Abstract
The postnatal development of the cerebellum is accomplished via a series of cytogenetic and morphogenetic events encoded in the genome. To decipher the underlying genetic basis of these events we have systematized the spatio-temporal gene expression profiles during mouse cerebellar development in the Cerebellar Development Transcriptome Database (CDT-DB). Using the CDT-DB, Ca2+-dependent activator protein for secretion 2 (CAPS2 or CADPS2) was identified as a developmentally regulated gene that is predominantly expressed in cerebellar granule cells (GCs) with an expression peak around the first or second postnatal week. CAPS2 protein is concentrated in parallel fiber (PF) terminals and is associated with secretory vesicles containing brain-derived neurotrophic factor (BDNF) and neurotrophin-3 (NT-3). CAPS2 enhances release of BDNF and NT-3, both of which are essential for normal cerebellar development. CAPS2-deficient (CAPS2−/−) mice show reduced secretion of BDNF and NT-3; consequently, the cerebella of these mice exhibit developmental deficits, such as delayed development and increased cell death in GCs, fewer branched dendrites on Purkinje cells (PCs), and loss of the intercrural fissure. The PF-PC synapses have aberrant cytoarchitectures and electrophysiological properties. These abnormal cellular and morphological phenotypes are more severe around the cerebellar vermis, in which hypoplasia has been reported in autism patients. Moreover, CAPS2−/− mice had fewer cortical and hippocampal parvalbumin-positive interneurons and some autistic-like behavioral phenotypes. In the CAPS2 genes of some autistic patients an aberrant splicing variant and non-synonymous SNPs have been identified. These recent studies implicate CAPS2 in autism susceptibility. Therefore, CAPS2−/− mice will be a useful model animal in which to study aspects of the neuropathology and behaviors characteristic of developmental disorders.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ca2+-dependent activator protein for secretion 2 (CAPS2 or CADPS2), one of the developmentally regulated genes identified in the cerebellar development transcriptome database (CDT-DB) project [1], is predominantly expressed in post-mitotic cerebellar granule cells (GCs) with an expression peak around the first or second postnatal week when various cellular developmental events are actively occurring. CAPS2 protein is associated with secretory vesicles containing neurotrophin-3 (NT-3) and brain-derived neurotrophic factor (BDNF) within parallel fiber (PF, axons of GCs) terminals and promotes release of NT-3 and BDNF in primary cerebellar cultures and in a heterologous expression system [2–4].
Several lines of in vivo evidence obtained by targeted gene disruption in mice have demonstrated that CAPS2 is involved in BDNF and NT-3 release: the CAPS2 deficiency causes impaired cerebellar development and function, including delayed development of GCs, increased cell death of GCs and Purkinje cells (PCs), atrophic dendrite arbors in PCs, impaired formation and electrophysiology of PF-PC synapses, hypoplasia of the vermis, and eye movement adaptation [5, 6]. Furthermore, CAPS2-deficient mice not only displayed aberrant cellular phenotypes in the cortex and hippocampus (fewer parvalbumin-positive interneurons), but also exhibited some autistic-like behavioral phenotypes (impaired social interaction, hyperactivity in a familiar environment, decreased exploratory behavior, and increased anxiety in an unfamiliar environment).
A study of the expression of CAPS2 in autism, a developmental disorder, showed that a subgroup of autistic patients express an aberrant alternatively spliced CAPS2 that is not properly transported into axons, presumably resulting in a deficit in local synaptic BDNF release [6]. In addition, seven missense single-nucleotide polymorphisms (SNPs) were identified in the CAPS2 genes of some autistic patients [6].
In this review, we provide an overview of the identification of CAPS2 based on its spatio-temporal expression profile during cerebellar development and demonstrate an indispensable role of CAPS2-mediated BDNF and NT-3 release in normal brain development. We also discuss the role of CAPS2 in autism susceptibility, the autistic-like phenotypes of CAPS2-deficient mice, and the aberrant splicing and SNPs of the CAPS2 genes of autistic individuals.
Identification of CAPS2 via a Cerebellar Development Transcriptome Study
Elucidating both spatial (cellular and regional) and temporal (developmental time series) gene expression patterns in developing brains is critical to understanding the brain development transcriptome. We investigated the gene expression profiles of mouse cerebellum during postnatal development by differential display and microarray (GeneChip) analyses for temporal expression profiling, and by in situ hybridization for spatial expression profiling [1]. Spatio-temporal gene expression profile data were systematized into the Cerebellar Development Transcriptome Database (CDT-DB). The CDT-DB indicates that the postnatal development of the mouse cerebellum is programmed by thousands of different genes, which exhibit differential expression patterns in time and space in developing mouse brains [1]. The CDT-DB will be useful to understand the transcriptomic bases underlying cerebellar development.
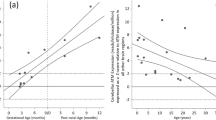
Another advantage of this systematic approach is to provide us with a foundation for mining potentially important gene candidates that have characteristic expression profiles in particular cell types or during a particular time window, and thus are implicated in particular brain development events. We have thus far identified various such brain development genes by CDT-DB searches for characteristic spatio-temporal expression patterns. CAPS2 (Fig. 1a) is expressed in post-mitotic GCs of the inner EGL (iEGL) and internal granular layer (IGL) (Fig. 1c) with an expression peak around postnatal day (P)7–12 (Fig. 1b) and is a BDNF release regulator that is potentially associated with autism [3, 4, 6, 7]. Cupidin/Homer2 is predominantly expressed in early-stage GCs and is a postsynaptic density scaffold protein that tethers mGluR1α/5, inositol 1,4,5-trisphosphate receptor, and Shank [8, 9]. p130Cas/Bcar1 is expressed in GCs in the developing EGL and IGL, and is a Src-type tyrosine kinase signaling adaptor that regulates growth cone extension [10]. Very-KIND is expressed in IGL GCs during the mid to late stages of cerebellar development, and is a brain-specific Ras guanine nucleotide exchanger that regulates dendrite growth [11]. Opalin/Tmem10 is up-regulated in the white matter and is an oligodendrocyte myelin glycoprotein [12, 13]. In this review, we focus on the expression pattern and function of CAPS2 and the autistic-like phenotypes of CAPS2-deficient mice.
Spatio-temporal expression of CAPS family members in the developing mouse cerebellum. a A schematic depiction of the predicted CAPS2 protein (aa 1–1,264). The dynactin1/p150Glued-interacting domain (DID), C2 domain (C2), pleckstrin homology domain (PH), and Munc13–1 homologous domain (MHD) are represented; these domains correspond to amino acid positions 134–331, 367–456, 494–596, and 677–1,166, respectively. b Time-series expression patterns (RT-PCR data) of CAPS2 mRNA and CAPS1 mRNA during postnatal cerebellar development. c Spatial cellular expression patterns (in situ hybridization data) of CAPS2 mRNA and CAPS1 mRNA in the whole brains, cerebella, and cerebellar cortices of mice at P7 and P21. Scale bars in panels of cerebellar cortex, 100 µm. These spatio-temporal expression data are deposited in the CDT-DB (http://www.cdtdb.brain.riken.jp). d Immunolocalization of CAPS2 protein (left) and CAP1 protein (right) in the same sagittal section of P8 (top panels) and P28 (bottom panels) wild-type cerebellum. Scale bars 20 µm. oEGL outer external granular layer, iEGL inner external granular layer, ML molecular layer, PCL Purkinje cell layer, IGL internal granular layer, WM white matter
Ca2+-Dependent Activator Protein for Secretion 2
CAPS2/CADPS2 (Fig. 1a) is a paralog of CAPS1/CADPS1 [3, 14, 15], which was first characterized as a neural/neuroendocrine-specific cytosolic component regulating Ca2+-triggered dense-core vesicle (DCV) exocytosis [16–20]. Recent studies have demonstrated that the two CAPS family members likely exert regulatory roles in the secretion of neuropeptides and monoaminergic transmitters from different neurons and neuroendocrine cells. CAPS2-mediated secretion plays an important role in the elaborate synapse and dendrite development of some types of cerebellar, neocortical and hippocampal neurons. Our study on CAPS2-knockout (KO) mice and autistic patients suggested that CAPS2 impairment may affect behavioral traits, including quality of social interaction behaviors.
CAPS2 is Concentrated in the Parallel Fibers, Whereas CAPS1 is Enriched in Glomeruli and Climbing Fibers
CAPS2 mRNA is predominantly expressed in post-mitotic GCs during postnatal cerebellar development, whereas CAPS1 mRNA is widely expressed in granule, Golgi, Purkinje, stellate, and basket cells in the cerebellum (Fig. 1c) [3, 7]. CAPS2 protein is abundantly localized in PFs, whereas CAPS1 protein is concentrated in climbing fiber (CF) terminals of the molecular layer (ML) and glomeruli of the internal granular layer (IGL) (Fig. 1d), indicating that the two CAPS family members are involved in secretions from different cerebellar neurons and afferent fibers, as well as from different cellular compartments (for example, presynaptic or somatodendritic).
CAPS Family Proteins Function in the Secretion Pathway
Many previous studies have shown that CAPS family proteins are involved in DCV exocytosis for various monoaminergic transmitters and neuropeptides (for example, release of norepinephrine from PC12 neuroendocrine cells [19] and rat brain synaptosomes [16, 17], release of catecholamine from mouse adrenal chromaffin cells [20], insulin release from mouse pancreatic β-cells [21, 22], and release of atrial natriuretic peptide (ANP) from mouse arterial myocytes [23]). It is of interest that reduced expression of CAPS1, together with many components of the catecholamine secretion pathway, was shown in pheochromocytoma of patients with von Hippel–Lindau syndrome [24].
CAPS1 interacts with membrane phosphatidylinositol 4,5-bisphosphate (PIP2) [25–28], which was suggested to be involved in the priming of DCV exocytosis [25, 26]. The sole CAPS ortholog UNC-31 in the Caenorhabditis elegans nervous system was also suggested to promote DCV exocytosis like the SV priming protein UNC-13 (Munc13 in mice) [29, 30]. There is, however, a debate regarding whether CAPS directly regulates DCV exocytosis. Recent studies on CAPS knockout mice suggested different function(s) by which CAPS regulates uptake or storage of catecholamine in the DCVs of chromaffin cells [31] and vesicular serotonin uptake in embryonic brains [32] and modulates priming, exocytosis, and stability of insulin granules in pancreatic β cells [22]. Moreover, a recent study on glutamatergic synapses of hippocampal neurons showed that CAPS is essential for the generation of readily releasable SVs by acting in the priming pathway like Munc13 [33]. On the contrary, several other studies support a role for CAPS proteins in exocytosis, but not in the loading of DCVs [34, 35] and a role in DCV exocytosis but not in SV exocytosis [29, 30]. Therefore, CAPS function is still a controversial issue.
Although the molecular mechanism underlying CAPS-mediated secretion has not been defined yet, as described above, it was shown that CAPS is genetically and/or functionally associated with the insulinoma-associated protein IA-2 [36], the syntaxin-interacting protein tomosyn [37], the Ras-like small GTPase RRP17/Rasl10b [23], the D2 dopamine receptor [38], the hetero-trimetric G-protein Gαs [39] and the Go2 pathway [32]. It should also be noted that immunoreactivity for CAPS2 was colocalized with that for the t-SNARE proteins VAMPs, the v-SNARE protein SNAP-25, and the endocytosis-related protein dynamin I [7]. Whether or not these vesicle trafficking-related proteins are commonly used by the CAPS2-associated secretion pathway remains to be determined.
CAPS2 is Involved in BDNF and NT-3 Release from Parallel Fibers
CAPS family proteins are involved in monoaminergic or neuropeptidergic secretion from various neurons and neuroendocrine cells. It is intriguing what kinds of secretory substances are released via the CAPS-mediated secretion pathway in the cerebellum. Cerebellar granule cells express neither tyrosine hydroxylase (TH), the first enzymatic step in catecholamine synthesis [40, 41], nor dopamine-β-hydroxylase [42], indicating that CAPS2 is not involved in catecholamine release in the cerebellum. On the other hand, Purkinje cells, which express CAPS1, express TH but have no catecholamine biosynthetic activity [40, 41]. A recent KO mouse study suggested that CAPS is associated with insulin granule secretion from pancreatic β-cells [22]. Although insulin stimulation was shown to induce tyrosine phosphorylation of the insulin receptor and its downstream signaling proteins SHC and GRB2 in mouse cerebellum [43, 44], it remains unclear not only whether any cerebellar neurons or afferent terminals release insulin, but also whether CAPS is associated with insulin secretion in the cerebellum.
It should be noted that the highest expression level of CAPS2 is present in the PFs of cerebellar granule cells (Fig. 1c, d). Immunogold electron microscopy revealed that CAPS2 protein is concentrated near vesicular and membranous structures within the PF terminals [3]. Immuno-screening for possible secretory substances within vesicle fractions immunoaffinity purified with anti-CAPS2 antibody intriguingly indicated the presence of two neurotrophins, BDNF, and NT-3, as well as the DCV marker chromogranin B [3]. These data suggest that CAPS2 protein is associated with BDNF- or NT-3-containing vesicles within PF terminals. Over-expression of exogenous CAPS2 not only enhances depolarization-induced (high KCl-induced) NT-3 and BDNF release from PC12 cells and cerebellar granule cells, but also promotes Purkinje cell survival [3]. In addition to BDNF release from cerebellar granule cells, BDNF is also thought to be released from CF terminals originating in the inferior olive [45]. CAPS1, but not CAPS2, is expressed in the inferior olive [7] and was found to be colocalized with VGLUT2, a CF terminal marker, around the proximal dendrites of Purkinje cells (Fig. 1d) [5], suggesting the possible involvement of CAPS1 in BDNF release from CF terminals.
Possible Role of CAPS2-Mediated BDNF and NT-3 Release in Cerebellar Development
During postnatal development, the mouse cerebellum undergoes a series of magnificent cellular events to accomplish the formation of cerebellar circuit architectures. Granule cells are generated by vigorous cell proliferation in the EGL to reach an immense cell number of approximately half the neurons in the brain, extend their PF axons, and migrate downward to form the IGL. Purkinje cells undergo extensive outgrowth of dendrites and form elaborate arbors with numerous synapses with the extending PFs of granule cells. During this period, NT-3 expression is down-regulated and BDNF expression is up-regulated [46–48]. In the mouse cerebellum, BDNF and NT-3 released by granule cells bind to their Trk receptors, TrkB and TrkC, respectively, on both postsynaptic Purkinje cells and presynaptic granule cells, thereby leading to Trk signaling in an autocrine-paracrine and anterograde fashion [49]. BDNF-TrkB and NT-3-TrkC signaling are essential for the differentiation and survival of both granule cells and Purkinje cells [50–55]. Mice genetically lacking either BDNF [56–58] or NT-3 [59] showed abnormal cerebellar morphology. Thus CAPS2-mediated NT-3 and BDNF release is expected to play an important role in postnatal cerebellar development, which has been shown by the analysis of CAPS2-deficient mice as described below.
Developmental and Functional Abnormalities in the Cerebella of CAPS2-Deficient Mice
Homozygous CAPS2-deficient (CAPS2−/−) pups are born at the expected Mendelian frequency [5, 6]. CAPS2−/− mice exhibit no obvious difference in life expectancy from control mice and have normal reproductive ability. The appearance of CAPS2−/− mice is normal except for slightly decreased body weights. In marked contrast, CAPS1-knockout (CAPS1−/−) mice die shortly after birth [31]. CAPS2−/− mice have complete loss of ability to produce CAPS2 protein (Fig. 2a) with no significant alteration in CAPS1 protein expression.
Cerebellar phenotypes of CAPS2−/− mice. a Western blot analysis of CAPS2 protein expression in the cerebella, cerebrums, and hippocampi of wild-type (+/+), heterozygote (+/−) and homozygote (−/−) CAPS2-deficient mice. b Delayed granule cell development in CAPS2−/− cerebellum. Cresyl violet-stained images of vermal lobule VI from sagittal sections of P17 wild-type (left) and CAPS2−/− (right) cerebella. The EGL is indicated by a square bracket. Scale bar 25 µm. c Scant outgrowth of Purkinje cell dendrites in CAPS2−/− cerebellum. Sagittal sections of P8 wild-type (left) and CAPS2−/− (right) cerebella were immunolabeled with an anti-calbindin antibody. Scale bars 50 µm. d Atrophic dendritic arbors of Purkinje cells in CAPS2−/− cerebellum. P8 wild-type (left) and CAPS2−/− (right) Purkinje cells in lobule V were visualized by Golgi staining. Scale bars 10 µm. e Enlarged presynaptic terminals and decreased vesicle distribution in the PF-PC synapses of CAPS2−/− mice. Electron micrographs of a PF-PC synapse in the P15 wild-type (left) and CAPS2−/− (right) cerebellum. Asterisks represent presynaptic terminals of PFs. Scale bars 200 nm. f Impaired short-term synaptic plasticity at PF-PC synapses of cerebellar lobules VI–VII at P15–P21.The mean paired-pulse facilitation (PPF) ratios of excitatory postsynaptic currents (EPSCs) are plotted against various inter-stimulus intervals (ms). Insets show representative traces of the first and second stimulation. *p < 0.05; **p < 0.01; ***p < 0.001 by the Mann–Whitney U test
Impaired Neurotrophin Signaling in the CAPS2−/− Cerebellum
In primary cerebellar cultures, the spontaneously secreted NT-3 level in the CAPS2−/− culture medium at 8 days in vitro (DIV) was only approximately 36% of the level in wild-type culture medium, but there was no significant difference between the CAPS2−/− and wild-type cultures in terms of the total amount of NT-3 in cell lysates [5]. Although both NT-3 and BDNF synaptically released from presynaptic granule cells are known to be internalized into postsynaptic Purkinje cells, NT-3 and BDNF immunoreactivities in Purkinje cells were diminished in CAPS2−/− mice. Moreover, although binding of NT-3 and BDNF leads to autophosphorylation of TrkC and TrkB receptors, respectively, the intense immunoreactivity to phosphorylated Trk normally observed in the upper MLs of wild-type mice was considerably reduced in those of CAPS2−/− mice. Taken together, these data indicated that loss of CAPS2 results in a deficit in NT-3 and BDNF release followed by Trk receptor activation in target cells.
Aberrant Cytoarchitecture and Increased Cell Death in the CAPS2−/− Cerebellum
CAPS2−/− mice exhibit no overt abnormality of gross brain anatomy, with the exception of the absence of the intercrural fissure between vermal lobules VI and VII [5]. However, development of cerebellar neurons is found to be severely impaired. At P8, the dendritic arbors of CAPS2−/− Purkinje cells are reduced to approximately half the average length of those of wild-type Purkinje cells (Fig. 2c). At P28, the thickness of the ML is almost indistinguishable between wild-type and CAPS2−/− cerebella, but less branched dendritic arbors are observed in many CAPS2−/− Purkinje cells (Fig. 2d). The EGL in the CAPS2−/− cerebellum is still thick at P17, when it is barely detectable in the wild-type cerebellum (Fig. 2b), although it is no longer detectable at P28, indicating a delay in granule cell development. In the CAPS2−/− cerebellum at P8, increased cell death is observed in the EGL of lobules VI–VII and lobule IX compared with wild-type cerebellum. Hypoplasia of cerebellar lobules VI and VII was also reported in a subpopulation of autistic patients that underwent magnetic resonance imaging (MRI) [60, 61]. There are considerably fewer calbindin-positive (Calb+) Purkinje cells in primary cultures of CAPS2−/− cerebellum, the number being approximately 41% of that in wild-type cultures. By culturing cells in BDNF-supplemented media, the survival rate of CAPS2−/− Purkinje cells was significantly increased in comparison with cells cultured in media containing no BDNF. A reduction in the number of Calb+ Purkinje cells to about 80% of the normal level was also observed in sections of the P28 cerebellum. These data indicate the functional importance of CAPS2 for cerebellar development.
Impaired Morphology and Electrophysiology in the PF-PC Synapses of CAPS2−/− Mice
In the PF-PC synapses of the CAPS2−/− cerebellum at P15, the vesicles within the PF terminals appear to be located normally around the active zones, but are considerably reduced in number around extrasynaptic sites in comparison with wild-type cerebellum (Fig. 2e) [5]. The terminal boutons of CAPS2−/− PFs are larger than those of wild-type PFs. At 12 weeks postnatal, the morphology of PF-PC synapses becomes indistinguishable between wild-type and CAPS2−/− cerebella, except in lobules VI and VII, in which morphological abnormalities of PF-PC synapses (enlarged presynaptic boutons and divided postsynaptic densities) remain obvious, even in the adult.
There is little difference between wild-type and CAPS2−/− mice in terms of the peak amplitudes of PF excitatory postsynaptic currents (PF-EPSCs). However, paired-pulse facilitation (PPF) of the PF-EPSC is impaired in the PF-PC synapses of CAPS2−/− mice. This impaired presynaptic function is observed in the anterior (lobules II–V), central (lobules VI–VII) (Fig. 2f), and posterior lobes (lobule IX), between P15 and 21, but only in lobules VI–VII between P43 and 54. Thus, lobules VI–VII seem to be more vulnerable to a loss of CAPS2 function. These results indicate that although basic synaptic transmission at PF-PC synapses of CAPS2−/− mice is unimpaired, PPF, a kind of short-term plasticity, is impaired.
Abnormal Cerebellum-Related Behaviors of CAPS2−/− Mice
In terms of coordinated motor performance on a rotarod treadmill test, young CAPS2−/− mice (P28) show low to moderate performance in comparison with their wild-type littermates [5]. However, this impairment tends to be relieved by P56, indicating that CAPS2−/− mice have an impairment of their motor coordination ability that is capable of recovery with advancing age of the animal, by an unknown mechanism that appears to compensate for the loss of CAPS2.
The vestibulo-ocular reflex (VOR) and optokinetic response (OKR) are controlled by the cerebellum. There were no significant differences between wild-type and CAPS2−/− mice in terms of the gains of the horizontal VOR (HVOR) or horizontal OKR (HOKR), and the phases of the HVOR were also almost the same in CAPS2−/− mice and their wild-type littermates. However, the loss of CAPS2 leads to an eye movement disorder characterized by impaired gain and adaptation of the HOKR [5].
Similar Abnormalities of Cerebellar Phenotypes in CAPS2−/−, BDNF−/−, and NT-3−/− Mice
CAPS2−/− mice have phenotypes similar to those observed in BDNF−/− mice (Table 1) [56–58] and cerebellum-specific NT-3−/− mice [59], namely, aberrant arborization of Purkinje cells in BDNF−/− mice [57, 58], a thicker EGL in P17 BDNF−/− mice [56, 57], increased apoptosis and a lobulation deficit in BDNF−/− mice [57] and NT-3−/− mice [59], presynaptic swelling of PF terminals in BDNF−/− mice [58], and impaired PPF at PF-PC synapses in BDNF−/− mice [58]. The enlarged PF terminal boutons of CAPS2−/− mice are also reminiscent of the enlarged presynaptic boutons reported in the hippocampal neurons of TrkB−/− animals [62]. Thus, these morphological and physiological phenotypes of CAPS2−/− mice imply that CAPS2 function is closely correlated with the neurotrophic actions of BDNF and NT-3 in the cerebellum. However, CAPS2−/− mice also exhibit several other phenotypes distinct from those of BDNF and NT-3 mutant mice: BDNF−/− mice have a short life span [56] and NT-3−/− mice die soon after birth [63], whereas CAPS2−/− mice are viable. The distributions of these proteins do not completely overlap throughout the brain, as BDNF in the hypothalamus is co-distributed with CAPS1 rather than with CAPS2 [7]. Therefore, in some brain regions, CAPS1 may be involved in secretion of BDNF and/or NT-3.
Association Between the CAPS2 Gene and Autism
Autism is a severe neurodevelopmental disorder marked by profound disturbances in social, communicative, and behavioral functioning [64, 65]. Epidemiological studies have shown a high prevalence of autism spectrum disorders (three to six affected per 1,000 people) [66]. The concordance rate in monozygotic twins is between 70% and 90% [67], suggesting that autism has a prominent genetic component [68]. One susceptibility locus for autism was mapped to human chromosome 7q31–q33 (autism susceptibility locus 1; AUTS1) [69] and the human CAPS2 gene is located on chromosome 7q31.32 within the AUTS1 locus [6]. The presence of de novo copy number variation owing to a microdeletion of the chromosomal region containing the CAPS2 gene has also been reported in some autistic patients [68]. Moreover, a subgroup of autistic patients exhibits several abnormalities in cerebellar morphology and function, including a significant loss of Purkinje cells [70, 71], hypoplasia of the cerebellar lobules [60, 61, 72] and impaired eye movement [73] and motor coordination [74], most of which are similarly characteristic of CAPS2−/− mice, as described above (also see Table 1). In addition, CAPS2−/− mice display some other cellular and behavioral phenotypes reminiscent of autism, as described below.
Cellular Deficits in the Cerebrum and Hippocampus of CAPS2−/− Mice
In addition to the cerebellum, BDNF release from primary cultures of CAPS2−/− neocortical neurons during 21 DIV was also reduced relative to the levels in wild-type cultures [6]. On the other hand, there was no significant difference in the levels of NT-3 or NGF between wild-type and CAPS2−/− neocortical culture media. Underdevelopment of GABAergic interneurons has been reported in autism patients [75], and differentiation of a subset of neocortical parvalbumin-positive (Pvalb+) GABAergic neurons is regulated by BDNF [56]. At P17, the CAPS2−/− mouse neocortex had significantly fewer Pvalb+ interneurons, but no fewer Calb+ interneurons, than the wild-type neocortex [6]. This decrease in the number of Pvalb+ interneurons in the CAPS2−/− neocortex at P17 could be rescued by BDNF injection at P5.
At P17, the CAPS2−/− hippocampus showed a reduction in the number of Pvalb+ interneurons in comparison with the wild-type hippocampus [6]; this reduction in the number of Pvalb+ interneurons was also reported in BDNF−/− mice [56]. Similarly, the reduced number of Pvalb+ interneurons in the CAPS2−/− hippocampus at P17 could be rescued by BDNF injection at P5, to a level comparable with that in the wild-type hippocampus.
Basic Sensory and Cognitive Functions of CAPS2−/− Mice
CAPS2−/− mice have almost unchanged basal sensory performances in comparison with their wild-type littermates. There is no significant difference between CAPS2−/− and wild-type mice in a visual test (visual recognition of surface to hold under tail suspension), olfactory test (scenting out hidden food), and auditory test (movements to sound stimulus). On the other hand, the Morris water maze test to evaluate the cognitive function of CAPS2−/− mice indicated impairments in spatial learning and memory [6].
Autistic-like Behaviors of CAPS2−/− Mice
Autism is characterized by abnormal behavioral characteristics, including impaired social interaction [64, 65], hyperactivity [76, 77], and augmented anxiety and/or reduced environmental exploration in a novel environment [61, 64, 65], and is frequently accompanied by an abnormal sleep-wake rhythm [78, 79]. Detailed behavioral analyses demonstrated several autistic-like behavioral phenotypes in CAPS2−/− mice [6]. First, social interaction among CAPS2−/− mice was impaired. Pairs of mice of the same genotype (CAPS2+/+ or CAPS2−/−) that had never met were placed in a neutral cage, and the frequency of interactions displayed by CAPS2−/− mice was significantly lower than that displayed by wild-type mice. Second, the CAPS2−/− mice exhibited hyperactivity in their home cages. Third, the CAPS2−/− mice tended to show decreased exploratory behavior and/or increased anxiety in a novel environment. CAPS2−/− mice showed almost normal locomotor activity in an open field in the light cycle and showed no significant anxiety-like behavior in the light/dark transition test in comparison with their wild-type littermates. However, CAPS2−/− mice became less active than wild-type mice when placed in an open field containing a novel object. Fourth, the CAPS2−/− mice had deficits in intrinsic sleep-wake regulation and circadian rhythm under constant dark conditions. In addition, maternal neglect of newborns is a striking feature of CAPS2−/− mothers. Maternal care is an important reciprocal interaction behavior. The newborns of a CAPS2−/− dam and wild-type sire rarely survived beyond postnatal day 1 because of a defect in the maternal behavior of CAPS2−/− mothers.
Aberrant Splicing of CAPS2 in Patients with Autism
Both CAPS2 and CAPS1 are expressed in histamine-positive basophilic leukocytes, a minor cell population in human blood, which enables us to analyze CAPS expression in individuals with autism. RT-PCR analysis of RNA samples from peripheral blood showed a shorter band in some autistic patients (four out of 16 patients tested). Cloning and sequencing analysis showed that the shorter band of 328 bp lacked the complete sequence of exon 3, suggesting aberrant alternative splicing of CAPS2 mRNA (Fig. 3a). By contrast, no exon 3 skipping was found in any of the 24 healthy control subjects tested. It is noteworthy that autistic monozygotic twins both expressed exon 3-skipped CAPS2 mRNA, while none of their parents or healthy family members (father, mother, older brother, and younger sister) was found to express the exon 3-skipped form.
Abnormality of CAPS2 in autistic patients. a Aberrant alternative splicing and single nucleotide polymorphisms (SNPs). Expression of the aberrantly spliced CAPS2 variant. Top wild-type CAPS2; middle aberrantly spliced CAPS2 variant, which has exon 3 skipping (Δexon3), observed in some autistic patients; bottom non-synonymous SNPs detected in the CAPS2 genes of autistic patients. b Impaired axonal distribution of CAPS2Δexon3 in autistic individuals. Subcellular localization of C-terminal HA-tagged CAPS2 (left) and CAPS2Δexon3 (right) proteins exogenously expressed in cerebellar primary cultures (7DIV). Immunostaining signals for HA-CAPS2 (white) were localized in soma and dendrites immunostained with MAP2ab (grey), but also in axons (long MAPab-negative neurites), whereas those for HA-CAPS2Δexon3 were not localized in axons. Scale bars 50 µm. c Schematic representation of CAPS2-mediated BDNF release from a neuron. Top local BDNF release from the somato-dendritic domain and presynaptic domain in wild-type neurons. Middle defect in BDNF release from CAPS2−/− neurons. Bottom impaired presynaptic local BDNF release from autistic CAPS2Δexon3-expressing neurons, which may cause a disturbance in functional neural circuit development and/or connectivity
Although the mechanism underlying exon 3 skipping remains elusive, no mutations were found around the splice donor site, acceptor site, or branchpoint in the genomic sequences of these patients. In addition to these authentic cis-elements for splicing, additional cis-elements, known as exonic or intronic splicing enhancers/silencers, regulate splicing events in some cases. Thus the possibility of a “cis-hypothesis” cannot be ruled out because mutation(s) might still be present somewhere in the unexplored genomic region within the CAPS2 genes of these patients. Alternatively, a “trans-hypothesis” may explain the underlying causality: for example, a polymorphism(s) in an unknown gene(s), which alters the splicing pattern of CAPS2 mRNA. Other potential mechanisms for pathological alternative splicing of CAPS2 in autistic patients include aberrant epigenetic regulation and de novo mutation. Future extensive studies are warranted to expose the precise pathological mechanisms.
The Exon 3-skipped CAPS2 Variant in Autistic Individuals has BDNF Releasing Activity, But is not Transported into Axons
The exon 3-skipped CAPS2 variant contains a predicted deletion of 111 amino-acid residues in the human CAPS2 protein. However, it retains almost normal BDNF releasing activity when exogenously expressed in either PC12 cells or neocortical primary cultures derived from CAPS2−/− mice [6]. On the other hand, the region encoded by exon 3 was found to be involved in the protein–protein interaction between CAPS2 and p150Glued, which is a subunit of the dynactin complex and known to be associated with bidirectional axonal transport [80, 81]. Exon 3-skipped CAPS2 expressed in either neocortical or cerebellar primary cultures was absent from axons, indicating a deficiency in the proper axonal transport of exon 3-skipped CAPS2 (Fig. 3b). These results suggest the loss of local synaptic BDNF release owing to the impaired transport of exon 3-skipped CAPS2 to the axon terminal (Fig. 3c).
Single-nucleotide Polymorphisms in the CAPS2 Gene in Individuals with Autism
By analyzing the CAPS2-coding sequence of 252 Caucasian autistic patients, seven non-synonymous SNPs were identified (Fig. 3a) [6]. These SNPs were not detected in the control subjects tested or in the public SNP databases available to date. Although the correlation between these SNPs and the pathology in autism remains to be studied, possible CAPS2 disturbance resulting from genetic/allelic heterogeneities owing to SNPs may predispose individuals to a higher risk for autism, at least in some cases.
Conclusion
Many brain development genes must be involved in the development of mouse cerebellar circuits. The spatio-temporal gene expression profile data systematized in the CDT-DB are a useful foundation not only to elucidate the underlying genetic basis of cerebellar development, but also to identify brain development genes. CAPS2 was identified as a developmentally regulated gene. CAPS2 is involved in BDNF and NT-3 release. A CAPS2−/− mouse study indicated that CAPS2-mediated BDNF and NT-3 release is indispensable for cerebellum-related cellular, morphological, physiological, and behavioral phenotypes, some of which are reminiscent of the cerebellar deficits observed in autistic patients. Moreover, a subset of autistic patients expresses aberrant non-axonal CAPS2 variants, owing to seven non-synonymous SNPs. The current data suggest that disturbances in localized (axonal) BDNF release caused by aberrant CAPS2 contribute to the abnormal circuit connectivity observed in autistic patients. Although autism is thought to be a neurodevelopmental disorder caused by the interaction of multiple susceptibility genes and/or environmental influences [68], CAPS2−/− mice will provide us with a useful model system in which to study some aspects of the neuropathology and behaviors of autistic patients.
References
Sato A, Sekine Y, Saruta C, Nishibe H, Morita N, Sato Y, Sadakata T, Shinoda Y, Kojima T, Furuichi T (2008) Cerebellar development transcriptome database (CDT-DB): profiling of spatio-temporal gene expression during the postnatal development of mouse cerebellum. Neural Netw 21:1056–1069
Sadakata T, Furuichi T (2006) Identification and mRNA expression of Ogdh, QP-C, and two predicted genes in the postnatal mouse brain. Neurosci Lett 405:217–222
Sadakata T, Mizoguchi A, Sato Y, Katoh-Semba R, Fukuda M, Mikoshiba K, Furuichi T (2004) The secretory granule-associated protein CAPS2 regulates neurotrophin release and cell survival. J Neurosci 24:43–52
Sadakata T, Washida M, Furuichi T (2007) Alternative splicing variations in mouse CAPS2: differential expression and functional properties of splicing variants. BMC Neurosci 8:25
Sadakata T, Kakegawa W, Mizoguchi A, Washida M, Katoh-Semba R, Shutoh F, Okamoto T, Nakashima H, Kimura K, Tanaka M, Sekine Y, Itohara S, Yuzaki M, Nagao S, Furuichi T (2007) Impaired cerebellar development and function in mice lacking CAPS2, a protein involved in neurotrophin release. J Neurosci 27:2472–2482
Sadakata T, Washida M, Iwayama Y, Shoji S, Sato Y, Ohkura T, Katoh-Semba R, Nakajima M, Sekine Y, Tanaka M, Nakamura K, Iwata Y, Tsuchiya KJ, Mori N, Detera-Wadleigh SD, Ichikawa H, Itohara S, Yoshikawa T, Furuichi T (2007) Autistic-like phenotypes in Cadps2-knockout mice and aberrant CADPS2 splicing in autistic patients. J Clin Invest 117:931–943
Sadakata T, Itakura M, Kozaki S, Sekine Y, Takahashi M, Furuichi T (2006) Differential distributions of the Ca2+-dependent activator protein for secretion family proteins (CAPS2 and CAPS1) in the mouse brain. J Comp Neurol 495:735–753
Shiraishi-Yamaguchi Y, Furuichi T (2007) The Homer family proteins. Genome Biol 8:206
Shiraishi Y, Mizutani A, Bito H, Fujisawa K, Narumiya S, Mikoshiba K, Furuichi T (1999) Cupidin, an isoform of Homer/Vesl, interacts with the actin cytoskeleton and activated rho family small GTPases and is expressed in developing mouse cerebellar granule cells. J Neurosci 19:8389–8400
Huang J, Sakai R, Furuichi T (2006) The docking protein Cas links tyrosine phosphorylation signaling to elongation of cerebellar granule cell axons. Mol Biol Cell 17:3187–3196
Huang J, Furuya A, Furuichi T (2007) Very-KIND, a KIND domain containing RasGEF, controls dendrite growth by linking Ras small GTPases and MAP2. J Cell Biol 179:539–552
Yoshikawa F, Sato Y, Tohyama K, Akagi T, Hashikawa T, Nagakura-Takagi Y, Sekine Y, Morita N, Baba H, Suzuki Y, Sugano S, Sato A, Furuichi T (2008) Opalin, a transmembrane sialylglycoprotein located in the central nervous system myelin paranodal loop membrane. J Biol Chem 283:20830–20840
Aruga J, Yoshikawa F, Nozaki Y, Sakaki Y, Toyoda A, Furuichi T (2007) An oligodendrocyte enhancer in a phylogenetically conserved intron region of the mammalian myelin gene Opalin. J Neurochem 102:1533–1547
Speidel D, Varoqueaux F, Enk C, Nojiri M, Grishanin RN, Martin TF, Hofmann K, Brose N, Reim K (2003) A family of Ca2+-dependent activator proteins for secretion: comparative analysis of structure, expression, localization, and function. J Biol Chem 278:52802–52809
Cisternas FA, Vincent JB, Scherer SW, Ray PN (2003) Cloning and characterization of human CADPS and CADPS2, new members of the Ca2+-dependent activator for secretion protein family. Genomics 81:279–291
Berwin B, Floor E, Martin TF (1998) CAPS (mammalian UNC-31) protein localizes to membranes involved in dense-core vesicle exocytosis. Neuron 21:137–145
Tandon A, Bannykh S, Kowalchyk JA, Banerjee A, Martin TF, Balch WE (1998) Differential regulation of exocytosis by calcium and CAPS in semi-intact synaptosomes. Neuron 21:147–154
Renden R, Berwin B, Davis W, Ann K, Chin CT, Kreber R, Ganetzky B, Martin TF, Broadie K (2001) Drosophila CAPS is an essential gene that regulates dense-core vesicle release and synaptic vesicle fusion. Neuron 31:421–437
Ann K, Kowalchyk JA, Loyet KM, Martin TF (1997) Novel Ca2+-binding protein (CAPS) related to UNC-31 required for Ca2+-activated exocytosis. J Biol Chem 272:19637–19640
Elhamdani A, Martin TF, Kowalchyk JA, Artalejo CR (1999) Ca2+-dependent activator protein for secretion is critical for the fusion of dense-core vesicles with the membrane in calf adrenal chromaffin cells. J Neurosci 19:7375–7383
Waselle L, Gerona RR, Vitale N, Martin TF, Bader MF, Regazzi R (2005) Role of phosphoinositide signaling in the control of insulin exocytosis. Mol Endocrinol 19:3097–3106
Speidel D, Salehi A, Obermueller S, Lundquist I, Brose N, Renstrom E, Rorsman P (2008) CAPS1 and CAPS2 regulate stability and recruitment of insulin granules in mouse pancreatic beta cells. Cell Metab 7:57–67
Rybkin II, Kim MS, Bezprozvannaya S, Qi X, Richardson JA, Plato CF, Hill JA, Bassel-Duby R, Olson EN (2007) Regulation of atrial natriuretic peptide secretion by a novel Ras-like protein. J Cell Biol 179:527–537
Eisenhofer G, Huynh TT, Elkahloun A, Morris JC, Bratslavsky G, Linehan WM, Zhuang Z, Balgley BM, Lee CS, Mannelli M, Lenders JW, Bornstein SR, Pacak K (2008) Differential expression of the regulated catecholamine secretory pathway in different hereditary forms of pheochromocytoma. Am J Physiol Endocrinol Metab 295:E1223–1233
Grishanin RN, Klenchin VA, Loyet KM, Kowalchyk JA, Ann K, Martin TF (2002) Membrane association domains in Ca2+-dependent activator protein for secretion mediate plasma membrane and dense-core vesicle binding required for Ca2+-dependent exocytosis. J Biol Chem 277:22025–22034
Grishanin RN, Kowalchyk JA, Klenchin VA, Ann K, Earles CA, Chapman ER, Gerona RR, Martin TF (2004) CAPS acts at a prefusion step in dense-core vesicle exocytosis as a PIP2 binding protein. Neuron 43:551–562
Osborne SL, Wallis TP, Jimenez JL, Gorman JJ, Meunier FA (2007) Identification of secretory granule phosphatidylinositol 4,5-bisphosphate-interacting proteins using an affinity pulldown strategy. Mol Cell Proteomics 6:1158–1169
James DJ, Khodthong C, Kowalchyk JA, Martin TF (2008) Phosphatidylinositol 4,5-bisphosphate regulates SNARE-dependent membrane fusion. J Cell Biol 182:355–366
Speese S, Petrie M, Schuske K, Ailion M, Ann K, Iwasaki K, Jorgensen EM, Martin TF (2007) UNC-31 (CAPS) is required for dense-core vesicle but not synaptic vesicle exocytosis in Caenorhabditis elegans. J Neurosci 27:6150–6162
Hammarlund M, Watanabe S, Schuske K, Jorgensen EM (2008) CAPS and syntaxin dock dense core vesicles to the plasma membrane in neurons. J Cell Biol 180:483–491
Speidel D, Bruederle CE, Enk C, Voets T, Varoqueaux F, Reim K, Becherer U, Fornai F, Ruggieri S, Holighaus Y, Weihe E, Bruns D, Brose N, Rettig J (2005) CAPS1 regulates catecholamine loading of large dense-core vesicles. Neuron 46:75–88
Brunk I, Blex C, Speidel D, Brose N, Ahnert-Hilger G (2008) Ca2+-dependent activator proteins of secretion promote vesicular monoamine uptake. J Biol Chem 284:1050–1056
Jockusch WJ, Speidel D, Sigler A, Sorensen JB, Varoqueaux F, Rhee JS, Brose N (2007) CAPS-1 and CAPS-2 are essential synaptic vesicle priming proteins. Cell 131:796–808
Fujita Y, Xu A, Xie L, Arunachalam L, Chou TC, Jiang T, Chiew SK, Kourtesis J, Wang L, Gaisano HY, Sugita S (2007) Ca2+-dependent activator protein for secretion 1 is critical for constitutive and regulated exocytosis but not for loading of transmitters into dense core vesicles. J Biol Chem 282:21392–21403
Zhou KM, Dong YM, Ge Q, Zhu D, Zhou W, Lin XG, Liang T, Wu ZX, Xu T (2007) PKA activation bypasses the requirement for UNC-31 in the docking of dense core vesicles from C. elegans neurons. Neuron 56:657–669
Cai T, Fukushige T, Notkins AL, Krause M (2004) Insulinoma-Associated Protein IA-2, a Vesicle Transmembrane Protein, Genetically Interacts with UNC-31/CAPS and Affects Neurosecretion in Caenorhabditis elegans. J Neurosci 24:3115–3124
Gracheva EO, Burdina AO, Touroutine D, Berthelot-Grosjean M, Parekh H, Richmond JE (2007) Tomosyn negatively regulates CAPS-dependent peptide release at Caenorhabditis elegans synapses. J Neurosci 27:10176–10184
Binda AV, Kabbani N, Levenson R (2005) Regulation of dense core vesicle release from PC12 cells by interaction between the D2 dopamine receptor and calcium-dependent activator protein for secretion (CAPS). Biochem Pharmacol 69:1451–1461
Charlie NK, Schade MA, Thomure AM, Miller KG (2006) Presynaptic UNC-31 (CAPS) is required to activate the Gαs pathway of the Caenorhabditis elegans synaptic signaling network. Genetics 172:943–961
Austin MC, Schultzberg M, Abbott LC, Montpied P, Evers JR, Paul SM, Crawley JN (1992) Expression of tyrosine hydroxylase in cerebellar Purkinje neurons of the mutant tottering and leaner mouse. Brain Res Mol Brain Res 15:227–240
Takada M, Sugimoto T, Hattori T (1993) Tyrosine hydroxylase immunoreactivity in cerebellar Purkinje cells of the rat. Neurosci Lett 150:61–64
Verney C, Grzanna R, Farkas E (1982) Distribution of dopamine-β-hydroxylase-like immunoreactive fibers in the rat cerebellar cortex during ontogeny. Dev Neurosci 5:369–374
Fernandes ML, Saad MJ, Velloso LA (2001) Effects of age on elements of insulin-signaling pathway in central nervous system of rats. Endocrine 16:227–234
de LAFML, Saad MJ, Velloso LA (1999) Insulin induces tyrosine phosphorylation of the insulin receptor and SHC, and SHC/GRB2 association in cerebellum but not in forebrain cortex of rats. Brain Res 826:74–82
Sherrard RM, Bower AJ (2002) Climbing fiber development: do neurotrophins have a part to play? Cerebellum 1:265–275
Katoh-Semba R, Takeuchi IK, Semba R, Kato K (1997) Distribution of brain-derived neurotrophic factor in rats and its changes with development in the brain. J Neurochem 69:34–42
Katoh-Semba R, Takeuchi IK, Semba R, Kato K (2000) Neurotrophin-3 controls proliferation of granular precursors as well as survival of mature granule neurons in the developing rat cerebellum. J Neurochem 74:1923–1930
Das KP, Chao SL, White LD, Haines WT, Harry GJ, Tilson HA, Barone S Jr (2001) Differential patterns of nerve growth factor, brain-derived neurotrophic factor and neurotrophin-3 mRNA and protein levels in developing regions of rat brain. Neuroscience 103:739–761
Lindholm D, Hamner S, Zirrgiebel U (1997) Neurotrophins and cerebellar development. Perspect Dev Neurobiol 5:83–94
Lindholm D, Castren E, Tsoulfas P, Kolbeck R, Berzaghi Mda P, Leingartner A, Heisenberg CP, Tessarollo L, Parada LF, Thoenen H (1993) Neurotrophin-3 induced by tri-iodothyronine in cerebellar granule cells promotes Purkinje cell differentiation. J Cell Biol 122:443–450
Segal RA, Pomeroy SL, Stiles CD (1995) Axonal growth and fasciculation linked to differential expression of BDNF and NT3 receptors in developing cerebellar granule cells. J Neurosci 15:4970–4981
Gao WQ, Zheng JL, Karihaloo M (1995) Neurotrophin-4/5 (NT-4/5) and brain-derived neurotrophic factor (BDNF) act at later stages of cerebellar granule cell differentiation. J Neurosci 15:2656–2667
Larkfors L, Lindsay RM, Alderson RF (1996) Characterization of the responses of Purkinje cells to neurotrophin treatment. J Neurochem 66:1362–1373
Doughty ML, Lohof A, Campana A, Delhaye-Bouchaud N, Mariani J (1998) Neurotrophin-3 promotes cerebellar granule cell exit from the EGL. Eur J Neurosci 10:3007–3011
Zhou P, Porcionatto M, Pilapil M, Chen Y, Choi Y, Tolias KF, Bikoff JB, Hong EJ, Greenberg ME, Segal RA (2007) Polarized signaling endosomes coordinate BDNF-induced chemotaxis of cerebellar precursors. Neuron 55:53–68
Jones KR, Farinas I, Backus C, Reichardt LF (1994) Targeted disruption of the BDNF gene perturbs brain and sensory neuron development but not motor neuron development. Cell 76:989–999
Schwartz PM, Borghesani PR, Levy RL, Pomeroy SL, Segal RA (1997) Abnormal cerebellar development and foliation in BDNF−/− mice reveals a role for neurotrophins in CNS patterning. Neuron 19:269–281
Carter AR, Chen C, Schwartz PM, Segal RA (2002) Brain-derived neurotrophic factor modulates cerebellar plasticity and synaptic ultrastructure. J Neurosci 22:1316–1327
Bates B, Rios M, Trumpp A, Chen C, Fan G, Bishop JM, Jaenisch R (1999) Neurotrophin-3 is required for proper cerebellar development. Nat Neurosci 2:115–117
Courchesne E, Yeung-Courchesne R, Press GA, Hesselink JR, Jernigan TL (1988) Hypoplasia of cerebellar vermal lobules VI and VII in autism. N Engl J Med 318:1349–1354
Pierce K, Courchesne E (2001) Evidence for a cerebellar role in reduced exploration and stereotyped behavior in autism. Biol Psychiatry 49:655–664
Martinez A, Alcantara S, Borrell V, Del Rio JA, Blasi J, Otal R, Campos N, Boronat A, Barbacid M, Silos-Santiago I, Soriano E (1998) TrkB and TrkC signaling are required for maturation and synaptogenesis of hippocampal connections. J Neurosci 18:7336–7350
Ernfors P, Lee KF, Kucera J, Jaenisch R (1994) Lack of neurotrophin-3 leads to deficiencies in the peripheral nervous system and loss of limb proprioceptive afferents. Cell 77:503–512
World Health Organization (1992) The ICD-10 classification of mental and behavioural disorders: clinical descriptions and diagnostic guidelines. WHO, Geneva
American Psychiatric Association., American Psychiatric Association. Task Force on DSM-IV (1994) Diagnostic and statistical manual of mental disorders: DSM-IV, 4th edn. American Psychiatric Association, Washington, DC
Muhle R, Trentacoste SV, Rapin I (2004) The genetics of autism. Pediatrics 113:e472–486
Folstein SE, Rosen-Sheidley B (2001) Genetics of autism: complex aetiology for a heterogeneous disorder. Nat Rev Genet 2:943–955
Abrahams BS, Geschwind DH (2008) Advances in autism genetics: on the threshold of a new neurobiology. Nat Rev Genet 9:341–355
IMGSAC (2001) Further characterization of the autism susceptibility locus AUTS1 on chromosome 7q. Hum Mol Genet 10:973–982
Bauman M, Kemper TL (1985) Histoanatomic observations of the brain in early infantile autism. Neurology 35:866–874
Whitney ER, Kemper TL, Bauman ML, Rosene DL, Blatt GJ (2008) Cerebellar Purkinje cells are reduced in a subpopulation of autistic brains: a stereological experiment using calbindin-D28k. Cerebellum 7:406–416
Courchesne E, Pierce K, Schumann CM, Redcay E, Buckwalter JA, Kennedy DP, Morgan J (2007) Mapping early brain development in autism. Neuron 56:399–413
Takarae Y, Minshew NJ, Luna B, Krisky CM, Sweeney JA (2004) Pursuit eye movement deficits in autism. Brain 127:2584–2594
Manjiviona J, Prior M (1995) Comparison of Asperger syndrome and high-functioning autistic children on a test of motor impairment. J Autism Dev Disord 25:23–39
Casanova MF, Buxhoeveden D, Gomez J (2003) Disruption in the inhibitory architecture of the cell minicolumn: implications for autisim. Neuroscientist 9:496–507
Aman MG (2004) Management of hyperactivity and other acting-out problems in patients with autism spectrum disorder. Semin Pediatr Neurol 11:225–228
Aman MG, Langworthy KS (2000) Pharmacotherapy for hyperactivity in children with autism and other pervasive developmental disorders. J Autism Dev Disord 30:451–459
Richdale AL, Prior MR (1995) The sleep/wake rhythm in children with autism. Eur Child Adolesc Psychiatry 4:175–186
Filipek PA, Accardo PJ, Ashwal S, Baranek GT, Cook EH Jr., Dawson G, Gordon B, Gravel JS, Johnson CP, Kallen RJ, Levy SE, Minshew NJ, Ozonoff S, Prizant BM, Rapin I, Rogers SJ, Stone WL, Teplin SW, Tuchman RF, Volkmar FR (2000) Practice parameter: screening and diagnosis of autism: report of the Quality Standards Subcommittee of the American Academy of Neurology and the Child Neurology Society. Neurology 55:468–479
Deacon SW, Serpinskaya AS, Vaughan PS, Lopez Fanarraga M, Vernos I, Vaughan KT, Gelfand VI (2003) Dynactin is required for bidirectional organelle transport. J Cell Biol 160:297–301
Waterman-Storer CM, Karki SB, Kuznetsov SA, Tabb JS, Weiss DG, Langford GM, Holzbaur EL (1997) The interaction between cytoplasmic dynein and dynactin is required for fast axonal transport. Proc Natl Acad Sci U S A 94:12180–12185
Acknowledgments
This study was supported by grants-in-aid for Scientific Research from the Japanese Ministry of Education, Culture, Sports, Science, and Technology (MEXT), the Japan Science and Technology Agency (JST), the Japan Society for the Promotion of Science (JSPS), and the Institute of Physical and Chemical Research (RIKEN).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sadakata, T., Furuichi, T. Developmentally Regulated Ca2+-Dependent Activator Protein for Secretion 2 (CAPS2) is Involved in BDNF Secretion and is Associated with Autism Susceptibility. Cerebellum 8, 312–322 (2009). https://doi.org/10.1007/s12311-009-0097-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-009-0097-5