Abstract
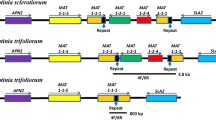
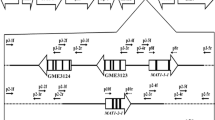
Homothallic Didymella zeae-maydis undergoes sexual reproduction by selfing. Sequence analysis of the mating type (MAT) locus from this fungus revealed that MAT carries both MAT1-1-1 and MAT1-2-1 genes found in heterothallic Dothideomycetes, separated by ∼1.0 kb of noncoding DNA. To understand the mechanistic basis of homothallism in D. zeae-maydis, each of the MAT genes was deleted and the effects on selfing and on ability to cross in a heterothallic manner were determined. The strain carrying an intact MAT1-1-1 but defective MAT1-2-1 gene (MAT1-1-1;ΔMAT1-2-1) was self-sterile, however strains carrying an intact MAT1-2-1 but defective MAT1-1-1 gene (ΔMAT1-1-1;MAT1-2-1), when selfed, showed delayed production of a few ascospores. Attempts to cross the two MAT deletion strains yielded fewer ΔMAT1-1-1;MAT1-2-1 than MAT1-1-1;ΔMAT1-2-1 progeny and very few ascospores overall compared to WT selfs. This study demonstrates that, as in the other homothallic Dothideomycetes, both MAT genes are required for full fertility, but that, in contrast to other cases, the presence of a single MAT1-2-1 gene can induce homothallism, albeit inefficiently, in D. zeae-maydis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ait Benkhali, J., Coppin, E., Brun, S., Peraza-Reyes, L., Martin, T., Dixelius, C., Lazar, N., van Tilbeurgh, H., and Debuchy, R. 2013. A network of HMG-box transcription factors regulates sexual cycle in the fungus Podospora anserina. PLoS Genet.9, e1003642.
Altschul, S.F., Gish, W., Miller, W., Myers, E.W., and Lipman, D.J. 1990. Basic local alignment search tool. J. Mol. Biol.215, 403–410.
Arie, T., Christiansen, S.K., Yoder, O.C., and Turgeon, B.G. 1997. Efficient cloning of ascomycete mating type genes by PCR amplification of the conserved MAT HMG box. Fungal Genet. Biol.21, 118–130.
Arie, T., Kaneko, I., Yoshida, T., Noguchi, M., Nomura, Y., and Yamaguchi, I. 2000. Mating-type genes from asexual phytopathogenic ascomycetes Fusarium oxysporum and Alternaria alternata. Mol. Plant-Microbe Interact.13, 1330–1339.
Aveskamp, M.M., de Gruyter, J., Woudenberg, J.H.C., Verkley, G.J.M., and Crous, P.W. 2010. Highlights of the Didymellaceae: A polyphasic approach to characterise Phoma and related pleosporalean genera. Stud. Mycol.65, 1–60.
Bennett, R.S., Yun, S.H., Lee, T.Y., Turgeon, B.G., Arseniuk, E., Cunfer, B.M., and Bergstrom, G.C. 2003. Identity and conservation of mating type genes in geographically diverse isolates of Phaeosphaeria nodorum. Fungal Genet. Biol.40, 25–37.
Bustin, M. 2001. Revised nomencalture for high mobility group (HMG) chromosomal proteins. Trends Biochem. Sci.26, 152–153.
Butler, G. 2007. The Evolution of MAT: The Ascomycetes, pp. 3–18. In Heitman, J., Kronstad, J.W., Taylor, J.W., and Casselton, L.A. (eds.), Sex in Fungi: Molecular Determination and Evolutionary Implications, ASM Press, Washington, D.C., USA.
Chi, M.H., Park, S.Y., and Lee, Y.H. 2009. A quick and safe method for fungal DNA extraction. Plant Pathology J.25, 108–111.
Comstock, J.C., Martinson, C.A., and Gengenbach, B.G. 1973. Host specificity of a toxin from Phyllosticta maydis for Texas cytoplasmically male sterile maize. Phytopathology63, 1357–1360.
Conde-Ferraez, L., Waalwijk, C., Canto-Canche, B.B., Kema, G.H., Crous, P.W., James, A.C., and Abeln, E.C. 2007. Isolation and characterization of the mating type locus of Mycosphaerella fijiensis, the causal agent of black leaf streak disease of banana. Mol. Plant Pathol.8, 111–120.
Condon, B.J. 2013. Genomic and molecular genetic analyses of secondary metabolism, toxin production, and iron homeostasis in Cochliobolus heterostrophus. Ph.D. thesis. Cornell Universiy, Ithaca, NY 14853, USA.
Danko, S.J., Kono, Y., Daly, J.M., Suzuki, Y., Takeuchi, S., and McCrery, D.A. 1984. Structural and biological activity of a host-specific toxin produced by the fungal corn pathogen Phyllosticta maydis. Biochemistry23, 759–766.
Debuchy, R., Berteaux-Lecellier, V., and Silar, P. 2010. Mating systems and sexual morphogenesis in ascomycetes, pp. 501–535. In Borkovich, K.A. (ed.), Cellular and Molecular Biology of Filamentous Fungi, ASM Press, Washington, D.C., USA.
Debuchy, R. and Turgeon, B. 2006. Mating-type structure, evolution, and function in Euascomycetes, pp. 293–323. In Kues, U. and Fischer, R. (eds.), The Mycota, Springer, Berlin, Germany.
Herskowitz, I. 1988. Life cycle of the budding yeast Saccharomyces cerevisiae. Microbiol. Rev.52, 536–553.
Herskowitz, I. 1989. A regulatory hierarchy for cell specialization in yeast. Nature342, 749–757.
Inderbitzin, P., Harkness, J., Turgeon, B.G., and Berbee, M.L. 2005. Lateral transfer of mating system in Stemphylium. Proc. Natl. Acad. Sci. USA102, 11390–11395.
Kim, H.K., Cho, E.J., Lee, S., Lee, Y.S., and Yun, S.H. 2012. Functional analyses of individual mating-type transcripts at MAT loci in Fusarium graminearum and Fusarium asiaticum. FEMS Microbiol. Lett.337, 89–96.
Klix, V., Nowrousian, M., Ringelberg, C., Loros, J.J., Dunlap, J.C., and Pöggeler, S. 2010. Functional characterization of MAT1-1-specific mating-type genes in the homothallic ascomycete Sordaria macrospora provides new insights into essential and nonessential sexual regulators. Eukaryot. Cell9, 894–905.
Kono, Y., Danko, S.J., Suzuki, J., Takeuchi, S., and Daly, J.M. 1983. Structure of the host-specific pathotoxins produced by Phyllosticta maydis. Tetrahedron Lett.24, 3803–3806.
Leach, J., Lang, B.R., and Yoder, O.C. 1982. Methods for selection of mutants and in vitro culture of Cochliobolous heterostrophus. J. Gen. Microbiol.128, 1719–1729.
Lee, J., Lee, T., Lee, Y.W., Yun, S.H., and Turgeon, B.G. 2003. Shifting fungal reproductive mode by manipulation of mating type genes: obligatory heterothallism of Gibberella zeae. Mol. Microbiol.50, 145–152.
Liu, Y.G. and Whittier, R.F. 1995. Thermal asymmetric interlaced PCR: automatable amplification and sequencing of insert end fragments from P1 and YAC clones for chromosome walking. Genomics25, 674–681.
Lu, S.W., Yun, S.H., Lee, T., and Turgeon, B.G. 2011. Altering sexual reproductive mode by interspecific exchange of MAT loci. Fungal Genet. Biol.48, 714–724.
Martin, T., Lu, S.W., van Tilbeurgh, H., Ripoll, D.R., Dixelius, C., Turgeon, B.G., and Debuchy, R. 2010. Tracing the origin of the fungal alpha1 domain places its ancestor in the HMG-box superfamily: implication for fungal mating-type evolution. PLoS One5, e15199.
McFeeley, J.C. 1971. Comparison of isolates causing yellow leaf blight of corn in Ohio. Plant Dis. Rep.55, 1064–1068.
Morita, A., Saitoh, Y., Izumitsu, K., and Tanaka, C. 2012. Molecular organization of the mating type (MAT) locus of Exserohilum monoceras (Setosphaeria monoceras), a bioherbicide agent for Echinochloa weeds. Mycoscience53, 92–101.
Mukunya, D.M. and Boothroyd, C.W. 1973. Mycosphaerella zeae-maydis sp. n., the sexual stage of Phyllosticta maydis. Phytopathology63, 529–532.
Sambrook, J. and Russell, D.W. 2001. Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, Plainview, USA.
Soullier, S., Jay, P., Poulat, F., Vanacker, J.M., Berta, P., and Laudet, V. 1999. Diversification pattern of the HMG and SOX family members during evolution. J. Mol. Evol.48, 517–527.
Turgeon, B.G., Bohlmann, H., Ciuffetti, L.M., Christiansen, S.K., Yang, G., Schöder, W., and Yoder, O.C. 1993. Cloning and analysis of the mating type genes from Cochliobolus heterostrophus. Mol. Gen. Genet.238, 270–284.
Turgeon, B.G. and Yoder, O.C. 2000. Proposed nomenclature for mating type genes of filamentous ascomycetes. Fungal Genet. Biol.31, 1–5.
von Arx, J.A. 1987. Plant pathogenic fungi. Beih. Nova Hedwigia.87, 1–288.
Waalwijk, C., Mendes, O., Verstappen, E.C., de Waard, M.A., and Kema, G.H. 2002. Isolation and characterization of the mating-type idiomorphs from the wheat septoria leaf blotch fungus Mycosphaerella graminicola. Fungal Genet. Biol.35, 277–286.
Whittle, C.A. and Johannesson, H. 2011. Evolution of mating-type loci and mating-type chromosomes in model species of filamentous ascomycetes. In Wöstemeyer, J. and Pöeggeler, S. (eds.), THE MYCOTA XIV, Evolution of fungi and fungal-like organisms, Springer Verlag, Heidelberg, Germany.
Wik, L., Karlsson, M., and Johannesson, H. 2008. The evolutionary trajectory of the mating-type (mat) genes in Neurospora relates to reproductive behavior of taxa. BMC Evol. Biol.8, 109.
Wirsel, S., Horwitz, B., Yamaguchi, K., Yoder, O.C., and Turgeon, B. 1998. Single mating type-specific genes and their 3 UTRs control mating and fertility in Cochliobolus heterostrophus. Mol. Gen. Genet.259, 272–281.
Wirsel, S., Turgeon, B.G., and Yoder, O.C. 1996. Deletion of the Cochliobolus heterostrophus mating-type (MAT) locus promotes the function of MAT transgenes. Curr. Genet.29, 241–249.
Woudenberg, J.H., De Gruyter, J., Crous, P.W., and Zwiers, L.H. 2012. Analysis of the mating-type loci of co-occurring and phylogenetically related species of Ascochyta and Phoma. Mol. Plant Pathol.13, 350–362.
Yoder, O.C. 1973. A selective toxin produced by Phyllosticta maydis. Phytopathology63, 1361–1365.
Yoder, O.C., Macko, V., Wolpert, T.J., and Turgeon, B.G. 1997. Cochliobolus spp. and their host-selective toxins, pp. 145–166. In Carroll, G. and Tudzynski, P. (eds.), The Mycota Vol. 5: Plant Relationships, Part A, Sringer-Verlag, Berlin, Germany.
Yun, S.H., Berbee, M.L., Yoder, O.C., and Turgeon, B.G. 1999. Evolution of the fungal self-fertile reproductive life style from self-sterile ancestors. Proc. Natil. Acad. Sci. USA96, 5592–5597.
Yun, S.H., Turgeon, B.G., and Yoder, O.C. 1998. REMI-induced mutants of Mycosphaerella zeae-maydis lacking the polyketide PM-toxin are deficient in pathogenesis to corn. Physiol. Mol. Plant Pathol.52, 53–66.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplemental material for this article may be found at http://www.springerlink.com/content/120956.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Yun, SH., Yoder, O.C. & Gillian Turgeon, B. Structure and function of the mating-type locus in the homothallic ascomycete, Didymella zeae-maydis . J Microbiol. 51, 814–820 (2013). https://doi.org/10.1007/s12275-013-3465-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-013-3465-2