Abstract
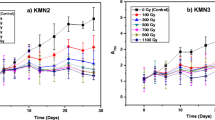
Microalgae have been drawing much attention as a platform for food supplements and biofuel production. Advance molecular tools are not available for manipulating microalgae, and therefore, methods for strain improvement mostly depend on random mutation. Radiation is frequently used mutagen in plant as well as microalgae breeding methods. In this study, the resistance of 7 microalgae species to ionizing irradiation was measured. To monitor the growth of microalgae, optical density and staining methods were used. Based on the D10 values, the dose required to reduce one log cycle of the cell population, Chlorella protothecoides, Zygnema circumcarinatum, and Spirogyra varians were shown to be highly resistant to ionizing radiation. The changes in protein expression levels in S. varians were further investigated. Using 2-dimensional electrophoresis and protein identification, it was shown that some proteins involved in energy and glyceride metabolisms were up-regulated. These results provide fundamental insights into metabolic changes that occur in a microalga species upon exposure to ionizing irradiation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Dang, N. M. and K. Lee (2018) Utilization of organic liquid fertilizer in microalgae cultivation for biodiesel production. Biotechnol. Bioproc. Eng. 23: 405–414.
Hong, S. J., Y. S. Park, M. A. Han, Z. H, Kim, B. K. Cho, H. Lee, H. K. Choi, and C. G. Lee (2017) Enhanced production of fatty acids in three strains of microalgae using a combination of nitrogen starvation and chemical inhibitors of carbohydrate synthesis. Biotechnol. Bioproc. Eng. 22: 60–67.
Ahloowalia, B. S. and M. Maluszynski (2001) Induced mutations–A new paradigm in plant breeding. Euphytica 118: 167–173.
Holzinger. A. and C. Lütz (2006) Algae and UV irradiation: Effects on ultrastructure and related metabolic functions. Micron 37: 190–207.
Rastogi R. P., R. P. Sinha, S. H. Moh, T. K. Lee, S. Kottuparambil, Y. J. Kim, J. S. Rhee, E. M. Choi, M. T. Brown, D. P. Häder, and T. Han (2014) Ultraviolet radiation and cyanobacteria. J. Photoch. Photobio. B 141:154–69.
Garcia, M. M., B. W. Brooks, R. B. Stewart, W. Dion, J. R. Trudel, and T. Ouwerkerk (1987) Evaluation of gamma radiation levels for reducing pathogenic bacteria and fungi in animal sewage and laboratory effluents. Can. J. Vet. Res. 51: 285–289.
Berberoglu, H., P. S. Gomez, and L. Pilon (2009) Radiation characteristics of Botryococcus braunii, Chlorococcum littorale, and Chlorella sp. Used for CO2 fixation and biofuel production. J. Quant. Spectrosc. Ra. 110: 1879–1893.
Rotman, B. and B.W. Papermaster (1996) Membrane properties of living mammalian cells as studied by enzymatic hydrolysis of fluorogenic esters. P. Natl. Acad. Sci. USA 55: 134–141.
Choi, J., M. Yoon, S. Lim, G. H. Kim, and H. Park (2015) Effect of gamma irradiation on physiological and proteomic changes of Arctic Zygnema sp. (Chlorophyta, Zygnematales). Phycologia 54: 333–341.
Yoon, M., J. Choi, G. H. Kim, D. H. Kim, and D. H. Park (2013) Proteomic analysis of Spirogyra varians mutant with high starch content and growth rate induced by gamma irradiation. Bioproc. Biosyst. Eng. 36: 757–763.
Joe, M. H., J. Y. Kim, S. Lim, D. H. Kim, S. Bai S., H. Park, S. G. Lee, S. J. Han, and J. Choi (2015) Microalgal lipid production using the hydrolysates of rice straw pretreated with gamma irradiation and alkali solution. Biotechnol. Biofuels 8: 125.
Choi, J., M. Yoon, M. Joe, H. Park, S. G. Lee, S. J. Han, and P. C. Lee (2014) Development of microalga Scenedesmus dimorphus mutant with higher lipid content by radiation breeding. Bioproc. Biosyst. Eng. 37: 2437–2444.
Baek, J., J. Choi, H. Park, S. Lim, and S. J. Park (2016) Isolation and proteomic analysis of a Chlamydomonas reinhardtii mutant with enhanced lipid production by the gamma irradiation method. J. Microbiol. Biotechnol. 26: 2076–2085.
Wu, H., J. V. Volponi, A. E. Oliver, A. N. Parikh, B. A. Simmons, and S. Singh (2011) P. Natl. Acad. Sci. USA 108: 3809–3814.
Fischer, M. and R. G. Sawers (2013) A universally applicable and rapid method for measuring the growth of streptomyces and other filamentous microorganisms by methylene blue adsorptiondesorption. Appl. Environ. Microb. 79: 4499–4502.
Rotman, B. and B. W. Papermaster (1966) Membrane properties of living mammalian cells as studied by enzymatic hydrolysis of fluorogenic esters. P. Natl. Acad. Sci. USA 55: 134–141.
Herburger, H. and A. Holzinger (2015) Localization and quantification of callose in the streptophyte green algae Zygnema and Klebsormidium: Correlation with desiccation tolerance. Plant and Cell Physiology 56: 2259–2270.
Germ, M., I. Kreft, and A. Gaberscik (2009) UV–B radiation and selenium affected energy availability in green alga Zygnema. Biologia 64: 676–679.
Huss, V. A., C. Ciniglia, P. Cennamo, S. Cozzolino, G. Pinto, and A. Pollio (2002) Phylogenetic relationships and taxonomic position of Chlorella–like isolates from low pH environments (pH < 3.0). BMC Evol. Biol. 2:13.
Slade, D. and M. Radman (2011) Oxidative Stress Resistance in Deinococcus radiodurans. Microbiol. Mol. Biol. R. 75: 133–191.
Singh H. (2018) Desiccation and radiation stress tolerance in cyanobacteria. J. Basic Microb. 58: 813–826.
Yoon, M., H. Y. Yang, S. S. Lee, D. H. Kim, G. H. Kim, and J. Choi (2013) Characterization of gamma radiation inducible thioredoxin h from Spirogyra varians. Enzyme Microb. Tech. 53: 136–142.
Ciereszko, I., H. Johansson, V. Hurry, and L. A. Kleczkowski (2001) Phosphate status affects the gene expression, protein content and enzymatic activity of UDP–glucose pyrophosphorylase in wild–type and pho mutants of Arabidopsis. Planta 212: 598–605.
Wang, S. B., F. Chen, and M. Sommerfeld (2004) Proteomic analysis of molecular response to oxidative stress by the green alga Haematococcus pluvialis (Chlorophyceae). Planta 220: 17–29.
Hasunuma, K., N. Yabe, Y. Yoshida, Y. Ogura, and T. Hamada (2003) Putative functions of nucleoside diphosphate kinase in plants and fungi. J. Bioenerg. Biomembr. 35:57–65.
Wei, S. J., C. S. Trempus, R. C. Ali, L. A. Hansen, and R. W. Tennant (2004) 12–O–tetradecanoylphorbol–13–acetate and UV radiationinduced nucleoside diphosphate protein kinase B mediates neoplastic transformation of epidermal cells. J. Biol. Chem. 279: 5993–6004.
Yasunobu, O., Y. Yoshida, N. Yabe, and K. Hasunuma (2001) A point mutation in nucleoside diphosphate kinase results in a deficient light response for perithecial polarity in Neurospora crassa. J. Biol. Chem. 276: 21228–21234.
Blomberg A. and L. Adler (1989) Roles of glycerol and glycerol–3–phosphate dehydrogenase (NAD+) in acquired osmotolerance of Saccharomyces cerevisiae. J. Bacteriol. 171: 1087–1092.
Wu, L., J. Wu, Y. Liu, X. Gong, J. Xu, D. Lin, and Y. Dong (2016) The rice pentatricopeptide repeat gene TCD10 is needed for chloroplast development under cold stress. Rice 9: 67.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, EJ., Choi, Ji. Resistance and Proteomic Response of Microalgae to Ionizing Irradiation. Biotechnol Bioproc E 23, 704–709 (2018). https://doi.org/10.1007/s12257-018-0468-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-018-0468-1