Abstract
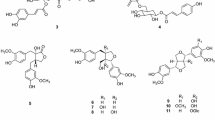
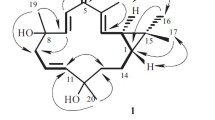
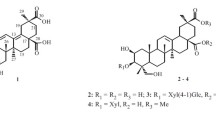
We isolated seven terpenes and five phenolic constituents from the aerial parts of Lactuca indica L. using column chromatographic separation of its MeOH extract. Their structures were determined by spectroscopic methods to be trans-phytol (1), 3β-hydroxyglutin-5-ene (2), 5,6-epoxy-3-hydroxy-7-megastigmen-9-one (3), 11β-13-dihydrolactucin (4), 2-phenylethyl β-d-glucopyranoside (5), cichorioside B (6), 1-hydroxylinaloyl-6-O-β-d-glucopyranoside (7), (6S,9S)-roseoside (8), benzyl-β-d-glucopyranoside (9), 2-(3′-O-β-d-glucopyranosyl-4′-hydroxyphenyl)-ethanol (10), 3-(β-d-glucopyranosyloxymethyl)-2-(4-hydroxy-3-methoxyphenyl)-5-(3-hydroxypropyl)-7-methoxy-dihydrobenzofuran (11), and (+)-taraxafolin-B (12). Compounds 1–3, 5, and 7–12 were isolated for the first time from this plant source. The isolated compounds were tested for cytotoxicity against four human tumor cell lines in vitro using a Sulforhodamin B bioassay.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Antinio, G. G., Esteban, A. F., and Angel, G. R., Triterpenes from Maytenus horrida. Phytochemistry, 26, 2785–2788 (1987).
Armandodoriano, B., Raffaele, A. M., Cristiana, M., Giovanni, R., Maria, L. S., Anna, S., and Nicola, U., Microcomponents of olive oil-III. glucosides of 2(3,4-dihydroxy-phenyl)ethanol. Food Chem., 63, 461–464 (1998).
Brigida, D., Marina, D., Antonio, F., Pietro, M., Palma, O., and Fabio, T., Structure elucidation and phytotoxicity of C13 nor-isoprenoids from Cestrum parqui. Phytochemistry, 65, 497–505 (2004).
Brown, G. D., Phytene-1,2-diol from Artemisia annua. Phytochemistry, 36, 1553–1554 (1994).
Chi, K. S. and Brian, M. L., Synthesis of 2-phenylethyl β-glucoside. J. Agric. Food Chem., 42, 1732–1733 (1994).
Chia, C. H., Shwu, J. L., Juei, T. C., and Feng, L. H., Antidiabetic dimeric guianolides and a lignan glycoside from Lactuca indica. J. Nat. Prod., 66, 625–629 (2003).
Funda, N. Y., Tayfun, E., Pmar, A., and Ihsan, C., Phenolic, megastigmane, nucleotide, acetophenon and monoterpene glycosides from Phlomis samia and P. carica. Turk. J. Chem., 27, 703–711 (2003).
Hui, W. H. and Lee, W. K., Triterpenoid and steroid constituents of some Latuca and Ageratum species of Hong Kong. Phytochemistry, 10, 899–901 (1971).
Kaoru, U., Itsuno, H., Toshio, M., Aktra, U., Setsuo, H., and Chizuko, K., Studies on the constituents of leaves of Citrus unshiu marcov. Chem. Pharm. Bull., 36, 5004–5008 (1988).
Kan, W. S., Pharmaceutical Botany, 7th ed. National Reasearch institute of Chinese Medicine, Taipei, Taiwan, ROC (1986).
Kim, K. H., Kim, Y. H., and Lee, K. R., Isolation of quinic acid derivatives and flavonoids from the aerial parts of Lactuca indica L. and their hepatoprotective activity in vitro. Bioorg. Med. Chem. Lett., 17, 6739–6743 (2007).
Makoto, K., Hiroshi, H., Seiichi, E., and Noboru, S., Identification of Flavones in Sixteen Compositae Species. Agric. Biol. Chem., 42, 475–477 (1978).
Mamoru, S., Toshio, M., Kaou, U., Akira, U., Yutaka, H., and Norio, O., Sesquiterpene lactones from Cichorium endivia L. and C. intybus L. and cytotoxicity activity. Chem. Pharm. Bull., 36, 2423–2429 (1988).
Park, H. J., Lee, M. S., Lee, E., Choi, M. Y., Cha, B. C., Jung, W. T., and Young, H. S., Serum cholesterol-lowering effects and triterpenoids of the herbs of Lactuca indica. Korean J. Pharmacog., 26, 40–46 (1995).
Sag, T. M., Omar, A. A., Khafagy, S. M., Grenz, M., and Bohlmann, F., 11β,13-Dihydrolactucin, a sesquiterpene lactone from Launaea mucronata. Phytochemistry, 21, 1163 (1982).
Sheng, Y. W., Hsing, N. C., Kai, T. L., Chiu, P. L., Ning, S. Y., and Lei, F. S., Antioxidant properties and phytochemical charateristics of extracts from Lactuca indica. J. Agric. Food Chem., 51, 1506–1512 (2003).
Skehan, P., Storeng, R., Scudiero, D., Monks, A., Mcmahon, J., Vistica, D., Warren, J. T., Bokesch, H., Kenney, S., and Boyd, M. R., New colorimetric cytotoxicity assay for anticancer-drug screening. J. Natl. Cancer Inst., 82, 1107–1112 (1990).
Stephen, J. P., Louise, N. J., and David, C. P., High-resolution 1H-and 13C-NMR. Spectra of d-glucopyranose, 2-acetamido-2-deoxy-d-glucopyranose, and related compounds in aqueous media. Carbohydr. Res., 59, 19–34 (1977).
Tsutomu, N., Naoki, I., Yuka, I., Hiroko, M., Akira, I., Jin, M., Frank, A. L., Munekazu, I., and Toshiyuki, T., Neolignan and flavonoid glycosides in Juniperus communis var. depressa. Phytochemistry, 65, 207–213 (2004).
Veronique, S., Francois, B., and Peter, G. W., Novel oligorhamnosides from the stem bark of Cleistopholis glauca. J. Nat. Prod., 63, 6–11 (2000).
Yann, L. L., Yu, L. W., Shih, C. H., and Li, S. S., Chemical constituents from root of Taraxacum formosanum. Chem. Pharm. Bull., 53, 853–855 (2005).
Yumiko, Y. and Masayoshi, I., Synthesis of optically active vomifoliol and roseoside stereoisomers. Chem. Pharm. Bull., 53, 541–546 (2005).
Yves, C., Gilles, C., Joseph, V., Daovy, P. A., and Albert, J. C., Norterpenoid and sesquiterpenoid glucosides from Juniperus phoenicea and Galega officinalis. Phytochemistry, 50, 1219–1223 (1999).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, K.H., Lee, K.H., Choi, S.U. et al. Terpene and phenolic constituents of Lactuca indica L.. Arch. Pharm. Res. 31, 983–988 (2008). https://doi.org/10.1007/s12272-001-1256-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-001-1256-8