agosti V, et al. Cognitive and affective theory of mind in patients with essential tremor. J Neurol. 2013;260:513–20. doi:10.1007/s00415-012-6668-2.
Adolphs R. Cognitive neuroscience of human social behaviour. Nat Rev Neurosci. 2003;4:165–78. doi:10.1038/nrn1056.
Krienen FM, Buckner RL. Segregated fronto-cerebellar circuits revealed by intrinsic functional connectivity. Cereb Cortex. 2009;19:2485–97. doi:10.1093/cercor/bhp135.
Chen SH, Desmond JE. Cerebrocerebellar networks during articulatory rehearsal and verbal working memory tasks. NeuroImage. 2005;24:332–8. doi:10.1016/j.neuroimage.2004.08.032.
Durisko C, Fiez JA. Functional activation in the cerebellum during working memory and simple speech tasks. Cortex; J Dev Study Nerv Syst Behav. 2010;46:896–906. doi:10.1016/j.cortex.2009.09.009.
Brune M, Brune-Cohrs U. Theory of mind--evolution, ontogeny, brain mechanisms and psychopathology. Neurosci Biobehav Rev. 2006;30:437–55. doi:10.1016/j.neubiorev.2005.08.001.
Dupuis MJ, Delwaide PJ, Boucquey D, Gonsette RE. Homolateral disappearance of essential tremor after cerebellar stroke. Mov Disord: Off J Mov Disord Soc. 1989;4:183–7. doi:10.1002/mds.870040210.
Constantino AE, Louis ED. Unilateral disappearance of essential tremor after cerebral hemispheric infarct. J Neurol. 2003;250:354–5.
Kim JS, Park JW, Kim WJ, Kim HT, Kim YI, Lee KS. Disappearance of essential tremor after frontal cortical infarct. Mov Disord: Off J Mov Disord Soc. 2006;21:1284–5. doi:10.1002/mds.20894.
Chahine LM, Ghosh D. Essential tremor after ipsilateral cerebellar hemispherectomy: support for the thalamus as the central oscillator. J Child Neurol. 2009;24:861–4. doi:10.1177/0883073808329528.
Molnar GF, Sailer A, Gunraj CA, Lang AE, Lozano AM, Chen R. Thalamic deep brain stimulation activates the cerebellothalamocortical pathway. Neurology. 2004;63:907–9.
Buijink AW, Caan MW, Contarino MF, Schuurman PR, van den Munckhof P, de Bie RM, et al. Structural changes in cerebellar outflow tracts after thalamotomy in essential tremor. Parkinsonism Relat Disord. 2014;20:554–7. doi:10.1016/j.parkreldis.2014.02.020.
Noroozian M. The role of the cerebellum in cognition: beyond coordination in the central nervous system. Neurol Clin. 2014;32:1081–104. doi:10.1016/j.ncl.2014.07.005.
Schmahmann JD. An emerging concept. The cerebellar contribution to higher function. Arch Neurol. 1991;48:1178–87.
Schmahmann JD. The role of the cerebellum in cognition and emotion: personal reflections since 1982 on the dysmetria of thought hypothesis, and its historical evolution from theory to therapy. Neuropsychol Rev. 2010;20:236–60. doi:10.1007/s11065-010-9142-x.
Schmahmann JD, Sherman JC. The cerebellar cognitive affective syndrome. Brain: J Neurol. 1998;121(Pt 4):561–79.
Schmahmann JD, Macmore J, Vangel M. Cerebellar stroke without motor deficit: clinical evidence for motor and non-motor domains within the human cerebellum. Neuroscience. 2009;162:852–61. doi:10.1016/j.neuroscience.2009.06.023.
Suenaga M, Kawai Y, Watanabe H, Atsuta N, Ito M, Tanaka F, et al. Cognitive impairment in spinocerebellar ataxia type 6. J Neurol Neurosurg Psychiatry. 2008;79:496–9. doi:10.1136/jnnp.2007.119883.
Feng L, Chen DB, Hou L, Huang LH, Lu SY, Liang XL, et al. Cognitive impairment in native Chinese with spinocerebellar ataxia type 3. Eur Neurol. 2014;71:262–70. doi:10.1159/000357404.
Troster AI, Fields JA, Pahwa R, Wilkinson SB, Strait-Troster KA, Lyons K, et al. Neuropsychological and quality of life outcome after thalamic stimulation for essential tremor. Neurology. 1999;53:1774–80.
Gasparini M, Bonifati V, Fabrizio E, Fabbrini G, Brusa L, Lenzi GL, et al. Frontal lobe dysfunction in essential tremor: a preliminary study. J Neurol. 2001;248:399–402.
Lombardi WJ, Woolston DJ, Roberts JW, Gross RE. Cognitive deficits in patients with essential tremor. Neurology. 2001;57:785–90.
Lacritz LH, Dewey Jr R, Giller C, Cullum CM. Cognitive functioning in individuals with “benign” essential tremor. J Int Neuropsychol Soc: JINS. 2002;8:125–9.
Sahin HA, Terzi M, Ucak S, Yapici O, Basoglu T, Onar M. Frontal functions in young patients with essential tremor: a case comparison study. J Neuropsychiatry Clin Neurosci. 2006;18:64–72. doi:10.1176/appi.neuropsych.18.1.64.
Higginson CI, Wheelock VL, Levine D, King DS, Pappas CT, Sigvardt KA. Cognitive deficits in essential tremor consistent with frontosubcortical dysfunction. J Clin Exp Neuropsychol. 2008;30:760–5. doi:10.1080/13803390701754738.
Kim J-S, Song I-U, Shim Y-S, Park J-W, Yoo J-Y, Kim Y-I, et al. Cognitive impairment in essential tremor without dementia. J Clin Neurol. 2009;5:81–4. doi:10.3988/jcn.2009.5.2.81.
Frisina PG, Tse W, Halbig TD, Libow LS. The pattern of cognitive-functional decline in elderly essential tremor patients: an exploratory-comparative study with Parkinson’s and Alzheimer’s disease patients. J Am Med Dir Assoc. 2009;10:238–42. doi:10.1016/j.jamda.2008.10.013.
Louis ED, Benito-León J, Vega-Quiroga S, Bermejo-Pareja F, Neurological Disorders in Central Spain Study G. Cognitive and motor functional activity in non-demented community-dwelling essential tremor cases. J Neurol Neurosurg Psychiatry. 2010;81:997–1001. doi:10.1136/jnnp.2009.202838.
Louis ED, Benito-León J, Vega-Quiroga S, Bermejo-Pareja F, Neurological Disorders in Central Spain Study G. Faster rate of cognitive decline in essential tremor cases than controls: a prospective study. Eur J Neurol: Off J Eur Fed Neurolo Soc. 2010;17:1291–7. doi:10.1111/j.1468-1331.2010.03122.x.
Cerasa A, Passamonti L, Novellino F, Salsone M, Gioia MC, Morelli M, et al. Fronto-parietal overactivation in patients with essential tremor during Stroop task. Neuroreport. 2010;21:148–51. doi:10.1097/WNR.0b013e328335b42c.
Appollonio IM, Grafman J, Schwartz V, Massaquoi S, Hallett M. Memory in patients with cerebellar degeneration. Neurology. 1993;43:1536–44.
Rapoport M, van Reekum R, Mayberg H. The role of the cerebellum in cognition and behavior: a selective review. J Neuropsychiatry Clin Neurosci. 2000;12:193–8.
Steinlin M, Styger M, Boltshauser E. Cognitive impairments in patients with congenital nonprogressive cerebellar ataxia. Neurology. 1999;53:966–73.
Honey GD, Bullmore ET, Sharma T. Prolonged reaction time to a verbal working memory task predicts increased power of posterior parietal cortical activation. NeuroImage. 2000;12:495–503. doi:10.1006/nimg.2000.0624.
Tomasi D, Caparelli EC, Chang L, Ernst T. fMRI-acoustic noise alters brain activation during working memory tasks. NeuroImage. 2005;27:377–86. doi:10.1016/j.neuroimage.2005.04.010.
Marvel CL, Desmond JE. The contributions of cerebro-cerebellar circuitry to executive verbal working memory. Cortex; J Dev Study Nerv Syst Behav. 2010;46:880–95. doi:10.1016/j.cortex.2009.08.017.
Desmond JE, Chen SH, DeRosa E, Pryor MR, Pfefferbaum A, Sullivan EV. Increased frontocerebellar activation in alcoholics during verbal working memory: an fMRI study. NeuroImage. 2003;19:1510–20.
Passamonti L, Novellino F, Cerasa A, Chiriaco C, Rocca F, Matina MS, et al. Altered cortical-cerebellar circuits during verbal working memory in essential tremor. Brain: J Neurol. 2011;134:2274–86. doi:10.1093/brain/awr164.
Benito-León J, Louis ED, Bermejo-Pareja F, Neurological Disorders in Central Spain Study G. Population-based case-control study of cognitive function in essential tremor. Neurology. 2006;66:69–74. doi:10.1212/01.wnl.0000192393.05850.ec.
Gottwald B, Wilde B, Mihajlovic Z, Mehdorn HM. Evidence for distinct cognitive deficits after focal cerebellar lesions. J Neurol Neurosurg Psychiatry. 2004;75:1524–31. doi:10.1136/jnnp.2003.018093.
Schmahmann JD. From movement to thought: anatomic substrates of the cerebellar contribution to cognitive processing. Hum Brain Mapp. 1996;4:174–98. doi:10.1002/(SICI)1097-0193(1996)4:3<174::AID-HBM3>3.0.CO;2-0.
Benito-León J, Alvarez-Linera J, Hernández-Tamames JA, Alonso-Navarro H, Jiménez-Jiménez FJ, Louis ED. Brain structural changes in essential tremor: voxel-based morphometry at 3-Tesla. J Neurol Sci. 2009;287:138–42. doi:10.1016/j.jns.2009.08.037.
Greicius MD, Srivastava G, Reiss AL, Menon V. Default-mode network activity distinguishes Alzheimer’s disease from healthy aging: evidence from functional MRI. Proc Natl Acad Sci U S A. 2004;101:4637–42. doi:10.1073/pnas.0308627101.
Acknowledgments
Dr. Benito-León is supported by the National Institutes of Health, Bethesda, MD, USA (NINDS #R01 NS39422), the Commission of the European Union (grant ICT-2011-287739, NeuroTREMOR), and the Spanish Health Research Agency (grant FIS PI12/01602).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflicts of interest.
Rights and permissions
About this article
Cite this article
Benito-León, J., Labiano-Fontcuberta, A. Linking Essential Tremor to the Cerebellum: Clinical Evidence. Cerebellum 15, 253–262 (2016). https://doi.org/10.1007/s12311-015-0741-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-015-0741-1