Abstract
Objective
It has been reported that D-galactose (D-gal) can model subacute aging, and aluminum (Al) acts as a neurotoxin, but combined effects of them have not been reported. The present work aimed to reveal the effect of combined administration of D-gal and Al in mice and compare the effect of D-gal treatment with that of Al treatment.
Methods
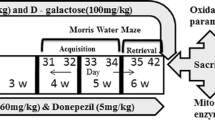
Al was intragastrically administered and D-gal was subcutaneously injected into Kunming mice for 10 consecutive weeks. Learning and memory, cholinergic systems, as well as protein levels of amyloid β (Aβ) and hyperphosphorylated tau were determined using Morri water maze test, biochemical assays and immunohistochemical staining, respectively.
Results
The mice with combined treatment had obvious learning and memory deficits, and showed decreases in brain acetylcholine (ACh) level and in activities of choline acetyltransferase (ChAT) and acetylcholinesterase (AChE). Formation of senile plaque (SP)-like and neurofibrillary tangle (NFT)-like structures was also observed. The behavioral and pathological changes persisted for at least 6 weeks after withdrawal of D-gal and Al.
Conclusion
Combined use of D-gal and Al is an effective way to establish the non-transgenic Alzheimer’s disease (AD) animal model, and is useful for studies of AD pathogenesis and therapeutic evaluation.
摘要
目的
D-半乳糖能制作亚急性衰老模型, 铝具有神经毒性, 但两者联合应用的作用未见报道。 本研究旨在探讨D-半乳糖和铝联合应用对动物学习记忆、 脑内生化和病理的影响, 以及与单独应用D-半乳糖或铝所制作的动物模型相比较。
方法|
昆明小鼠单独皮下注射D-半乳糖、 单独灌胃铝以及既注射D-半乳糖又灌胃铝, 制作动物模型, 共给药8周或10周, 10周后再停用药物6周。 在第8、 10、 16周末, 采用Morris水迷宫检测小鼠学习记忆能力, 生化学方法检测脑内乙酰胆碱能系统, 免疫组化法检测老年斑和神经原纤维缠结的形成。
结果
联合应用D-半乳糖和铝后, 小鼠表现出明显的学习和记忆力障碍, 并且其脑内乙酰胆碱水平降低, 乙酰胆碱转移酶和胆碱脂酶活性下降, 出现老年斑样和神经原纤维缠结样病理改变。 停止给药后, 其行为学、 生化和病理改变至少能维持6周以上。
结论
小鼠中D-半乳糖和铝联合应用是一个有效的非转基因阿尔茨海默病(Alzheimer’s disease, AD)模型, 可用于AD病理研究和相关治疗药物的评价。
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Whitehouse PJ, Price DL, Struble RG, Clark AW, Coyle JT, Delon MR. Alzheimer’s disease and senile dementia: loss of neurons in the basal forebrain. Science 1982, 215(4537): 1237–1239.
Selkoe, DJ. Alzheimer’s disease: Genes, proteins, and therapy. Physiological Rev 2001, 81(2): 741–766.
Duff K, Hardy J. Alzheimer’s disease: mouse model made. Nature 1995, 373(6514): 476–477.
Kawabata S, Higgins GA, Gordon JW. Amyloid plaques, neurofibrillary tangles and neuronal loss in brains of transgenic mice overexpressing a C-terminal fragment of human amyloid precursor protein. Nature 1991, 354(6353): 476–478.
Quon D, Wang Y, Catalano R, Scardina JM, Murakami K, Cordell B. Formation of beta-amyloid protein deposits in brains of transgenic mice. Nature 1991, 352(6332): 239–241.
Ho SC, Liu JH, Wu RY. Establishment of the mimetic aging effect in mice caused by D-galactose. Biogerontology 2003, 4(1): 15–18.
Zhang Q, Li X, Cui X, Zuo P. D-galactose injured neurogenesis in the hippocampus of adult mice. Neurol Res 2005, 27(5): 552–556.
Wei H, Li L, Song Q, Ai H, Chu J, Li W. Behavioural study of the D-galactose induced aging model in C57BL/6J mice. Behav Brain Res 2005, 57(2): 245–251.
Cui X, Wang L, Zuo P, Han Z, Fang Z, Li W, et al. D-galactosecaused life shortening in Drosophila melanogaster and Musca domestica is associated with oxidative stress. Biogerontology 2004, 5(5): 317–325.
Ida H, Ishibashi K, Reiser K, Hjelmeland LM, Handa JT. Ultrastructural aging of the RPE-Bruch’s membrane-choriocapillaris complex in the D-galactose-treated mouse. Invest Ophthalmol Vis Sci 2004, 45(7): 2348–2354.
Song X, Bao M, Li D, Li YM. Advanced glycation in D-galactoseinduced mouse aging model. Mech Ageing Dev 1999, 108(3): 239–251.
Moumen R, Ait-Oukhatar N, Bureau F, Fleury C, Bouglé D, Arhan P, et al. Aluminium increases xanthine oxidase activity and disturbs antioxidant status in the rat. J Trace Elem Med Biol 2001, 15(2–3): 89–93.
Platt B, Fiddler G, Riedel G, Henderson Z. Aluminium toxicity in the rat brain: histochemical and immunocytochemical evidence. Brain Res Bull 2001, 55(2): 257–267.
Boni UD, Otvos A, Scott JW, Crapper DR. Neurofibrillary degeneration induced by systemic aluminum. Acta Neuropathol (Berl) 1976, 35(4): 285–294.
Muller JP, Bruinink A. Neurotoxic effects of aluminium on embryonic chick brain cultures. Acta Neuropathol (Berl) 1994, 88(4): 359–366.
Hermenegildo C, Saez R, Minoia C, Manzo L, Felipo V. Chronic exposure to aluminium impairs the glutamate-nitric oxide-cyclic GMP pathway in the rat in vivo. Neurochem Int 1999, 34(3): 245–253.
Henderson AS. Epidemiology of dementia disorders. In: Wurtman RJ, Corkin S, Growdon JH, Ritter-Walker E (Eds). Alzheimer’s disease. Vol. 51 Advances in neurology. New York: Raven Press, 1990: 15–25.
Luo HM, Xiao F. Alzheimer-like pathological changes of mice induced by D-galactose and aluminum trichloride. Chin J Pharmacol Toxicol 2004, 18(1): 22–26.
Luo HM, Xiao F. Preparing method for Alzheimer disease animal model [P]. CN, CN1278603.2006-10-11.
Litchfield S, Nagy Z. New temperature modification makes the Bielschowsky silver stain reproducible. Acta Neuropathologica 2001, 101(1): 17–21.
Andrasi E, Pali N, Molnar Z, Kösel S. Brain aluminum, magnesium and phosphorus contents of control and Alzheimer-diseased patients. J Alzheimers Dis 2005, 7(4): 273–284.
Fattoretti P, Bertoni-Freddari C, Balietti M, Giorgetti B, Solazzi M, Zatta P. Chronic aluminum administration to old rats results in increased levels of brain metalions and enlarged hippocampal mossy fibers. Ann N Y Acad Sci 2004, 1019(8): 44–47.
Deloncle R, Guillard O. Mechanism of Alzheimer’s disease: arguments for a neurotransmitter-aluminium complex implication. Neurochem Res 1990, 15(12): 1239–1245.
McLachlan DR, Lukiw WJ, Cho HJ, Carp RI, Wisniewski H. Chromatin structure in scrapie and Alzheimer’s disease. Can J Neurol Sci 1986, 13(4 Suppl): 427–431.
Yokel RA. Aluminum produces age related behavioral toxicity in the rabbit. Neurotoxicol Teratol 1989, 11(3): 237–242.
Guo GW, Wu YL, Yang XH, Guo LN, Yang YX. Effects of aluminum chloride on amyloid β-protein precursor and glial fibrillary acidic protein expression in rat cortex. Chin J Pharmacol Toxicol 1999, 13(3): 227–230.
Qian YH, Yang J, Ren HM, Hu HT, Zhang ZJ. Immunocytochemical study of amyloid protein accumulation in dorsal hippocampal formation of a rat model of dementia. J Xian Medi Univ 1997, 18(3): 304–307.
McLachlan DR, Kruck TP, Lukiw WJ, Krishnan SS. Would decreased aluminum ingestion reduce the incidence of Alzheimer’s disease? CMAJ 1991, 145(7): 793–804.
Pepeu G, Giovannini MG. Changes in acetylcholine extracellular levels during cognitive processes. Learn Mem 2004, 11(1): 21–27.
Author information
Authors and Affiliations
Additional information
These authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Xiao, F., Li, XG., Zhang, XY. et al. Combined administration of D-galactose and aluminium induces Alzheimerlike lesions in brain. Neurosci. Bull. 27, 143–155 (2011). https://doi.org/10.1007/s12264-011-1028-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12264-011-1028-2