Abstract
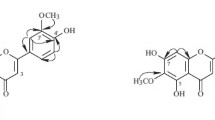
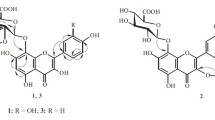
A new C-alkylated flavonoid (5,7-dihydroxy-3′-(4″-acetoxy-3″-methylbutyl)-3,6,4′-trimethoxyflavone (1), along with two known C-alkylated flavonoids (5,7-dihydroxy-3′-(3-hydroxymethylbutyl)-3,6,4′-trimethoxyflavone (2), 5,7,4′-trihydroxy-3′-(3-hyroxymethylbutyl)-3,6-dimethoxyflavone (3) and two new source C-alkylated flavonoids (5,7-dihydroxy-3′-(2-hydroxy-3-methyl-3-butenyl)-3,6,4′-trimethoxyflavone (4), 5,7,4′-trihydroxy-3,6-dimethoxy-3′-isoprenyl-flavone (5) were isolated from the aerial parts of Dodonaea viscosa. The structures of all compounds were established on the basis of 1D and 2D NMR spectroscopy and mass spectrometry. The isolated compounds were evaluated for their inhibitory effect on urease and α-chymotrypsin enzyme. All the compounds (1–5) exhibited mild inhibition against urease but remained recessive in case of α-chymotrypsin.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Abdullah, P., Sapindaceae. In Nasir, E. and Ali, S. I. (Eds). Flora of West Pakistan No. 39. Stewart Herbarium, Gordon College, Rawalpindi, pp. 1–3, (1973).
Anis, I., Ahmed, S., Malik, A., Yasin, A., and Choudhary, M. I., Enzyme inhibitory constituents from Duranta repens. Chem. Pharm. Bull., 50, 515–518 (2002).
Arfan, M., Ali, M., Anis, I., Ahmad, H., Choudhary, M. I., Khan, A., and Shah, M. R., Urease inhibitors from Hypericum oblongifolium Wall. J. Enzym. Inhib. Med. Chem., 25, 296–299 (2010).
Arun, M. and Asha, V. V., Gastroprotective effect of Dodonaea viscosa on various experimental ulcer models. J. Ethnopharmacol., 118, 460–465 (2008).
Atta-ur-Rahman and Choudhary, M. I., Bioactive natural products as a potential source of new pharmacophores. A theory of memory. Pure Appl. Chem., 73, 555–560 (2001).
Azhar-ul-Haq, Malik, A., Khan, A. U., Shah, M. R., and Muhammad, P., Spinoside, new coumaroyl flavone glycoside from Amaranthus spinosus. Arch. Pharm. Res., 27, 1216–1219 (2004).
Cannell, R. J. P., Kellam, S. J., Owsianka, A. M., and Walker, J. M., Results of a Large Scale Screen Microalgae for the production of protease inhibitors. Planta Med., 54, 10–14 (1988).
Clarkson, C., Maharaj, V. J., Crouch, N. R., Grace, O. M., Pillay, P., Matsabisa, M. G., Bhagwandin, N., Smith, P. J., and Folb, P. I., In vitro antiplasmodial activity of medicinal plants native to or naturalized in South Africa. J. Ethnopharmacol., 92, 177–191 (2004).
Coblentz, A. and Coblentz, D. R., Treatment of peptic ulcer with chymotrypsin and an antibiotic. Mil. Med., 131, 150–154 (1966).
Coblentz, A., Treatment of geriatric peptic ulcer with chymotrypsin and an antibiotic. J. Am. Geriatr. Soc., 16, 1039–1046 (1968).
Ferheen, S., Ahmed, E., Afza, N., Malik, A., Shah, M. R., Nawaz, S. A., and Choudhary, M. I., Haloxylines A and B, antifungal and cholinesterase inhibiting piperidine alkaloids from Haloxylon salicornicum. Chem. Pharm. Bull., 53, 570–572 (2005).
Hussain, J., Ullah, F., Hussain, H., Hussain, S. T., and Shah, M. R., Nepetolide: A new diterpene from Nepeta suavis. Z. Naturforsch. B, 63, 591–594 (2008).
Hussain, J., Jamila, N., Khan, F. U., Devkota, K. P., Shah, M. R., and Anwar, S., Nepetanal and nepetanoate: A new diterpene aldehyde and a benzene derivative ester from Nepeta juncea. Magn. Reson. Chem., 47, 625–627 (2009a).
Hussain, J., Khan, F. U., Rehman, N. U., Ullah, R., Mohmmad, Z., Tasleem, S., Naeem, A., and Shah, M. R., One new triterpene ester from Nepeta suavis. J. Asian Nat. Prod. Res., 11, 997–1000 (2009b).
Iqbal, K., Malik, A., Mukhtar, N., Anis, I., Nahar, S., and Choudhary, M. I., α-Glucosidease inhibitory constituents of Duranta repens. Chem. Pharm. Bull., 52, 785–789 (2004).
Jan, A. K., Shah, M. R., Anis, I., and Marwat, I. K., In vitro antifungal and antibacterial activities of extracts of Galium tricornutum subsp Longipedunculatum. J. Enzym. Inhib. Med. Chem., 24, 192–196 (2009).
Khan, S. B., Azhar-Ul-Haq, Afza, N., Malik, A., Khan, M. T., Shah, M. R., and Choudhary, M. I., Tyrosinase-inhibitory long-chain esters from Amberboa ramosa. Chem. Pharm. Bull., 53, 86–89 (2005a).
Khan, S. B., Azhar-ul-Haq, Perveen, S., Afza, N., Malik, A., Nawaz, S. A., Shah, M. R., and Choudhary, M. I., Butyrylcholinesterase inhibitory guaianolides from Amberboa ramosa. Arch. Pharm. Res., 28, 172–176 (2005b).
Khurram, M., Khan, M. A., Hameed, A., Abbas, N., Qayum, A., and Inayat, H., Antibacterial activities of Dodonaea viscosa using contact bioautography technique. Molecules, 14, 1332–1341 (2009).
Mohammad, A., Shah, M. R., Anis, I., McKee, V., and Frese, W. E. J., 7-[2-(3-Furyl)ethyl]-7,8-dimethyl-3,5,6,6a,7,8,9,10-octahydro-1H-naphtho[1,8a-c]furan-3-one. Acta Crystallogr. Sect. E Struct. Rep. Online, 66, o2529 (2010a).
Mohammad, A., Shah, M. R., Anis, I., McKee, V., and Frese, W. E. J., 5,7-Dihydroxy-3,6-dimethoxy-2-(4-methoxyphenyl)-4Hchromen-4-one monohydrate. Acta Crystallogr. Sect. E Struct. Rep. Online, 66, o2716–o2717 (2010b).
Nisar, M., Tariq, S. A., Marwat, I. K., Shah, M. R., and Khan, I., Antibacterial, antifungal, insecticidal, cytotoxicity and phytotoxicity studies on Indigofera gerardiana. J. Enzym. Inhib. Med. Chem., 24, 224–229 (2009).
Pirzada, A. J., Shaikh, W., Usmanghani, K., and Mohiuddin, E., Antifungal activity of Dodonaea viscosa Jacq extract on pathogenic fungi isolated from superficial skin infection. Pak. J. Pharm. Sci., 23, 337–340 (2010).
Riaz, N., Anis, I., Khan, P. M., Shah, R., and Malik, A., A new triterpene from Alysicarpus monolifer. Nat. Prod. Lett., 16, 415–418 (2002).
Sachdev, K. and Kulshreshtha, D. K., Aliarin, A new flavonoid from Dodonaea viscosa Linn. Indian J. Chem. B, 21, 798–799 (1982).
Sachdev, K. and Kulshreshtha, D. K., Flavonoids from Dodonaea viscosa. Phytochemistry, 22, 1253–1256 (1983).
Sharif, A., Ahmed, E., Malik, A., Riaz, N., Afza, N., Nawaz, S. A., Arshad, M., Shah, M. R., and Choudhary, M. I., Lipoxygenase inhibitory constituents from Indigofera oblongifolia. Arch. Pharm. Res., 28, 761–764 (2005).
Veerapur, V. P., Prabhakar, K. R., Parihar, V. K., Bansal, P., Srinivasan, K. K., Priyadarsini, K. I., and Unnikrishnan, M. K., Antidiabetic, hypolipidaemic and antioxidant activity of Dodonaea viscosa aerial parts in streptozotocininduced diabetic rats. Int. J. Phytomed., 2, 59–70 (2010).
Wagner, H., Ludwig, C., Grotjahn, L., and Khan, M. S. Y., Biologically active saponins from Dodonaea viscosa. Phytochemistry, 26, 697–701 (1987).
Wollenweber, E. and Roitman, J. N., New reports on surface flavonoids from Chaaebatiaria (Rosaceae), Dodonaea (Sapindaceae), Elsholtzia (Lamiaceae), and Silphium (Asteraceae). Nat. Prod. Commun., 2, 385–389 (2007).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Muhammad, A., Anis, I., Khan, A. et al. Biologically active C-alkylated flavonoids from Dodonaea viscosa . Arch. Pharm. Res. 35, 431–436 (2012). https://doi.org/10.1007/s12272-012-0305-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-012-0305-6