Abstract
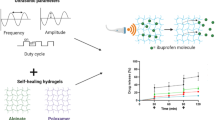
Hydrogels that can respond to dynamic forces either from endogenous biological activities or from external mechanical stimuli show great promise as novel drug delivery systems (DDS). However, it remains challenging to engineer hydrogels that specifically respond to externally applied mechanical forces with minimal basal drug leakage under normal stressful physiological conditions. Here we present an ultrasound responsive hydrogel-based DDS with special dual-crosslinked nanoscale network architecture. The covalent crosslinks endow the hydrogel high mechanical stability and greatly suppress deformation-triggered drug release. Meanwhile, the dynamic covalent boronate ester linkages between hydrogel backbone and the anti-inflammation compound, tannic acid (TA), allow effective ultrasound-triggered pulsatile release of TA. As such, the hydrogel shows distinct drug release profiles under compression and ultrasound. A proof-of-principle demonstration of the suppression of inflammation activation of macrophage upon ultrasound-triggered release of TA was also illustrated. We anticipate that this novel hydrogel-based drug delivery system can be used for the treatment of inflammatory diseases on load-bearing tissues, such as muscle and cartilage.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Lu, Y.; Aimetti, A. A.; Langer, R.; Gu, Z. Bioresponsive materials. Nat. Rev. Mater. 2016, 2, 16075.
Drury, J. L.; Mooney, D. J. Hydrogels for tissue engineering: Scaffold design variables and applications. Biomaterial. 2003, 24, 4337–4351.
Wang, H. Y.; Heilshorn, S. C. Adaptable hydrogel networks with reversible linkages for tissue engineering. Adv. Mater. 2015, 27, 3717–3736.
Slaughter, B. V.; Khurshid, S. S.; Fisher, O. Z.; Khademhosseini, A.; Peppas, N. A. Hydrogels in regenerative medicine. Adv. Mater. 2009, 21, 3307–3329.
Jayakumar, R.; Prabaharan, M.; Sudheesh Kumar, P. T.; Nair, S. V.; Tamura, H. Biomaterials based on chitin and chitosan in wound dressing applications. Biotechnol. Adv. 2011, 29, 322–337.
Koehler, J.; Brandl, F. P.; Goepferich, A. M. Hydrogel wound dressings for bioactive treatment of acute and chronic wounds. Eur. Polym. J. 2018, 100, 1–11.
Xue, B.; Qin, M.; Wang, T. K.; Wu, J. H.; Luo, D. J.; Jiang, Q.; Li, Y.; Cao, Y.; Wang, W. Electrically controllable actuators based on supramolecular peptide hydrogels. Adv. Funct. Mater. 2016, 26, 9053–9062.
Pei, Z. Q.; Yang, Y.; Chen, Q. M.; Terentjev, E. M.; Wei, Y.; Ji, Y. Mouldable liquid-crystalline elastomer actuators with exchangeable covalent bonds. Nat. Mater. 2014, 13, 36–41.
Fratzl, P.; Barth, F. G. Biomaterial systems for mechanosensing and actuation. Natur. 2009, 462, 442–448.
Li, J. Y.; Mooney, D. J. Designing hydrogels for controlled drug delivery. Nat. Rev. Mater. 2016, 1, 16071.
Zhang, S. F.; Ermann, J.; Succi, M. D.; Zhou, A.; Hamilton, M. J.; Cao, B.; Korzenik, J. R.; Glickman, J. N.; Vemula, P. K.; Glimcher, L. H. et al. An inflammation-targeting hydrogel for local drug delivery in inflammatory bowel disease. Sci. Transl. Med. 2015, 7, 300ra128.
Zhang, X. L.; Dong, C. M.; Huang, W. Y.; Wang, H. M.; Wang, L.; Ding, D.; Zhou, H.; Long, J. F.; Wang, T. L.; Yang, Z. M. Rational design of a photo-responsive UVR8-derived protein and a self-assembling peptideprotein conjugate for responsive hydrogel formation. Nanoscal. 2015, 7, 16666–16670.
Zhang, Y. Q.; Yu, J. C.; Bomba, H. N.; Zhu, Y.; Gu, Z. Mechanical forcetriggered drug delivery. Chem. Rev. 2016, 116, 12536–12563.
Sverdlova, N. S.; Witzel, U. Principles of determination and verification of muscle forces in the human musculoskeletal system: Muscle forces to minimise bending stress. J. Biomech. 2010, 43, 387–396.
Mansour, J. M. Biomechanics of cartilage. In Kinesiology: The Mechanics and Pathomechanics of Human Movement. Oatis, C. A., Ed.; Wolter Kluwer: Philadelphia. 2003; pp 66–79.
Zamir, M. Shear forces and blood vessel radii in the cardiovascular system. J. Gen. Physiol. 1977, 69, 449–461.
Barnes, L. A.; Marshall, C. D.; Leavitt, T.; Hu, M. S.; Moore, A. L.; Gonzalez, J. G.; Longaker, M. T.; Gurtner, G. C. Mechanical forces in cutaneous wound healing: Emerging therapies to minimize scar formation. Adv. Wound Car. 2018, 7, 47–56.
Lee, K. Y.; Peters, M. C.; Anderson, K. W.; Mooney, D. J. Controlled growth factor release from synthetic extracellular matrices. Natur. 2000, 408, 998–1000.
Van Der Schaft, D. W. J.; Van Spreeuwel, A. C. C.; Van Assen, H. C.; Baaijens, F. P. T. Mechanoregulation of vascularization in aligned tissueengineered muscle: A role for vascular endothelial growth factor. Tissue Eng. Part. 2011, 17, 2857–2865.
Holme, M. N.; Fedotenko, I. A.; Abegg, D.; Althaus, J.; Babel, L.; Favarger, F.; Reiter, R.; Tanasescu, R.; Zaffalon, P. L.; Ziegler, A. et al. Shear-stress sensitive lenticular vesicles for targeted drug delivery. Nat. Nanotechnol. 2012, 7, 536–543.
Korin, N.; Kanapathipillai, M.; Matthews, B. D.; Crescente, M.; Brill, A.; Mammoto, T.; Ghosh, K.; Jurek, S.; Bencherif, S. A.; Bhatta, D. et al. Shear-activated nanotherapeutics for drug targeting to obstructed blood vessels. Scienc. 2012, 337, 738–742.
Lu, Y.; Hu, Q. Y.; Lin, Y. L.; Pacardo, D. B.; Wang, C.; Sun, W. J.; Ligler, F. S.; Dickey, M. D.; Gu, Z. Transformable liquid-metal nanomedicine. Nat. Commun. 2015, 6, 10066.
Di, J.; Yu, J. C.; Wang, Q.; Yao, S. S.; Suo, D. J.; Ye, Y. Q.; Pless, M.; Zhu, Y.; Jing, Y.; Gu, Z. Ultrasound-triggered noninvasive regulation of blood glucose levels using microgels integrated with insulin nanocapsules. Nano Res. 2017, 10, 1393–1402.
Ye, Y. Q.; Wang, J. Q.; Hu, Q. Y.; Hochu, G. M.; Xin, H. L.; Wang, C.; Gu, Z. Synergistic transcutaneous immunotherapy enhances antitumor immune responses through delivery of checkpoint inhibitors. ACS Nan. 2016, 10, 8956–8963.
Wang, C.; Sun, W. J.; Wright, G.; Wang, A. Z.; Gu, Z. Inflammation-triggered cancer immunotherapy by programmed delivery of CpG and anti-PD1 antibody. Adv. Mater. 2016, 28, 8912–8920.
Hu, Q. Y.; Qian, C. G.; Sun, W. J.; Wang, J. Q.; Chen, Z. W.; Bomba, H. N.; Xin, H. L.; Shen, Q. D.; Gu, Z. Engineered nanoplatelets for enhanced treatment of multiple myeloma and thrombus. Adv. Mater. 2016, 28, 9573–9580.
Sirsi, S. R.; Borden, M. A. State-of-the-art materials for ultrasoundtriggered drug delivery. Adv. Drug. Deliv. Rev. 2014, 72, 3–14.
Di, J.; Price, J.; Gu, X.; Jiang, X. N.; Jing, Y.; Gu, Z. Ultrasound-triggered regulation of blood glucose levels using injectable nano-network. Adv. Healthc. Mater. 2014, 3, 811–816.
Huebsch, N.; Kearney, C. J.; Zhao, X. H.; Kim, J.; Cezar, C. A.; Suo, Z. G.; Mooney, D. J. Ultrasound-triggered disruption and self-healing of reversibly cross-linked hydrogels for drug delivery and enhanced chemotherapy. Proc. Natl. Acad. Sci. US. 2014, 111, 9762–9767.
Wang, J. L.; Kaplan, J. A.; Colson, Y. L.; Grinstaff, M. W. Mechanoresponsive materials for drug delivery: Harnessing forces for controlled release. Adv. Drug. Deliv. Rev. 2017, 108, 68–82.
Thévenot, J.; Oliveira, H.; Sandre, O.; Lecommandoux, S. Magnetic responsive polymer composite materials. Chem. Soc. Rev. 2013, 42, 7099–7116.
Dai, Q.; Nelson, A. Magnetically-responsive self assembled composites. Chem. Soc. Re. 2010, 39, 4057–4066.
Yu, J. C.; Zhang, Y. Q.; Sun, W. J.; Wang, C.; Ranson, D.; Ye, Y. Q.; Weng, Y. Y.; Gu, Z. Internalized compartments encapsulated nanogels for targeted drug delivery. Nanoscal. 2016, 8, 9178–9184.
Lu, Y.; Sun, W. J.; Gu, Z. Stimuli-responsive nanomaterials for therapeutic protein delivery. J. Control. Releas. 2014, 194, 1–19.
Hu, Q. Y.; Katti, P. S.; Gu, Z. Enzyme-responsive nanomaterials for controlled drug delivery. Nanoscal. 2014, 6, 12273–12286.
Mayumi, K.; Marcellan, A.; Ducouret, G.; Creton, C.; Narita, T. Stressstrain relationship of highly stretchable dual cross-link gels: Separability of strain and time effect. ACS Macro Lett. 2013, 2, 1065–1068.
Kampa M.; Nifli, A. P.; Notas G.; Castanas E. Polyphenols and cancer cell growth. In Reviews of Physiology, Biochemistry and Pharmacology. Amara, S.; Bamberg, E.; Fleischmann, B.; Gudermann, T.; Hebert, S. C.; Jahn, R.; Lederer, W. J.; Lill, R.; Miyajima, A.; Offermanns, S. et al., Eds.; Springer: Berlin, Heidelberg. 2007.
Shukla, M.; Gupta, K.; Rasheed, Z.; Khan, K. A.; Haqqi, T. M. Consumption of hydrolyzable tannins-rich pomegranate extract suppresses inflammation and joint damage in rheumatoid arthritis. Nutritio. 2008, 24, 733–743.
Rasheed, Z.; Anbazhagan, A. N.; Akhtar, N.; Ramamurthy, S.; Voss, F. R.; Haqqi, T. M. Green tea polyphenol epigallocatechin-3-gallate inhibits advanced glycation end product-induced expression of tumor necrosis factor-a and matrix metalloproteinase-13 in human chondrocytes. Arthritis Res. Ther. 2009, 11, R71.
Yesilyurt, V.; Webber, M. J.; Appel, E. A.; Godwin, C.; Langer, R.; Anderson, D. G. Injectable self-healing glucose-responsive hydrogels with pH-regulated mechanical properties. Adv. Mater. 2016, 28, 86–91.
Dong, Y. Z.; Wang, W. H.; Veiseh, O.; Appel, E. A.; Xue, K.; Webber, M. J.; Tang, B. C.; Yang, X. W.; Weir, G. C.; Langer, R. et al. Injectable and glucose-responsive hydrogels based on boronic acid–glucose complexation. Langmui. 2016, 32, 8743–8747.
Bapat, A. P.; Roy, D.; Ray, J. G.; Savin, D. A.; Sumerlin, B. S. Dynamiccovalent macromolecular stars with boronic ester linkages. J. Am. Chem. Soc. 2011, 133, 19832–19838.
Cromwell, O. R.; Chung, J.; Guan, Z. B. Malleable and self-healing covalent polymer networks through tunable dynamic boronic ester bonds. J. Am. Chem. Soc. 2015, 137, 6492–6495.
He, L. H.; Fullenkamp, D. E.; Rivera, J. G.; Messersmith, P. B. pH responsive self-healing hydrogels formed by boronate–catechol complexation. Chem. Commun. 2011, 47, 7497–7499.
Leslie, K. O.; Trahan, S.; Gruden, J. Pulmonary pathology of the rheumatic diseases. Semin. Resp. Crit. Car. 2007, 28, 369–378.
Acknowledgements
This research is supported mainly by the National Natural Science Foundation of China (Nos. 21522402, 11674153, 81622033 and 21774057) and the Fundamental Research Funds for the Central Universities (No. 020414380080).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Sun, W., Jiang, H., Wu, X. et al. Strong dual-crosslinked hydrogels for ultrasound-triggered drug delivery. Nano Res. 12, 115–119 (2019). https://doi.org/10.1007/s12274-018-2188-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-018-2188-4