Abstract
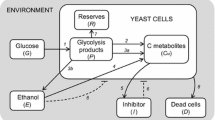
Production of Saccharomyces cerevisiae yeast for applications in the food industry or in bioethanol production still presents important techno-economic challenges as an industrial bioprocess. Mathematical modeling of cellular metabolism in biological production usually improves process yields, though for industrial applications, the model should be as simple as possible in order to sustain model usefulness and technical feasibility. A comparative analysis between a black box description and a simple metabolic network accounting for the main metabolic events involved in yeast growth and bioethanol production is proposed here. In both cases, a thorough analysis of reaction rates allowed for the ethanol concentrations produced in fed-batch yeast cultures, although our results showed more accurate estimations with the metabolic flux balance methodology. Moreover, an interpretation of the yeast physiological state in fed-batch cultures at different glucose feed concentrations was accomplished by means of a stoichiometric analysis linked to the simplified metabolic network. The results confirmed that increasing glucose uptake rates, controlled mainly by the glucose concentration in the input flow, produced an up-regulation in reductive catabolism, resulting in higher ethanol excretion. The biomass production relied mostly on oxidative catabolism, which is controlled by the glucose and oxygen uptake rates. Thus, ethanol or biomass production is strongly dependent on the physiological state of yeast in the culture, which can be inferred from a suitable metabolic flux balance approach.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Jørgensen, H., L. Olsson, B. Rønnow, and E. A. Palmqvist (2002) Fed-batch cultivations of baker’s yeast followed by a nitrogen or carbon starvation: Effects on fermentative capacity and content of trehalose and glycogen. App. Microbiol. Biotechnol. 59: 310–317.
Antoni, D., V. Zverlov, and W. H. Schwarz (2007) Biofuels from microbes. App. Microbiol. Biotechnol. 77: 23–35.
Cannizzaro, C., S. Valentinotti, and U. von Stockar (2004) Control of yeast fed-batch process through regulation of extracellular ethanol concentration. Bioproc. Biosys. Eng. 26: 377–383.
Kwiatkowski, J. R., A. J. McAloon, F. Taylor, and D. B. Johnston (2006) Modeling the process and costs of fuel ethanol production by corn dry-grind process. Indus. Crops Prod. 23: 288–296.
Hahn-Hägerdal, B., M. Galbe, M. F. Gorwa-Grauslung, G. Lidén, and G. Zacchi (2006) Bio-ethanol — the fuel of tomorrow from the residues of today. Trends Biotechnol. 24: 549–556.
Hill, J., E. Nelson, D. Tilman, S. Polasky, and D. Tiffanty (2005) Environmental, economic, and energetic costs and benefits of biodiesel and ethanol biofuels. Proc. Natl. Acad. Sci. USA 103: 11206–11210.
Wong, W. Ch., H. -S. Song, J. H. Lee, and D. Ramkrishna (2010) Hybrid cybernetic model-based simulation of continuous producqtion of lignocellulosic ethanol: Rejecting abruptly changing feed conditions. Contrl. Eng. Pract. 18: 177–189.
Wu, W. H., F. Sh. Wang, and M. Sh. Chang (2008) Dynamic sensitivity analysis of biological systems. BMC Bioinformatics 9: 1–17.
Pricen, N. D., J. L. Reed, and B. O. Palsson (2004) Genome-scale models of microbial cells: Evaluating the consequences of constraints. Nat. Rev. Microbiol. 2: 886–897.
Mahadevan, R., A. P. Burgard, I. Famili, S.van Dien, and Ch. H. Schilling (2005) Applications of metabolic modelling to drive process development for the production of value-added chemicals. Biotechnol. Bioproc. Eng. 10: 408–417.
Hjersted, J. L., M. A. Henson, and R. Mahadevan (2007) Genome-scale analysis of Saccharomyces cerevisiae metabolism and ethanol production in fed-batch culture. Biotechnol. Bioeng. 97: 1190–1204.
Bro, Ch., B. Regenberg, J. Förster, and J. Nielsen (2006) In silico aided metabolic engineering of Saccharomyces cerevisiae for improved bioethanol production. Metabolic Eng. 8: 102–111.
Shiyota, S., H. Shimizu, T. Hirasawa, K. Nagahisa, C. Furusawa, G. Pandey, and Y. Katakura (2007) Metabolic pathway recruiting through genome data analysis for industrial application of Saccharomyces cerevisiae. Biochem. Eng. J. 36: 28–37.
Burgard, A. P., S. Vaidyaraman, and C. D. Maranas (2001) Minimal reaction sets for Eschericha coli metabolism under different growth requirements and uptake environments. Biotechnolg. Prog. 17: 791–797.
Aranda-Barradas, J. S., C. Garibay-Orijel, J. A. Badillo-Corona, and E. Salgado-Manjarrez (2010) A stochiometric analysis of biological xylitol production. Biochem. Eng. J. 50: 1–9.
Herwig, Ch. and U. von Stockar (2002) A small metabolic flux model to identify transient metabolic regulations in Saccharomyces cerevisiae. Bioproc. Biosys. Eng. 24: 395–403.
Merchuk, J. C., S. Yona, M. H. Siegel, and A. Ben Zvi (1989) On the first order approximation to the response of dissolved oxygen electrodes for dynamic k L a estimation. Biotechnol. Bioeng. 35: 1161–1163.
Scargiali, F., A. Busciglio, F. Grisafi, and A. Brucato (2010) Simplified dynamic pressure method for k L a measurements in aerated bioreactors. Biochem. Eng. J. 49: 165–172.
Moresi, M. and M. Patete (1988) Prediction of k L a in conventional stirred fermenters. J. Chem. Tech. Biotechnol. 42: 197–210.
Nielsen, J. and J. Villadsen (1994) Bioreaction Engineering Principles. pp. 100–102. Plenum Press, NY, USA.
Soto-Cruz, O. and J. Páez-Lerma (2005) Fermentation process balances: Consistency and metabolic flux analysis. Rev. Mex. Ing. Quim. 4: 59–74.
van der Heijden, R. T. J. M., B. Romein, J. J. Heijnen, C. Hellinga, and K. Luyben (1994) Linear constraint relations in biochemical reaction system: II. Diagnosis and estimation of gross errors. Biotechnol. Bioeng. 43: 11–20.
Nielsen, J. and J. Villadsen (1994) Bioreaction Engineering Principles. pp. 138–141. Plenum Press, NY, USA.
Stephanopoulos, G. N., A. A. Aristidou, and J. Nielsen (1998) Metabolic Engineering Principles and Methodologies. pp. 138–139. Academic Press, NY, USA.
Llaneras, F. and J. Picó (2008) Stoichiometric modelling of cell metabolism. J. Biosci. Bioeng. 105: 1–11.
Nielsen, J. and J. Villadsen (1994) Bioreaction Engineering Principles. pp. 111–116. Plenum Press, NY, USA.
Fiechter, A., G. F. Furhmann, and O. Käppeli (1981) Regulation of glucose metabolism in growing yeast cells. Adv. Microbiol. Physiol. 22: 123–183.
Cooney, Ch. L., H. Y. Wang, and D. I. C. Wang (1977) Computeraided baker’s yeast fermentations. Biotech. Bioeng. 19: 69–86.
Gancedo, J. M. (1998) Yeast carbon catabolite repression. Microbiol. Mol. Biol. Rev. 62: 334–361.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Barrera-Martínez, I., Axayácatl González-García, R., Salgado-Manjarrez, E. et al. A simple metabolic flux balance analysis of biomass and bioethanol production in Saccharomyces cerevisiae fed-batch cultures. Biotechnol Bioproc E 16, 13–22 (2011). https://doi.org/10.1007/s12257-010-0176-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-010-0176-y