Abstract
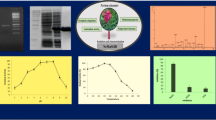
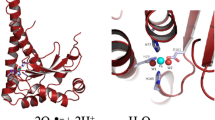
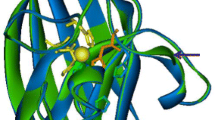
A superoxide dismutase (SOD) gene of Thermoascus aurantiacus var. levisporus, a thermophilic fungus, was cloned, sequenced, and expressed in Pichia pastoris and its gene product was characterized. The coding sequence predicted a 231 residues protein with a unique 35 amino acids extension at the N-terminus indicating a mitochondrial-targeting sequence. The content of Mn was 2.46 μg/mg of protein and Fe was not detected in the purified enzyme. The enzyme was found to be inhibited by NaN3, but not by KCN or H2O2. These results suggested that the SOD in Thermoascus aurantiacus var. levisporus was the manganese superoxide dismutase type. In comparison with other MnSODs, all manganese-binding sites were also conserved in the sequence (H88, H136, D222, H226). The molecular mass of a single band of the enzyme was estimated to be 21.7 kDa. The protein was expressed in tetramer form with molecular weight of 68.0 kDa. The activity of purified protein was 2,324 U/mg. The optimum temperature of the enzyme was 55°C and it exhibited maximal activity at pH 7.5. The enzyme was thermostable at 50 and 60°C and the half-life at 80°C was approximately 40 min.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Amo, T., H. Atomi, and T. Imanaka. 2003. Biochemical properties and regulated gene expression of the superoxide dismutase from the facultatively aerobic hyperthermophile Pyrobaculum calidifontis. J. Bacteriol. 185, 6340–6347.
Angelova, M., P. Dolashka-Angelova, E. Ivanova, J. Serkedjieva, L. Slokoska, S. Pashova, R. Toshkova, S. Vassilev, I. Simeonov, H-J. Hartman, S. Stoeva, U. Weser, and W. Voelter. 2001. A novel glycosylated Cu/Zn-containing superoxide dismutase: production and potential therapeutic effect. Microbiology 147, 1641–1650.
Bannister, J.V., W.H. Bannieter, and G. Rotiliom. 1987. Aspects of the structure, function and application of superoxide dismutase. CRC. Crit. Rev. Biochem. 22, 111–180.
Beauchamp, C. and I. Fridovich. 1971. Superoxide dismutase: Improved assays and an assay applicable to acrylamide gels. Anal. Biochem. 44, 276–287.
Bell, K. and V. Smith. 1994. Occurrence and distribution of antioxidant enzymes in the haemolymph of the shore crab Carcinus maenas. Mar. Biol. 123, 829–836.
Bendich, A. 1993. Physiological role of antioxidants in the immune system. J. Dairy Sci. 76, 2789–2794.
Benov, L., H. Sage, and I. Fridovich. 1997. The Copper- and Zinc-containing superoxide dismutase from E. coil: Molecular weight and stability. Arch. Biochem. Biophys. 340, 305–310.
Beyer, W., J. Imlay, and I. Fridovich. 1991. Superoxide dismutases. Prog. Nucleic Acid Res. Mol. Biol. 40, 221–253.
Brouwer, M., T.H. Brouwer, W. Grater, J.J. Enghild, and I.B. Thogersen. 1997. The paradigm that all oxygen-respiring eukaryotes have cytosol CuZn-superoxide dismutase and that Mn-superoxide dismutase is localized tonic the mitochondria does not apply to a large group of marine arthropods. Biochemistry 36, 13381–13388.
Brouwer, M., T.H. Brouwer, W. Grater, and P.N. Brown. 2003. Replacement of a cytosolic copper/zinc superoxide dismutase by a novel cytosolic manganese superoxide dismutase in crustaceans that use copper (haemocyanin) for oxygen transport. Biochem. J. 374, 219–228.
Culotta, V.C., M. Yang, and T.V. O’Halloran. 2006. Activation of superoxide dismutases: Putting the metal to the pedal. Biochim. Biophys. Acta. 1763, 747–758.
E, S.J., F.X. Guo, S. Liu, J. Chen, Y. Wang, and D.C. Li. 2007. Purification, characterization, and molecular cloning of a thermostable superoxide dismutase from Thermoascus aurantiacus. Biosci. Biotechnol. Biochem. 71, 1090–1093.
Ekanayake, P.M., H.S. Kang, D.M. Zyosa, Y.H. Jee, Y.H. Lee, and J. Lee. 2006. Molecular cloning and characterization of Mn-superoxide dismutase from disk abalone (Haliotis discus discus). Comp. Biochem. Physiol. 145, 318–324.
Halliwell, B. and J.M.C. Gutteridge. 1999. Free Radicals in Biology and Medicine. Oxford Science, Oxford, UK.
Holdom, M.D., R.J. Hay, and A.J. Hamilton. 1995. Purification, N-terminal amino acid sequence and partial characterization of a Cu,Zn superoxide dismutase from the pathogenic fungus Aspergillus fumigatus. Free Rad. Res. 22, 519–531.
Holdom, M.D., R.J. Hay, and A.J. Hamilton. 1996. The Cu,Zn superoxide dismutases of Aspergillus flavus, Aspergillus niger, Aspergillus nidulans, and Aspergillus terreus: purification and biochemical comparison with the Aspergillus fumigatus Cu, Zn superoxide dismutase. Infect. Immun. 64, 3326–3332.
Gorecki, M., Y. Beck, J.R. Hartman, M. Fischer, L. Weiss, Z. Tochner, S. Slavin, and A. Nimrod. 1991. Recombinant human superoxide dismutases: production and potential therapeutical uses. Free Radic. Res. Commun. 12–13, 401–410.
Ken, C.F., C.C. Lee, K.J. Duan, and C.T. Lin. 2005. Unusual stability of manganese superoxide dismutase from a new species, Tatumella ptyseos ct: its gene structure, expression and enzyme properties. Protein. Expr. Purif. 40, 42–50.
Knapp, S., S. Kardinahl, N. Hellgren, G. Tibbelin, G. Schafer, and R. Ladenstein. 1999. Refined crystal structure of a superoxide dismutase from the hyperthermophilic archaeon Sulfolobus acidocaldarius at 2.2 A resolution. J. Mol. Biol. 285, 689–702.
Kowluru, R.A., V. Kowluru, Y. Xiong, and Y.S. Ho. 2006. Overexpression of mitochondrial superoxide dismutase in mice protects the retina from diabetes-induced oxidative stress. Free Radic. Biol. Med. 41, 1191–1196.
Laemmli, U.K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–685.
Li, D.C., J. Gao, Y.L. Li, and J. Lu. 2005. A thermostable manganese-containing superoxide dismutase from the thermophilic fungus Thermomyces lanuginosus. Extremophiles 9, 1–6.
Liau, Y.J., L.S. Wen, J.F. Shaw, and C.T. Lin. 2007. A highly stable cambialistic-superoxide dismutase from Antrodia camphorata: Expression in yeast and enzyme properties. J. Biotechnol. 131, 84–91.
Lim, J.H., Y.G. Yu, Y.S. Han, S. Cho, B.Y. Ahn, S.H. Kim, and Y. Cho. 1997. The crystal struture of an Fe-superoxide dismutase from the hyperthermophile Aquifex pyrophilus at 1.9 A resolution: structural basis for thermostability. J. Mol. Biol. 270, 259–274.
Lowry, O.H., N.J. Rosebrough, A.L. Farr, and R.J. Randall. 1951. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193, 265–275.
Maheshwari, R., G. Bharadwaj, and M.K. Bhat. 2000. Thermophilic fungi: Their physiology and enzymes. Microbiol. Mol. Rev. 64, 461–488.
Mates, J.M. and F.M. Sanchez-Jimenez. 2000. Role of active oxygen species in apoptosis: implications for cancer therapy. Int. J. Biochem. Cell. Biol. 32, 157–170.
Mezes, M., P. Surai, G. Salyi, B. Speake, T. Gaal, and A. Maldjian. 1997. Nutritional metabolic diseases of poultry and disorders of the biological antioxidant defense system. Acta. Vet. Hung. 45, 349–360.
Pipe, R., C. Porte, and D. Livingstone. 1993. Antioxidant enzymes associated with the blood cells and haemolymph of the mussel Mytilus edulis. Fish. Shellfish. Immunol. 3, 221–233.
Plantivaux, A., P. Furla, D. Zoccola, G. Garello, D. Forcioli, S. Richier, P.L. Merle, E. Tambutte, S. Tambutte, and D. Allemand. 2004. Molecular characterization of two Cu Zn-superoxide dismutases in a sea anemone. Free Radic. Biol. Med. 37, 1170–1181.
Rudneva, I.I. 1999. Antioxidant system of Black Sea animals in early development. Comp. Biochem. Physiol. 122, 265–271.
Sambrook, J., E.F. Fritsch, and T. Maniatis. 1989. Molecular Cloning: A Laboratory Manual, 2nd ed. Cold Spring Harbor Laboratory, Cold Spring Harbor, New York, N.Y., USA.
Stewart, R.C. and J.D. Bewley. 1980. Lipid peroxidation associated with accelerated aging of soybean axes. Plant Physiol. 65, 245–248.
Trotti, A. 1997. Toxicity antagonists in cancer therapy. Curr. Opin. Oncol. 9, 569–578.
Yamano, S. and T. Maruyama. 1999. An azide-insensitive superoxide dismutase from a hyperthermophilic archaeon, Sulfolobus solfataricus. J. Biochem. 125, 186–193.
Yamano, S., Y. Sako, N. Nomura, and T. Maruyama. 1999. A cambialistic SOD in a strictly aerobic hyperthermophilic archaeon, Aeropyrum pernix. J. Biochem. 126, 218–225.
Youn, H.D., E.J. Kim, J.H. Roe, Y.C. Hah, and S.O. Kang. 1996. A novel nickel-containing superoxide dismutase from Streptomyces spp. Biochem. J. 318, 889–896.
Zhang, Q.L., F.H. Lia, B. Wang, J.Q. Zhang, Y.C. Liu, Q. Zhou, and J.H. Xiang. 2007. The mitochondrial manganese superoxide dismutase gene in Chinese shrimp Fenneropenaeus chinensis: Cloning, distribution and expression. Dev. Comp. Immunol. 31, 429–440.
Zhong, W., L.W. Oberly, T.D. Oberly, and D.K.St. Clair. 1997. Suppression of the malignant phenotype of human glioma cells by overexpression of manganese superoxide dismutase. Oncogene 14, 481–490.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Song, NN., Zheng, Y., E, SJ. et al. Cloning, expression, and characterization of thermostable Manganese superoxide dismutase from Thermoascus aurantiacus var. levisporus . J Microbiol. 47, 123–130 (2009). https://doi.org/10.1007/s12275-008-0217-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-008-0217-9