Abstract
Depth of colonization (Z c) is a useful seagrass growth metric that describes seagrass response to light availability. Similarly, percent surface irradiance at Z c (% SI) is an indicator of seagrass light requirements with applications in seagrass ecology and management. Methods for estimating Z c and % SI are highly variable making meaningful comparisons difficult. A new algorithm is presented to compute maps of median and maximum Z c, Z c,med, and Z c,max, respectively, for four Florida coastal areas (Big Bend, Tampa Bay, Choctawhatchee Bay, Indian River Lagoon). Maps of light attenuation (K d) based on MODIS satellite imagery, PAR profiles, and Secchi depth measurements were combined with seagrass growth estimates to produce maps of % SI at Z c,med and Z c,max. Among estuary segments, mean Z c,med varied from (±SE) 0.80 ± 0.13 m for Old Tampa Bay to 2.33 ± 0.26 m for Western Choctawhatchee Bay. Standard errors for Z c,med were 1–10% of the segment means. Percent SI at Z c,med averaged 18% for Indian River Lagoon (range = 9–24%), 42% for Tampa Bay (37–48%), and 58% for Choctawhatchee Bay (51–75%). Estimates of % SI were significantly lower in Indian River Lagoon than in the other estuaries, while estimates for Tampa Bay and Choctawhatchee Bay were higher than the often cited estimate of 20%. Spatial gradients in depth of colonization and % SI were apparent in all estuaries. The analytical approach could be applied easily to new data from these estuaries or to other estuaries and could be incorporated routinely in assessments of seagrass status and condition.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Seagrasses are ecologically valuable components of aquatic ecosystems and have a critical role in shaping aquatic habitat. These “ecosystem engineers” influence multiple characteristics of aquatic systems through interactions with many biological and abiotic components (Jones et al. 1994; Koch 2001). For example, seagrasses reduce wave action, stabilize sediments, and provide habitat and refuge for invertebrates and juvenile fish (Williams and Heck 2001; Hughes et al. 2009). Seagrasses also respond to changes in light attenuation. Seagrass coverage declines with water depth due to light attenuation and declines more rapidly in productive aquatic ecosystems where light attenuation is relatively high (Duarte 1995). The light-limited maximum depth to which seagrass grows is variously called “depth limits” or “depth of colonization.” Empirical relationships between nutrient loading, light attenuation, and depth of colonization have been identified (Duarte 1991; Kenworthy and Fonseca 1996; Choice et al. 2014) and have been used to characterize light regimes and other water quality requirements to maintain seagrass habitat (Janicki and Wade 1996; Steward et al. 2005). Seagrasses may be particularly useful in this respect because they respond to water quality over relatively long time scales, “integrating” over time their exposure to stress. Thus, time scales of seagrass response better match time scales for monitoring and assessment of responses to nutrient management (Duarte 1995; Burkholder et al. 2007).
A variety of approaches have been used to estimate seagrass depth of colonization. A common in situ approach is to sample seagrass along depth transects until the maximum depth is adequately characterized (e.g., Spears et al. 2009; Choice et al. 2014). Alternative techniques include underwater photos or videos, aquascope identification, or hydroacoustic assessments (Zhu et al. 2007; Søndergaard et al. 2013). These are especially useful for site-specific evaluations where the analysis needs are driven by local questions (e.g., Iverson and Bittaker 1986; Hale et al. 2004). Availability of estuary-scale geospatial data for seagrass coverage, based on photo-interpretation of aerial imagery, suggests that standardized techniques can be applied at different spatial scales and would be valuable for a variety of scientific and policy applications. For large-scale evaluation, it is often useful to subdivide water bodies into segments for the purpose of analysis and policy development (e.g., Steward et al. 2005; Schaeffer et al. 2012; US EPA 2012). One challenge in doing so is that estuaries are often characterized by gradients in water quality, such that any segmentation scheme is likely to include within-segment gradients in water quality and associated seagrass depth of colonization. Local conditions may require adaptive assessment approaches that can address interactive effects of environmental variables at different spatial scales.
An example from the Big Bend region of Florida illustrates the issue of scale in analyzing seagrass depth of colonization (Fig. 1a). In a segment from this region, the highest depth of colonization largely occurs around the outer perimeter of the mapped seagrass coverage (red line in Fig. 1a). However, depth-dependent seagrass growth patterns are also evident at smaller spatial scales within the segment, wherein the segment-scale depth of colonization overestimates the depth distribution near the outflow of the Steinhatchee River, where high concentrations of colored dissolved organic matter reduce water clarity locally (personal communication, N. Wellendorf, Florida Department of Environmental Protection). An improved method for estimating depth of colonization should have sufficient flexibility to characterize seagrass responses at a large scale, such as the segment average, while still resolving important patterns at smaller scales, such as the local impact of a river outflow. The method should also have the potential to be applied widely using available data sets to support seagrass conservation. Developing and demonstrating this capability was one objective of this study.
Examples of data and grid locations for estimating seagrass depth of colonization for a region of the Big Bend, Florida. a Seagrass coverage and depth contours at 2-m intervals, including the whole-segment estimate for depth of colonization and location of the outflow of the Steinhatchee River (x); b a grid of sampling locations with sampling radii for estimating Z c and seagrass depth points derived from bathymetry and seagrass coverage layers; c an example of sampled seagrass depth points for a test location
Another objective of this study was to combine spatially resolved estimates of depth of colonization with light attenuation measures at the same spatial scales to characterize the pattern and range of seagrass light requirements in estuaries. In this paper, we operationally define “light requirements” as the average percentage of incident light present at the depth of colonization, which is similar to PLW min defined by Kemp et al. (2004) and also the definition suggested by Dennison et al. (1993). This approach is practical and relates well to the data most often available for policy and conservation applications, but comes with assumptions and caveats which we appropriately address. Regardless of such complexity, the spatial distribution of submerged aquatic plants is often associated with changes in water depth and light availability (Barko et al. 1982; Hall et al. 1999; Dennison et al. 1993), wherein depth of colonization is controlled by light requirements and average light attenuation. Published estimates of seagrass light requirements are species specific and quite variable. Duarte (1991) reported that seagrasses can extend to depths receiving an average of 11% of surface irradiance, while estimates of light requirements for seagrass in Chesapeake Bay were about 20% (Batiuk et al. 1992). Dennison et al. (1993) reported minimum light requirements, which they defined similarly as the percent light at the maximum depth limit, ranging from less than 5% to greater than 30% depending on site conditions. Estimates of ~20% are common in the literature, approximating a value in the middle of published estimates (see also Kemp et al. 2004). An estimate of ~20% for seagrass in Florida estuaries has been applied for management purposes in Tampa Bay, Choctawhatchee Bay, and elsewhere in Florida (e.g., FDEP 2012; US EPA 2012) and can be traced to the estimate of 22.5% for lower Tampa Bay from Dixon and Leverone (1995).
Sources of variation in estimates of seagrass light requirements are numerous and include physiological differences among seagrass species, differences in attenuation on the leaf surface by attached algae and detritus (i.e., K e in Kemp et al. 2004), and variations in other physiological stressors such as salinity or water temperature (Kenworthy and Haunert 1991; Kemp et al. 2004; Choice et al. 2014). For example, long-term or seasonal increases in water temperature could increase estimates of light requirements due to increased metabolic rates (Masini and Manning 1997). Differences in operational definitions and method of estimation are also likely to contribute to differences in reported values. For example, Dennison et al. (1993) defined minimal light requirements as the percent light at the maximal depth limit for seagrass—where depth limit was defined variously by different included studies. Choice et al. (2014) applied a statistical method to data from individual stations seeking to find the percentage of surface irradiance linked to seagrass percent cover or shoot density of zero. In this study, we sought to generate more comparable estimates of depth of colonization and light requirements by broadly applying the same method to characterize seagrass depth of colonization and relating it to estimates of light attenuation at similar scales. To quantify light attenuation at temporal and spatial scales relevant to understanding seagrass distributions in coastal ecosystems, we used estimates derived from satellite remote sensing along with more conventional observations of water clarity, including profiles using underwater radiometers and Secchi depth. Although in situ measures provide locally precise estimates, ocean color data from satellite remote sensing can provide consistent estimates of light attenuation across a large spatial extent, often with a high return frequency (e.g., ~8 days) and long-term data collection, which is useful for characterizing average attenuation (Woodruff et al. 1999; Chen et al. 2007).
The overall goal of this study is to present an algorithm for estimating seagrass depth of colonization and light requirements using geospatial datasets describing seagrass coverage and satellite remote sensing data of light attenuation in the water column. The approach allowed us to generate consistent estimates of seagrass depth of colonization and light requirements, enabling meaningful comparisons of each across space and time. This supports the management need to evaluate status and trends, and to predict how water quality changes could affect seagrass extent and distribution given existing relationships with light attenuation. Specific objectives were to (1) describe the method for estimating seagrass depth of colonization, (2) apply the technique to target locations in four Florida estuaries to illustrate quantification of seagrass growth patterns, and (3) develop a spatial description of relationships among depth limits and light attenuation, characterizing patterns in light requirements in each case study and between regions. The analytical approach was also automated for use in the R statistical programming language (RDCT 2016), which allowed us to evaluate changes in seagrass light requirements in Tampa Bay over a period of 25 years.
Methods
Study Sites and Data Sources
Study sites included four coastal areas in Florida: the Big Bend region (northeast Gulf coast), Choctawhatchee Bay (panhandle), Tampa Bay (central Gulf coast), and Indian River Lagoon (Atlantic coast; Table 1 and Fig. 2). Florida’s estuaries and coastal waters are partitioned into segments following a scheme developed by the US Environmental Protection Agency for numeric nutrient criteria (US EPA 2012). The method for estimating depth of colonization was evaluated initially using one segment in each of the four areas, chosen based on geographic coverage in Florida coastal areas and availability of seagrass data. These segments included Big Bend (BB), Western Choctawhatchee Bay (WCB), Old Tampa Bay (OTB), and Upper Indian River Lagoon (UIRL). The analysis was then expanded to quantify spatially resolved seagrass depth limits and associated light requirements for all the segments in three of the estuaries, omitting the Big Bend from further analysis due to an insufficient number of light attenuation measurements.
Locations and seagrass coverage of estuary segments used to evaluate depth of colonization estimates. Seagrass coverage layers are from 2006 (BB Big Bend), 2010 (OTB Old Tampa Bay), 2009 (UIRL Upper Indian R. Lagoon), and 2007 (WCB Western Choctawhatchee Bay). SR Steinhatchee River outflow, MI Merritt Island National Wildlife Refuge
Geospatial data describing seagrass coverage and bathymetry were used to estimate depth of colonization. These data are available for coastal regions of Florida from the US Geological Survey, Florida Department of Environmental Protection, Florida Fish and Wildlife Conservation Commission, and Florida’s watershed management districts. Seagrass coverage maps were obtained for a recent year in each of the study sites (Table 1), except for Tampa Bay, for which coverages were obtained for every available year from 1988 to 2014. The original coverage maps were produced by photo-interpreting aerial images to categorize seagrass as absent, discontinuous (patchy), or continuous. We aggregated to two categories, present (continuous and patchy) and absent.
Bathymetry data were obtained from the National Oceanic and Atmospheric Administration’s (NOAA) National Geophysical Data Center (http://www.ngdc.noaa.gov/) as either Digital Elevation Models or as bathymetric sounding data from hydroacoustic or other surveys. Tampa Bay bathymetry provided by the Tampa Bay National Estuary Program are described in Tyler et al. (2007). Bathymetry for the Indian River Lagoon were obtained from the St. John’s Water Management District (CPE 1997). Because the vertical datum (i.e., MLLW, NAVD88, etc.) varied, all bathymetric data were vertically adjusted to local mean sea level using the NOAA VDatum tool (http://vdatum.noaa.gov/). Adjusted data were combined with seagrass coverage layers using standard union techniques for raster and vector layers in ArcMap 10.1 (ESRI 2012). To reduce computation time, bathymetry layers were first masked to remove observations more than 1 km from any seagrass polygon. Raster bathymetric layers were converted to point layers to combine with seagrass coverage maps, as described below.
Quantifying Light Attenuation
Satellite remote sensing imagery was used to create a gridded 1-km2 map of estimated light attenuation for Tampa Bay and Choctawhatchee Bay. Secchi depth measurements were used to quantify attenuation for the Indian River Lagoon because light scattering from bottom reflectance and insufficient grid resolution prevented use of satellite remote sensing measurements in this very narrow, back-bay estuary.
For Tampa Bay, daily MODIS (Aqua level-2) satellite data were downloaded from the NASA website (http://oceancolor.gsfc.nasa.gov/) for the 5 years preceding the year the seagrass imagery was acquired. Images were reprocessed using the SeaWiFS Data Analysis System software (SeaDAS, version 7.0). For Tampa Bay, light attenuation was derived from daily MODIS images using a previously developed algorithm that estimates Secchi depth using a regression relating field observations and satellite-derived estimates of the diffuse attenuation coefficient at 490 nm (K d (490), Chen et al. 2007). Monthly and annual mean Secchi depth was estimated from the daily images and then averaged to create a single layer.
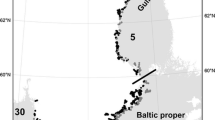
Similarly, K d for Choctawhatchee Bay was derived from MODIS using the QAA algorithm (Lee et al. 2005), which was also used by Chen et al. 2007 to estimate K d (490). Monthly field measurements of K d obtained in 2010 at ten optically deep locations in Choctawhatchee Bay were used to correct annual means of the un-validated satellite values (K d,MODIS) to match annual means of in situ measurements (Fig. S1a). The satellite estimates of K d were corrected by comparing regression curves of in situ data and satellite estimates from corresponding pixels versus cumulative frequency of each type of measurement (Cumulative Freq = −0.34 + 1.75 ∙ K d, r 2 = 0.93 for in situ model; Cumulative Freq = −0.35 + 1.15 ∙ K d,MODIS, r 2 = 0.95 for satellite model). For any uncorrected satellite estimate (Fig. S1b), the corresponding cumulative estimate on the regression curve from the satellite data was identified, matched with the corresponding percentile for the in situ data, and then related to the associated in situ K d value to yield the corrected satellite estimate. Annual means were used to create the regressions and corrections for the remaining years of satellite data from Choctawhatchee Bay. These were preferred rather than daily match-ups because many of the in situ observations would not generate match-ups due to cloud cover and because the annual mean was the time scale of interest. Field K d estimates were estimated as the slope relating log irradiance and log depth (i.e., logI Z = logI O − K d Z) measured using a 4 pi Biospherical PAR sensor on a SBE25 CTD. The empirical relationship for the 2010 data was applied to all 5 years of annual mean satellite-derived K d data prior to averaging to create a single layer for further analysis.
For Indian River Lagoon, Secchi depth data (meters, Z Secchi) collected within 10 years prior to the seagrass coverage data (i.e., 1999–2009) were obtained from the state of Florida’s Impaired Waters Rule (IWR) database, update 40. A 10-year averaging period was used for Indian River Lagoon to compensate for uneven temporal coverage, whereas 5-year averages were used for the other estuaries. Stations with less than five observations were removed as were observations flagged because the Secchi disk was visible on the bottom. As an additional data quality screen, Secchi data were compared with bathymetry to ensure that the reported Secchi depth was less than the water depth.
A land mask was applied to MODIS data for Tampa Bay and Choctawhatchee Bay, and pixels were further masked where water depth was less than the local estimate of seagrass depth of colonization thereby eliminating data from optically shallow water. For all three estuaries, light attenuation in deeper water adjacent to seagrass beds was assumed to be similar to attenuation at the deep-edge of seagrass. In general, Secchi depth cannot be used to estimate water clarity directly in a seagrass bed because the disk is visible on the bottom. Bottom reflectance similarly limits application of remote sensing, with some exceptions (Barnes et al. 2013). Although PAR profiles can in principle be made in shallow water, these often give less precise estimates of K d than deeper profiles. Many studies have similarly used open-water estimates to infer water clarity in adjacent seagrass beds (e.g., Kemp et al. 2004; Biber et al. 2005; Corbett and Hale 2006).
Estimating Seagrass Depth of Colonization
Seagrass depth of colonization (Z c) was computed by overlaying seagrass coverage maps and bathymetry data to generate a point shapefile with attributes of location, depth (m), and seagrass presence or absence. To relate depth and seagrass presence/absence, the proportion of points with seagrass present was computed as a function of depth within discrete depth bins and parameters describing the distribution were computed from the resulting function. The analysis was repeated for observations within a particular search radius of grid nodes spanning the study areas, generating a map of depth of colonization (Fig. 1). The analysis was also repeated with a large radius encompassing entire segments, resulting in whole-segment estimates. Further details are provided below regarding search radius, depth bins, and computing depth of colonization metrics.
Depth of colonization at particular locations was computed using observations found within a search radius, which was 0.04 degrees for Choctawhatchee Bay, 0.1 degrees for Tampa Bay, and 0.15 degrees for Indian River Lagoon. Analyses that focused on an individual segment at each study location used a common search radius of 0.02 degrees for visual and scale comparisons in the resulting figures. Geospatial data were imported and processed using functions in the rgeos and sp packages in R (Bivand et al. 2008; Bivand and Rundel 2014). Although no particular radius is “wrong,” selection involves several trade-offs. A large search radius improves the precision of estimates by finding more data, but may encompass areas that are ecologically dissimilar. As an example, the radius to characterize depth of colonization at the outflow of the Steinhatchee River in Fig. 1 was large enough to describe variation in growth affected by local water quality, but small enough to not include observations well outside the influence of the river outflow. Moreover, a radius much larger than the grid spacing inflates the computational requirements with little benefit. A smaller search radius and closer grid spacing provides more spatial resolution, but also finds less data, increasing uncertainty. A small radius may also encompass little or no depth gradient, in which case the relationship between seagrass and depth cannot be quantified. An appropriate search radius will in many cases result in a plot illustrating a decreasing proportion of points with seagrass with increasing depth (Fig. 3). The appropriate scale may be related to the size of the estuary. A larger radius was selected for Tampa Bay, which is the largest of the water bodies in our study.
Methods for estimating seagrass depth of colonization using sampled seagrass depth points around a single location. Three depth estimates (Z c,min, Z c,med, Z c,max) are based on a linear curve through the inflection point of a logistic curve. The curve is defined by the parameters α, β, and γ and describes the decrease in the proportion of sample points with seagrass as a function of depth below mean tide level (MTL). a The estimation method when the linear curve intercepts α at a depth greater than zero. b The alternative method when the linear curve intercepts α at depth less than zero
Depth bins, within which seagrass presence/absence proportions were computed, had variable widths <0.5 m defined to include 1 to 50 bathymetric soundings (i.e., if a 0.5-m bin includes more than 50 observations, the bin width is reduced). For whole-segment estimates, these parameters were changed to 0.1 m and 1 to 1000 points. This method created many narrow bins if a large number of soundings were present and fewer, wider bins when fewer observations were available. The result is that, other factors being equal, having more data resulted in lower estimates of parameter uncertainty.
Three depth of colonization metrics were derived from the function relating seagrass presence versus depth (Fig. 3). These included minimum (Z c,min), median (Z c,med), and maximum (Z c,max) depth of colonization. These terms each describe meaningful points on the depth distribution of the seagrass coverage map. Z c,max is the deepest depth at which mappable seagrasses occurred, excluding isolated patches (or outliers) at deeper depths. Z c,med is the median depth of the deep water edge of seagrass. Finally, Z c,min approximates the depth at which seagrass percent cover begins to decline with increasing depth, which may occur at Z = 0 m.
At each sampled location, a logistic function was fitted to the extracted depth points using non-linear regression, quantifying the decrease in seagrass cover with respect to depth (Eq. 1; Fig. 3):
where the proportion of points with seagrass present, P, within each depth bin, centered at Z, was defined by a logistic curve with an upper asymptote α, an inflection point β, and a scale parameter γ. The curve was fitted by minimizing the residual sums-of-squares with the Gauss-Newton algorithm (Bates and Chambers 1992). Initial parameter values for fitting were estimated as α = max (P), β = median (Z), and γ = 75th percentile (Z) − median (Z). The maximum rate of decrease in seagrass coverage with respect to Z is −α/4γ and occurs at Z = β. The tangent at Z = β passes through the line P = α at Z = β − 2γ and through P = 0 at Z = β + 2γ. The three seagrass depth of colonization metrics were defined in terms of these reference values as follows:
Several quality control measures were implemented to reduce spurious estimates. First, Z c parameters (i.e., depth of colonization) were estimated only if the number of seagrass depth points was sufficient for the logistic curve to be estimable. Second, estimates were provided only if the fitted value for β, the inflection point on the logistic curve, was within the range of depth, Z, in the data. It was possible for β − 2γ to be less than zero, and this was common when seagrass cover declined immediately as depth increased from zero. In these cases, Z c,min = 0 and Z c,med is half the depth from zero to Z c,max (Fig. 3b).
Estimates of parameter uncertainty from the logistic model were also used to evaluate the quality and variability associated with individual depth of colonization estimates. Elements of the model-estimated variance–covariance matrix for the model parameters α, β, and γ were used to estimate the variance of the depth of colonization parameters using equations for sums and differences of normal random variables (Ku 1966):
We then estimated 95% prediction intervals for each parameter as the estimate \( 1.96\pm \sqrt{\sigma^2} \) where σ 2 is the appropriate estimate from Eqs. 5, 6, or 7. The value of \( {\sigma}_{Z_{\mathrm{c}, \min}}^2 \) is not defined when β − 2γ < 0 because Z c,min is fixed at zero (Fig. 3b). Given the estimated prediction intervals, we also considered depth of colonization to be inestimable if the 95% prediction interval for Z c,max included zero.
Given estimates of depth of colonization for grid nodes, calculated as above, segment means and standard errors were computed using an intercept-only spatial mixed model, implemented using the nlme package in R (Pinheiro et al. 2016), thereby accounting for spatial autocorrelation among the gridded estimates. An isotropic Gaussian correlation structure with a nugget effect was selected from among other options based on the Akaike Information Criterion. This approach made it possible to separate variability within the segment from uncertainty regarding either individual node estimates or segment means. Whole-segment estimates, calculated using a single node with a large search radius, also provided an estimate of uncertainty, but not an estimate of variability within the segment.
Seagrass Light Requirements
Seagrass light requirements were estimated as the average fraction of surface irradiance (PAR) reaching the depth of colonization, which was quantified for this purpose as Z c,med. The same calculations using Z c,max are included as supplemental information. Light attenuation was quantified on the same grid as depth of colonization, which was selected to maximize the number of matches between depth of colonization and light attenuation measurements. Grid cells centered more than 1 km from seagrass were not included, preventing spurious comparison of seagrass depth of colonization with light attenuation far from shorelines. The percentage of surface irradiance (% SI) at the median depth of colonization was computed using
where the light extinction coefficient (K d) was obtained as a remote sensing product (Choctawhatchee Bay, Tampa Bay) or from field-based Secchi depth measurements (Indian River Lagoon). Surface irradiance at the seagrass edge was not estimated from remote sensing data if the associated depth of colonization estimate exceeded the actual bottom depth. These locations were removed to reduce potential bias from bottom reflectance in shallow waters (10% of locations in Choctawhatchee Bay, 13% in Tampa Bay). Where K d was derived from Secchi depth (Z Secchi), it was computed using K d ∙ Z Secchi = 1.7. The product K d ∙ Z Secchi varies in relation to the ratio of scattering to absorption, with higher values for the product associated with greater scattering. A range of 1.1 to 2.0 (Liu et al. 2005) encompasses the values commonly applied in estuaries. An analysis of 504 PAR profiles and corresponding Secchi depth measurements from Pensacola Bay, FL (Murrell et al. 2009), had a mean (±SE) K d ∙ Z Secchi of 1.63 ± 0.03, which likely reflects a relatively low ratio of scattering to absorption in the Florida estuaries (Davies-Colley and Vant 1988). The % SI at the maximum depth of colonization (Z c,max) was also estimated and is reported as supplemental information.
Segment means and standard errors for light requirements were computed from the spatially correlated gridded estimates using an intercept-only spatial mixed model, as for depth of colonization. Tests for differences in mean light requirements between estuary segments were also implemented using a spatial mixed model using the nlme package in R, in this case with a single categorical fixed effect (estuary segment) and a Gaussian spatial correlation structure with a nugget effect.
Trends in Seagrass Light Requirements in Tampa Bay
To demonstrate a useful application of our analysis, we estimated changes in depth of colonization and light requirements in Tampa Bay, where seagrass recovery has been a focus of management efforts for decades (Greening et al. 2014) and nominally biennial seagrass surveys were available for 1988 to 2014 (1988, 1990, 1992, 1994, 1996, 1999, 2001, 2004, 2006, 2008, 2010, 2012, and 2014). Light attenuation was estimated from monthly Secchi depth (TBEP 2011, Fig. S2), rather than satellite estimates, which were not available for the full extent of the seagrass time series. Secchi depth at each station was averaged by year then translated to light attenuation using K d ∙ Z Secchi = 1.7, as above. Seagrass light requirements were estimated at each monitoring station for each year with seagrass coverage, then evaluated to describe trends.
Results
Segment Characteristics and Seagrass Depth of Colonization Estimates
The surface area of the study regions ranged from 59 km2 for Western Choctawhatchee Bay (WCB) to 271 km2 for the Big Bend (BB) (Table 1). Mean depth was less than 5 m in each segment, except for WCB, which was slightly deeper (5.3 m). Maximum depth was greater in WCB (11.9 m) and Old Tampa Bay (OTB, 10.4 m) compared to BB (3.6 m) and Upper Indian River Lagoon (UIRL, 3.7 m) segments. Seagrass coverage was extensive in BB (74.8% of total segment area), less in UIRL (32.8%) and OTB (11.9% in 2010), and very sparse in WCB (5.9%), where most seagrass was in a large patch located just west of the inlet to the Gulf of Mexico (Fig. 2). Seagrasses were distributed throughout BB except for a noticeable area of decreased coverage near the outflow of the Steinhatchee River (Fig. 2, upper left). Seagrasses in OTB and UIRL were distributed narrowly along the shorelines, consistent with strong depth-dependence. Seagrass coverage declined toward the northern ends of both OTB and UIRL.
Whole-segment estimates (±prediction interval) based on single locations with large radii for Z c,med varied from 0.95 ± 0.07 m in OTB to 2.29 ± 0.19 m in BB, with 95% prediction intervals equal to ±1% to 10% of the estimate (Table 2). Estimates of Z c,max varied from 1.1 to 3.8 m and were somewhat less precise, with 95% prediction intervals equal to 3 to 20% of the estimate (Table 2). Means of spatially resolved estimates of Z c,med for a grid of points in each segment varied from 2% more to 15% less than the whole-segment estimates (Table 2), within the margin of uncertainty for each, suggesting that the two approaches gave comparable results. The difference was largest for BB, where the gridded estimates of depth of colonization had a bi-modal distribution (i.e., lower estimates near the Steinhatchee outflow vs. higher values distant from the river outflow; Fig. 4, upper left). The whole-segment estimate of Z c,max for BB was 3.8 m, 65% more than the 2.3 m average of the gridded estimates, reflecting a greater influence of the deeper-distributed seagrass on the whole-segment calculation (Table 2; Fig. 4). Estimates for Z c,min were as low as zero in BB and OTB (Table 2), indicating that seagrass coverage decreased immediately with any increase in depth. The highest values for Z c,min were associated with relatively deep seagrass distributions, as in WCB. In these cases, coverage increased initially with increasing depth, rather than decreasing. Seagrass percent cover often increased initially with increasing depth, likely reflecting stressors such as wave energy or desiccation during extreme low tides affecting the shallow margin of the seagrass bed.
Spatially resolved estimates of maximum seagrass depth of colonization (m) for four coastal segments of Florida. Estimates are assigned to grid locations for each segment, where grid spacing was fixed at 0.01 decimal degrees. Radii for sampling seagrass bathymetric data around each grid location were fixed at 0.02 decimal degrees. BB Big Bend, OTB Old Tampa Bay, UIRL Upper Indian River Lagoon, WCB Western Choctawhatchee Bay
Spatial heterogeneity of the gridded estimates for depth of colonization was particularly apparent for segments BB and UIRL (Fig. 4). As previously mentioned, depth of colonization in the Big Bend segment was reduced in the vicinity of the Steinhatchee River discharge. Seagrasses were also limited to shallower depths at the north end of the Upper Indian River Lagoon segment, but grew at maximum depths up to 2.2 m on the eastern portion of the Upper Indian River Lagoon segment near the Merritt Island National Wildlife Refuge (Fig. 2). Seagrasses in Old Tampa Bay grew to slightly greater depth in the eastern and southern portion of the segment and were limited to shallower depths near freshwater inflow channels on the northern margin (Fig. 4). The deepest growing seagrass in Choctawhatchee Bay was closest to Destin Pass, where regular tidal exchange with Gulf of Mexico waters keeps light attenuation low (Fig. 4). Z c could not be estimated where seagrasses were sparse or absent as in the center of Old Tampa Bay and western Choctawhatchee Bay, or where there was an insufficient gradient in water depth, as in several areas of the Big Bend segment (Fig. 4). However, depth of colonization can be estimated for locations that lack seagrass if sufficient coverage is present within the search radius. As a result, gridded maps illustrate the spatial patterns of depth of colonization and do not repeat the spatial patterns of seagrass coverage (i.e., Fig. 4 does not always resemble coverage in Fig. 2).
Gridded estimates for all segments in each bay (excluding Big Bend) provided further information on seagrass depth of colonization within and among segments and the average depth of colonization in each estuary (Table 3; Figs. 7, 8, and 9). Seagrass Z c estimates were computed for 338 locations in Choctawhatchee Bay, 252 locations in Tampa Bay, and 47 locations in the Indian River Lagoon. Mean Z c,med (±SE) was 1.43 ± 0.38 for Choctawhatchee Bay, 0.91 ± 0.31 for Tampa Bay, and 1.12 ± 0.26 m for Indian River Lagoon. Mean Z c,med was not significantly different between any of the bays. Means for segments within estuaries varied, although statistical differences were difficult to determine in some locations due to low sample size (i.e., Indian River Lagoon segments, Table 3). In Tampa Bay, Z c,med was ~0.5 m less in Old Tampa Bay than in the Lower or Middle Tampa Bay segments, but was only significantly different (p < 0.05) compared with the latter. Similarly, Z c,med in the eastern segment of Choctawhatchee Bay was 1.1 and 1.5 m less than Z c,med in the central and western bay, respectively, although only the eastern and western estimates were significantly different (p < 0.05). No statistical differences were observed between segments of the Indian River Lagoon despite higher Z c,med in more southern segments (Fig. 9).
Seagrass Light Requirements
Estimates of light attenuation, seagrass depth of colonization, and corresponding light requirements for all locations in Choctawhatchee Bay, Tampa Bay, and the Indian River Lagoon indicated substantial variation, both between and within the different bays. Satellite-derived estimates for Choctawhatchee Bay and Tampa Bay resolved spatial variation in average light attenuation (Figs. 5 and 6). For Choctawhatchee Bay, K d increased from the western and central segments toward the eastern segment, which is adjacent to the Choctawhatchee River discharge (Fig. 5). Similarly, attenuation increased from lower and central Tampa Bay into Old Tampa Bay and Hillsborough Bay. Attenuation was also less in the central area of the lower bay segments (Fig. 6). Secchi depth was highest in the southern Indian River Lagoon and decreased to the north. Relatively few Secchi depth measurements were available for the Upper Indian River Lagoon and Banana River segments, likely because maximum water clarity exceeded the maximum depth in shallow areas, resulting in right-censored measurements.
Satellite estimated light attenuation for Choctawhatchee Bay as an average of years from 2003 to 2007. See Fig. 7 for segment identification
Satellite estimated light attenuation for Tampa Bay as an average of years from 2006 to 2010. See Fig. 8 for segment identification
Whole-estuary means for percent surface irradiance (% SI) at Z c,med (i.e., seagrass light requirement) was (mean ± SE) 58 ± 3.2% SI for Choctawhatchee Bay, 42 ± 2.6% SI for Tampa Bay, and 18 ± 3.0% SI for Indian River Lagoon. Based on Tukey contrasts, average light requirements for seagrass were significantly different between all estuaries, particularly for Indian River Lagoon. Despite some apparent spatial patterns in seagrass light requirements (Figs. 7, 8, and 9), significant differences were not found between segments of a single estuary, except for a marginally significant difference between eastern and western Choctawhatchee Bay (p = 0.04). In Tampa Bay, the segment mean (±SE) % SI at Z c,med ranged from 36 ± 5.5% SI to 48 ± 5.6% SI (Table 3, Fig. 8). For Choctawhatchee Bay, segment means were 50 ± 5.7% SI to 75 ± 8.7% SI, with the apparently higher values in eastern Choctawhatchee Bay (Table 3, Fig. 7). For Indian River Lagoon, segment means ranged from 9 ± 7.1% SI to 24 ± 8.6% SI (Table 3, Fig. 9). Either small sample sizes, as for Indian River Lagoon, or a small number of effectively independent samples, given the spatial correlation of residuals, reduced the statistical significance of apparent spatial differences in light requirements between segments.
Median depth of seagrass colonization (Z c,med, m) and light requirements (% surface irradiance at Z c,med) for multiple locations in Choctawhatchee Bay, Florida. Each location has light attenuation from satellite observations and an estimate of seagrass depth of colonization with a search radius of 0.04 degrees. Box plots show the 25th percentile, median, and 75th percentile. Whiskers extend to the 5th and 95th percentiles with outliers beyond. CCB Central Choctawhatchee Bay, ECB East Choctawhatchee Bay, WCB West Choctawhatchee Bay
Median depth of seagrass colonization (Z c,med, m) and light requirements (% surface irradiance at Z c,med) for multiple locations in Tampa Bay, Florida. Each location has light attenuation from satellite observations and an estimate of seagrass depth of colonization with a search radius of 0.1 degrees. Box plots show 25th percentile, median, and 75th percentile. Whiskers extend to the 5th and 95th percentiles with outliers beyond. HB Hillsborough Bay, LTB Lower Tampa Bay, MTB Middle Tampa Bay, OTB Old Tampa Bay
Median depth of seagrass colonization (Z c,med, m) and light requirements (% surface irradiance at Z c,med) for multiple locations in Indian River Lagoon, Florida. Each location has an average Secchi depth observation and an estimate of seagrass depth of colonization with a search radius of 0.15 degrees. Map locations are georeferenced observations of Secchi depth. Box plots show 25th percentile, median, and 75th percentile. Whiskers extend to the 5th and 95th percentiles with outliers beyond. BR Banana River, LCIRL Lower Central Indian River Lagoon, LIRL Lower Indian River Lagoon, LML Lower Mosquito Lagoon, UCIRL Upper Central Indian River Lagoon, UIRL Upper Indian River Lagoon, UML Upper Mosquito Lagoon
The final analysis illustrated the temporal evolution from 1988 to 2014 of seagrass depth of colonization and light attenuation (Fig. 10, upper panel), and resulting expression as light requirements (Fig. 10, lower panel) in Tampa Bay. Depth of colonization decreased in Hillsborough Bay and Middle Tampa Bay during the early 1990s before rebounding strongly toward the end of the time series (Fig. 10, upper panel). Median light attenuation varied from year to year, but decreased overall. We applied spatial mixed models to evaluate pairwise comparisons at 18 grid nodes covering the whole Bay (Fig. S2) using the terminal ends of the time series to evaluate overall changes from 1988 to 2014. Depth of colonization increased by 3 to 30 cm between 1988 and 2014 (mean = 16 cm, p < 0.01). Light attenuation decreased by 0.04 to 0.5 m−1. Changes in light requirements (reflecting the interaction of both trends) were positive at 17 of 18 nodes and varied from −3.5 to 14% of surface irradiance between years. The largest increases in depth of colonization were near the boundary between lower and middle Tampa Bay and in Old Tampa Bay. The largest increases in light requirements were in Hillsborough Bay, where light requirements were lowest early in the time series (Fig. 10, lower panel).
Annual changes in median light attenuation (K d, m−1) and depth of colonization (Z c,med, m) in segments of Tampa Bay from 1988 to 2014 (upper panels) and resulting changes in percent of surface irradiance at the depth of colonization (lower panel). Contours in upper panels illustrate isopleths of percent surface irradiance at depth of colonization (Eq. 8). Box plots show the distribution of light requirements at locations in each segment shown in Fig. S2. HB Hillsborough Bay, LTB Lower Tampa Bay, MTB Middle Tampa Bay, OTB Old Tampa Bay
As a final note, the estimates of light requirements for 2010 shown in Fig. 10 (lower panel) differ from those in Fig. 8. These relate to the small number of locations for which Secchi depth data were consistently available from 1988 through 2014 (Fig. S2), compared to the virtually complete spatial coverage obtained from the satellite remote sensing data. Although the time series values are not as representative of all values in the segment (i.e., as in Fig. 8), they are consistent in each year and, thus, are a valid representation of change over time at those locations.
Discussion
Seagrass depth of colonization is an important measure of the status and condition of seagrass communities in estuaries because it relates to light attenuation and related anthropogenic water quality changes, especially eutrophication caused by excess nutrient loading (Dennison et al. 1993; Short and Wyllie-Echeverria 1996; Burkholder et al. 2007). Because seagrasses are ecologically important and sensitive to water quality changes, both seagrass coverage and depth of colonization have been used to define water quality management objectives (Steward et al. 2005; US EPA 2012; Greening et al. 2014). For applications to water quality management, deriving estimates and associated estimates of uncertainty at appropriate scales using a consistent methodology is particularly important (US EPA 2012). The methods developed and demonstrated in this study are a rigorous, yet efficient and practical approach for computing seagrass depth of colonization at a large scale using widely available geospatial data sets describing seagrass areal extent and bathymetry. The method is automated via R code and, thus, not especially labor intensive. Because it is automated, it is reproducible and can be applied to new data from the studied estuaries or other estuaries with appropriate data. To demonstrate this, we applied the analysis to 13 seagrass coverage maps for Tampa Bay, quantifying changes in depth of colonization and light requirements from 1988 to 2014, which can be interpreted in the context of long-term water quality changes (e.g., Beck and Hagy 2015). The method provides maps of depth of colonization, resolving both means and spatial gradients at a range of scales from an individual measurement to the whole estuary. Uncertainty is estimated for individual estimates of depth of colonization, while both variability (SD) and uncertainty (SE) are quantified for segments of estuaries, whole estuaries, and temporal changes. Given these characteristics, our approach is a novel tool for assessment of seagrass distribution with respect to water depth at different spatial scales. Resolving spatial differences in depth of colonization and light requirements provides valuable information to support further investigation of the causes and mechanisms affecting the extent and spatial distribution of seagrass habitats and also informs policy development and evaluation of ecosystem responses to water quality management.
Maps illustrating spatial patterns in depth of colonization quantified expected but previously unquantified spatial patterns, wherein seagrasses grew to greater depth when closer to ocean passes, where water was clearer (Figs. 7, 8, and 9). Differences among segment means were mostly not statistically different, reflecting variability within segments in addition to variability among segments. Evaluations of temporal changes, which may be even more important for policy applications, could be evaluated without segmentation as pairwise differences of estimates by grid node. These revealed statistically significant changes in depth of colonization and light requirements, as well as spatial patterns in such changes. Application to other estuaries with substantial time series of water quality and seagrass distributions, such as Chesapeake Bay (Orth et al. 2010), could glean valuable new insights from these data.
For the first time, we also combined estimates of light attenuation for estuaries based on satellite remote sensing and Secchi depth measurements to resolve spatial patterns in seagrass light requirements. Like depth of colonization, maps of these values quantify gradients in light requirements, even if “segments” delineated for management purposes did not clearly identify regions that differed by light requirements. We would expect light requirements, as we defined them, to be higher at locations closer to freshwater and nutrient sources because epiphytic algal growth, salinity variations, color, or other factors such as sediment geochemistry could impose constraints on seagrass growth beyond those imposed by light attenuation in the water column (Hemminga 1998; Kemp et al. 2004). The results neither conflicted with nor definitively supported this expectation. For example, the highest light requirements in each of the estuaries were furthest from tidal exchange and closest to sources of freshwater and nutrients (Figs. 7, 8, and 9). However, the differences were subtle and, given variations within segments, did not emerge as significant differences among segment means. Alternatively, light requirements for seagrass in Indian River Lagoon were significantly less than for seagrass in Choctawhatchee Bay and Tampa Bay. Light requirements for Choctawhatchee Bay and Tampa Bay were both more than the 20% estimate that has been referenced broadly in water quality management (Batiuk et al. 1992; Dennison et al. 1993; Kemp et al. 2004) and locally within Florida (Dixon and Leverone 1995; US EPA 2012). Light requirements for Tampa Bay were similar to 20% in only a few areas of the Bay. We further note the differences in light requirements for Tampa Bay using grid-based estimates at a uniform and fine spatial scale in Fig. 8, compared to those based on relatively few locations at routine monitoring stations (Figs. 10 and S2). This highlights the need to consider sampling regime and relevant scales for estimates of light requirements that apply to an entire estuary. Regardless, our estimates are not outside the norm given the broad range in published estimates of seagrass light requirements (Dennison et al. 1993).
Some of the differences in light requirements that we observed may relate to differences in species composition since the physiology of seagrass is known to vary among seagrass species. For example, Halodule wrightii is the most abundant seagrass in western Choctawhatchee Bay (Yarbro and Carlson 2015) and has higher light requirements than several other abundant species in Florida (Choice et al. 2014) including Thalassia testudinum, which dominates the more oceanic areas of Tampa Bay. Choice et al. (2014) found that light requirements for Syringodium filiforme were much less, as low as 8–15% SI, although Kenworthy and Fonseca (1996) found that the depth distribution of H. wrightii and S. filiforme were similar in the lower Indian River Lagoon, implying their light requirements may be similar. Estimates of light requirements for several species of Halophila indicate a potential to grow at 5% SI or less (Kenworthy and Haunert 1991), consistent with some of our lowest estimates from Lower Indian River Lagoon (Fig. 9). Neither S. filiforme nor any of the Halophila spp. appear to be dominant species in Tampa Bay or Choctawhatchee Bay, perhaps limiting seagrass distributions in those estuaries to higher light environments compared with lower Indian River Lagoon. Although we cannot be certain the extent to which species composition can explain the differences we observed in % SI at the depth of colonization, the key observation is that differences were observed that seagrass species vary in their physiology and responses to a range of factors and that, therefore, it may be useful to understand and manage seagrass habitats utilizing local information where possible. Another consideration related to species composition is that our estimates are likely driven by the deepest growing species. Light attenuation changes could alter competitive relationships among species within the mappable seagrass area, which would not be apparent in our analysis.
Our estimates of % SI at the depth of colonization necessarily also depend on our approach to estimating depth of colonization, as do others in the literature. Depth of colonization is a reference point along a gradient of decreasing seagrass cover (presumably) associated with increasing light limitation and related physiological stress (e.g., figure 3 in Hemminga 1998). In some studies, percent cover was estimated by diver observation (Choice et al. 2014), enabling a statistical approach (e.g., moving split window) that directly resolves a threshold for rapid decline in percent cover with respect to % SI at the scale of a single quadrat. To scale up the analysis, we needed to use seagrass coverage maps based on photointerpretation, which imposes a binary classification (present/absent). By inferring the probability of seagrass presence conditional on depth, however, we obtained an estimate analogous to that of Choice et al. (2014), with the parameter β (Fig. 3) estimating the threshold for most rapid decline in seagrass presence. However, it is still unavoidable that seagrass will be both present at greater depths and stressed by light limitation at lesser depths. In this regard, a strength of our approach is that we can estimate the % SI associated with both the local extremes of the mappable seagrass distribution (i.e., Z c,max; Figs. S3, S4, and S5) and the center of that depth distribution (i.e., Z c,med). Moreover, by being linked to aerial coverage data, the estimates are available for a range of spatial scales, are comparable across all those scales, and can be quickly re-computed when new surveys are completed.
Our estimates also depend on an accurate characterization of average light attenuation, something that will always be challenging in the context of seagrass ecology. For example, since %SI = exp(−K d ∙ Z c), K d = K Secchi/Z Secchi, and K Secchi is generally between 1 and 2, Secchi depth in seagrass habitats is often similar to the depth of colonization, potentially leading to right censoring of Secchi measurements when the disk would be visible on the bottom. Accurate light profiling is possible but is also difficult in shallow water. Limitations on boat operations also favor sampling during calm winds, perhaps leading to under-sampling when sediment resuspension is above average. Quantifying light attenuation via satellite remote sensing has advantages but also presents similar and new challenges. For example, concern regarding bottom reflectance led Chen et al. (2007) to exclude data if water depth was <2 m, excluding nearly all seagrass areas. Light attenuation estimates for seagrass areas may therefore be based on nearby but deeper waters, whether measured via satellite remote sensing or boat-based estimates. If light attenuation is lower in open water, this will tend to increase the estimate of % SI at the depth of colonization. Conversely, it may not be preferable to measure attenuation on the interior of a seagrass beds since seagrass feedback effects may decrease light attenuation there (Gurbisz and Kemp 2014) and light attenuation at the deeper perimeter has more relevance to seagrass depth of colonization. Thus, using established satellite remote sensing methods, despite challenges and limitations, offers the advantage of uniform and sustained spatial and temporal coverage.
Sustained trends in water quality are another factor that can affect estimates of light requirements because seagrasses can be both slow to recover following disturbance and resistant to stress in the first place. In particular, species such as T. testudinum display a phalanx growth strategy and buffer against periods of low light by tapping into below ground reserves, making them slow to achieve a light-limited equilibrium distribution in the presence of water quality trends. Improving trends in light attenuation, accompanied by a lagging response in depth of colonization, as we observed for Tampa Bay, could explain our increasing estimates of light requirements, whereas the opposite may be true with declining trends in clarity. Epiphytes on seagrasses can also account for a significant fraction of total attenuation of light reaching seagrass leaves, and epiphyte growth may also respond directly to the availability of light in the water (e.g., Stankelis et al. 2003; Kemp et al. 2004). As a result, simultaneously considering changes in depth of colonization, light attenuation, and apparent light requirements may be useful for understanding the status and trends related to seagrass habitats.
This study has implications for both seagrass ecology and environmental management. Scientifically, the ability to resolve patterns in several parameters related to depth of colonization as well as % SI at the depth of colonization could be useful for generating testable hypotheses. For example, persistent differences in spatial patterns of depth distributions may suggest hypotheses regarding the causes and could stimulate research to identify local drivers. Similarly, we could seek to better understand temporal changes in depth of colonization, but without a consistent approach for quantifying, we may not be aware of such changes. For example, despite extensive documentation of changes in the area of seagrass habitat in Tampa Bay (Greening et al. 2014) and Chesapeake Bay (Orth et al. 2010), little attention has been given to associated trends in the depth distribution. Our results in Fig. 10 demonstrate the novelty of our approach and its potential to describe these previously undocumented changes in seagrass recovery from eutrophication impacts.
There are several important management implications related to our method and results. Localized patterns in depth of colonization, such as in the case of the Steinhatchee River outflow, illustrate that management goals related to seagrass depth distribution and light attenuation may not be applicable in water quality segments that are drawn without considering local drivers. At a slightly larger scale, differences among segments and among entire estuaries show that it can be both important and possible to consider local differences in the water quality requirements for seagrasses when developing and evaluating water quality goals over time. Even though seagrasses are affected by factors other than light attenuation, resistance and resilience in the face of multiple stressors can be influenced by the physiological and energetic changes affected by light availability (Burkholder et al. 2007). In the case of Tampa Bay, light availability generally exceeds seagrass light requirements estimated in the early 1990s. This may have sustained the seagrass recovery, which accelerated following a brief El Niño-Southern Oscillation (ENSO)-related period of increased river flow and increased light attenuation in the late 1990s (Greening et al. 2014).
References
Barko, J.W., D.G. Hardin, and M.S. Matthews. 1982. Growth and morphology of submersed freshwater macrophytes in relation to light and temperature. Canadian Journal of Botany 60: 877–887.
Barnes, B.B., C. Hu, B.A. Schaeffer, Z. Lee, D.A. Palandro, and J.C. Lehrter. 2013. MODIS-derived spatiotemporal water clarity patterns in optically shallow Florida keys waters: a new approach to remove bottom contamination. Remote Sensing of Environment 134: 377–391.
Bates, D.M., and J.M. Chambers. 1992. Nonlinear models. In Statistical models in S, ed. J.M. Chambers and T.J. Hastie, 421–454. Pacific Grove, California: Wadsworth and Brooks/Cole.
Batiuk, R.A., R.J. Orth, K. Moore, W.C. Dennison, J.C. Stevenson, L.W. Staver, V. Carter, N.B. Rybicki, R.E. Hickman, S. Kollar, S. Bieber, and P. Heasly. 1992. Chesapeake Bay submerged aquatic vegetation habitat requirements and restoration targets: a technical synthesis. Annapolis: US Environmental Protection Agency, Chesapeake Bay Program Office 190 pp.
Beck, M.W., and J.D. Hagy III. 2015. Adaptation of a weighted regression approach to evaluate water quality trends in an estuary. Environmental Modeling & Assessment 20 (6): 637–655.
Biber, P.D., H.W. Paerl, C.L. Gallegos, and W.J. Kenworthy. 2005. Evaluating indicators of seagrass stress to light. In Estuarine indicators, ed. S.A. Barton, 193–209. Boca Raton: CRC Press.
Bivand, R., and C. Rundel. 2014. rgeos: interface to geometry engine—open source (gEOS), R package version 0.3–8. http://CRAN.R-project.org/package=rgeos. Accessed July 1, 2015.
Bivand, R.S., E.J. Pebesma, and V. Gómez-Rubio. 2008. Applied spatial data analysis with R. New York: Springer.
Burkholder, J.M., D.A. Tomasko, and B.W. Touchette. 2007. Seagrasses and eutrophication. Journal of Experimental Marine Biology and Ecology 350 (1–2): 46–72.
Chen, Z., F.E. Muller-Karger, and C. Hu. 2007. Remote sensing of water clarity in Tampa Bay. Remote Sensing of Environment 109: 249–259.
Choice, Z.D., T.K. Frazer, and C.A. Jacoby. 2014. Light requirements of seagrasses determined from historical records of light attenuation along the Gulf coast of peninsular Florida. Marine Pollution Bulletin 81: 94–102.
CPE (Coastal Planning and Engineering). 1997. Indian River lagoon bathymetric survey. A final report to St. John’s river water Management District. Contract 95W142. Palatka: Coastal Planning and Engineering.
Davies-Colley, R.J., and W.N. Vant. 1988. Estimation of optical properties of water from Secchi disk depths. Water Resources Bulletin 24 (6): 1329–1335.
Dennison, W.C., R.J. Orth, K.A. Moore, J.C. Stevenson, V. Carter, S. Kollar, P.W. Bergstrom, and R.A. Batiuk. 1993. Assessing water quality with submersed aquatic vegetation. Bioscience 43: 86–94.
Dixon, L.K., and J.R. Leverone. 1995. Light requirements of Thalassia testudinum in Tampa Bay, Florida. In Technical report number 425. Sarasota: Mote Marine Lab.
Duarte, C.M. 1991. Seagrass depth limits. Aquatic Botany 40: 363–377.
Duarte, C.M. 1995. Submerged aquatic vegetation in relation to different nutrient regimes. Ophelia 41: 87–112.
ESRI (Environmental Systems Research Institute). 2012. ArcGIS v10.1. Redlands: Environmental Systems Research Institute.
FDEP (Florida Department of Environmental Protection). 2012. Site-specific information in support of establishing numeric nutrient criteria for Choctawhatchee Bay. Tallahassee: Florida Department of Environmental Protection 121 pp.
Greening, H., A. Janicki, E.T. Sherwood, R. Pribble, and J.O.R. Johansson. 2014. Ecosystem responses to long-term nutrient management in an urban estuary: Tampa Bay, Florida, USA. Estuarine, Coastal and Shelf Science 151: A1–A16.
Gurbisz, C., and W.M. Kemp. 2014. Unexpected resurgence of a large submersed plant bed in Chesapeake Bay: analysis of time series data. Limnology and Oceanography 59 (2): 482–494.
Corbett, C.A., and J.A. Hale. 2006. Development of water quality targets for Charlotte Harbor, Florida using seagrass light requirements. Florida Scientist 69 (Supplement 2): 36–50.
Hale, J.A., T.K. Frazer, D.A. Tomasko, and M.O. Hall. 2004. Changes in the distribution of seagrass species along Florida’s central Gulf Coast: Iverson and Bittaker revisited. Estuaries 27: 36–43.
Hall, M.O., M.J. Durako, J.W. Fourqurean, and J.C. Zieman. 1999. Decadal changes in seagrass distribution and abundance in Florida Bay. Estuaries 22 (2B): 445–459.
Hemminga, M.A. 1998. The root/rhizome system of seagrasses: an asset and a burden. Journal of Sea Research 39 (3–4): 183–196.
Hughes, A.R., S.L. Williams, C.M. Duarte, K.L. Heck, and M. Waycott. 2009. Associations of concern: declining seagrasses and threatened dependent species. Frontiers in Ecology and the Environment 7: 242–246.
Iverson, R.L., and H.F. Bittaker. 1986. Seagrass distribution and abundance in eastern Gulf of Mexico coastal waters. Estuarine, Coastal and Shelf Science 22: 577–602.
Janicki, A., and D. Wade. 1996. Estimating critical external nitrogen loads for the Tampa Bay estuary: an empirically based approach to setting management targets. Technical report 06-96. St. Petersburg: Tampa Bay National Estuary Program 200 pp.
Jones, C.G., J.H. Lawton, and M. Shachak. 1994. Organisms as ecosystem engineers. Oikos 69: 373–386.
Kemp, W.C., R. Batiuk, R. Bartleson, P. Bergstrom, V. Carter, C.L. Gallegos, W. Hunley, L. Karrh, E.W. Koch, J.M. Landwehr, K.A. Moore, L. Murray, M. Naylor, N.B. Rybicki, J.C. Stevenson, and D.J. Wilcox. 2004. Habitat requirements for submerged aquatic vegetation in Chesapeake Bay: water quality, light regime, and physical–chemical factors. Estuaries 27: 363–377.
Kenworthy, W.J., and M.S. Fonseca. 1996. Light requirements of seagrasses Halodule wrightii and Syringodium filiforme derived from the relationship between diffuse light attenuation and maximum depth distribution. Estuaries 19: 740–750.
Kenworthy, W.J., and D.E. Haunert, eds. 1991. The light requirements of seagrasses: proceedings of a workshop to examine the capability of water quality criteria, standards and monitoring programs to protect seagrasses. Beaufort: NOAA Technical Memorandum NMFS-SEFC-287 181 pp.
Koch, E.W. 2001. Beyond light: physical, geological, and geochemical parameters as possible submersed aquatic vegetation habitat requirements. Estuaries 24: 1–17.
Ku, H.H. 1966. Notes on the use of propagation of error formulas. Journal of Research of the National Bureau of Standards—C. Engineering and Instrumentation 70C (4): 263–273.
Lee, Z.P., K.P. Du, and R. Arnone. 2005. A model for the diffuse attenuation of downwelling irradiance. Journal of Geophysical Research 110: 1–15.
Liu, W.C., M.H. Hsu, S.Y. Chen, C.R. Wu, and A.Y. Kuo. 2005. Water column light attenuation in Danshuei River estuary, Taiwan. Journal of the American Water Resources Association 41: 425–435.
Masini, R.J., and C.R. Manning. 1997. The photosynthetic responses to irradiance and temperature of four meadow-forming seagrasses. Aquatic Botany 58: 21–36.
Murrell, M.C., J.G. Campbell, J.D. Hagy III, and J.M. Caffrey. 2009. Effects of irradiance on benthic and water column processes in a Gulf of Mexico estuary: Pensacola Bay, Florida, USA. Estuarine, Coastal and Shelf Science 81: 501–512.
Orth, R.J., M.R. Williams, S.R. Marion, D.J. Wilcox, T.J.B. Carruthers, K.A. Moore, W.M. Kemp, W.C. Dennison, N.B. Rybicki, P. Bergstrom, and R.A. Batiuk. 2010. Long-term trends in submersed aquatic vegetation (SAV) in Chesapeake Bay, USA, related to water quality. Estuaries and Coasts 33 (5): 1144–1163.
Pinheiro, J., D. Bates, S. Deb Roy, D. Sarkar, and R Core Team. 2016. nlme: linear and nonlinear mixed effects models. R package version 3.1-128. https://CRAN.R-project.org/package=nlme. Accessed July 1, 2015.
RDCT (R Development Core Team). 2016. R: a language and environment for statistical computing, v3.2.0. Vienna: R Foundation for Statistical Computing.
Schaeffer, B., J.D. Hagy III, R. Conmy, J.C. Lehrter, and R. Stumpf. 2012. An approach to developing numeric water quality criteria for coastal waters using the sea WiFS satellite data record. Environmental Science & Technology 46: 916–922.
Short, F.T., and S. Wyllie-Echeverria. 1996. Natural and human-induced disturbances of seagrass. Environmental Conservation 23: 17–27.
Søndergaard, M., G. Phillips, S. Hellsten, A. Kolada, F. Ecke, H. Mäemets, M. Mjelde, M.M. Azzella, and A. Oggioni. 2013. Maximum growing depth of submerged macrophytes in European lakes. Hydrobiologia 704: 165–177.
Spears, B.M., I.D.M. Gunn, L. Carvalho, I.J. Winfield, B. Dudley, K. Murphy, and L. May. 2009. An evaluation of methods for sampling macrophyte maximum colonisation depth in Loch Leven, Scotland. Aquatic Botany 91: 75–81.
Stankelis, R.M., M.D. Naylor, and W.R. Boynton. 2003. Submerged aquatic vegetation in the mesohaline region of the Patuxent estuary: past, present, and future status. Estuaries 26 (2A): 186–195.
Steward, J.S., R.W. Virnstein, L.J. Morris, and E.F. Lowe. 2005. Setting seagrass depth, coverage, and light targets for the Indian River lagoon system, Florida. Estuaries 28: 923–935.
TBEP (Tampa Bay Estuary Program) 2011. Tampa Bay Water Atlas. http://www.tampabaywateratlas.usf.edu. Accessed January 30, 2017.
Tyler, D., D.G. Zawada, A. Nayegandhi, J.C. Brock, M.P. Crane, K.K. Yates, and K.E.L. Smith. 2007. Topobathymetric data for Tampa Bay, Florida. Open-file report 2007-1051 (revised). St. Petersburg: US Geological Survey, US Department of the Interior. (poster).
US EPA (US Environmental Protection Agency) 2012. Technical support document for U.S. EPA’s proposed rule for numeric nutrient criteria for Florida’s estuaries, coastal waters, and south Florida inland flowing waters. Volume 1: Estuaries. November 30, 2012. EPA-HQ-OW-2010-0222-0002. 365 pp.
Williams, S.L., and K.L. Heck. 2001. Seagrass community ecology. In Marine community ecology, ed. M.D. Bertness, S.D. Gaines, and M.E. Hay. Sunderland: Sinauer Associates.
Woodruff, D.L., R.P. Stumpf, J.A. Scope, and H.W. Paerl. 1999. Remote estimation of water clarity in optically complex estuarine waters. Remote Sensing of Environment 68: 41–52.
Yarbro, L.A., and P.R. Carlson Jr. 2015. Seagrass integrated mapping and monitoring program, mapping and monitoring report no. 1.1. FWRI technical report TR-17. St. Petersburg: Florida Fish and Wildlife Conservation Commission 10 pp.
Zhu, B., D.G. Fitzgerald, S.B. Hoskins, L.G. Rudstam, C.M. Mayer, and E.L. Mills. 2007. Quantification of historical changes of submerged aquatic vegetation cover in two bays of Lake Ontario with three complementary methods. Journal of Great Lakes Research 33: 122–135.
Acknowledgments
We thank Dr. Peter Tango and two anonymous reviewers for helpful comments on the manuscript. The views expressed in this article are those of the authors and do not necessarily reflect the views or policies of the U.S. Environmental Protection Agency.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Richard C. Zimmerman
Electronic supplementary material

Fig. S1
Uncorrected satellite estimates of light attenuation in Choctawhatchee Bay (a) and example of correction with in situ data (b). In situ data of light attenuation were estimated as an annual average (2010) for monthly data at the sampling sites labeled as points in (a) (mean depth 7.2 m, minimum 3.5 m). The corresponding satellite data in the same grid cells were compared to the in situ data based on regressions of each dataset with frequency estimates for both. An example correction is shown in (b) where for any uncorrected satellite estimate (point 1), the corresponding frequency estimate on the regression curve from the satellite data was identified (point 2), matched with the corresponding frequency for the in situ data (point 3), and then related to the associated in situ light attenuation value (point 4) to yield the corrected satellite estimate (GIF 63.2 kb)

Fig. S2
Locations of selected water quality stations monitored by the Hillsborough County Environmental Protection Commission (TBEP 2011). Secchi observations at each station were used to evaluate changes in light requirements of seagrass at approximate biennial intervals from 1988 to 2014. Stations are labeled by their designation and were chosen based on continuity of data for the period of interest. HB Hillsborough Bay, LTB Lower Tampa Bay, MTB Middle Tampa Bay, OTB Old Tampa Bay (GIF 40.1 kb)

Fig. S3
Maximum depth of seagrass colonization (Z c,max, m) and light requirements (% surface irradiance at Z c,max) for multiple locations in Choctawhatchee Bay, Florida. Each location has light attenuation from satellite observations and an estimate of seagrass depth of colonization with a search radius of 0.04 degrees. Box plots show the 25th percentile, median, and 75th percentile. Whiskers extend to the 5th and 95th percentiles with outliers beyond. CCB Central Choctawhatchee Bay, ECB East Choctawhatchee Bay, WCB West Choctawhatchee Bay (GIF 107 kb)

Fig. S4
Maximum depth of seagrass colonization (Z c,max, m) and light requirements (% surface irradiance at Z c,max) for multiple locations in Tampa Bay, Florida. Each location has light attenuation from satellite observations and an estimate of seagrass depth of colonization with a search radius of 0.1 degrees. Box plots show 25th percentile, median, and 75th percentile. Whiskers extend to the 5th and 95th percentiles with outliers beyond. HB Hillsborough Bay, LTB Lower Tampa Bay, MTB Middle Tampa Bay, OTB Old Tampa Bay (GIF 104 kb)

Fig. S5
Maximum depth of seagrass colonization (Z c,max, m) and light requirements (% surface irradiance at Z c,max) for multiple locations in Indian River Lagoon, Florida. Each location has an average Secchi depth observation and an estimate of seagrass depth of colonization with a search radius of 0.15 degrees. Map locations are georeferenced observations of Secchi depth. Box plots show 25th percentile, median, and 75th percentile. Whiskers extend to the 5th and 95th percentiles with outliers beyond. BR Banana River, LCIRL Lower Central Indian River Lagoon, LIRL Lower Indian River Lagoon, LML Lower Mosquito Lagoon, LSL Lower St. Lucie, UCIRL Upper Central Indian River Lagoon, UIRL Upper Indian River Lagoon, UML Upper Mosquito Lagoon (GIF 68.9 kb)
Rights and permissions
About this article
Cite this article
Beck, M.W., Hagy, J.D. & Le, C. Quantifying Seagrass Light Requirements Using an Algorithm to Spatially Resolve Depth of Colonization. Estuaries and Coasts 41, 592–610 (2018). https://doi.org/10.1007/s12237-017-0287-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-017-0287-1