Abstract
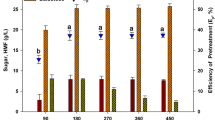
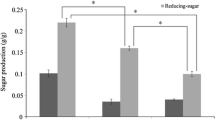
Saccharina japonica (Sea tangle, Dasima), a seaweed, was fermented in order to produce bioethanol after thermal hydrogen peroxide (H2O2) hydrolysis pretreatment and enzymatic saccharification. The optimal pretreatment conditions of 1% (v/v) H2O2 (28%, Dustan Pure Chemicals Co., Ltd, Ansan, Korea) and 10% (w/v) seaweed slurry at 121°C for 60 min were determined using the Response Surface Method (RSM). A reducing sugar yield of 33.4% (w/w) and a viscosity of 520 cP were obtained. Enzymatic saccharification was then carried out; a monosaccharide concentration of 28.5 g/L with a 40.5% (w/w) theoretical yield was obtained after the addition of 2-mL Celluclast® 1.5L to 100 g/L of seaweed slurry after thermal H2O2 hydrolysis. Fermentation of a two-stage ethanol production was carried out using Saccharomyces cerevisiae KCCM 1129 in order to ferment glucose in the first stage, and a high level of mannitol-acclimated Pichia angophorae KCTC 17574 to ferment mannitol in the second stage. Acclimation of yeast effectively slowed the uptake of sugar in ethanol fermentation. The overall ethanol yield from S. japonica after the two-stage fermentation was 9.9 g/L.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Lin, Y. and S. Tanaka (2006) Ethanol fermentation from biomass resources; current state and prospects. Appl. Microbiol. Biotechnol. 69: 627–642.
Horn, S. J., I. M. Aasen, and K. Østgaard (2000) Ethanol production from seaweed extract. J. Ind. Microbiol. Biotechnol. 25: 249–254.
Ge, L. W., W. Peng, and M. Haijin (2011) Study on saccharification techniques of seaweed wastes for the transformation of ethanol. Renew Energy. 36: 84–89.
Cardona, C. A. and O. J. Sanchez (2007) Fuel ethanol production: Process design trends and integration opportunities. Bioresour. Technol. 98: 2415–2457.
Saha, B. C. and M. A. Cotta (2006) Ethanol production from alkaline peroxide pretreated enzymatically saccharified wheat straw. Biotechnol. Prog. 22: 449–453.
Kang, H. K., N. M. Kim, and D. M. Kim (2011) Enhanced saccharification of rice straw using hypochlorite-hydrogen peroxide. Biotechnol. Bioproc. Eng. 16: 273–281.
AOAC (Association of Official Analytical Chemists) (1995) In: Cunniff, P. (ed.). Official methods of analysis of the association of official analytical chemists. 16th ed. Association of Official Analytical Chemists, Arlington, VA.
Yoo, C. G., C. W. Lee, and T. H. Kim (2011) Optimization of two-stage fraction process for lignocellulosic biomass using response surface methodology (RSM). Biomass & Bioenergy 35: 4901–4909.
Jang, J. S., Y. K. Cho, G. T. Jeong, and S. K. Kim (2012) Optimization of saccharification and ethanol production by simultaneous saccharification and fermentation (SSF) from seaweed, Saccharina japonica. Bioproc. Biosyst. Eng. 35: 11–18.
Irfan, M., M. Gulsher, S. Abbas, Q. Syed, M. Nadeem, and S. Baig (2011) Effect of various pretreatment conditions on enzymatic saccharification. Songklanakarin. J. Sci. Technol. 33: 397–404.
Lee, S. M. and J. H. Lee (2012) Ethanol fermentation for main sugar components of brown-algae using various yeasts. Ind. Eng. Chem. 18: 16–18.
Ernandes, J. R., J. W. Williams, and G. G. Stewart (1992) Simultaneous utilization of galactose and glucose by Saccharomyces spp. Biotechnol. Techniques. 6: 233–238.
Horn, S. J., I. M. Aasen, and K. Østgaard (2000) Production of ethanol from mannitol by Zymobacter palmae. J. Ind. Microbiol. Biotechnol. 24: 51–57.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ra, C.H., Kim, SK. Optimization of pretreatment conditions and use of a two-stage fermentation process for the production of ethanol from seaweed, Saccharina japonica . Biotechnol Bioproc E 18, 715–720 (2013). https://doi.org/10.1007/s12257-013-0019-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-013-0019-8