Abstract
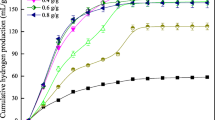
The optimum conditions for biological hydrogen production from food waste by Clostridium beijerinckii KCTC 1875 were investigated. The optimum initial pH and fermentation temperature were 7.0 and 40°C, respectively. When the pH of fermentation was controlled to 5.5, a maximum amount of hydrogen could be obtained. Under these conditions, about 2,737 mL of hydrogen was produced from 50 g COD/L of food waste for 24 h, and the hydrogen content in the biogas was 38%. Hydrogen production rate and yield were about 108 mL/L·h and 128 mL/g CODdegraded, respectively. High concentrations of acetic (< 5,000 mg/L) or butyric acid (< 3,000 mg/L) significantly inhibited hydrogen production.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Ming, L., Z. Youcai, G. Qiang, Q. Xiaoqing, and N. Dongjie (2008) Bio-hydrogen production from food waste and sewage sludge in the presence of aged refuse excavated from refuse landfill. Renewable Energy doi:10.1016/j.renene.2008.02.018.
Schnackenberg, J., R. Schulz, and H. Senger (1993) Characterization and purification of a hydrogenase from the eukaryotic green alga Scenedesmus obliquus. FEBS Lett. 327: 21–24.
Shi, D. J., M. Brouers, D. O. Hall, and R. J. Rubins (1987) The effects of immobilization on the biochemical, physiological and morphological features of Anabaena azollae. Planta 172: 298–308.
Banerjee, M., A. Kumar, and H. D. Kumar (1989) Factors regulating nitrogenase activity and hydrogen evolution in Azolla-Anabaena symbiosis. Int. J. Hydrogen Energy 14: 871–879.
Tsygankov, A. S., L. T. Serebryakova, D. A. Sueshnikov, K. K. Rao, I. N. Gogotov, and D. O. Hall (1997) Hydrogen photoproduction by three different nitrogenases in whole cells of Anabaena variabilis and dependence on pH. Int. J. Hydrogen Energy 22: 859–867.
Krahn, E., K. Schneider, and K. Muller (1996) Comparative characterization of H2 production by the conventional Mo nitrogenase and the alternative “irononly” nitrogenase of Rhodobacter capsulatus hup-mutants. Appl. Microbiol. Biotechnol. 46: 285–290.
Fascetti, E., E. D’Addario, O. Todini, and A. Robertiello (1998) Photosynthetic hydrogen evolution with volatile organic acids derived from the fermentation of source selected municipal solid wastes. Int. J. Hydrogen Energy 23: 753–760.
Oh, Y. K., Y. J. Kim, J. Y. Park, T. H. Lee, and M. S. Kim, and S. Park (2005) Biohydrogen production from carbon monoxide and water by Rhodopseudomonas palustris P4. Biotechnol. Bioprocess Eng. 10: 270–274.
Chen, W. H., S. Y. Chen, S. K. Khanal, and S. Sung (2006) Kinetic study of biological hydrogen production by anaerobic fermentation. Int. J. Hydrogen Energy 31: 2170–2178.
Taguchi, F., N. Mizukami, K. Hasegawa, T. Saito-Taki, and M. Morimoto (1994) Effect of amylase accumulation on hydrogen production by Clostridium beijerinckii, strain AM21B. J. Ferment. Bioeng. 77: 565–567.
Oh, Y. K., M. S. Park, E. H. Seol, S. J. Lee, and S. Park (2003) Isolation of hydrogen-producing bacteria from granular sludge of an upflow anaerobic sludge blanket reactor. Biotechnol. Bioprocess Eng. 8: 54–57.
Shin, H. S., J. H. Youn, and S. H. Kim (2004) Hydrogen production from food waste in anaerobic mesophilic and thermophilic acidogenesis. Int. J. Hydrogen Energy 29:1355–1363.
Noike, T., I. B. Ko, S. Yokoyama, Y. Kohno, and Y. Y. Li (2005) Continuous hydrogen production from organic waste. Water Sci. Technol. 52: 145–151.
Fan, K. S., N. Kan, and J. Lay (2006) Effect of hydraulic retention time on anaerobic hydrogenesis in CSTR. Bioresour. Technol. 97: 84–89.
Benemann, J. (1996) Hydrogen biotechnology: progress and prospects. Nat. Biotechnol. 14: 1101–1103.
Nandi, R. and S. Sengupta (1998) Microbial production of hydrogen: an overview. Crit. Rev. Microbiol. 24: 61–84.
Levin, D. B., L. Pitt, and M. Love (2004) Biohydrogen production: prospects and limitations to practical application. Int. J. Hydrogen Energy 29: 173–185.
Lin, P. Y., L. M. Whang, Y. R. Wu, W. J. Ren, C. J. Hsiao, S. L. Li, and J. S. Chang (2007) Biological hydrogen production of the genus Clostridium: Metabolic study and mathematical model simulation. Int. J. Hydrogen Energy 32: 1728–1735.
Fang, H. H. P., T. Zhang, and H. Liu (2002) Microbial diversity of a mesophilic H2-producing sludge. Appl. Microbiol. Biotechnol. 58: 112–118.
Fang, H. H. P., H. Liu, and T. Zhang (2002) Characterization of a hydrogen-producing granular sludge. Biotechnol. Bioeng. 78: 44–52.
Iyer, P., M. A. Bruns, H. Zhang, S. Van Ginkel, and B. E. Logan (2004) H2-producing bacterial communities from a heat-treated soil inoculum. Appl. Microbiol. Biotechnol. 66: 166–173
Wu, S. Y., C. H. Hung, C. N. Lin, H. W. Chen, A. S. Lee, and J. S. Chang (2006) Fermentative hydrogen production and bacterial community structure in high-rate anaerobic bioreactors containing silicone-immobilized and self-flocculated sludge. Biotechnol. Bioeng. 93: 934–946.
Taguchi, F., J. D. Chang, S. Takiguchi, and M. Morimoto (1992) Efficient hydrogen production from starch by a bacterium isolated from termites. J. Ferment. Bioeng. 73: 244–245.
Gómez, X., A. Morán, M. J. Cuetos, and M. E. Sánchez (2006) The production of hydrogen by dark fermentation of municipal solid wastes and slaughterhouse waste: A two-phase process. J. Power Sources 157: 727–732.
Chen, W. M., Z. J. Tseng, K. S. Lee, and J. S. Chang (2005) Fermentative hydrogen production with Clostridium butyricum CGS5 isolated form anaerobic sewage sludge. Int. J. Hydrogen Energy 30: 1063–1070.
Lay, J. J., K. S. Fan, J. Chang, and C. H. Ku (2003) Influence of chemical nature of organic wastes on their conversion to hydrogen by heat-shock digested sludge. Int. J. Hydrogen Energy 28: 1361–1367.
Hatch, J. L. and K. T. Finneran (2008) Influence of reduced electron shuttling compounds on biological H2 production in the fermentative pure culture Clostridium beijerinckii. Curr. Microbiol. 56: 268–273.
Jeong, T. Y., G. C. Cha, S. H. Yeom, and S. S. Choi (2008) Comparison of hydrogen production by four representative hydrogen-producing bacteria. J. Ind. Eng. Chem. 14: 333–337.
Lasko, D. R., N. Zamboni, and U. Sauer (2000) Bacterial response to acetate challenge: a comparison of tolerance among species. Appl. Microbiol. Biotechnol. 54:243–247.
Wang, G. and D. I. C. Wang (1984) Elucidation of growth inhibition and acetic acid production by Clostridium thermoaceticum. Appl. Environ. Microbiol. 47: 294–298.
Booth, I. R. (1985) Regulation of cytoplasmic pH in bacteria. Microbiol. Rev. 49: 359–378.
Donnison, A. M., C. M. Brockelsby, H. W. Morgan, and R. M. Daniel (1989) The degradation of lignocellulosics by extremely thermophilic microorganisms. Biotechnol. Bioeng. 33: 1495–1499.
van Niel, E. W. J., P. A. M. Claassen, and A. J. M. Stams (2003) Substrate and product inhibition of hydrogen production by the extreme thermophile, Caldicellulosiruptor saccharolyticus. Biotechnol. Bioeng. 81: 255–262.
Khanal, S. K., W. H. Chen, L. Li, and S. Sung (2004) Biological hydrogen production: effects of pH and intermediate products. Int. J. Hydrogen Energy 29: 1123–1131.
Liu, D., D. Liu, R. J. Zeng, and I. Angelidaki (2006) Hydrogen and methane production from household solid waste in the two-stage fermentation process. Water Res. 40: 2230–2236.
Ueno, Y., H. Fukui, and M. Goto (2007) Operation of a two-stage fermentation process producing hydrogen and methane from organic waste. Environ. Sci. Technol. 41:1413–1419.
Kim, S. H., S. K. Han, and H. S. Shin (2008) Optimization of continuous hydrogen fermentation of food waste as a function of solids retention time independent of hydraulic retention time. Process Biochem. 43: 213–218.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, J.K., Nhat, L., Chun, Y.N. et al. Hydrogen production conditions from food waste by dark fermentation with Clostridium beijerinckii KCTC 1785. Biotechnol Bioproc E 13, 499–504 (2008). https://doi.org/10.1007/s12257-008-0142-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-008-0142-0