Abstract
Alantolactone is a sesquiterpene lactone isolated from Inula helenium L. Although alantolactone possesses anti-inflammation and apoptosis-induction activities, the underlying mechanism of anti-cancer effect on human breast cancer cells remains largely unknown. In this study, we explored the possibility of alantolactone as an apoptosis-inducing cytotoxic agent using MDA-MB-231 cells as in vitro model. Alantolactone significantly induced its apoptosis, demonstrated by cell cycle analysis, annexin V-APC/7-AAD double staining and dUTP nick end labeling. Additionally, alantolactone triggered the mitochondrial-mediated caspase cascade apoptotic pathway, which was confirmed by increased Bax/Bcl-2 ratio, loss of MMP, release of cytc from mitochondria to cytoplasm, activation of caspase 9/3, and subsequent cleavage of PARP. Z-VAD-FMK partially prevented apoptosis induced by alantolactone. Alantolactone provoked the production of ROS, while NAC (a scavenger of ROS) reversed alantolactone-mediated depolarization of MMP and apoptosis. Alantolactone modulated the activities of MAPKs. As expected, cotreatment with SB203580, SP600125 or U0126 could reduced the apoptotic rate. Furthermore, alantolactone decreased the protein expressions of p-NF-kB p65 and p-STAT3, increased p-c-Jun level in a dose-dependent manner. These findings suggested that alantolactone possessed anticancer activity via ROS-mediated mitochondrial dysfunction involving MAPK pathway, and had an effect on the transcription factors of NF-kB, AP-1 and STAT3.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Breast cancer is one of the most common types of malignancy and is the second leading cause of cancer death for women worldwide (Sharma et al. 2012; Sarma et al. 2015; Kim et al. 2016). It will be useful to understand the molecular biology of breast cancer in developing new therapeutic schemes, which could specifically target to the abnormal molecular. Although there have been many treatment modalities of breast cancer, such as hormonal therapy, radiotherapy, chemotherapy and surgery, the mortality rates are still high (Izdebska et al. 2016). Nowadays, the chemotherapy with drugs of natural origination is an attractive option in breast cancer because of its low toxicity, high efficiency and less side effects (Rates 2001).
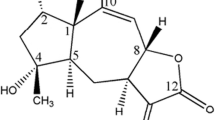
Alantolactone (molecular weight = 232.31, Fig. 1a), an allergenic sesquiterpene lactone, is mainly extracted from the roots of Inula helenium L. Alantolactone is a well known medicinal plant officially listed in some European pharmacopeias as elecampane (Stojakowska et al. 2006; Trendafilova et al. 2010). A large body of pharmacological studies suggested that alantolactone possessed various pharmacologic activities, such as anti-inflammatory, hepatoprotective, antifungal, antibacterial and antitumor effects, which were responsible for its beneficial effects (Dirsch et al. 2001; Lawrence et al. 2001; Konishi et al. 2002; Stojakowska et al. 2006). Sesquiterpene lactones, containing an α-methylene-γ-lactone group, have been demonstrated to execute anti-cancer ability through apoptisis induction (Zhang et al. 2005). Interestingly, alantolactone is a sesquiterpene lactone containing the α-methylene-γ-lactone group. However, the complicated mechanisms underlying induction of breast cancer apoptosis by alantolactone remain largely unknown.
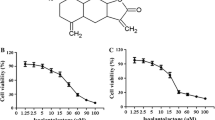
The chemical structure of alantolactone and the cytotoxity of alantolactone against two breast cancer cells: MDA-MB-231 cells and MCF-7 cells. a The chemical structure of alantolactone. b MDA-MB-231 cells were treated with different concentration of alantolactone for 24, 48 or 72 h and cell death ratio was evaluated by MTT assay. c MCF-7 cells were treated with different concentration of alantolactone for 24, 48 or 72 h and cell death ratio was evaluated by MTT assay. All data were expressed as mean ± SD (n = 3)
Apoptosis, a type of programmed cell death, is an important pathway for regulating homeostasis and morphogenesis by eliminating redundant and abnormal cells, and is associated with various diseases, especially cancer (Tang et al. 2016). The typical characteristics of carcinoma is deregulation of apoptosis, so apoptosis induction by kinds of cytotoxic anticancer agents is one of the most effective methods for cancer therapy. Apoptosis is controlled by two principal pathways, including the death receptor-mediated (extrinsic) pathway and the mitochondria-dependent (intrinsic) pathway. Numerous studies have demonstrated that mitochondria actes as a central executioner of apoptosis (Robertson and Orrenius 2002; Cui et al. 2011). The mitochondrial apoptosis pathway starts from mitochondrial depolarization, which is regulated by members of the Bcl-2 protein family, triggering the release of cytochrome c (cytc) into cytosol, thereby activates caspase-9 and caspase-3, ultimately leads to apoptosis (Cory and Adams 2002). ROS formation is tightly coupled with a number of downstream events in apoptosis, including the activation of mitogen-activated protein kinases (MAPKs) (Hsin et al. 2008; Mao et al. 2008).
The present study was designed to explore the effect of alantolactone on cell proliferation and apoptosis induction in human breast cancer cell line, MDA-MB-231. We found that alantolactone increased the cell death ratio as well as apoptosis in MDA-MB-231 cells, and induced ROS generation. Furthermore, we investigated the underlying mechanism of cytotoxic effects of alantolactone to confirm the downstream effectors of ROS accumulation in the apoptotic process.
Materials and methods
Reagents and materials
Alantolactone (purity ≥ 98%), 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), N-acetyl cysteine (NAC), diphenyleneiodonium (DPI), the general inhibitor of caspases (Z-VAD-FMK) were provided by Sigma Chemical Co. (St. Louis, MO). L-15 cell culture medium and fetal bovine serum (FBS) were purchased from Gibco (USA). Alantolactone was dissolved in dimethyl sulfoxide (DMSO) for the further experiments. The primary antibodies for PARP, cleaved PARP, caspase 9, cleaved caspase 9, caspase 3, cleaved caspase 3, Bax, Bcl-2, cytochrome c (cytc), p38 MAPK, p-p38 MAPK, JNK1/2, p-JNK1/2, p-ERK1/2, ERK1/2, p-NF-kB, NF-kB, p-c-Jun, c-Jun, p-STAT3, STAT3 and β-actin were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). ROS detection kit and mitochondrial membrane potential assay kit (JC-1) were obtained from Jiangsu Beyotime Institute of Biotechnology. Matrigel and Annexin V-APC/7-AAD kit were purchased from Becton–Dickinson Biosciences (San Jose, CA, USA). All other chemicals were obtained from commercial sources.
Cell culture
Human breast cancer MDA-MB-231 cell and MCF-7 cell were obtained from the Cell Bank of Shanghai Institute of Cell Biology (Shanghai, China). MDA-MB-231 cells were grown in L-15 medium supplemented with 10% heat-inactivated FBS, 100 units/ml penicillin/streptomycin at 37 °C with 5% CO2 in a humidified atmosphere. MCF-7 cells were grown in DMEM media containing 10% FBS, 100 units/ml penicillin/streptomycin in a humidified atmosphere containing 5% CO2 at 37 °C. Cells were seeded into multi-well plates at appropriate densities for the experiment alanalyses at a generating of 80%–90% confluent layer.
Cell viability assay
The cytotoxicity activity of alantolactone on MDA-MB-231 cells and MCF-7 cell were evaluated by the MTT assay (Chen et al. 2016). Cells (1 × 104 cells/well) were seeded in 96-well plates and cultured in complete growth medium overnight, and then treated with various concentrations of alantolactone. After the required time point, the medium was changed and 10 μL of MTT reagent solution (5 mg/ml) was added to each well and the cells were incubated for an additional 4 h at 37 °C. The purple formazan crystals were dissolved in 100 μL DMSO. After mixing for 10 min, the plates were read at 570 nm on a SpectraMax 190 (Molecular Devices, California, Sunnyvale, USA). All the experiments were performed in triplicate.
Preparation of mitochondrial and cytosolic fractions
Preparation of mitochondrial and cytosolic fractions was carried out as previously described (Eum et al. 2011). Cells were washed with ice-cold PBS and then resuspended in isotonic homogenization buffer. After 80 strokes in a Dounce homogenizer, the unbroken cells were spun down at 30 g for 5 min. The mitochondrial fractions were fractionated at 800 g for 10 min and 14,000 g for 30 min from the supernatant, respectively. For cytosolic fractionation, after ten strokes with a loose homogenizer, the collected supernatant was spun down at 800 g for 10 min and 14,000 g for 30 min.
Cell cycle analysis
Cells were cultured in the presence or absence of various concentrations of alantolactone (5, 10 and 15 μM) for 24 h. Then, cells were washed with phosphate-buffered saline (PBS), and harvested by Trypsin/EDTA digestion at 37 °C for 5 min. Subsequently, cells were fixed in ice-cold 75% ethanol and stained with hypotonic propidium iodide (PI) solution (100 μg/ml RNase and 10 μg/ml PI in PBS). The percentage of cells percentage of cells was determined using a flow cytometric system (FACSCalibur, Becton–Dickinson, USA).
Detection of apoptosis by flow cytometry
Cells were pretreated with a pan-caspase inhibitor (Z-VAD-FMK, 50 μM) or the scavenger of ROS (NAC, 1 mM; DPI, 1 μM) for 1 h, and then incubated with alantolactone (5, 10, and 15 μM) for additional 24 h. MDA-MB-231 cells were also coincubated with 15 μM alantolactone and 15 μM SB203580, 15 μM SP600125 or 15 μM U0126 for 24 h. Both adherent and floating cells were harvested and washed three times with ice-cold PBS. Subsequently, the cells were resuspended in binding buffer and incubated with Annexin V-APC and 7-AAD for 10 min at room temperature in the dark. Finally, the apoptotic cells were quantified by a FACScan Flow Cytometer (Becton–Dickinson, USA).
TUNEL analysis
TUNEL staining was performed using the in situ Cell Death Detection Kit according to the manufacturer’s protocol (Keygen Biotech. Co., LTD, Nanjing, China). Briefly, Cells were treated with alantolactone (5, 10, and 15 μM) for 24 h after the fixation in paraformaldehyde. Then, cells were permeabilized with Triton-X 100 at 4 °C for 5 min. Cells were then washed and labeled with TUNEL reaction mixture at 37 °C for 1 h. The number of TUNEL-positive cells was counted in five random fields under ×40 magnifications. All samples were observed using fluorescence microscopy, and representative fields were captured.
Measurement of changes in mitochondrial membrane potential (MMP)
Cells were preincubated with 1 mM NAC, 1 μM DPI or 5 μM proton gradient uncoupler (FCCP) for 1 h, and treated with alantolactone (5, 10 and 15 μM) overnight. Then, cells were harvested and resuspended in PBS supplemented with JC-1 working solution. After the removal of JC-1, the fluorochrome-conjugated cells were analyzed by flow cytometric assay.
Measurement of intracellular ROS generation
Cells were treated with different concentration of alantolactone for 12 h in the presence or absence of 1 mM NAC or 1 μM DPI. The intracellular ROS mediated oxidation of DCFH to the fluorescent compound 2′,7′-dichlorofluorescein (DCF). The fluorescence of samples was monitored using a spectrophotometer at excitation and emission wavelengths of 480 and 525 nm, respectively, by FACScan flow cytometer as previously described (Martinez-Outschoorn et al. 2011).
Western blot analysis
After incubated with alantolactone (5, 10 and 15 μM) for 24 h, cells were washed with ice-cold PBS and lysed in lysis buffer (50 mM Tris–Cl, pH 7.6, 150 mM NaCl, 1 mM EDTA, 1% (m/v) NP-40, 0.2 mM PMSF, 0.1 mM NaF and 1.0 mM DTT) on ice for 30 min. Equal amounts of supernatant protein (50 μg) were loaded, separated onto 10% sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto nitrocellulose blotting membranes with a semi-dry blot system. Furthermore, membranes were blocked with 5% skim milk and then incubated with the primary antibodies against PARP (1:1000), cleaved PARP (1:1000), Bax (1:1000), Bcl-2 (1:1000), caspase 9 (1:1000), cleaved caspase 9 (1:1000), caspase 3 (1:1000), cleaved caspase 3 (1:1000), p-NF-kB (1:1000), NF-kB (1:1000), p-c-Jun (1:1000), c-Jun (1:1000), p-STAT3 (1:1000), STAT3 (1:1000) and β-actin (1:1000) overnight at 4 °C. Blots were then washed three times with TBS containing 0.1% (w/v) Tween 20 and incubated with horseradish peroxidase-conjugated secondary antibodies (1:5000) in a shaker for 2 h at room temperature. Finally, membranes were washed for four times (10 min/time) and then chemo luminescence reagents (0.1 ml/cm2) were added for the visualization. Experiments were replicated at least three times, and representative blots were shown in the figures. The quantification of proteins was analyzed with Image-Pro Plus (IPP) software.
Statistical analysis
Data are mean ± standard deviation (SD) from three independently performed experiments and analyzed statistically by one-way analysis of variance (ANOVA) with the SPSS software. P values less than 0.05 were considered statistically significant.
Results
Alantolactone showed potent cytotoxic effect
To investigate the effect of alantolactone on breast cancer cell viability, cells were cultured with alantolactone (5, 10, 15 μM) for 24, 48 and 72 h. The cell viability was assessed by MTT assay. As shown in Fig. 1b, alantolactone decreased MDA-MB-231 cell viability in a concentration- and time-dependent manner. The half maximal inhibitory concentration (IC50) was approximately 20.6 μM at 24 h, 10.7 μM at 48 h and 7.6 μM at 72 h, respectively. As shown in Fig. 1c, alantolactone also decreased MCF-7 cell viability in a concentration- and time-dependent manner.
Alantolactone promoted apoptosis
Flow cytometry analysis was carried out to examine the effect of alantolactone on cell cycle of MDA-MB-231 cells. Alantolactone induced a decrease in the population of cells in G1 and S phase, and a remarkable accumulation of cells in G2/M phase in a concentration-dependent manner (Fig. 2a), suggesting that MDA-MB-231 cells were arrested in G2/M phase. To further demonstrate alantolactone could cause cell death, flow cytometric analysis was employed to evaluate the early apoptosis and late apoptosis. As shown in Fig. 2b, alantolactone induced MDA-MB-231 cell apoptosis in a concentration-dependent manner. Compared with control group, alantolactone remarkably triggered breast cancer cell apoptosis from 7.7 ± 0.6% to 51.7 ± 3.4% (p < 0.01). The induction of apoptotic effect of alantolactone was more deeply clarified by TUNEL assay. As shown in Fig. 2c, alantolactone explicitly induced MDA-MB-231 cell apoptosis, whereas there were few TUNEL positive cells in control group. Similar results could be seen in MCF-7 cells, when it was treated with different concentrations of isoalantolactone (Data were not shown). The important characteristics of apoptosis have been obtained from the in situ apoptosis assays, which evidently indicated that alantolactone-mediated suppression of MDA-MB-231 cells viability was closely associated with the increased apoptosis in cancer cells.
Alantolactone induced MDA-MB-231 cells apoptosis. a cell cycle was performed by flow cytometry. a control, b alantolactone 5 μM, c alantolactone 10 μM, d alantolactone 15 μM. b The cells were treated with different concentration of alantolactone for 24 h to estimate the early apoptosis and late apoptosis. a control, b alantolactone 5 μM, c alantolactone 10 μM, d alantolactone 15 μM. c The TUNEL assay was performed to determine the induction of apoptotic effect of alantolactone (The green spots represented apoptotic cells). a control, b alantolactone 5 μM, c alantolactone 10 μM, d alantolactone 15 μM
Alantolactone reduced mitochondrial membrane potential (MMP)
The down-regulation of MMP was associated with apoptosis-releated mitochondrial dysfunction (Chipuk et al. 2006). In order to identify the molecular basis of the apoptosis mediated by alantolactone, the changes of MMP in MDA-MB-231 cells were determined by a FACScan flow cytometer. As shown in Fig. 3a, the MMP in MDA-MB-231 cells was decreased gradually when treated with alantolactone. The ratios of cells with green light were 8.89, 22.60 and 38.76% when cells were incubated with 5, 10 and 15 μM alantolactone for 24 h, respectively (Fig. 3b). The obtained observations indicated that alantolactone caused the loss of MMP in MDA-MB-231 cells, which was similar to the results generated by FCCP. However, pretreatment with 1 mM NAC or 1 μM DPI for 1 h remarkably reversed the depolarization of MMP induced by alantolactone. NAC and DPI alone group was presented in Fig. 3a and b, the result indicated that NAC and DPI alone has no effect on the MMP value in MDA-MB-231 cells, further confirming that alantolactone affect MMP via ROS pathway. Additionally, we estimated the release of cytc following alantolactone treatment, and results exhibited a remarkable up-regulation in the protein expression of cytc in the cytosol (Fig. 3c, d). These data indicated that alantolactone mediated apoptosis potently via the mitochondrial pathway.
Alantolactone reduced MMP of MDA-MB-231 cells and the release of cytc following alantolactone treatment. a Cells were cultured with alantolactone for 24 h or pretreated with 1 mM NAC or 1 μM DPI for 1 h. The changes of MMP in MDA-MB-231 cells were determined by a FACScan flow cytometer. a control, b alantolactone 5 μM, c alantolactone 10 μM, d alantolactone 15 μM, e alantolactone 15 μM + NAC 1 mM, f alantolactone 15 μM + DPI 1 μM, g NAC 1 mM, h DPI 1 μM, i FCCP. b The value of MMP was analyzed. Data are presented as mean ± SD (n = 3), **p < 0.01, alantolactone 15 μM + NAC 1 mM versus alantolactone 15 μM; ##p < 0.01, alantolactone 15 μM + DPI 1 μM versus alantolactone 15 μM. c The cytc protein expressions both in cytoplasm and mitochondria were detected by western blot analysis. d The cytc protein expression was semi-quantified. Data are presented as mean ± SD (n = 3), *p < 0.05, **p < 0.01, alantolactone versus control
Alantolactone increased Bax/Bcl-2 ratio and activated caspases
Bcl-2 family members was involved to keep cell viability by preventing depolarization of MMP, and regulated the release of mitochondrial cytc in apoptosis (Gross et al. 1999). To investigate the possible role of Bcl-2 family members in apoptosis induced by alantolactone, we assessed the effects of alantolactone on the expression of Bax and Bcl-2 proteins by western blot analysis (Fig. 4a, b). When compared with the control group, treatment of alantolactone caused an obvious up-regulation of Bax/Bcl-2 ratio.
Alantolactone increased Bax/Bcl-2 ratio, activated caspase cascade and induced apoptosis on caspase-dependent manner. a, b MDA-MB-231 cells were treated with alantolactone for 24 h, the expression of Bax and Bcl-2 proteins were analyzed by western blotting. Data are presented as mean ± SD (n = 3), *p < 0.05, alantolactone versus control. c, d MDA-MB-231 cells were treated with alantolactone for 24 h, the expression of caspase cascade (caspase 9, cleaved-caspase 9, caspase 3, cleaved-caspase 3, PARP, cleaved PARP) were analyzed by western blotting. Data are presented as mean ± SD (n = 3), *p < 0.05, **p < 0.01, alantolactone versus control. e,f MDA-MB-231 cells were treated with alantolactone in the presence or absence of NAC, MAPK inhibitor (SB203580, SP600125 or U0126), the expression of bcl-2, cleaved caspase 9, cleaved caspase 3, cleaved PARP proteins were determined by western blotting. Data are presented as mean ± SD (n = 3), *p < 0.05, **p < 0.01, alantolactone + NAC versus alantolactone; #p < 0.05, ##p < 0.01, alantolactone + SB203580 versus alantolactone; &p < 0.05, &&p < 0.01, alantolactone + SP600125 versus alantolactone; $p < 0.05, $$p < 0.01, alantolactone + U0126 versus alantolactone. g Alantolactone-induced apoptosis was caspase-dependent, (a) control; (b) alantolactone 15 μM; (c) Z-VAD-FMK 15 μM; (d) Z-VAD-FMK 15 μM + alantolactone 15 μM. *p < 0.05, alantolactone versus control
It has been reported that caspases played a critical role in the terminal, execution phase of apoptosis induced by diverse stimuli (Salvesen and Dixit 1997). Once releasing cytoc in the cytoplasm, it activated caspase-9 and then caspase-3, which was a well known intrinsic cell apoptotic pathway via mitochondria. As shown in Fig. 4c and d, after alantolactone treatment (5, 10 and 15 μM) for 24 h, the levels of cleaved-caspase 9 and cleaved-caspase 3 protein were remarkably increased. To demonstrate the activation of the caspase cascade, one of caspase downstream effectors (PARP) was estimated by western blot analysis. As expected, the expression of cleaved-PARP protein was significantly enhanced after MDA-MB-231 cells exposure to alantolactone. To investigate the effect of ROS and MAPKs on caspases and bcl-2 protein levels, MDA-MB-231 cells were treated with alantolactone (15 μM) in the presence or absence of NAC (1 mM) or MAPK inhibitor (SB203580, SP600125 or U0126, 15 μM). As shown in Fig. 4e and f, the bcl-2 protein level was increased to different degree when cells were treated with alantolactone and NAC or MAPK inhibitor, compared with alantolactone alone. On the contrary, the expression of cleaved caspase 9, cleaved caspase 3 and cleaved PARP proteins were decreased when cells were treated with alantolactone and NAC or MAPK inhibitor, compared with alantolactone alone. There was no significant difference between control and NAC alone group on the protein expressions of Bcl-2, cleaved caspase 9, cleaved caspase 3 and cleaved PARP. And these protein expressions have no obvious defference between control group and MAPKs inhibitor (SB203580, SP600125, U0126) group. These results indicated that ROS and MAPKs played an important role in the alantolactone′s effect on caspases and bcl-2 protein levels.
To further confirm whether alantolactone-induced apoptosis was caspase-dependent, MDA-MB-231 cells were pretreated with Z-VAD-FMK (a pan-caspase inhibitor, 15 μM) for 1 h. As shown in Fig. 4g, Z-VAD-FMK alone has no effect on cell apoptosis, but the co-treatment of Z-VAD-FMK and alantolactone could effectively but not completely suppressed alantolactone-induced apoptosis. We therefore speculated that alantolactone might induce apoptosis both through caspase-dependent and—independent mechanisms.
Alantolactone increased ROS levels
It was reported that ROS played a vital role in apoptosis of multiple types of cell. To investigate whether ROS were involved in alantolactone-mediated MDA-MB-231 cells apoptosis, intracellular ROS generation was determined by the fluorescence dye DCF-DA after exposure of cells to alantolactone. The results showed that incubation with 15 μM alantolactone rapidly induced ROS generation until 6 h after treatment when compared with control group. Then, its level was further up-regulated over the following 18 h (Fig. 5a). As depicted in Fig. 5b, the treatment of alantolactone for 12 h increased ROS level in a dose-dependent manner. To further demonstrate that ROS played a role of initiator in alantolactone-induced apoptosis, MDA-MB-231 cells were pretreated with 1 mM NAC or 1 μM DPI, and cultured with 15 μM alantolactone for 12 h. As we expected, both ROS scavengers significantly down-regulated ROS level (p < 0.01). The result exhibited in Fig. 5c indicated that NAC alone had no effect on cell apptosis, but when cells were co-cultured with alantolactone and NAC, the apoptosis was down-regulated from 51.7 ± 4.3% to 27.9 ± 2.5% (p < 0.01). Similarly, DPI alone had no effect on cell apptosis, the co-treatment of alantolactone and DPI remarkably decrease the apoptosis, compared with alantolactone (p < 0.01). Additionally, the co-treatment of alantolactone and NAC or DPI impressively decreased cell death ratio, compared with alantolactone alone (Fig. 5d). When MCF-7 cells were treated with alantolactone, the intracellular ROS level was also up-regulated, which was alleviated by the addition of NAC. The ROS inhibitor also suppressed alantolactone-induced apoptosis and cell death in MCF-7 cells (Data were not shown). All the above results indicated that ROS was involved in the apoptosis mediated by alantolactone in breast cancer cells.
Alantolactone induced MDA-MB-231 cells apoptosis via increasing ROS level. a Alantolactone stimulated the production of ROS for the marked time periods. MDA-MB-231 cells were treated with 15 μM alantolactone for the marked time periods. b MDA-MB-231 cells were pretreated with 1 mM NAC or 1 μM DPI for 1 h and then incubated with 15 μM alantolactone for 12 h. Then, the production of ROS was determined. Data are presented as mean ± SD (n = 3), **p < 0.01, alantolactone 15 μM + NAC 1 mM versus alantolactone; &&p < 0.01, alantolactone 15 μM + DPI 1 μM versus alantolactone. c The apoptotic induction of alantolactone on MDA-MB-231 cells. Cells were pretreated with or without 1 mM NAC or 1 μM DPI for 1 h and then incubated with 15 μM alantolactone for 24 h. a control; b NAC 1 mM; c 1 μM DPI; d alantolactone 15 μM; e alantolactone 15 μM + NAC 1 mM; f alantolactone 15 μM + DPI 1 μM. D, MDA-MB-231 cells death was evaluated by MTT assay. Data are presented as mean ± SD (n = 3), *p < 0.01, alantolactone 15 μM + NAC 1 mM versus alantolactone; &&p < 0.01, alantolactone 15 μM + DPI 1 μM versus alantolactone
Alantolactone activated MAPKs pathway
To examine the potential involvement of MAPKs in alantolactone-mediated apoptosis in MDA-MB-231 cells, western blot analysis was carried out to determine the expression of p38 MAPK, JNK and ERK proteins. As shown in Fig. 6a and b, compared with the control group, the phosphorylation of p38 MAPK, JNK and ERK in MDA-MB-231 cells was increased after alantolactone treatment. In contrast, the level of non-phosphorylated p38 MAPK, JNK and ERK was not affected by alantolactone. The protein expression of p-p38, p-JNK or p-ERK was significantly decreased when cells were cultured with alantolactone (15 μM) and MAPK inhibitor (SB203580, SP600125, U0126, 15 μM), compared with alantolactone treatment, and there was no difference on the expression of p-p38, p-JNK or p-ERK from inhibitor alone group versus the control group (Fig. 6c–e). When cells were co-treated with alantolactone and NAC or DPI, the activation of p38 MAPK, JNK and ERK by alantolactone was significantly inhibited. However, the expression of p38 MAPK, JNK, ERK, p-p38 MAPK, p-JNK and p-ERK proteins has no change when cells were treated with NAC or DPI alone (Fig. 7a, b). To demonstrate the role of MAPKs pathway in the process of apoptosis, MDA-MB-231 cells were coincubated with alantolactone and SB203580 (p38 MAPK specific inhibitor), SP600125 (JNK specific inhibitor) or U0126 (ERK specific inhibitor) for 24 h. As shown in Fig. 6f, the cell apoptosis in SB203580, SP600125 or U0126 alone group was 11.2 ± 1.1%, 10.8 ± 0.8% and 10.3 ± 0.9%, respectively, has no obvious difference with control group (7.7 ± 0.6%). However, when cells were co-treated with these specific inhibitors and alantolactone, the apoptosis was effectively attenuated, compared with alantolactone alone. The similar results were observed in MCF-7 cells (Data were not shown). These findings suggested that alantolactone induced breast cancer cells apoptosis at least partially depending on MAPK pathway.
The MAPKs, including p38, JNK, and ERK, were involved in alantolactone-mediated apoptosis. a, b MDA-MB-231 cells were cultured with 15 μM alantolactone up to 24 h, and western blotting was carried out to analyze p38, p-p38, JNK, p-JNK, ERK, and p-ERK protein expression at indicated times. Data are presented as mean ± SD (n = 3), *p < 0.05, **p < 0.01, alantolactone versus control. c, d, e Cells were cultured with 15 μM alantolactone in the presence or absence of 15 μM MAPK inhibitors, then the protein expression of p-p38, p-JNK or p-ERK was determined by western blotting. Data are presented as mean ± SD (n = 3), **p < 0.01, alantolactone versus control; #p < 0.05, alantolactone + MAPK inhibitor versus alantolactone. f MDA-MB-231 cells were incubated with SB203580 (15 μM), SP600125 (15 μM) or U0126 (15 μM) in the presence or absence of alantolactone (15 μM) for 24 h, and flow cytometric analysis was employed to evaluate the early apoptosis and late apoptosis. a control; b alantolactone; c alantolactone + SB203580; d alantolactone + SP600125; e alantolactone + U0126; f SB203580; g SP600125; h U0126
The releationship between ROS and MAPKs on MDA-MB-231 cells. a, b MDA-MB-231 cells were preincubated with NAC 1 mM or DPI 1 μM for 1 h, and then treated with alantolactone 15 μM for 24 h. The expressions of p38, p-p38, JNK, p-JNK, ERK, and p-ERK were detected by western blot analysis. Data are presented as mean ± SD (n = 3), **p < 0.01, alantolactone versus control; #p < 0.05, alantolactone + NAC versus alantolactone; &p < 0.05, alantolactone + DPI versus alantolactone
The effect of alantolactone on the transcription factors of NF-kB, AP-1 and STAT3
To investigate the effect of alantolactone on the transcription factors under the condition of apoptosis in alantolactone-treated MDA-MB-231 cells, we tested the expression levels of p-NF-kB, p-c-Jun and p-STAT3. As shown in Fig. 8, alantolactone significantly decreased the protein expressions of p-NF-kB and p-STAT3, increased p-c-Jun level in a dose-dependent manner.
The effect of alantolactone on the transcription factors. a MDA-MB-231 cells were treated with different concentrations of alantolactone for 24 h. The protein expressions of p-NF-kB, NF-kB, p-c-Jun, c-Jun, p-STAT3 and STAT3 were detected by western blotting. b The quantitation of western blot analysis. β-actin was used as a loading control. Data are presented as mean ± SD (n = 3). *p < 0.05, **p < 0.01, alantolactone versus control
Discussion
Apoptosis induction by various cytotoxic anticancer agents was one of the most effective methods for cancer treatment (Yang et al. 2016). Recently, the apoptosis-inducing activity of plant-derived natural products has attracted much attention. Alantolactone was a sesquiterpene lactone extracted from Inula helenium L. It has been reported that alantolactone could induce apoptosis of leukemia T cells, human hepatoma cells, glioblastoma cells, and so on (Dirsch et al. 2001; Khan et al. 2012; Lei et al. 2012). In this study, the results of cell cycle analysis, annexin V-APC/7-AAD double staining, and dUTP nick end labeling experiments demonstrated that alantolactone could induce apoptosis of MDA-MB-231 cells. Our study indicated that alantolactone might be a promising candidate against breast cancer through apoptosis pathway.
It was well known that mitochondria played an important role in the signal transduction of apoptosis (van Loo et al. 2002). The Bcl-2 family participated in the modulation of apoptosis and was a significant target of treating cancer (Del Poeta et al. 2003; Jung et al. 2016). Bcl-2 was an anti-apoptotic protein, whereas Bax was a pro-apoptotic protein. The imbalance between the two proteins was closely related to apoptosis. In the present study, alantolactone reduced MMP but upregulated the ratio of Bax to Bcl-2, leading to the following efflux of mitochondrial cytc to cytosol and activation of caspase 9, caspase 3 and its downstream substrate PARP. However, the pan-caspase inhibitor (Z-VAD-FMK) could partially alleviate alantolactone-induced apoptosis. Therefore, we speculated that alantolactone potentially induced apoptosis both via caspase-dependent and -independent pathway.
Growing evidence suggested that low level of ROS promoted cell proliferation, whereas excessive accumulation of ROS led to cell apoptosis or necrosis (Wang et al. 2008; Circu and Aw 2010). In the present study, alantolactone stimulated the generation of ROS to a high value in MDA-MB-231 cells. The level of ROS was in a state of continuous change in biological system. Contradictory comments were present in literatures with respect to the relationship between ROS and mitochondria. Some studies demonstrated that ROS was released from mitochondria when it was damaged, because mitochondria was the major source of ROS. On the other hand, some researchers supported the opinion that excessive accumulation of ROS damaged mitochondria, leading to the down-regulation of MMP, stimulated the release of cytc from mitochondria into cytoplasm, and ultimately induced cells apoptosis (Ricci et al. 2003). In the present study, we found that NAC (a scavenger of ROS) effectively suppressed the depolarization of MMP. Additionally, NAC blocked alantolactone induced production of ROS and cytotoxicity in MDA-MB-231 cells. These observations indicated that alantolactone could stimulate the mitochondria-induced apoptosis pathway through a ROS-dependent mechanism in MDA-MB-231 cells.
Several reports suggested that MAPKs were involved in the signaling pathways in response to various extracellular stimuli, resulting in apoptosis (Xia et al. 1995). There was no dispute that p38 and JNK both were involved in apoptosis-induction of cancer cells. And our present study demonstrated that alantolactone could increase the levels of p-p38 and p-JNK protein in MDA-MB-231 cells. When cells were co-treated with p38 or JNK and alantolactone, the apoptosis mediated by alantolactone was remarkably abrogated. The results revealed that the activation of p38 and JNK was involved in apoptosis-induction of alantolactone. A previous study demonstrated that ERK was an important downstream target of the ROS-mediated apoptotic process (Han et al. 2016). Although the ERK pathway was usually related to enhanced cell proliferation, conversely, many literatures indicated that ERK activation increased expressions of apoptotic genes which contributed to cell death (Cagnol et al. 2010). Recent study revealed that activation of ERK by quercetin resulted in antiproliferative effects such as apoptosis, senescence, or autophagy in cancer cells (Kim et al. 2008). Park et al. reported that MAPK proteins were activated upon Toyocamycin treatment, the p38 and ERK activities were regulated by ROS-mediated signaling pathway underlying the Toyocamycin-induced apoptosis (Park et al. 2017). Sangivamycin, an adenosine analog, induced apoptosis by inhibiting ERK phosphorylation in primary effusion lymphoma cells (Wakao et al. 2014). Compared with these, our result showed that the effect of alantolactone had a critical role in ERK activation in MDA-MB-231 cells, suggesting that the impact of ERK signaling may depend on the type and status of tumor and stimuli. Taken together, all of these results demonstrated that the activation of p38, JNK and ERK was involved in apoptosis-induction of alantolactone.
ROS signaling stimulated the activation of mitochondrial-dependent cell death pathway through activation of MAPK pathway, resulting in up-regulated pro-apoptotic protein expression and down-regulated anti-apoptotic protein level, followed by subsequent mitochondrial membrane permeabilization and cell death (Hseu et al. 2012). Three classic MAPK pathways have been identified in mammalian cells, namely, ERK, JNK, and p38. These parallel pathways may be independent or overlapping (Saito 2010). Baohuoside I has been reported to elevate ROS levels, JNK and p38 activation, mitochondrial cytc release into the cytosol, loss of MMP during apoptosis of human adenocarcinoma alveolar basal epithelial A549 cells (Song et al. 2012). It has been reported that doxycycline could induce intra-cellular ROS production, JNK activation at an early stage of treatment, a remarkable increase of cytc protein level in cytosol, and MMP change during melanoma cell apoptosis (Shieh et al. 2010). 7-O-Geranylquercetin was reported to trigger ROS generation, activate p38 and JNK signaling pathways, subsequently lead to mitochondrial impairment by regulating the expression of Bcl-2, Bcl-xl and Bax, finally promote cytc release and caspases activation to induce gastric cancer cells apoptosis (Zhu et al. 2017). Interestingly, alantolactone has been previously reported to significantly inhibit MAPKs phophorylation in LPS-stimulated RAW 264.7 cells (Chun et al. 2012). The involvement of MAPK during alantolactone-induced MDA-MB-231 cell apoptosis has not been reported so far. Herein, we attempted to confirm whether the MAPK pathways were involved in the process. The results indicated that alantolactone was indeed an activator of p38, JNK and ERK. Additionally, the specific inhibitors SB203580, SP600125 and U0126 could potently attenuate the apoptosis via inhibition of p38, JNK and ERK signaling. NAC or DPI could also suppress MAPK pathway activation. In all, we further conclude that the promoted ROS generation by alantolactone is the upstream signaling event leading to parallel p38, JNK and ERK activation.
Transcription factors, such as NF-kB, AP-1, and STAT3, played important roles in apoptosis-induction by natural products from plants at the molecular level. The antitumor activity of alantolactone has been well-documented in a wide spectrum of tumor cells, and the underlying mechanisms of which were involved in inducing apoptosis by suppression of NF-kB activation (Ding et al. 2016; Wei et al. 2013). We also found that alantolactone could inhibit the protein expression of p-NF-kB p65 in MDA-MB-231 cells. It was reported that AP-1 induces apoptosis in the nervous system, and it blocks apoptosis in IL-4-deprived T cells by upregulating the expression of an anti-apoptotic protein, Bcl-3 (Rebollo et al. 2000). Our experiments indicated that alantolactone increased the protein expression of AP-1 under its apoptosis-induction in MDA-MB-231 cells, consistented with the reported results by other researchers (Na et al. 2012; Ma et al. 2016; Ren et al. 2013). The activation of STAT3 was found in many cancer cells including multiple myeloma, leukemia, prostate cancer, breast cancer, colon cancer and so on (Hsiao et al. 2003; Schuringa et al. 2000; Mora et al. 2002; Dolled-Filhart et al. 2003; Lin et al. 2005). In particular, recent studies have reported the potential of many compounds from natural products in cancer treatment by targeting STAT3. For instances, curcumin inhibited constitutive and IL-6-inducible STAT3 phosphorylation in human multiple myeloma cells (Bharti et al. 2003). Similarly, betulinic acid and ursolic acid suppressed STAT3 activation pathway through induction of SHP-1 in multiple myeloma cells (Pandey et al. 2009). Khan et al. reported that alantolactone induced apoptosis in HepG2 cells through inhibition of STAT3 activation (Khan et al. 2012). And we found that alantolactone suppressed the protein level of STAT3 in MDA-MB-231 cells.
In summary, our results confirmed that alantolactone-induced apoptosis in human breast cancer MDA-MB-231 cells was mediated by the activation of mitochondria-dependent (intrinsic) pathway, which needed ROS generation. Additionally, the ROS/MAPK signaling pathway was an initiator of cell apoptosis, suggesting a novel insight into the molecular mechanism of alantolactone-induced anticancer activity. Furthermore, our present findings provided a rationale for the development of alantolactone as an alternative for the treatment of human breast cancer in an appropriate dosage.
References
Bharti AC, Donato N, Aggarwal BB (2003) Curcumin (diferuloylmethane) inhibits constitutive and IL-6-inducible STAT3 phosphorylation in human multiple myeloma cells. J Immunol 171:3863–3871
Cagnol S, Chambard JC (2010) ERK and cell death: mechanisms of ERK-induced cell death–apoptosis, autophagy and senescence. FEBS J 277:2–21
Chen X, Luo J, Meng L, Pan T, Zhao B, Tang ZG, Dai Y (2016) Dracorhodin perchlorate induces the apoptosis of glioma cells. Oncol Rep 35:2364–2372
Chipuk JE, Bouchier-Hayes L, Green DR (2006) Mitochondrial outer membrane permeabilization during apoptosis: the innocent bystander scenario. Cell Death Differ 13:1396–1402
Chun J, Choi RJ, Khan S, Lee DS, Kim YC, Nam YJ, Lee DU, Kim YS (2012) Alantolactone suppresses inducible nitric oxide synthase and cyclooxygenase-2 expression by down-regulating NF-κB, MAPK and AP-1 via the MyD88 signaling pathway in LPS-activated RAW 264.7 cells. Int Immunopharmacol 14:375–583
Circu ML, Aw TY (2010) Reactive oxygen species, cellular redox systems, and apoptosis. Free Radic Biol Med 48:749–762
Cory S, Adams JM (2002) The Bcl2 family: regulators of the cellular life-or-death switch. Nat Rev Cancer 2:647–656
Cui M, Zhang Y, Liu S, Xie W, Ji M, Lou H, Li X (2011) 1-oxoeudesm-11(13)-ene-12,8alpha-lactone-induced apoptosis via ROS generation and mitochondria activation in MCF-7 cells. Arch Pharm Res 34:1323–1329
Del Poeta G, Venditti A, Del Principe MI, Maurillo L, Buccisano F, Tamburini A, Cox MC, Franchi A, Bruno A, Mazzone C, Panetta P, Suppo G, Masi M, Amadori S (2003) Amount of spontaneous apoptosis detected by Bax/Bcl-2 ratio predicts outcome in acute myeloid leukemia (AML). Blood 101:2125–2131
Ding Y, Gao H, Zhang Y, Li Y, Vasdev N, Gao Y, Chen Y, Zhang Q (2016) Alantolactone selectively ablates acute myeloid leukemia stem and progenitor cells. J Hematol Oncol 22(9):93
Dirsch VM, Stuppner H, Vollmar AM (2001) Cytotoxic sesquiterpene lactones mediate their death-inducing effect in leukemia T cells by triggering apoptosis. Planta Med 67:557–559
Dolled-Filhart M, Camp RL, Kowalski DP, Smith BL, Rimm DL (2003) Tissue microarray analysis of signal transducers and activators of transcription 3 (Stat3) and phospho-Stat3 (Tyr705) in node-negative breast cancer shows nuclear localization is associated with a better prognosis. Clin Cancer Res 9:594–600
Eum DY, Byun JY, Yoon CH, Seo WD, Park KH, Lee JH, Chung HY, An S, Suh Y, Kim MJ, Lee SJ (2011) Triterpenoid pristimerin synergizes with taxol to induce cervical cancer cell death through reactive oxygen species-mediated mitochondrial dysfunction. Anticancer Drugs 22:763–773
Gross A, McDonnell JM, Korsmeyer SJ (1999) BCL-2 family members and the mitochondria in apoptosis. Genes Dev 13:1899–1911
Han MH, Park C, Lee DS, Hong SH, Choi IW, Kim GY, Choi SH, Shim JH, Chae JI, Yoo YH, Choi YH (2017) Cytoprotective effects of esculetin against oxidative stress are associated with the upregulation of Nrf2-mediated NQO1 expression via the activation of the ERK pathway. Int J Mol Med 39:380–386
Hseu YC, Lee MS, Wu CR, Cho HJ, Lin KY, Lai GH, Wang SY, Kuo YH, Kumar KJ, Yang HL (2012) The chalcone flavokawain B induces G2/M cell-cycle arrest and apoptosis in human oral carcinoma HSC-3 cells through the intracellular ROS generation and downregulation of the Akt/p38 MAPK signaling pathway. J Agric Food Chem 60:2385–2397
Hsiao JR, Jin YT, Tsai ST, Shiau AL, Wu CL, Su WC (2003) Constitutive activation of STAT3 and STAT5 is present in the majority of nasopharyngeal carcinoma and correlates with better prognosis. Br J Cancer 89:344–349
Hsin YH, Chen CF, Huang S, Shih TS, Lai PS, Chueh PJ (2008) The apoptotic effect of nanosilver is mediated by a ROS- and JNK-dependent mechanism involving the mitochondrial pathway in NIH3T3 cells. Toxicol Lett 179:130–139
Izdebska M, Grzanka D, Gagat M, Halas-Wisniewska M, Grzanka A (2016) Downregulation of importin-9 protects MCF-7 cells against apoptosis induced by the combination of garlic-derived alliin and paclitaxel. Oncol Rep 35:3084–3093
Jung H, Bae J, Ko SK, Sohn UD (2016) Ultrasonication processed Panax ginseng berry extract induces apoptosis through an intrinsic apoptosis pathway in HepG2 cells. Arch Pharm Res 39:855–862
Khan M, Yi F, Rasul A, Li T, Wang N, Gao H, Gao R, Ma T (2012) Alantolactone induces apoptosis in glioblastoma cells via GSH depletion, ROS generation, and mitochondrial dysfunction. IUBMB Life 64:783–794
Kim YH, Lee DH, Jeong JH, Guo ZS, Lee YJ (2008) Quercetin augments TRAIL-induced apoptotic death: involvement of the ERK signal transduction pathway. Biochem Pharmacol 75:1946–1958
Kim KH, Kim JY, Kwak JH, Kim BO, Pyo S (2016) Different apoptotic effects of saxifragifolin C in human breast cancer cells. Arch Pharm Res 39:577–589
Konishi T, Shimada Y, Nagao T, Okabe H, Konoshima T (2002) Antiproliferative sesquiterpene lactones from the roots of Inula helenium. Biol Pharm Bull 25:1370–1372
Lawrence NJ, McGown AT, Nduka J, Hadfield JA, Pritchard RG (2001) Cytotoxic Michael-type amine adducts of alpha-methylene lactones alantolactone and isoalantolactone. Bioorg Med Chem Lett 11:429–431
Lei JC, Yu JQ, Yin Y, Liu YW, Zou GL (2012) Alantolactone induces activation of apoptosis in human hepatoma cells. Food Chem Toxicol 50:3313–3319
Lin Q, Lai R, Chirieac LR, Li C, Thomazy VA, Grammatikakis I, Rassidakis GZ, Zhang W, Fujio Y, Kunisada K, Hamilton SR, Amin HM (2005) Constitutive activation of JAK3/STAT3 in colon carcinoma tumors and cell lines: inhibition of JAK3/STAT3 signaling induces apoptosis and cell cycle arrest of colon carcinoma cells. Am J Pathol 167:969–980
Ma W, Zhang X, Li M, Ma XL, Huang BR, Chen H, Chen D (2016) Proapoptotic RYBP interacts with FANK1 and induces tumor cell apoptosis through the AP-1 signaling pathway. Cell Signal 28:779–787
Mao X, Yu CR, Li WH, Li WX (2008) Induction of apoptosis by shikonin through a ROS/JNK-mediated process in Bcr/Abl-positive chronic myelogenous leukemia (CML) cells. Cell Res 18:879–888
Martinez-Outschoorn UE, Lin Z, Trimmer C, Flomenberg N, Wang C, Pavlides S, Pestell RG, Howell A, Sotgia F, Lisanti MP (2011) Cancer cells metabolically “fertilize” the tumor microenvironment with hydrogen peroxide, driving the Warburg effect: implications for PET imaging of human tumors. Cell Cycle 10:2504–2520
Mora LB, Buettner R, Seigne J, Diaz J, Ahmad N, Garcia R, Bowman T, Falcone R, Fairclough R, Cantor A, Muro-Cacho C, Livingston S, Karras J, Pow-Sang J, Jove R (2002) Constitutive activation of Stat3 in human prostate tumors and cell lines: direct inhibition of Stat3 signaling induces apoptosis of prostate cancer cells. Cancer Res 62:6659–6666
Na HK, Kim EH, Choi MA, Park JM, Kim DH, Surh YJ (2012) Diallyl trisulfide induces apoptosis in human breast cancer cells through ROS-mediated activation of JNK and AP-1. Biochem Pharmacol 84:1241–1250
Pandey MK, Sung B, Ahn KS, Aggarwal BB (2009) Butein suppresses constitutive and inducible signal transducer and activator of transcription (STAT) 3 activation and STAT3-regulated gene products through the induction of a protein tyrosine phosphatase SHP-1. Mol Pharmacol 75:525–533
Park SG, Kim SH, Kim KY, Yu SN, Choi HD, Kim YW, Nam HW, Seo YK, Ahn SC (2017) Toyocamycin induces apoptosis via the crosstalk between reactive oxygen species and p38/ERK MAPKs signaling pathway in human prostate cancer PC-3 cells. Pharmacol Rep 69:90–96
Rates SM (2001) Plants as source of drugs. Toxicon 39:603–613
Rebollo A, Dumoutier L, Renauld JC, Zaballos A, Ayllon V, Martinez AC (2000) Bcl-3 expression promotes cell survival following interleukin-4 deprivation and is controlled by AP1 and AP1-like transcription factors. Mol Cell Biol 20:3407–3416
Ren XX, Song W, Liu WJ, Guan X, Miao F, Miao SY, Wang LF (2013) Rhomboid domain containing 1 inhibits cell apoptosis by upregulating AP-1 activity and its downstream target Bcl-3. FEBS Lett 587:1793–1798
Ricci JE, Gottlieb RA, Green DR (2003) Caspase-mediated loss of mitochondrial function and generation of reactive oxygen species during apoptosis. J Cell Biol 160:65–75
Robertson JD, Orrenius S (2002) Role of mitochondria in toxic cell death. Toxicology 181–182:491–496
Saito H (2010) Regulation of cross-talk in yeast MAPK signaling pathways. Curr Opin Microbiol 13:677–683
Salvesen GS, Dixit VM (1997) Caspases: intracellular signaling by proteolysis. Cell 91:443–446
Sarma P, Bag I, Ramaiah MJ, Kamal A, Bhadra U, Pal Bhadra M (2015) Bisindole-PBD regulates breast cancer cell proliferation via SIRT-p53 axis. Cancer Biol Ther 16:1486–1501
Schuringa JJ, Wierenga AT, Kruijer W, Vellenga E (2000) Constitutive Stat3, Tyr705, and Ser727 phosphorylation in acute myeloid leukemia cells caused by the autocrine secretion of interleukin-6. Blood 95:3765–3770
Sharma G, Kar S, Palit S, Das PK (2012) 18beta-glycyrrhetinic acid induces apoptosis through modulation of Akt/FOXO3a/Bim pathway in human breast cancer MCF-7 cells. J Cell Physiol 227:1923–1931
Shieh JM, Huang TF, Hung CF, Chou KH, Tsai YJ, Wu WB (2010) Activation of c-Jun N-terminal kinase is essential for mitochondrial membrane potential change and apoptosis induced by doxycycline in melanoma cells. Br J Pharmacol 160:1171–1184
Song J, Shu L, Zhang Z, Tan X, Sun E, Jin X, Chen Y, Jia X (2012) Reactive oxygen species-mediated mitochondrial pathway is involved in Baohuoside I-induced apoptosis in human non-small cell lung cancer. Chem Biol Interact 199:9–17
Stojakowska A, Michalska K, Malarz J (2006) Simultaneous quantification of eudesmanolides and thymol derivatives from tissues of Inula helenium and I. royleana by reversed-phase high-performance liquid chromatography. Phytochem Anal 17:157–161
Tang X, Tang J, Liu X, Zeng L, Cheng C, Luo Y, Li L, Qin SL, Sang Y, Deng LM, Lv XB (2016) Downregulation of miR-129-2 by promoter hypermethylation regulates breast cancer cell proliferation and apoptosis. Oncol Rep 35:2963–2969
Trendafilova A, Chanev C, Todorova M (2010) Ultrasound-assisted extraction of alantolactone and isoalantolactone from Inula helenium roots. Pharmacogn Mag 6:234–237
Van Loo G, Saelens X, van Gurp M, MacFarlane M, Martin SJ, Vandenabeele P (2002) The role of mitochondrial factors in apoptosis: a Russian roulette with more than one bullet. Cell Death Differ 9:1031–1042
Wakao K, Watanabe T, Takadama T, Ui S, Shigemi Z, Kagawa H, Higashi C, Ohga R, Taira T, Fujimuro M (2014) Sangivamycin induces apoptosis by suppressing Erk signaling in primary effusion lymphoma cells. Biochem Biophys Res Commun 444:135–140
Wang CC, Chiang YM, Sung SC, Hsu YL, Chang JK, Kuo PL (2008) Plumbagin induces cell cycle arrest and apoptosis through reactive oxygen species/c-Jun N-terminal kinase pathways in human melanoma A375.S2 cells. Cancer Lett 259:82–98
Wang Y, Xu L, Lou LL, Song SJ, Yao GD, Ge MY, Hayashi T, Tashiro SI, Onodera S, Ikejima T (2017) Timosaponin AIII induces apoptosis and autophagy in human melanoma A375-S2 cells. Arch Pharm Res 40:69–78
Wei W, Huang H, Zhao S, Liu W, Liu CX, Chen L, Li JM, Wu YL, Yan H (2013) Alantolactone induces apoptosis in chronic myelogenous leukemia sensitive or resistant to imatinib through NF-κB inhibition and Bcr/Abl protein deletion. Apoptosis 18:1060–1070
Xia Z, Dickens M, Raingeaud J, Davis RJ, Greenberg ME (1995) Opposing effects of ERK and JNK-p38 MAP kinases on apoptosis. Science 270:1326–1331
Yang F, Tang XY, Liu H, Jiang ZW (2016) Inhibition of mitogen-activated protein kinase signaling pathway sensitizes breast cancer cells to endoplasmic reticulum stress-induced apoptosis. Oncol Rep 35:2113–2120
Zhang S, Won YK, Ong CN, Shen HM (2005) Anti-cancer potential of sesquiterpene lactones: bioactivity and molecular mechanisms. Curr Med Chem Anticancer Agents 5:239–249
Zhu Y, Jiang Y, Shi L, Du L, Xu X, Wang E, Sun Y, Guo X, Zou B, Wang H, Wang C, Sun L, Zhen Y (2017) 7-O-Geranylquercetin induces apoptosis in gastric cancer cells via ROS-MAPK mediated mitochondrial signaling pathway activation. Biomed Pharmacother 87:527–538
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Nos. 81403121 and 81503306), Young Medical Talent Project of Jiangsu Province (QNRC2016636) and the Natural Science Foundation of Jiangsu Province (BK20151045).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
All the authors declare that they have no conflicts of interest to disclose.
Rights and permissions
About this article
Cite this article
Cui, L., Bu, W., Song, J. et al. Apoptosis induction by alantolactone in breast cancer MDA-MB-231 cells through reactive oxygen species-mediated mitochondrion-dependent pathway. Arch. Pharm. Res. 41, 299–313 (2018). https://doi.org/10.1007/s12272-017-0990-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-017-0990-2