Abstract
Agriculture that depends upon irrigation is challenged by the increasing scarcity of fresh water and global climate change, and increasing human populations aggravate this situation. The potato crop depends on a regular water supply to secure high quality yields. Abiotic stress factors, such as drought, heat and salinity, have severe, adverse effects on growth and yield. In this review, different approaches to cope with water stress are presented. These approaches include altering morphological, physiological and genetic characteristics of potato and the use of biotechnology. For example, native potato and alien genes have been identified by transcriptomics and may provide useful candidates for deployment against stress. Transgenic potato cultivars harboring many of these genes have been evaluated and show promise for future release as new, stress tolerant cultivars. Potential management tools for economizing water use include efficient irrigation systems and precision agriculture. The use of alternative water resources, such as greywater, recycled wastewater, agricultural drainage water, and desalinated water will contribute to the water requirements of the potato crop and should help meet future challenges.
Resumen
La agricultura que depende de riego se enfrenta al reto de la escasez cada vez mayor de agua dulce y al cambio climático global, y con el aumento de la población humana se agrava esta situación. El cultivo de la papa depende de un suministro regular de agua para asegurar rendimientos de alta calidad. Los factores del agobio abiótico, tales como la sequía, el calor y la salinidad, tienen efectos severos adversos en el crecimiento y en rendimiento. En esta revisión se presentan diferentes enfoques para lidiar con el agobio hídrico. Estos enfoques incluyen alteraciones de características morfológicas, fisiológicas y genéticas de la papa y el uso de la biotecnología. Por ejemplo, papa silvestre y genes ajenos se han identificado por transcriptómica y pudieran proporcionar candidatos útiles para ubicación contra el agobio. Se han evaluado variedades de papa transgénicas que contienen muchos de estos genes y se muestran promisorias para futura liberación como variedades nuevas tolerantes al agobio. Las herramientas potenciales de manejo para economizar el uso de agua incluyen sistemas eficientes de riego y agricultura de precisión. El uso de fuentes alternativas de agua, tales como aguas negras, agua residual reciclada, agua del drenaje agrícola, y agua desalinizada, contribuirán a los requerimientos hídricos del cultivo de la papa y pudieran encarar retos futuros.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Extreme weather events such as floods in Pakistan, heat waves in Russia, tornados in the southeastern United States and devastating droughts in Africa support the idea of climate change with potentially devastating effects on global freshwater resources (Bates et al. 2008; Qi and Luo 2005). Predicted climate changes over the next 50 years include increased global temperatures, frequent extreme temperatures (especially heat waves), increased ozone at crop level, increased carbon dioxide, and drought in many crucial crop production regions (Gornall et al. 2010; Jaggard et al. 2010; http://www.global-warming-forecasts.com/). These projected changes will show wide regional variation (Hansen et al. 2006; Scholze et al. 2006; Wolf and van Oijen 2002).
Increasing human population, urbanization and dietary trends are straining land and water resources, adversely affecting the environment globally (Foley et al. 2011). Many semi-arid and arid regions are already experiencing moderate to severe water shortages and a decline in water quality. These developments lead to reduced crop productivity of irrigated and rain fed agriculture (Anonymous 2006; Kotb et al. 2000; Qi and Luo 2005; Turral et al. 2011).
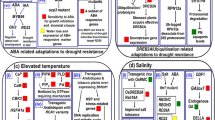
Potatoes are an important staple food worldwide and climate change will adversely affect potato production primarily in warm, dry climates. A modeling study carried out at the International Potato Center (Hijmans 2003) indicated most potato producing countries would experience considerable declines in potato yield when temperature change was the major driver. Hijmans (2003) stated that improving the adaptation of potato to heat stress could alleviate yield loss. Multidisciplinary science can contribute to the development of cultivars with enhanced tolerance to heat, drought and salinity (Howden et al. 2007). Understanding the physiological and genetic basis of adaptation and employing modern breeding techniques are essential to the development of stress tolerant varieties (Reynolds and Ortiz 2010).
This review will examine the potato’s responses to drought and salinity stress. The responses will then be examined in the context of needed strategies and how change can be implemented so that future potato crops can respond to the developing challenges posed by global climate change.
Potato Responses to Drought Stress
Climate change will affect potato production in complex and location dependent ways. For example, increased CO2 levels may lead to increased yield even if the plants were to be exposed to moderate levels of drought stress (Fleisher et al. 2008a). Drought may be the most important abiotic stress factor for future potato production (Burton 1981). Although potatoes are an irrigated crop and can use water effectively as reflected by a high harvest index (Vos and Haverkort 2007), current potato cultivars are extremely sensitive to drought stress as well as heat stress (e.g., Iwama and Yamaguchi 2006; Levy and Veilleux 2007). In temperate climates, where potatoes are grown during the rainy summer season, dry spells during various growing stages may lead to transient water shortage and drought stress. In many semi-arid and arid regions where irrigation is obligatory, transient wilting under high temperature and low air humidity is a common phenomenon. Transient stress can result in loss of yield and tuber quality. Although these two abiotic stress factors have been shown to induce distinct defensive responses in Arabidopsis when they occur simultaneously (Rizhsky et al. 2004), this joint defensive response remains to be examined in potato.
Burton (1981) noted that production of dry matter by CO2 uptake is accompanied by loss of water through transpiration. He estimated potential loss of water “on a hot day in Idaho” through stomatal and cuticular transpiration of 100,000 kg of water per hectare per day. When water is limiting, stomatal conductance decreased at leaf water potentials of −0.3 to −0.4 MPa (Iwama and Yamaguchi 2006). This situation results in a reduction in photosynthesis although photosynthetic rate changes in a curvilinear fashion to stomatal conductance (Vos and Groenwold 1989b). Since maximum CO2 fixation is essential to optimal crop production in potato (Blum 2009), restriction of transpiration as a drought avoidance mechanism may not be a realistic option for production systems. However, reduced transpiration leading to sub-optimal yield at relatively consistent production levels may be acceptable for subsistence farming in drought prone environments (Sinclair 2011).
Potential mechanisms for drought resistance involve avoidance or tolerance (Jordan et al. 1983; Turner 1982). Avoidance responses will require fine-tuning potato phenology to the weather patterns of a growing site (Rockström 2003). Incorporating drought tolerance or coping mechanisms into potato through traditional or analytical breeding with gene transfer will require a coordinated, long-term commitment to new cultivar development based on an in-depth understanding of the plant’s response to adverse growing conditions (Araus et al. 2008; Blum 2011; Moffat 2002).
Drought Escape
The development of early maturing cultivars has been a successful avoidance strategy for short season areas. This approach allows the plants to escape adverse climatic conditions that are often encountered during the latter part of a growing season although the major drawback is lower cumulative photosynthesis (Tardieu 2012). An essential first step in breeding for drought prone areas will involve the synchronization of crop growth and development with the predicted water supply (Blum 2009).
Future potato production may benefit from extended crop duration provided by global warming (Tardieu 2012). This situation could lead to the use of cultivars with a longer development cycle and provide greater flexibility for selecting an appropriate, site-specific cultivar. However, increased temperature, greater temperature extremes and a longer growing season could also significantly increase irrigation requirements (Gornall et al. 2010).
Irrigation has often been cited as a solution for sub-optimal soil water (Pehrson et al. 2010; Walworth and Carling 2002). Optimized irrigation practices currently use best management practices (BMPs) that schedule watering routines based on repeated measurements of crop evapotranspiration (ET) and soil water tension (SWT) (Shock et al. 2007). However, agriculture is responsible for at least 75 % of global water use (Araus et al. 2008). With increased water use by society and reduced water availability for irrigation, future approaches to potato production will need to rely more heavily on the innate capabilities of the potato plant.
Potato Roots
The potato root system in tuber-based seed systems is composed of branched fibrous roots that primarily originate from adventitious roots associated with developing shoots (Cutter 1978). Functional roots also form in association with developing stolons and tubers (Kratzke and Palta 1985). Many potato cultivars are shallow rooted and often produce most of their roots in the plow layer (Iwama 2008; Iwama and Yamaguchi 2006; Lesczynski and Tanner 1976). This situation may be due to inhibition of root growth by high soil strength greater than 1 MPa (Miller and Martin 1987).
In some cases, management practices can overcome this limitation. For example, deep plowing or sub-soiling with destruction of the pan layer has led to greater exploitation of soil moisture by the potato plant (Bishop and Grimes 1978; Tanner et al. 1982) although the result depended on soil and weather conditions (Miller and Martin 1987; Parker et al. 1989). These results emphasize the importance of creating favorable soil environments that avoid impeding potato root growth in order to maximize the depth of water extraction (Stalham and Allen 2004). However, soil strength can still increase substantially under dry conditions when the matric potential of the soil increases (Bengough et al. 2011).
Root mass may be a useful trait for selecting drought tolerant germplasm. However, recording root mass under field conditions is laborious and inaccurate. Fortunately, new methods continue to be developed that may assist in selection of germplasm with high drought tolerance (e.g., Iwama 2008).
Increased Root Efficiency
As the evidence for global warming becomes more convincing, long-term solutions to drought tolerance will be required. For example, increased efficacy of the root system may be an effective means of avoiding or significantly delaying the effects of drought stress (Burton 1981; Porter and Semenov 2005). Under conditions of low soil strength (<1 MPa), root penetration to soil depths exceeding 100 cm has been observed (Stalham and Allen 2001). Since there are genotypic differences in rooting depth and the amount of root growth, it should be possible to select for potato plants with superior capabilities to penetrate high strength soils and extract water in drought prone environments (Lahlou and Ledent 2005; Taylor 1983). However, selection of superior genotypes must also take into account such factors as root growth and root system architecture (Tardieu 2012).
Small root length in the soil profile may be an important component of the potato plant’s sensitivity to stress (Iwama and Yamaguchi 2006). Other plant traits necessary to overcome mechanical impedance, penetrate strong soil layers and extract water may include root diameter, growth rate, root hair morphology, root tip shape, border cells and mucilage production (Bengough et al. 2011; Clark et al. 2003; Whitmore and Whalley 2009). These root traits should be examined for possible use in selecting drought resistant potato germplasm.
Drought stress can enhance abscisic acid (ABA) production within root systems of numerous species. The ABA, in turn, can promote root growth and increase root hydraulic conductivity while simultaneously closing stomata. However, at present, this area may not offer practical approaches to breeding for increased drought resistance (Neumann 2008).
Water uptake by roots and its movement within crop plants is tightly regulated by and integrated with shoot physiology (Maurel et al. 2010). The limited ability of potato roots to transport water has been suggested as the basis of potato’s drought sensitivity (Gregory and Simmonds 1992; Weisz et al. 1994). However, under moderate soil moisture deficit, deep potato roots were capable of withdrawing water “from considerable distances ahead of the rooting front” (Stalham and Allen 2004). Potato roots were also capable of sensing soil water deficits although this potential control point remains to be fully described in potato (Schafleitner 2009). A major control feature may occur in water channel proteins called aquaporins that regulate the hydraulic conductivity of roots (Brodribb et al. 2010). However, increased aquaporin expression has not lead to improved drought resistance and may not be a practical goal for breeders currently looking for easily screened drought resistant traits (Neumann 2008). Nevertheless, an increase in the hydraulic resistance of roots can lead to increased wheat yields (Richards 2006). Breeders using new genomic technologies should examine this response in potato at the physiological and molecular levels for possible future use (Witcombe et al. 2008).
Tubers and Abiotic Stresses
With the imposition of drought stress, accumulation of tuber dry matter and final tuber yield have been shown to decline (Levy 1985) although the effect may depend on the stage of potato development during the stress period (Schafleitner 2009). In addition, the presence of tubers may alter the photosynthetic response of drought stressed potato plants (Basu et al. 1999).
When heat stress accompanies drought stress, pronounced decline in tuber yield and tuber quality have been evident with notable differences in heat tolerance among potato cultivars (Ahn et al. 2004; Levy 1986). Tubers of drought sensitive cultivars accumulated proline in response to drought (Levy 1983b). Decreased water content, altered dry matter assimilation and increased sucrose levels have also been observed after imposition of heat or drought stress (Gawronska et al. 1992; Lafta and Lorenzen 1995). These changes affect subsequent storage performance of tubers (Bethke et al. 2009; Eldredge et al. 1996). The amount of time seed tubers have been in storage may also affect the subsequent response of potato plants to drought stress through effects on altered root and shoot development (Coleman 1988).
Leaf and Canopy Responses
Drought affects potato canopy architecture by decreasing leaf size and leaf expansion rate while limiting formation of new leaves and increasing the rate of senescence (Fleisher et al. 2008b). Since potato cultivars that are capable of maintaining above ground biomass in response to drought can produce larger yields (Schittenhelm et al. 2006), this area should be explored further.
When drought affects the potato crop, the first morphological effect is reduction in leaf size (Jefferies 1993; Jefferies and Mackerron 1993). This effect is more apparent in reduced leaf area than in leaf dry weight (Vos and Haverkort 2007). The smaller leaf area induced by drought stress in a range of species has been associated with greater vein density that may contribute to increased drought tolerance (Scoffoni et al. 2011). In many species, vein density has been correlated with hydraulic conductivity of water and maximum photosynthetic rate in leaves. This observation supports the hypothesis that vein location affects photosynthesis due to its effect on leaf hydraulic efficiency. (Brodribb et al. 2007). The role of vein density in drought tolerance should be examined in potato.
Tissue water potential lower than −0.4 MPa has been linked to complete inhibition of leaf and tuber growth (Gandar and Tanner 1976). Water potentials below −2.0 MPa can lead to loss of viability (Ackerson et al. 1977; Shimshi et al. 1983) although Vos and Haverkort (2007) noted the permanent wilting point of young potato crops was −0.6 and fell to −1.0 MPa in older crops. Jefferies (1989) indicated that the relative extension rate of potato leaves decreased to zero at leaf water potentials below −1.0 MPa. The relative water content (RWC) at a leaf water potential of −1.0 MPa was effective in differentiating drought resistance of potato cultivars (Coleman 1986). The more drought resistant cultivar, Raritan, had greater RWC in young and mature leaves than the drought sensitive cultivar, Shepody. The greater level of cellular hydration as exemplified by a higher RWC level may have been due to osmotic adjustment (OA).
Osmotic Adjustment
Although Vos and Haverkort (2007) suggested there was no evidence for osmotic adjustment in potato, other studies have indicated its occurrence and potential importance for dealing with drought stress (Coleman 1986; Heuer and Nadler 1998; Jefferies 1993; Levy 1983a, 1992). In semi-arid and arid climates, potato crops are exposed to various degrees of transient drought and heat stress resulting from high ambient temperatures and low air humidity. These conditions may occur at different stages of plant growth. Partial leaf wilting is commonly seen and may lead to loss of tuber yield and quality. Osmotic adjustment may help maintain leaf turgor and promote faster recovery upon stress relief. In numerous species, the accumulation of solutes during a decline in leaf water potential is essential for maintaining leaf hydration, avoiding cellular desiccation damage and promoting drought resistance in crop plants (Blum 2011; Chaves and Oliveira 2004; Girma and Krieg 1992; Jones and Turner 1978; Oosterhuis and Wullschleger 1987; Verslues and Bray 2004). For example, in wheat, the accumulation of potassium under drought stress conditions (controlled by a recessive osmoregulation gene, or) has been associated with greater yield (Richards 2006). In addition to OA, the protective roles played by small proteins such as the dehydrins remain to be examined in potato (Cellier et al. 1998, 2000; Han and Kermode 1996). These studies should be accompanied by appropriate water status measurements (Jones 2007).
Transpiration Efficiency
Climate change and increasing population pressure will require the development of potato crops with the ability to produce increased yield per unit of water used. Greater water use efficiency (WUE) is regulated by transpiration that is, in turn, primarily controlled by stomata. Increased transpiration efficiency has often been cited as a useful goal, provided harvest index does not decrease significantly (Yoo et al. 2009). Since maximizing yield under drought stress requires optimal soil moisture acquisition in order to support stomatal transpiration as well as minimized water loss by soil evaporation, this potentially useful goal is described as the effective use of water (EUW) and can be used for developing superior cultivars (Blum 2009).
The selection for high WUE in breeding for water-limited growing conditions may lead to reduced drought tolerance and to reduced yields (Blum 2009). An interesting program has incorporated carbon isotope discrimination (strongly associated with increased transpiration efficiency) for detecting superior yield responses in sunflower and wheat under drought conditions (Richards 2006). However, isotope discrimination may be a positive attribute for crop yield under extremely dry conditions and a negative feature under mild water deficits (Tardieu 2012). Although work with potato indicates that carbon isotope analysis may be a useful indirect screening method (Vos and Groenwold 1989a), additional studies in potato are necessary.
Stomata and Cuticle
Stomatal closure is sensitive to increasing levels of drought stress as demonstrated by falling transpiration rates when the leaf water potential was lower than −0.1 MPa. This condition can develop quickly when soil moisture decreases or on days of high potential evaporation (Harris 1992). Since small decreases in stomatal conductance can affect water loss by transpiration more than carbon assimilation, WUE will improve (Yoo et al. 2009). While most water loss by transpiration occurs during the day, transpiration during the night can account for 5–30 % of diurnal water loss depending on the species (Yoo et al. 2009). This feature should be considered in any physiological study of CO2 assimilation and transpiration in potato plants subjected to drought stress. In addition, the timing and degree of drought stress will be critical for defining stomatal and growth responses (Tardieu 2012).
Although stomata serve as the primary regulator of water loss to the environment, the cuticle also plays an important role. When stomata close during drought stress, the waxy cuticle can become the controlling factor in water loss and, to a lesser extent, CO2 exchange (Boyer et al. 1997; Yoo et al. 2009). When potato cultivars were exposed to mild drought stress, the drought resistant cv. Raritan doubled its epicuticular wax level whereas the drought sensitive cv. Shepody increased its wax level by less than 20 % (Coleman 1986).
Leaf Senescence
Resistance to drought-induced leaf senescence has been outlined for sorghum lines (the “stay green” lines). These genotypes have resulted in greater grain yield under drought conditions (Harris et al. 2007; Richards 2006). However, stay-green mutations in soybean have led to increased drought stress susceptibility (Luquez and Guiamét 2002). Since drought stress often results in extensive leaf loss in potato (see “Leaf and Canopy Responses” above), resistance to stress induced leaf loss and increased functional leaf life should be assessed for its usefulness in breeding for drought resistance in potato (Burton 1981).
Addressing the Problem—Irrigation Management
In view of the great susceptibility of potato to drought and the decrease in water resources, farmers are challenged to save water without sacrificing yield or quality. Optimized water use, avoiding wastage and leakage, and employment of efficient irrigation protocols have been recommended. The introduction of efficient irrigation technologies such as micro and trickle irrigation systems has been suggested (Lamm et al. 2007). The low pressure (LPS) drip irrigation system designed to operate at low pressures (0.14–0.21 kg/cm2) and with drippers of low flow rates was shown to conserve 40 % of the irrigation water in potatoes grown in Mexico compared to the common flooding irrigation system (Dowgert et al. 2006). In the arid Arava region of Israel, where the annual winter rain amounts to only ca 20 mm, and where the main water sources for irrigation are underground saline reservoirs, potatoes and other vegetables are grown successfully on sandy soils. Subsurface low pressure/low flow drippers are employed emitting 0.6 l/h (compared to 3.5 l/h commonly used). Up to 30 % of the water input was conserved with no reduction of tuber yields or tuber quality (personal communication, D. Levy).
Attempts to save water have prompted the assessment of deficit irrigation protocols and development in precision agriculture for reducing inputs and increasing resource use efficiency to improve sustainability (Fereres and Soriano 2007; Sepaskhah and Ahmadi 2010).
Deficit Irrigation (DI) and Partial Root Zone Drying (PRD)
Under conditions of limited water availability, DI and PRD allow the farmer to increase cultivated irrigated land and contribute to the efficient use of machinery and irrigation equipment. In addition, farmers may consider reduced application of nutrients, pesticides and water and accept reduced yields to improve cost efficiency. An important tool required for successful implementation of DI is crop production modeling for potato where the interactions between the various crop inputs, the productivity gains and the operating costs can be quantified (Campbell et al. 1976; Haverkort and MacKerron 1995).
Studies of DI under field conditions have become increasingly important in areas that rely heavily on fresh water supply. For example, the Columbia basin in the U.S. Reclamation’s 2011 SECURE Water Act Report (www.usbr.gov/climate) identified pending climate challenges for the region. Projections show that temperatures throughout the Columbia River Basin above the Dalles Dam may increase steadily by 3–4 °C during the 21st century. Increased wintertime runoff and reduction in runoff during spring and summer are likely to translate into water supply reduction for meeting irrigation demands.
Reduced supplies of irrigation water can result in decreased crop yield and emphasize the importance of DI studies. For example, in Oregon, Shock and Feibert (2002) studied three DI treatments. At each irrigation application 100, 70, and 50 % of the accumulated evapotranspiration (ETc) were applied. In experiments conducted over 3 years, the potato crop lost total yield and grade under DI with reduced profitability. The results suggested that DI for potato growing in the semi-arid environment of eastern Oregon was not a viable management tool. Similar results were found by Alva (2008) in Prosser, Washington working with cv. Ranger Russet.
Best practices include scheduling irrigation for the period from tuber initiation through mid-bulking, and selecting cultivars that use less water and/or are less sensitive to water stress. Geerts and Raes (2009) discussed DI as a strategy to maximize crop water productivity in dry areas. They state that no single DI strategy applies to all crops and that the levels of tolerance to drought differ among crops and at the various stages of plant development for each crop.
Deblonde et al. (1999) working in Belgium under temperate climate conditions, tested six potato cultivars in three irrigation treatments: fully irrigated by rainfall with complementary irrigation, rainfall only and drought treatment imposed by covering plots with plastic sheets from 50 % emergence until 55–57 days post-emergence and then removed. Tuber dry yield and harvest index were significantly reduced under the drought treatment in comparison with full irrigation. The later cultivars were better able to withstand drought stress during the early season than early cultivars under these experimental conditions.
Fabeiro et al. (2001), working in a semi-arid climate in Spain, also studied DI in potato and found that tuber yield was correlated with the amount of water applied. The most adverse effect on yield occurred when water shortage was imposed during the bulking and ripening stages. However, Darwish et al. (2006) did not observe reduced tuber yield of potato cv. Santana under DI in Lebanon using 80 % of measured evapotranspiration compared to the control of 100 %. Similarly, Nimah and Bashour (2010) found that irrigating 50 to 75 % of the root zone of potato and pepper gave better yield than the control in a study of potato cv. Spunta and pepper cv. Andalus in Lebanon. Water saving of up to 195 mm for potato and 192 mm for pepper was obtained without reduction in total yield for both crops by limiting water penetration to the top 50 % of the root zone. Irrigating 50 % of the root zone resulted in better WUE amounting to 5.46 kg/m3 and 6.46 kg/m3 for potato in 2007 and 2008, respectively.
Partial root zone drying (PRD) is a modified form of deficit irrigation. This procedure involves wetting only one part of the root zone in each irrigation event, letting the other part of the root zone dry and then rewetting it by shifting water application to the dry side and letting the other side dry (Ahmadi et al. 2010a, b; Sepaskhah and Ahmadi 2010). Ahmadi et al. (2010a and b) working in Arhus, Denmark under temperate climate conditions investigated the effect of DI and PRG (65 % of FI) compared to full irrigation (FI) on potato cv. Folva grown in drainable concrete lysimeters filled with three soil types: sandy loam, loamy sand and coarse sand. The lysimeters were equipped with an automatic mobile roof to protect the experiments from rainfall. The authors concluded that neither DI nor PRD could be recommended due to yield loss of 28 % compared to FI under the conditions of their experiment.
Xie et al. (2012), working in a semi-arid climate in northern China, found that reducing irrigation to 50 % of the control did not affect tuber yield, whereas WUE was significantly increased, regardless of irrigation method. A partial root-zone drying (PRD50) irrigation procedure, where furrows were alternatively watered and dried in each irrigation cycle, gave the best results. Yield differences among cultivars and locations were indicative of a G×E interaction. These interactions probably explain the conflicting data and suggest that DI needs to be assessed under local climatic and management conditions. In addition, monitoring soil matric potential (SMP) in the field could be helpful when DI is attempted. Wang et al. (2007) found that SMP of −25 kPa was the most favorable setting for potato production, while −15 kPa was too high and −45 kPa lead to severe water stress.
Muñoz-Carpena et al. (2005) found that irrigating tomato with a low-volume/high frequency (LVHF) soil moisture-based drip irrigation on shallow sandy soil reduced water use while not significantly affecting tomato yield. Tensiometers at the −15 kPa setpoint resulted in 73 % reduction in water use compared to a control irrigation treatment, or 50 % with respect to the historical evapotranspiration demand of the area (ET-100 %). These authors (Muñoz-Carpena et al. 2008) reported the development of an inexpensive irrigation controller that reduced water use up to 61 % compared to evapotranspiration-based application and up to 79 % when using switching tensiometers. Potato fields fully covered with green active canopy are commonly irrigated with amounts attempting to restore evapotranspiration demand of the area (ET-100 %). The data presented here suggest that the crop may have been over-irrigated in some cases and that employing soil-moisture sensors at the root zone can save water. Additional studies are needed to investigate opportunities to conserve water using DI and PRG under different growing conditions and with different cultivars.
Precision Agriculture (PA)
The concepts of precision farming or PA have been devised to increase farming efficiency and profitability and enhance environmental sustainability. PA allows site-specific management to account for variation in soil properties, crop requirements, pests and diseases (Cassman 1999). This technology offers improved efficiency of tillage, application of seed, nutrients, water, and pest control.
PA requires the integration and implementation of technologies that depend upon computer software, a global positioning system (GPS), geographic information systems (GIS), soil and crop sensors, and application control (Pierce and Nowak 1999). The variable application of inputs and management operations is mostly relevant to large scale farms, where field size and within-field variation justify the costs (Cassman 1999). PA is expected to restrict the excess use of inputs, compared to a uniform application to an entire field, thereby increasing efficiency and environmental quality. Advances in PA include tillage, seeding and harvest with GPS guided machinery that improves accuracy, avoids overlap, and saves seed as well as time and energy. Demonstrations of this technology are available online.
Spatial knowledge of soil conditions is important for management decisions. Current practice is to analyze soil samples, taken at several locations in a field, for physical properties. To obtain adequate coverage, many soil samples must be analyzed. Van Bergeijk et al. (2001) proposed a method to gather information on topsoil clay content automatically during plowing. This system uses a minimal set of sensors added to the plow and functions as a plow draught recording system in an on-farm precision agriculture research program. Spatial variation in topsoil clay content was correlated with spatial variation of crop yield. The spatial resolution of soil physical properties could be important for site-specific irrigation management. Persson et al. (2005) investigated the relationship between yield of potato and topographical factors in central Sweden. Up to 20 % of the yield could be explained in the final model for one of the two fields studied.
Khosla et al. (2008) studied precise nutrient management strategies for semi-arid irrigated cropping systems. Their site-specific management zones (SSMZ) approach was obtained by dividing the fields into smaller units that differed with regard to productivity potential. AgriTrak ProfessionalTM1 software was used to delineate management zones (Fleming and Westfall 2000). Maize yield and N uptake were statistically different between the low and high productivity management zones for most site-years and N fertilizer rates (p ≤ 0.05). The authors suggested that N fertilizer use could be optimized by using site-specific management zones as a basis for variable-rate N fertilizer application, thus improving economic return by as much as $679 USD ha−1. Overall, SSMZs are a practical approach to precise N management in the Western Great Plains of the US. Inexpensive site-specific management zones provide a pragmatic approach to precise N management in irrigated maize.
Nitrogen application in potato fields based on spectral indices has also been studied (Cohen et al. 2010). The main objective was to test the feasibility of determining the relationships among leaf spectral reflectance, simulated bands of the VENμS satellite, and nitrogen levels in potato petioles and leaves. The VENμS satellite has considerable potential for mapping spatial and temporal changes in leaf-N percentage since it can provide images of large areas every 2 days at low cost. The correlation between petiole NO3–N and leaf-N percentage was statistically significant but moderate (r 2 = 0.63). However, clouds interfere with satellite performance. Higher resolutions and additional improvements are required prior to the implementation of satellite data on a commercial scale.
Identification of pest infections at an early stage is essential to protect the potato crop and can be integrated into PA, as in winter wheat, for powdery mildew (Blumeria graminis) and leaf rust (Puccinia recondita) pathogens, by using remote sensing images (Franke and Menz 2007). High-resolution, multi-spectral data were more suitable to detect variation in crop vigor than for early detection of crop infections. Dammer and Ehlert (2006) developed real-time technology to apply fungicides to cereals. A CROP-Meter, a real-time sensor that measures crop biomass density that could be correlated with the Leaf Area Index was used. Field scale strip trials were conducted with the sensor-operated field sprayer to analyze the yield response of the crop. Higher, lower, as well as similar yield levels were obtained in the variable-rate plots in comparison with the uniform plots. In the 5-year experiments conducted in 11 field trials, average fungicide savings of 22 % were achieved.
Precision Irrigation (PI)
Precision irrigation is defined as timely and accurate water application in accordance with spatial and temporal soil properties in response to plant requirements during different growth stages (Al-Kufaishi et al. 2006). Smith et al. (2010) reviewed the role of PI with regard to PA concluding that PI will need technological improvement before widespread application. The recent development of crop simulation models may enable identification of optimal strategies. Zhu et al. (2011) acknowledged crop and agro-ecosystem models, including de Wit School, DSSAT series and APSIM series models, which have contributed substantially to improvement of agricultural practices. However, these models are weak in predicting performance of crops under field conditions. This situation limits their application in PI.
Al-Kufaishi et al. (2006) used a simulation model to calculate the amount of water required by sugar beets in a field experiment in Germany. The soil water balance was simulated based on 21 soil-sampling sites in the field. Spatially variable irrigation applied by a central pivot system was compared to uniform irrigation. When the soil moisture content dropped below 50 %, the model automatically triggered a sequence of irrigation to apply 20, 30 or 40 mm of water to raise soil moisture content to 80 % of field capacity. The comparison revealed that loss of water was greater for uniform applications than variable rate applications (VRA). The VRA scenario of 20 mm water was best for water conservation.
A prototype real-time, smart sensor array for measuring soil moisture and soil temperature that used off-the-shelf components was developed and evaluated for scheduling irrigation in cotton with a center-pivot variable-rate irrigation (VRI) system (Vellidis et al. 2008). The VRI system worked by cycling sprinklers on and off and by adjusting the travel speed of the center pivot. Their smart sensor array was a low cost system to monitor spatially variable soil water status in field crops. The authors indicated that developing a smart sensor array to interface with VRI could provide a fully automated, closed-loop irrigation system.
Shinghal et al. (2010) reported the application of wireless sensor network (WSN) technology to improve potato crop production in India. Li et al. (2011) developed a wireless sensor and actuator network (WSAN) for soil monitoring and irrigation control with a laboratory supervising system formed by a web-server. The system could automatically sample soil properties including soil moisture, electrical conductivity, and near surface temperature at four different depths and transmit field data through a cellular network to the web-server. Although data accuracy rate was greater than 97 %, the system needed further improvements prior to implementation.
In these studies, water status has been based on soil samples, soil sensors and climate data sensors. However, for measurements of crop and soil responses across large and variable fields, effective non-invasive sensing techniques and software capable of integrating data at relevant spatial scales are essential for PI systems. Jackson et al. (1981) claimed that irrigation-water distribution could be improved by directly monitoring crop water status rather than by measuring soil water content. Remote thermal imagery provides spatial information of surface temperature and enables mapping of canopy temperature variability over large areas. To make these thermal images useful for supporting VRI in space and time, Alchanatis et al. (2010) identified a requirement for assignment of irrigation zones. The recent development of un-cooled thermal imaging systems reduced the cost of the systems and, more importantly, made them easy to use in field conditions. Their relatively high spatial and thermal resolution enabled radiometric measurement of temperature with a typical sensitivity of 0.1 °C. Normalization is required to use TC as a water status indicator to account for varying environmental conditions. The crop water stress index (CWSI) has been used for normalization (Idso et al. 1981; Jones et al. 2002):
where Twet is the temperature of a leaf transpiring at the maximum potential rate and Tdry is the temperature of a non-transpiring leaf. Twet and Tdry may be computed by laying a set of assumptions and inversely solving for the canopy energy balance. A simple empirical determination of Twet and Tdry was used in several crops and orchards including cotton, grape, and olive (Cohen et al. 2005). The relatively low spatial resolution of the thermal images necessitated the use of high spatial resolution of red, green and blue (RGB) images taken at the same site to accurately extract the canopy fraction from each pixel (Moller et al. 2007). Laborious manual alignment of each pair of thermal and digital RGB images has mostly been used.
Zakaluk and Sri Ranjan (2006) investigated the feasibility of using a 5-megapixel digital camera to determine the leaf water potential (ΨL) of potato plants by capturing red, green, blue (RGB) digital images in the visible region of the electromagnetic spectrum. Their work showed promise of using RGB to assess leaf water potential and soil nitrate status. Wang et al. (2010) presented a computationally efficient algorithm for alignment of optical (RGB) and thermal (IR) image pairs. This algorithm was designed and packaged as a software application that provided an intervention free process for extracting plant water stress information to be fed into an automated irrigation scheduling program.
In Israel, Alchanatis et al. (2010) used thermal images of cotton plots to assess leaf water potential as an indicator of plant water status to control irrigation (Fig. 1). The method used an infrared (IR) thermal camera (ThermaCAM model PM545, FLIR Systems, Sweden) equipped with a 320 × 240 pixel microbolometer sensor, sensitive in the spectral range of 7.5–13 μm and a lens with a field of view of 240 mounted 5–20 m aboveground, pointing vertically downwards. A meteorological station positioned within the experimental plot accumulated data of global solar radiation, wind speed, air temperature and relative humidity. A model was developed based on vertically acquired high-resolution images that could be applied to lower-resolution, obliquely acquired images to produce a reliable map of the water status of a wider field area. However, to validate this model, larger scale measurements were needed, in different locations under different meteorological conditions.
Leaf water potential (LWP) map of experimental plots of cotton obtained with an infrared thermal camera. Digits indicate irrigation treatments: 1—fully watered (100 % of the required amount), 2—moderatey stressed (81 % of the required amount) and 3—highly stressed (63 % of the required amount). Grid lines indicate borders of irrigation treatments, and inner squares indicate areas that were sampled for calculation of average LWP values (Alchanatis et al. 2010)
Padhi et al. (2012) measured soil water content by thermal images of cotton using an infrared camera (NEC TH7800 model, NEC, Japan) as well as neutron probe data. Leaf water potential and stomatal conductance were measured on selected occasions. All measurements were made at fixed locations within three replicate plots of an irrigation experiment that consisted of four soil-water deficit treatments. Canopy temperature related well with soil water within the root zone of cotton as well as a stomatal conductance index derived from canopy temperature. Similarities in the pattern of spatial variation in canopy temperature and soil water over the experimental field indicated that thermography could be used with a stomatal conductance index to assess soil water deficit. This approach also allowed irrigation scheduling in order to apply water to areas where it was most needed.
In Israel, ground and aerial thermal images have been tested for water status mapping of potato crops on a field scale (Fig. 2). Analysis of the ground thermal and RGB images from three dates during each of two growing seasons showed highly significant correlations between CWSI and stomatal conductance during the tuber bulking stage (R 2 = 0.8–0.96, p < 0.01). Aerial thermal and RGB images revealed significant correlation between CWSI and stomatal conductance (R 2 = 0.87, p < 0.01).
Thermal images of the canopy of crop plants seem to offer a useful tool for the control of irrigation. Thermal images recorded by airplane photography can provide images of large fields at a reasonable resolution. Decision-making software, possibly coupled with automatic control operation, are also needed. A demonstration of VRI with pivot irrigation in field crops and in potato can be obtained at the Zimmatic website (www.zimmatic.com — accessed 5/28/2012). However, additional improvements in the PA and PI are expected to assure farmers of its reliability in securing yields and quality on a large scale operation.
Alternative Water Sources for Irrigation
Limited resources of fresh water have stimulated the search for alternative water sources for irrigation. Water resources other than natural fresh water have become an important source for irrigation in many parts of the world (Turral et al. 2011). This includes various forms of recycled water, saline and desalinized water.
Agricultural Drainage Water
About 40 % of the water applied for irrigation is discharged as drainage water to the shallow groundwater (Rhoades et al. 1989). Considerable supplies of drainage water are available throughout the world (Rhoades et al. 1992). This agricultural drainage water may be reused and has already become an important source of irrigation (Grattan 2002; Rhoades et al. 1989). Management practices for the control of water and soil salinity are essential components of irrigation management with marginal quality water. For example, agricultural drainage water may contain pesticides and herbicides, as well as elevated salinity. Alternatively, drainage water may contain beneficial nutrients. It is important to monitor the levels of solutes in the recycled water supply to meet crop requirements. Hence, the water properties must be controlled for appropriate irrigation and soil management practices.
Recycled Wastewater
In Australia where fresh water is scarce and precious, irrigators have often experienced drastic cuts in water allocations, affecting food availability and prices. The National Program for Sustainable Irrigation Land & Water Australia (Anonymous 2009) published a comprehensive document on using recycled (reclaimed) water for irrigation, mentioning the various sources of wastewater that are subject to collection, treatment and recycling. Depending on its source, wastewater can contain a range of chemicals, including surfactants, hydrocarbons, oils, industrial chemicals and highly toxic substances. Effluent and other wastewaters can contain a mixture of pharmaceuticals, including hormones, and other endocrine disruptors that may lead to reproductive problems in humans, animals and fish. Disease agents such as viruses, parasites and bacteria may also be transported in recycled water, detrimentally affecting animals, plants and humans. Recently, treated wastewater has been found to contain methicillin-resistant Staphylococcus aureus organisms that pose public health concerns (Goldstein et al. 2012). Treatments such as filtration, UV radiation or chlorination should guard against the spread of plant diseases in recycled drainage water although this area needs further study.
Use of recycled water for crop irrigation must be regulated and controlled, meeting the recommended microbiological and chemical quality guidelines to ensure the safety of farmers and consumers (Pescod 1992). Wastewater treatment facilities and protocols that are both low cost and low maintenance are required for wider adaptation. Recycled water must be completely separate from drinking water, and worldwide standards have been established whereby pipes and fittings for recycled water are colored lilac or carry a lilac stripe to avoid misuse or contamination (Anonymous 2009).
Chemical and physical characteristics of irrigation water have been discussed by Ayers and Westcot (1985) including descriptions of terminology, units, and key parameters. Examples of the use of saline water in the US, India, Tunisia, Egypt and Israel have been documented by Rhoades et al. (1992). Management practices have been discussed by Shalhevet (1994), Rhoades (1999), Sharma and Minhas (2005) and Qadir et al. (2010).
In many countries, the regulated safe use of recycled water has increased steadily. Vigneswaran and Sundaravadivel (2004) described the use of recycled water for various purposes including irrigation and the standards for irrigation of crops in different countries. In the US, Florida is now the largest producer of this water, which has an excellent safety record and has been used successfully for more than 40 years. Although reclaimed water in Florida was initially promoted to improve surface water quality, it has now become an important alternate source of water for irrigation. Citrus irrigated with high quality recycled water on well-drained Florida sands showed increased tree growth and fruit production compared to those irrigated with ground water. The higher ratings were attributed to consistently greater soil water content in the orchards irrigated with reclaimed water due to the increased irrigation frequency or irrigation quantities (Morgan et al. 2008).
The California Leafy Green Marketing Agreement (LGMA 2009) has provided a set of best practices developed by the growers and accepted by the state to assure high quality and safety of recycled water, allowing reclaimed water to become a major source of irrigation for food crops (Parsons et al. 2010). In Israel, the largest wastewater treatment plant (Shafdan) is located in the heavily populated area along the Mediterranean coast. The plant treats 130 million cubic meters of wastewater annually. The recycled water is transferred to the arid regions of the country for agricultural use through a series of separate (purple colored) pipes 90 km in length. The quality of the reclaimed water is high, making it suitable for irrigation of orange, carrot, potato, lettuce, wheat and flowers (WATEC 2007). Calculations by the Center for Strategic and International Studies (CSIS) show that, at present, wastewater treatment has proven a much cheaper process and thus suitable for use in agriculture compared to desalinating water (CSIS 2011).
Greywater
Greywater (untreated household effluent from baths, showers, kitchen and hand-wash basins, laundry, i.e., all non-toilet sources) has been used for irrigation by small-scale farmers exercising peri-urban agriculture near urban markets (Rodda et al. 2011). Swiss chard and carrot exhibited faster growth, increased yield and greater plant nutrient content when irrigated with greywater compared to crops irrigated with tap water. Soil irrigated with greywater showed increased electrical conductivity and increased concentrations of metals over time, coupled with an increase in sodium and metal concentrations in crops.
The Salinity Challenge
Many water logging and salinity problems occurring in irrigated agriculture throughout the world have resulted from the use of fresh irrigation water resources (Table 1). The Aral Sea environmental disaster where water diversion for irrigation purposes resulted in shrinkage of the sea to two-fifths of its original size, with its water quality ruined by salt accumulation and pesticide residue, demonstrates that management of water resources requires a coordinated action on national and regional levels (Micklin 2007). This includes agricultural and rural planning, enhanced water harvesting and storage systems, water recycling, water desalinization and water transport systems (UN Water 2006).
Many water scarce areas have aquifers of marginal quality, such as those that contain saline and/or alkali (sodic) waters. Saline water contains excess levels of salts, while sodic water contains elevated levels of Na+ compared to other cations. Marginal-quality groundwater resources without appropriate soil, crop and irrigation management strategies pose considerable risks, in terms of the development of salinity, sodicity, ion-specific toxicity, and nutrient imbalances in soil (Sharma and Minhas 2005).
Direct contact of saline water with leaf tissue may cause severe damage to many crops while their roots are much less sensitive. Hence, drip or trickle irrigation is employed for their production. Under drip irrigation, the salt content is usually lowest in the soil immediately below, adjacent to the emitters and greatest in the periphery of the wet zone. Removal of salt that has accumulated in this wet zone “front” must be addressed in the long-term. Therefore, irrigation with saline water requires leaching, i.e., irrigating with excessive water to remove salt from the root zone to deeper soil (Grattan 2002). The need to use excessive amounts of water and especially the long-term risk to the soil are not sustainable. Rapidly developing desalination technologies along with decreasing costs offer a sustainable solution to areas where saline water are the only available resource for irrigation.
Solar Energy Based Desalination
The alternative water resources used for irrigation, agricultural drainage and recycled water, contain elevated salt concentrations. Appropriate irrigation management and growing salinity tolerant crops provide opportunities for a certain period, but the continuous use of such water resources poses agriculture and environmental risks due to soil and water quality degradation. The long-term solution requires the removal of salts. Indeed, the agricultural use of desalinated water is practiced in Spain (Downward and Taylor 2007), the United Arab Emirates (Sanz et al. 2007), and Israel (Ghermandi and Messalem 2009). In Israel, desalinated water is supplied via the fresh water system, mixed with fresh water and used for irrigation. However, the high energy requirement of conventional technologies accounts for 40–45 % of the total costs of desalination (Betts 2004). Cost effective wastewater and saline water treatment suited for irrigation are fundamental.
Attempts to harness solar energy for the benefit of farmers are ongoing. For example, a solar-powered water supply and irrigation system were installed in a scarce and variable rainfall area near Lake Victoria in Tanzania (Knutson and Chen 2010). Burney et al. (2010) demonstrated the feasibility of using solar powered drip irrigation in the rural Sudano–Sahel to enhance food security. The rapid development of this technology in recent years may become useful especially in arid regions where radiation is ample (Ghermandi and Messalem 2009). These authors designed a hybrid solar-powered nanofiltration (NF) desalination plant that resulted in 40 % savings in operation costs compared to conventional membrane filtration.
An impressive desalination operation was constructed in Saudi Arabia where agriculture has depended on nonrenewable fossil water. According to estimates, about 70 % of ancient fossil aquifers in Saudi Arabia have been depleted in recent decades (Lee 2010). To secure water and food security, Saudi Arabia has embraced large-scale, oil-powered desalination. Desalinated water is transported to cities 400 km inland and accounts for 70 % of the need (CSIS 2011). However, prices for desalinated water have been trending upward in recent years. Hence, Saudi Arabia is investing in solar-powered desalination plants hoping to reduce water and energy costs by 40 %. In 2010, 24 million cubic meters per day of solar-powered desalinated water were being produced (Lee 2010). Solar-powered water desalination technology is both environmentally friendly and sustainable. As this green technology improves (Chung et al. 2012), the expense is expected to decrease, offering an elegant solution to alleviating water distress in agriculture.
Potato Response to Salinity
Presently, potato and other crops are irrigated in many parts of the world with water of various salinity levels. Salinity has adverse effects on growth and productivity of many crop plants (Maas and Hoffman 1977). Use of saline water for irrigation results in greater osmotic potential of the soil solution, restricting water uptake by the roots, and it has an adverse effect on soil structure, reducing water infiltration rate, soil aeration and oxygen supply to the roots (Ayers and Westcot 1985). Elevated concentrations of some ions (Na, Mg, Ca, Cl, HCO3, SO4 and B) in the soil solution have an adverse effect on plant stand; high concentrations of specific ions may cause physiological disorders in plant tissue.
Plants exposed to salinity commonly respond by reduced growth, stunting and reduced dry matter yield. Feigin (1988) stated that the decline in growth and yield reported for most crop plants was due mainly to the influence of the osmotic potential of the nutrient/salt solutions, and that standard fertilization management was usually satisfactory for crops irrigated with saline water. Saline water increases the proportion of exchangeable sodium ions in the soil solution, leading to formation of sodium carbonate, which raises the pH. These alkaline conditions reduce availability of nutrients, such as phosphate, iron, zinc and manganese, to the plants. In soils rich with calcium carbonate, this damaging process is inhibited. It is also well accepted that standard fertilization recommendations for nonsaline conditions may also be followed under most saline conditions and can reduce metabolic disturbance (Feigin 1985; Kafkafi 1994). Elkhatib et al. (2004b) demonstrated that potassium fertilizer up to 600 kg ha−1 increased the yield of four potato cultivars irrigated with saline water.
Potato is considered moderately sensitive to soil salinity (Bernstein et al. 1951; Maas and Hoffman 1977). The adverse effects of salinity stress on the potato plant have been described as follows (Levy and Veilleux 2007):
-
reduced growth of stems (stunting), leaves and tubers
-
leaf chlorosis (yellowing), tip burn and leaf burn
-
restricted water uptake by roots
-
enhanced plant senescence
-
reduced tuber yield
-
browning and cracking of tuber surface.
Potato leaves are severely damaged by overhead irrigation with saline water (Maas 1986; Meiri and Plaut 1985). Uptake of chlorine and sodium by leaves may induce toxicity, exhibited as leaf burn along the margins. Potato leaves are most sensitive to salt applied at the beginning of tuber formation (Bruns and Caesar 1990). Fidalgo et al. (2004) reported that salt stress negatively affected relative water content, leaf stomatal conductance and transpiration rate of cv. Desiree. At the ultrastructural level, thylakoid swelling and decreased grana stacking have been observed (Bruns and Hechtbuchholz 1990; Fidalgo et al. 2004). Changes to the chloroplast structure presumably affect photosynthesis, resulting in increased starch in leaves, suppression of nitrate reductase activity and reduced growth and dry matter in tubers (Ghosh et al. 2001). Hence, overhead irrigation of potato and other susceptible crops with saline water should be avoided (Maas 1986; van der Zaag 1991). Furrow and trickle irrigation result in considerably less damage (Meiri and Plaut 1985). However, roots of potato absorb chlorine, sodium and boron. Elevated levels of boron, greater than 1 to 2 mEq L−1 in the irrigation water, can be toxic. The symptoms in potato are yellowing of older leaves, drying leaf tissue at the tips and margins, and interveinal chlorosis (Ayers and Westcot 1985).
Breeding for Tolerance to Drought and Salinity
Varietal differences in field tolerance to drought have been observed in potato (van Loon 1981). Major criteria used for the selection of promising clones by breeders are yield potential, tuber qualities and resistance to pests. Tolerance to abiotic stress has been a rare objective used in traditional breeding programs. Tolerance to drought stress seems to involve many complex relationships (Vasquez-Robinet et al. 2008). An adapted genotype is expected to have a combination of genes for adaptation and tolerance to drought, resulting in superior performance in the field. Hassanpanah (2010) evaluated six cultivars under three different drought stress treatments and reported that cvs. Caesear and Kennebec were more tolerant to drought than the others.
A drought tolerance index was developed by Sharma et al. (2011) to evaluate the relative tolerance of potato cultivars to drought stress. Several characteristics, including plant morphology and size, root morphology, depth and size, stomatal behavior, osmotic adjustment and other metabolic changes are involved in adaptation and tolerance to drought (Valliyodan and Nguyen 2006). Several of these attributes may appear at first to attribute to drought tolerance but when their effect is considered over the whole growing season, the impact may be detrimental instead of advantageous (Spitters and Schapendonk 1990). For instance, early stomatal closure in response to drought may save water for later growth, but at the expense of actual growth.
Richards (2006) outlined traits that have contributed to productive breeding in a range of crops for drought-prone regions. They included: matching and optimizing crop phenology with existing rainfall patterns and hence avoiding stress; slowing water use by increasing root hydraulic resistance through reduced xylem vessel diameter; selection for low carbon isotope discrimination that correlates with high transpiration efficiency; increased osmoregulation; and, development of leaves resistant to drought-induced senescence. He noted the difficulties associated with assessing root traits although, in crops such as potato, root mass and rooting depth may be useful for selecting drought tolerant germplasm (Cattivelli et al. 2008).
Selection for drought tolerance under field conditions, a costly and laborious procedure, is further complicated by the fact that drought stress varies with duration and severity of the stress, which may differ from field to field and from year to year. White et al. (2012) defined key criteria, experimental approaches, equipment and data analysis tools required for robust, high-throughput field-based phenotyping (FBP). This system focused on simultaneous proximal sensing for spectral reflectance, canopy temperature, and plant architecture where a vehicle carrying replicated sets of sensors recorded data on multiple plots, with the potential to record data throughout the crop life cycle. The potential to assess traits, such as adaptation to water deficits or acute heat stress, several times during a single diurnal cycle is especially valuable for quantifying stress recovery.
In potato, breeding for drought tolerance is hindered by “low heritability, polygenic control, and epistasis of many drought tolerance traits” (Schafleitner 2009). Recently however, high heritability has been found in diploid potato for some drought response traits by Anithakumari et al. (2011). In view of the biodiversity of Solanum species (Hawkes 1990; Hijmans and Spooner 2001), the use of wild potato germplasm should facilitate the acquisition of useful traits such as drought and heat resistance (Coleman 2008; Ekanayake and de Jong 1992; Rizhsky et al. 2004; Spooner and Salas 2006). Certainly, the use of “crop wild relatives” has had a long and successful history of improving crop performance (Hajjar and Hodgkin 2007; Reynolds et al. 2007; Cabello et al. 2012). The use of marker-assisted selection (MAS) based on marker-trait associations or quantitative trait loci (QTLs) should improve breeding for drought resistance (Araus et al. 2008; Habash et al. 2009, 2010; Harris et al. 2007; Ribaut and Ragot 2007). As Cattivelli et al. (2008) noted, “QTLs for drought-related traits coincident with QTLs for yield potential should be considered as priority targets for MAS.” The availability of the potato genome should also benefit future breeding efforts (The Potato Genome Sequencing Consortium 2011).
The differential response of potato cultivars and of wild potato species to salinity stress and procedures for the selection for tolerance to salinity have been described earlier (Levy and Veilleux 2007; Levy and Tai 2013). However, mechanisms associated with tolerance to salinity are attracting much attention. Munns and Tester (2008) described three distinct mechanisms of plant adaptation to salinity: osmotic stress tolerance, Na+or Cl− exclusion, and the tolerance of tissue to accumulated Na+ or Cl−. The authors summarized the various tolerance mechanisms and their effect on plant and tissue processes, e.g., shoot growth, stomatal aperture, photosynthesis, osmotic adjustment and the accumulation of compatible solutes. They also listed a few candidate genes and suggested that molecular genetics and functional genomics provide a new opportunity to use molecular and physiological knowledge to improve the salinity tolerance of plants relevant to food production and environmental sustainability (Munns and Tester 2008). Recently, the contribution of Na+ exclusion trait found within ancestral wheat germplasm to salt tolerance of bread wheat has been reported (Munns et al. 2012).
Microarrays have provided a means to assess global changes in gene expression in response to stress, allowing the discovery of down- and up-regulated genes at particular times after imposition of the stress. Although various methodologies have been used to subject plants to drought or salt stress, some common trends have been observed. Evers et al. (2010) sampled leaves for RNA extraction of two potato cultivars (Sullu and SS2613, both S. tuberosum Group Andigenum) differing in drought tolerance 28 and 49 days after the imposition of drought compared to continuously irrigated controls in field grown plants. Both photosynthesis and carbohydrate related genes were strongly down-regulated in both cultivars, although the onset of repression differed between them; there was greater accumulation of osmotically active solutes in the drought tolerant cultivar.
In a subsequent study on growth chamber grown plants, Evers et al. (2012) found that salt stress likewise affected genes and proteins involved in photosynthesis and carbohydrate metabolism using both transcriptomic and proteomic analyses; some common stress response genes, including S-adenosylmethionine decarboxylase and δ-1-pyrroline-5-carboxylate synthetase, were up-regulated. Watkinson et al. (2006) also used microarrays to determine differential gene expression of potato accessions with different tolerance to drought. A proteomic analysis of the salt sensitive cv. Concord compared to the salt tolerant cv. Kennebec grown under in vitro salt stress conditions revealed that genes related to photosynthesis and protein synthesis were drastically down-regulated. However, osmotin-like proteins, heat shock proteins and calreticulin were up-regulated in the tolerant cultivar under salt stress, suggesting that up-regulation of defense-related proteins may confer salt tolerance (Aghaei et al. 2008).
The role of microRNAs as post-transcriptional regulators of gene expression has been recently revealed. In potato cv. Choobaek exposed to polyethylene glycol treatment in hydroponic culture, several families of microRNAs were associated with the stress response (Hwang et al. 2011a, b, c). Transcriptome analysis of potato cv. Kennebec exposed to various stress treatments including salt and drought has provided an array of candidate genes for investigation of abiotic stress response (Rensink et al. 2005a, b) and the possibility of introducing these genes transgenically into potato cultivars.
Transgenics
Introduction of single genes with the hope of delivering drought tolerance to potato through transgenic research has been attempted with a range of genes from various organisms. Kondrák et al. (2011) introduced the yeast trehalose-6-phosphate synthase 1 gene into potato cv. White Lady and observed improved drought tolerance in conjunction with a host of undesirable pleiotropic effects under non-stress conditions; microarray analysis revealed differential expression of 99 genes, 46 of which could be assigned into 16 functional groups. Kim et al. (2011) cloned a peroxiredoxin gene (2-Cys Prx) from Arabidopsis and used two different promoters, constitutive P35S and the stress inducible PSWPA2, from sweet potato, to drive expression of this antioxidant in potato cv. Atlantic. They found that the transgenic Atlantic were more tolerant of heat shock (42 °C for 10 h) and recovered more quickly than untransformed controls and that the stress inducible promoter resulted in greater expression of the transgene under methylviologen (paraquat herbicide that induces reactive oxygen species, mimicking drought stress) than P35S. No adverse pleiotropic effects were noted.
A transcription factor, StMYB1R-1, which had been identified as a putative stress response gene in potato (up-regulated by cold, drought and salt stress), was placed under the control of P35S and transformed into potato cv. Superior (Shin et al. 2011). Three selected transgenics and a control were subjected to drought stress (water was withheld for 15 days in a greenhouse trial); the transgenics were significantly decreased in their water loss and exhibited greater survival after the drought treatment than wild type controls. The biomass, tuber shape and size of the transgenics were not adversely affected.
A dehydroascorbate reductase gene (AtDHAR1) from Arabidopsis was transformed into cv. Atlantic resulting in plants with greater physiological indications (less ion leakage under drought, greater chlorophyll content, greater ascorbic acid than wild-type) of drought tolerance (Eltayeb et al. 2011). Likewise, a betaine aldehyde dehydrogenase (BADH) gene from spinach, selected for its production of the osmolyte, glycine betaine, was overexpressed in potato cv. Gannongshu 2, resulting in plants with slightly greater height and fresh weight under NaCl and polyethylene glycol to mimic drought stress on plants grown in vermiculite in the greenhouse (Zhang et al. 2011).
The influence of overexpression of a D-galacturonic acid reductase gene from strawberry was examined on the in vitro tuberization response of transgenic potato cv. Taedong Valley (Hemavathi et al. 2011) after demonstrating improved stress tolerance of the transgenics to methyl viologen, sodium chloride and mannitol in detached leaf disc assays (Hemavathi et al. 2009). Stress conditions were imposed by addition of methyl viologen, sodium chloride or zinc chloride to the microtuberization medium containing 9 % sucrose. Microtuberization and various enzyme activities were increased in the transgenics compared to nontransgenic controls, implying that the increased antioxidants of the trangenics improved their performance. Using a similar experimental system except that the gene selected for overexpression was a rat L-gulono-γ-lactone oxidase, Hemavathi et al. (2010) reported improved stress tolerance of the transgenics.
Transgenic potato overexpressing the Arabidopsis thaliana glutathione reductase gene (AtGR1) exhibited enhanced tolerance to methylviologen and recovered more rapidly after drought stress than nontransgenic controls (Eltayeb et al. 2010). Waterer et al. (2010) deployed an array of transgenes, including wheat mitochondrial Mn superoxide dismutase (SOD3:1), barley dehydrin 4 (DHN 4), a canola transcriptional factor (DREB/CBF1) or a bromegrass stress inducible ROB5 gene into potato cv. Desiree, placing the transgenes under the control of either constitutive P35S promoter or a stress inducible Arabidopsis COR78 promoter. Under drought stress in greenhouse trials, the promoter/transgene combinations, COR78:DHN4 and COR78:ROB5 were the most promising to enhance tuber yield under drought stress imposed at tuber initiation.
Stacking antioxidant genes may provide even greater protection against multiple stress conditions, including drought, than deploying single genes, as demonstrated by Ahmad et al. (2010) who used a combination of choline oxidase (Ahmad et al. 2008a), superoxide dismutase and ascorbate peroxidase to transform potato cv. Superior. After 2 weeks of withholding water followed by re-watering of the multigene transgenics, the plants recovered completely when control plants died under the same conditions. Antioxidant enzyme activity was greater for the transgenics under stress conditions.
Given the improved tolerance of potato transformed with any number of genes identified in stress tolerance of various organisms, it seems likely that a new array of drought tolerant potato will be available as GMOs in the coming years. Marker-free transgenic potatoes with improved drought stress due to chloroplast-targeted superoxide dismutase (SOD) and ascorbate peroxidase (APX) genes have already been developed in anticipation of objection to genetically modified plants (Ahmad et al. 2008b).
Improvement of salt stress tolerance of potato has also been the target of considerable transgenic research in recent years. Bayat et al. (2010) transformed two potato cultivars with a barley antiporter gene HvNHX2 and found that in vitro plantlets of five of six transgenic lines of one cultivar could survive on medium supplemented with 200 mM NaCl. Overexpression of the GalUR gene, an ascorbic acid pathway enzyme, provided potato cv. Taedong Valley with enhanced tolerance of salt stress measured by growth and microtuberization of in vitro plantlets (Upadhyaya et al. 2011).
The transcription factor, AtDREB1A, from Arabidopsis has been placed under control of a stress-inducible promoter to generate salt and drought resistant potato transgenics that displayed greater abundance of stress-induced genes (Watanabe et al. 2011). These initial studies of transgenics using stress response genes identified through the application of transcriptomic and proteomic tools in model plants as well as in potato suggest a new wave of commercial transgenic potatoes with wide environmental adaptation in the near future.
Conclusions and Prospects
A comprehensive public management strategy is necessary in the face of the projected decrease of fresh water resources. Improvements of irrigation management systems provide the farmer with tools for the increased efficiency of water use. To conserve water, farmers need to implement efficient irrigation systems such as micro-irrigation and drip irrigation. Soil moisture sensors may prove useful to monitor and control irrigation schedules and quantities. The potato could probably benefit from a low-volume/high frequency (LVHF) soil moisture-based drip irrigation system (Muñoz-Carpena et al. 2005), and the low pressure/low flow drip irrigation system (Dowgert et al. 2006) also deserves additional studies.
Precision irrigation is expected to be capable of sensing differences in water requirements among cultivars with differential tolerance to drought and to marginal water quality. It is expected to be adapted to manage any irrigation regime chosen by the farmer and provide variable water amounts to plants affected by uneven field conditions, such as topography, microclimate, soil gradients, etc. and respond in real time to the crop’s water requirements at different growth stages. This approach is meant to apply accurate water quantities in accordance with spatial and temporal soil properties, and avoid drought stress while conserving water.
Wastewater of various sources, mostly urban recycled water, will probably become a major source of water available for irrigation. Tertiary treatment, a process that consists of flocculation basins, clarifiers, filters, and chlorine basins or ozone or ultraviolet radiation, will be required to sustain soil structure and ensure the quality and safety of yields.
Various saline water sources are an important source of irrigation in many regions. However, this poses a serious risk to the soil and usually results in yield decrease. Desalination of wastewater and saline water is required to ensure sustainability. Improvements to desalination technologies and solar energy systems are expected to lower the costs of treatment and transport of these water resources, offering an affordable alternative to fresh water for farming.
Although the potato crop is an irrigated crop, it is often exposed to a midday transient drought stress during the sensitive period of tuber bulking. This phenomenon is common in semi-arid and arid climates. In addition, in many regions, saline water is the only available source for irrigation and potatoes are grown under various degrees of salinity stress. Hence, improved tolerance to drought and to salinity is of great importance to growers. Modern molecular tools offer considerable promise for breeding stress tolerant potato cultivars and will provide another approach to cope with water scarcity and climate challenge.
References
Ackerson, R.C., D.R. Krieg, T.D. Miller, and R.G. Stevens. 1977. Water relations and physiological activity of potatoes. Journal of the American Society for Horticultural Science 102: 572–575.
Aghaei, K., A.A. Ehsanpour, and S. Komatsu. 2008. Proteome analysis of potato under salt stress. Journal of Proteome Research 7: 4858–4868.
Ahmad, R., M.D. Kim, K.H. Back, H.S. Kim, H.S. Lee, S.Y. Kwon, N. Murata, W.I. Chung, and S.S. Kwak. 2008a. Stress-induced expression of choline oxidase in potato plant chloroplasts confers enhanced tolerance to oxidative, salt, and drought stresses. Plant Cell Reports 27: 687–698.
Ahmad, R., Y.H. Kim, M.D. Kim, M.N. Phung, W.I. Chung, H.S. Lee, S.S. Kwak, and S.Y. Kwon. 2008b. Development of selection marker-free transgenic potato plants with enhanced tolerance to oxidative stress. Journal of Plant Biology 51: 401–407.
Ahmad, R., Y.H. Kim, M.D. Kim, S.Y. Kwon, K. Cho, H.S. Lee, and S.S. Kwak. 2010. Simultaneous expression of choline oxidase, superoxide dismutase and ascorbate peroxidase in potato plant chloroplasts provides synergistically enhanced protection against various abiotic stresses. Physiologia Plantarum 138: 520–533.
Ahmadi, S.H., M.N. Anderson, F. Plauborg, R.T. Poulsen, C.R. Jensen, A.R. Sepakhah, and S. Hansen. 2010a. Effect of irrigation strategies and soils on field grown potatoes: Gas exchange and xylem ABA. Agricultural Water Management 97: 1486–1494.
Ahmadi, S.H., M.N. Anderson, F. Plauborg, R.T. Poulsen, C.R. Jensen, A.R. Sepakhah, and S. Hansen. 2010b. Effect of irrigation strategies and soils on field grown potatoes: Yield and water productivity. Agricultural Water Management 97: 1923–1930.
Ahn, Y.J., K. Claussen, and J.L. Zimmerman. 2004. Genotypic differences in the heat-shock response and thermotolerance in four potato cultivars. Plant Science 166: 901–911.
Alchanatis, V., Y. Cohen, S. Cohen, M. Moller, M. Sprinstin, M. Meron, J. Tsipris, Y. Saranga, and E. Sela. 2010. Evaluation of different approaches for estimating and mapping crop water status in cotton with thermal imaging. Precision Agriculture 11: 27–41.
Al-Kufaishi, S.A., B.S. Blackmore, and H. Sourell. 2006. The feasibility of using variable rate water application under a central pivot irrigation system. Irrigation and Drainage Systems 20: 317–327.
Alva, A.K. 2008. Setpoints for potato irrigation in sandy soils using real-time, continuous monitoring of soil-water content in soil profile. Journal of Crop Improvement 21: 117–137.
Anithakumari, A.M., O. Dolstra, B. Vosman, R.G.F. Visser, and C.G. van der Linden. 2011. In vitro screening and QTL analysis for drought tolerance in diploid potato. Euphytica 181: 357–369.
Anonymous. 2006. Coping with water scarcity: A strategic issue and priority for system-wide action. http://www.unwater.org, Accessed April 30, 2011
Anonymous. 2009. Using recycled water for irrigation. National Program for Sustainable Irrigation Land & Water Australia. http://lwa.gov.au/files/products/national-program-sustainable-irrigation/pn30123/pn30123.pdf, Accessed May 2, 2012
Araus, J.L., G.A. Slafer, C. Royo, and M.D. Serret. 2008. Breeding for yield potential and stress adaptation in cereals. Critical Reviews in Plant Sciences 27: 377–412.
Ayers, R.S., and D.W. Westcot. 1985. Water quality for agriculture. FAO Irrigation and Drainage Paper (FAO) No 29 (rev 1), p 186 pp.
Basu, P.S., A. Sharma, I.D. Garg, and N.P. Sukumaran. 1999. Tuber sink modifies photosynthetic response in potato under water stress. Environmental and Experimental Botany 42: 25–39.
Bates, B.C., Z.W. Kundzewicz, S. Wu, and J.P. Palutikof (eds) (2008) Climate Change and Water. Technical Paper of the Intergovernmental Panel on Climate Change. IPCC Secretariat, Geneva, 210 pp.
Bayat, F., B. Shiran, D.V. Belyaev, N.O. Yur’eva, G.I. Sobol’kova, H. Alizadeh, M. Khodambashi, and A.V. Babakov. 2010. Potato plants bearing a vacuolar Na+/H+ antiporter HvNHX2 from barley are characterized by improved salt tolerance. Russian Journal of Plant Physiology 57: 696–706.
Bengough, A.G., B.M. McKenzie, P.D. Hallett, and T.A. Valentine. 2011. Root elongation, water stress, and mechanical impedance: A review of limiting stresses and beneficial root tip traits. Journal of Experimental Botany 62: 59–68.
Bernstein, L., A.D. Ayers, and C.H. Wadleigh. 1951. The salt tolerance of White Rose potatoes. Proceedings American Society for Horticultural Science 57: 231–236.
Bethke, P.C., R. Sabba, and A.J. Bussan. 2009. Tuber water and pressure potentials decrease and sucrose contents increase in response to moderate drought and heat stress. American Journal of Potato Research 86: 519–532.
Betts, K. 2004. Desalination, desalination everywhere. Environmental Science & Technology 38: 246A–247A.
Bishop, J.C., and D.W. Grimes. 1978. Precision tillage effects on potato root and tuber production. American Potato Journal 55: 65–71.
Blum, A. 2009. Effective use of water (EUW) and not water-use efficiency (WUE) is the target of crop yield improvement under drought stress. Field Crops Research 112: 119–123.
Blum, A. 2011. Drought resistance—is it really a complex trait? Functional Plant Biology 38: 753–757.
Boyer, J.S., S.C. Wong, and G.D. Farquhar. 1997. CO2 and water vapor exchange across leaf cuticle (epidermis) at various water potentials. Plant Physiology 114: 185–191.
Brodribb, T.J., T.S. Feild, and G.J. Jordan. 2007. Leaf maximum photosynthetic rate and venation are linked by hydraulics. Plant Physiology 144: 1890–1898.
Brodribb, T.J., T.S. Feild, and L. Sack. 2010. Viewing leaf structure and evolution from a hydraulic perspective. Functional Plant Biology 37: 488–498.
Bruns, S., and K. Caesar. 1990. Shoot development and tuber yield of several potato cultivars under high salt concentrations at different stages of development. Potato Research 33: 23–32.
Bruns, S., and C. Hechtbuchholz. 1990. Light and electron microscope studies on the leaves of several potato cultivars after application of salt at various developmental stages. Potato Research 33: 33–41.
Burney, J., L. Woltering, M. Burke, R. Naylor, and D. Pasternak. 2010. Solar-powered drip irrigation enhances food security in the Sudano-Sahel. Proceedings of the National Academy of Sciences of the United States of America 107: 1848–1853.
Burton, W.G. 1981. Challenges for stress physiology in potato. American Potato Journal 58: 3–14.
Cabello, R., F. De Mendiburu, M. Bonierbal, P. Monneveux, W. Roca, and E. Chujoy. 2012. Large scale evaluation of potato improved varieties, genetic stocks and landraces for drought tolerance. American Journal of Potato Research 89: 400–410.
Campbell, M.D., G.S. Campbell, R. Kunkel, and R.I. Papendick. 1976. A model describing soil–plant–water relations for potatoes. American Potato Journal 53: 431–441.
Cassman, K.G. 1999. Ecological intensification of cereal production systems: Yield potential, soil quality, and precision agriculture. Proceedings of the National Academy of Sciences of the United States of America 96: 5952–5959.
Cattivelli, L., F. Rizza, F.W. Badeck, E. Mazzucotelli, A.M. Mastrangelo, E. Francia, C. Maré, A. Tondelli, and A.M. Stanca. 2008. Drought tolerance improvement in crop plants: An integrated view from breeding to genomics. Field Crops Research 105: 1–14.
Cellier, F., G. Conéjéro, J.C. Breitler, and F. Casse. 1998. Molecular and physiological responses to water deficit in drought-tolerant and drought-sensitive lines of sunflower: Accumulation of dehydrin transcripts correlates with tolerance. Plant Physiology 116: 319–328.
Cellier, F., Conéjére, and F. Casse. 2000. Dehydrin transcript fluctuations during a day/night cycle in drought-stressed sunflower. Journal of Experimental Botany 51: 299–304.
Chaves, M.M., and M.M. Oliveira. 2004. Mechanisms underlying plant resilience to water deficits: Prospects for water-saving agriculture. Journal of Experimental Botany 55: 2365–2384.
Chung, I., B. Lee, J. He, R.P.H. Chang, and M.G. Kanatzidis. 2012. All-solid-state dye-sensitized solar cells with high efficiency. Nature 485: 486–490.
Clark, L.J., W.R. Whalley, and P.B. Barraclough. 2003. How do roots penetrate strong soil? Plant and Soil 255: 93–104.
Cohen, Y., V. Alchanatis, M. Meron, Y. Saranga, and J. Tsipris. 2005. Estimation of leaf water potential by thermal imagery and spatial analysis. Journal of Experimental Botany 56: 1843–1852.
Cohen, Y., V. Alchanatis, Y. Zusman, Z. Dar, D.J. Bonfil, A. Karnieli, A. Zilberman, A. Moulin, V. Ostrovsky, A. Levi, R. Brikman, and M. Shenker. 2010. Leaf nitrogen estimation in potato based on spectral data and on simulated bands of the VEN mu S satellite. Precision Agriculture 11: 520–537.
Coleman, W.K. 1986. Water relations of the potato (Solanum tuberosum L.) cultivars Raritan and Shepody. American Potato Journal 63: 263–276.
Coleman, W.K. 1988. Tuber age as a contributory factor in the water relations of potato (Solanum tuberosum L.). American Potato Journal 65: 109–118.
Coleman, W.K. 2008. Evaluation of wild Solanum species for drought resistance. 1. Solanum gandarillasii Cardenas. Environmental and Experimental Botany 62: 221–230.
CSIS. 2011. Water and national strength in Saudi Arabia. Center for Strategic and International Studies: Middle East Program. http://csis.org/files/publication/110405_Water%20and%20national%20strength%20in%20Saudi%20Arabia.pdf, Accessed May 23, 2012
Cutter, E.G. 1978. Structure and development of the potato plant. In The Potato Crop, ed. P.M. Harris, 70–152. London: Chapman & Hall.
Dammer, K.H., and D. Ehlert. 2006. Variable-rate fungicide spraying in cereals using a plant cover sensor. Precision Agriculture 7: 137–148.
Darwish, T.M., T.W. Atallah, S. Hajhasan, and A. Haidar. 2006. Nitrogen and water use efficiency of fertigated processing potato. Agricultural Water Management 85: 95–104.
Deblonde, P.M.K., A.J. Haverkort, and J.F. Ledent. 1999. Responses of early and late potato cultivars to moderate drought conditions: Agronomic parameters and carbon isotope discrimination. European Journal of Agronomy 11: 91–105.
Dowgert, M., B. Marsh, R. Hutmacher, T.L. Thompson, D. Hannford, J. Phene, J. Anshutz, and C. Phene. 2006. Low pressure drip irrigation lessens agricultural inputs. irrigationtoolbox.com/ReferenceDocuments/…/IA/2006/012.pdf (Accessed 6 Jan 2013).
Downward, S.R., and R. Taylor. 2007. An assessment of Spain’s Programa AGUA and its implications for sustainable water management in the province of Almería, southeast Spain. Journal of Environmental Management 82: 277–289.
Ekanayake, I.J., and J.P. de Jong. 1992. Stomatal response of some cultivated and wild-bearing potatoes in warm tropics as influenced by water deficits. Annals of Botany 70: 53–60.
Eldredge, E.P., Z.A. Holmes, A.R. Mosley, C.C. Shock, and T.D. Stieber. 1996. Effects of transitory water stress on potato tuber stem-end reducing sugar and fry color. American Potato Journal 73: 517–530.
Elkhatib, H.A., E.A. Elkhatib, A.M.K. Allah, and A.M. El-Sharkawy. 2004. Yield response of salt-stressed potato to potassium fertilization: A preliminary mathematical model. Journal of Plant Nutrition 27: 111–122.
Eltayeb, A.E., S. Yamamoto, M.E.E. Habora, Y. Matsukubo, M. Aono, H. Tsujimoto, and K. Tanaka. 2010. Greater protection against oxidative damages imposed by various environmental stresses in transgenic potato with higher level of reduced glutathione. Breeding Science 60: 101–109.
Eltayeb, A.E., S. Yamamoto, M.E.E. Habora, L.N. Yin, H. Tsujimoto, and K. Tanaka. 2011. Transgenic potato overexpressing Arabidopsis cytosolic AtDHAR1 showed higher tolerance to herbicide, drought and salt stresses. Breeding Science 61: 3–10.
Evers, D., I. Lefèvre, S. Legay, D. Lamoureux, J.F. Hausman, R.O.G. Rosales, L.R.T. Marca, L. Hoffmann, M. Bonierbale, and R. Schafleitner. 2010. Identification of drought-responsive compounds in potato through a combined transcriptomic and targeted metabolite approach. Journal of Experimental Botany 61: 2327–2343.
Evers, D., S. Legay, D. Lamoureux, J.F. Hausman, L. Hoffmann, and J. Renaut. 2012. Towards a synthetic view of potato cold and salt stress response by transcriptomic and proteomic analyses. Plant Molecular Biology 78: 503–514.
Fabeiro, C., F.M.D. Olalla, and J.A. de Juan. 2001. Yield and size of deficit irrigated potatoes. Agricultural Water Management 48: 255–266.
Feigin, A. 1985. Fertilization management of crops irrigated with saline water. Plant and Soil 89: 285–299.
Feigin, A. 1988. Fertilization for increasing salt tolerance of agricultural crops. Israel Agresearch 2: 99–138.
Fereres, E., and M.A. Soriano. 2007. Deficit irrigation for reducing agricultural water use. Journal of Experimental Botany 58: 147–159.
Fidalgo, F., A. Santos, I. Santos, and R. Salema. 2004. Effects of long-term salt stress on antioxidant defence systems, leaf water relations and chloroplast ultrastructure of potato plants. Annals of Applied Biology 145: 185–192.
Fleisher, D.H., D.J. Timlin, and V.R. Reddy. 2008a. Elevated carbon dioxide and water stress effects on potato canopy gas exchange, water use, and productivity. Agricultural and Forest Meteorology 148: 1109–1122.
Fleisher, D.H., D.J. Timlin, and V.R. Reddy. 2008b. Interactive effects of carbon dioxide and water stress on potato canopy growth and development. Agronomy Journal 100: 711–719.
Fleming, K.L., and D.G. Westfall. 2000. Evaluating management zone technology and grid soil sampling for variable rate nitrogen application. In Proceedings of the 5th International Conference on Precision Agriculture, ed. P.C. Robert, R.H. Rust, and W.E. Larson. Madison: ASA/CSSA/SSSA.
Foley, J.A., N. Ramankutty, K.A. Brauman, E.S. Cassidy, J.S. Gerber, M. Johnston, N.D. Mueller, C. O’Connell, D.K. Ray, P.C. West, C. Balzer, E.M. Bennett, S.R. Carpenter, J. Hill, C. Monfreda, S. Polasky, J. Rockström, J. Sheehan, S. Siebert, D. Tilman, and D.P.M. Zaks. 2011. Solutions for a cultivated planet. Nature 478: 337–342.
Franke, J., and G. Menz. 2007. Multi-temporal wheat disease detection by multi-spectral remote sensing. Precision Agriculture 8: 161–172.
Gandar, P.W., and C.B. Tanner. 1976. Leaf growth, tuber growth, and water potential in potatoes. Crop Science 16: 534–538.
Gawronska, H., M.K. Thornton, and R.B. Dwelle. 1992. Influence of heat stress on dry matter production and photoassimilate partitioning by four potato clones. American Potato Journal 69: 653–665.
Geerts, S., and D. Raes. 2009. Deficit irrigation as an on-farm strategy to maximize crop water productivity in dry areas. Agricultural Water Management 96: 1275–1284.
Ghassemi, F., A.J. Jakeman, and H.A. Nix. 1995. Salinization of land and water resources: Human causes, extent, management and case studies. Canberra: University of New South Wales Press, Ltd.
Ghermandi, A., and R. Messalem. 2009. The advantages of NF desalination of brackish water for sustainable irrigation: The case of the Arava Valley in Israel. Desalination and Water Treatment 10: 101–107.
Ghosh, S.C., K. Asanuma, A. Kusutani, and M. Toyota. 2001. Effect of salt stress on some chemical components and yield of potato. Soil Science and Plant Nutrition 47: 467–475.
Girma, F.S., and D.R. Krieg. 1992. Osmotic adjustment in Sorghum. I. Mechanisms of diurnal osmotic potential changes. Plant Physiology 99: 577–582.
Goldstein, R.E.R., S.A. Micallef, S.G. Gibbs, J.A. Davis, X. He, A. George, L.M. Kleinfelter, N.A. Schreiber, S. Mukherjee, A. Sapkota, S.W. Joseph, and A.R. Sapkota. 2012. Methicillin-resistant Staphylococcus aureus (MRSA) detected at four U.S. wastewater treatment plants. Environmental Health Perspectives 120: 1551–1558.
Gornall, J., R. Betts, E. Burke, R. Clark, J. Camp, K. Willett, and A. Wiltshire. 2010. Implications of climate change for agricultural productivity in the early twenty-first century. Philosophical Transactions of the Royal Society of London, Series B: Biological Sciences 365: 2973–2989.
Grattan, S.R. 2002. Irrigation water sailinity and crop production, University of California Division of Agriculture and Natural Resources. Oakland: Farm Water Quality Planning Publication 8066.
Gregory, P.J., and L.P. Simmonds. 1992. Water relations and growth of potatoes. In The potato crop: The scientific basis for improvement, ed. P.M. Harris, 214–246. London: Chapman and Hall.
Habash, D.Z., Z. Kehel, and M. Nachit. 2009. Genomic approaches for designing durum wheat ready for climate change with a focus on drought. Journal of Experimental Botany 60: 2805–2815.
Habash, D.Z., Z. Kehel, and M. Nachit. 2010. Genomic approaches for designing durum wheat ready for climate change with a focus on drought (vol 60, pg 2805, 2009). Journal of Experimental Botany 61: 1249–1249.
Hajjar, R., and T. Hodgkin. 2007. The use of wild relatives in crop improvement: A survey of developments over the last 20 years. Euphytica 156: 1–13.
Han, B., and A.R. Kermode. 1996. Dehydrin-like proteins in castor bean seeds and seedlings are differentially produced in response to ABA and water-deficit-related stresses. Journal of Experimental Botany 47: 933–939.
Hansen, J., M. Sato, R. Ruedy, K. Lo, D.W. Lea, and M. Medina-Elizade. 2006. Global temperature change. Proceedings of the National Academy of Sciences of the United States of America 103: 14288–14293.
Harris, P.M. 1992. The potato crop: The scientific basis for improvement. London: Chapman and Hall.
Harris, K., P.K. Subudhi, A. Borrell, D. Jordan, D. Rosenow, H. Nguyen, P. Klein, R. Klein, and J. Mullet. 2007. Sorghum stay-green QTL individually reduce post-flowering drought-induced leaf senescence. Journal of Experimental Botany 58: 327–338.
Hassanpanah, D. 2010. Evaluation of potato cultivars for resistance against water deficit stress under in vivo conditions. Potato Research 53: 383–392.
Haverkort, A.J., and D.K.L. MacKerron. 1995. Potato ecology and modelling of crops under conditions limiting growth. Proceedings of the 2nd International Potato Modeling Conference. 3, 396. Wageningen: Kluwer.
Hawkes, J.G. 1990. The potato: Evolution, biodiversity and genetic resources. Bellhaven Press, London and Smithsonian Institution Press, Washington, 259 pp.
Hemavathi, C.P., K.E. Upadhyaya, N. Young, H.S. Akula, J.J. Kim, O.M. Heung, C.R. Oh, S.C. Aswath, D.H.Kim Chun, and S.W. Park. 2009. Over-expression of strawberry D-galacturonic acid reductase in potato leads to accumulation of vitamin C with enhanced abiotic stress tolerance. Plant Science 177: 659–667.
Hemavathi, C.P., N. Upadhyaya, K.E. Akula, S.C. Young, D.H.Kim Chun, and S.W. Park. 2010. Enhanced ascorbic acid accumulation in transgenic potato confers tolerance to various abiotic stresses. Biotechnology Letters 32: 321–330.
Hemavathi, C.P., N. Upadhyaya, H.S. Akula, J.H. Kim, O.M. Jeon, S.C. Ho, D.H.Kim Chun, and S.W. Park. 2011. Biochemical analysis of enhanced tolerance in transgenic potato plants overexpressing D-galacturonic acid reductase gene in response to various abiotic stresses. Molecular Breeding 28: 105–115.
Heuer, B., and A. Nadler. 1998. Physiological response of potato plants to soil salinity and water deficit. Plant Science 137: 43–51.
Hijmans, R.J. 2003. The effect of climate change on global potato production. American Journal of Potato Research 80: 271–279.
Hijmans, R.J., and D.M. Spooner. 2001. Geographic distribution of wild potato species. American Journal of Botany 88: 2101–2112.
Howden, S.M., J.F. Soussana, F.N. Tubiello, N. Chhetri, M. Dunlop, and H. Meinke. 2007. Adapting agriculture to climate change. Proceedings of the National Academy of Sciences of the United States of America 104: 19691–19696.
Hwang, E.W., S.J. Shin, and H.B. Kwon. 2011a. Identification of microRNAs and their putative targets that respond to drought stress in Solanum tuberosum. Journal of the Korean Society for Applied Biological Chemistry 54: 317–324.
Hwang, E.W., S.J. Shin, S.C. Park, M.J. Jeong, and H.B. Kwon. 2011b. Identification of miR172 family members and their putative targets responding to drought stress in Solanum tuberosum. Genes & Genomics 33: 105–110.
Hwang, E.W., S.J. Shin, B.K. Yu, M.O. Byun, and H.B. Kwon. 2011c. miR171 family members are involved in drought response in Solanum tuberosum. Journal of Plant Biology 54: 43–48.
Idso, S.B., R.D. Jackson, P.J. Pinter, R.J. Reginato, and J.L. Hatfield. 1981. Normalizing the stress-degree-day parameter for environmental variability. Agricultural Meteorology 24: 45–55.
Iwama, K. 2008. Physiology of the potato: New insights into root system and repercussions for crop management. Potato Research 51: 333–353.
Iwama, K., and J. Yamaguchi. 2006. Chapter 7. Abiotic Stresses. In Handbook of potato production, improvement, and postharvest management, ed. J. Gopal and S.M.P. Khurana, 231–278. New York: The Haworth Press.
Jackson, R.D., S.B. Idso, R.J. Reginato, and P.J. Pinter. 1981. Canopy temperature as a crop stress indicator. Water Resources Research 17: 1133–1138.
Jaggard, K.W., A.M. Qi, and E.S. Ober. 2010. Possible changes to arable crop yields by 2050. Philosophical Transactions of the Royal Society of London, Series B: Biological Sciences 365: 2835–2851.
Jefferies, R.A. 1989. Water stress and leaf growth in field-grown crops of potato (Solanum tuberosum L.). Journal of Experimental Botany 40: 1375–1381.
Jefferies, R.A. 1993. Responses of potato genotypes to drought. I. Expansion of individual leaves and osmotic adjustment. Annals of Applied Biology 122: 93–104.
Jefferies, R.A., and D.K.L. MacKerron. 1993. Responses of potato genotypes to drought. II. Leaf area index, growth and yield. Annals of Applied Biology 122: 105–112.
Jones, H.G. 2007. Monitoring plant and soil water status: Established and novel methods revisited and their relevance to studies of drought tolerance. Journal of Experimental Botany 58: 119–130.
Jones, M.M., and N.C. Turner. 1978. Osmotic adjustment in leave of sorghum in response to water deficits. Plant Physiology 61: 122–126.
Jones, H.G., M. Stoll, T. Santos, C. de Sousa, M.M. Chaves, and O.M. Grant. 2002. Use of infrared thermography for monitoring stomatal closure in the field: Application to grapevine. Journal of Experimental Botany 53: 2249–2260.
Jordan, W.R., W.A. Dugas, and P.J. Shouse. 1983. Strategies for crop improvement for drought-prone regions. Agricultural Water Management 7: 281–299.
Kafkafi, U. 1994. Combined irrigation and fertilization in arid zones. Israel Journal of Plant Sciences 42: 301–320.
Khosla, R., D. Inman, D.G. Westfall, R.M. Reich, M. Frasier, M. Mzuku, B. Koch, and A. Hornung. 2008. A synthesis of multi-disciplinary research in precision agriculture: Site-specific management zones in the semi-arid western Great Plains of the USA. Precision Agriculture 9: 85–100.
Kim, M.D., Y.H. Kim, S.Y. Kwon, B.Y. Jang, S.Y. Lee, D.J. Yun, J.H. Cho, S.S. Kwak, and H.S. Lee. 2011. Overexpression of 2-cysteine peroxiredoxin enhances tolerance to methyl viologen-mediated oxidative stress and high temperature in potato plants. Plant Physiology and Biochemistry 49: 891–897.
Knutson, C.L., and S. Chen. 2010. UNDP Community Water Initiative: Fostering Water Security and Climate Change Adaptation and Mitigation. United Nations Development Programme (UNDP), GEF Small Grants Programme (GEF SGP), New York. http://www.undp.org/content/dam/undp/library/Environment%20and%20Energy/Local%20Development/CWI_Fostering_Water_Security_and_Adaptation.pdf, Accessed May 1, 2012
Kondrák, M., F. Marincs, B. Kalapos, Z. Juhász, and Z. Bánfalvi. 2011. Transcriptome analysis of potato leaves expressing the trehalose-6-phosphate synthase 1 gene of yeast. Plos ONE 6: e23466.
Kotb, T.H.S., T. Watanabe, Y. Ogino, and K.K. Tanji. 2000. Soil salinization in the Nile Delta and related policy issues in Egypt. Agricultural Water Management 43: 239–261.
Kratzke, M.G., and J.P. Palta. 1985. Evidence for the existence of functional roots on potato tubers and stolons: Significance in water transport to the tuber. American Potato Journal 62: 227–236.
Lafta, A.M., and J.H. Lorenzen. 1995. Effect of high temperature on plant growth and carbohydrate metabolism in potato. Plant Physiology 109: 637–643.
Lahlou, O., and J.F. Ledent. 2005. Root mass and depth, stolons and roots formed on stolons in four cultivars of potato under water stress. European Journal of Agronomy 22: 159–173.
Lamm, F.R., J.E. Ayars, and F.S. Nakayama. 2007. Microirrigation for crop production: Design, operation, and management. Amsterdam: Elsevier.
Lee, E. 2010. Saudi Arabia and desalinization. Harvard International Review. 32, http://hir.harvard.edu/pressing-change/saudi-arabia-and-desalination-0. Accessed May 1, 2012
Lesczynski, D.B., and C.B. Tanner. 1976. Seasonal variation of root distribution of irrigated, field-grown Russet Burbank potato. American Potato Journal 53: 69–78.
Levy, D. 1983a. Varietal differences in the response of potatoes to repeated short periods of water stress in hot climates. I. Turgor maintenance and stomatal behavior. Potato Research 26: 303–313.
Levy, D. 1983b. Water deficit enhancement of proline and alpha amino nitrogen accumulation in potato plants and its association with susceptibility to drought. Physiologia Plantarum 57: 169–173.
Levy, D. 1985. The response of potatoes to a single transient heat or drought stress imposed at different stages of tuber growth. Potato Research 28: 415–424.
Levy, D. 1986. Genotypic variation in the response of potatoes (Solanum tuberosum L.) to high ambient temperatures and water deficit. Field Crops Research 15: 85–96.
Levy, D. 1992. Osmotic potential of potatoes subjected to a single cycle of water deficit. Potato Research 35: 17–24.
Levy, D., and G.C.C. Tai. 2013. Differential response of potatoes (Solanum tuberosum L.) to salinity in an arid environment and field performance of the seed tubers grown with fresh water in the following season. Agricultural Water Management 116: 122–127.
Levy, D., and R.E. Veilleux. 2007. Adaptation of potato to high temperatures and salinity—A review. American Journal of Potato Research 84: 487–506.
LGMA. 2009. California leafy green marketing agreement. California Leafy Green Products. http://www.caleafygreens.ca.gov/, Accessed May 29, 2012.
Li, Z., N. Wang, T.S. Hong, A. Franzen, and J.N.A. Li. 2011. Closed-loop drip irrigation control using a hybrid wireless sensor and actuator network. Science China-Information Sciences 54: 577–588.
Luquez, V.M., and J.J. Guiamét. 2002. The stay green mutations d1 and d2 increase water stress susceptibility in soybeans. Journal of Experimental Botany 53: 1421–1428.
Maas, E.V. 1986. Salt tolerance of plants. Applied Agricultural Research 1: 12–25.
Maas, E.V., and G.J. Hoffman. 1977. Crop salt tolerance—Current assessment. Journal of the Irrigation and Drainage Division, American Society of Civil Engineers 103: 115–134.
Maurel, C., T. Simonneau, and M. Sutka. 2010. The significance of roots as hydraulic rheostats. Journal of Experimental Botany 61: 3191–3198.
Meiri, A., and Z. Plaut. 1985. Crop production and management under saline conditions. Plant and Soil 89: 253–271.
Micklin, P. 2007. The Aral Sea disaster. Annual Review of Earth and Planetary Sciences 35: 47–72.
Miller, D.E., and M.W. Martin. 1987. The effect of irrigation regime and subsoiling on yield and quality of three potato cultivars. American Potato Journal 64: 17–25.
Moffat, A.S. 2002. Finding new ways to protect drought-stricken plants. Science 296: 1226–1229.
Moller, M., V. Alchanatis, Y. Cohen, M. Meron, J. Tsipris, A. Naor, V. Ostrovsky, M. Sprintsin, and S. Cohen. 2007. Use of thermal and visible imagery for estimating crop water status of irrigated grapevine. Journal of Experimental Botany 58: 827–838.
Morgan, K.T., T.A. Wheaton, L.R. Parsons, and W.S. Castle. 2008. Effects of reclaimed municipal waste water on horticultural characteristics, fruit quality, and soil and leaf mineral concentration of citrus. HortScience 43: 459–464.
Munns, R., and M. Tester. 2008. Mechanisms of salinity tolerance. Annual Review of Plant Biology 59: 651–681.
Munns, R., R.A. James, B. Xu, A. Athman, S.J. Conn, C. Jordans, C.S. Byrt, R.A. Hare, S.D. Tyerman, M. Tester, D. Plett, and M. Gilliham. 2012. Wheat grain yield on saline soils is improved by an ancestral Na+ transporter gene. Nature Biotechnology 30: 360–U173.
Muñoz-Carpena, R., M.D. Dukes, Y.C.C. Li, and W. Klassen. 2005. Field comparison of tensiometer and granular matrix sensor automatic drip irrigation on tomato. HortTechnology 15: 584–590.
Muñoz-Carpena, R., M.D. Dukes, Y. Li, and W. Klassen. 2008. Design and field evaluation of a new controller for soil-water based irrigation. Applied Engineering in Agriculture 24: 183–191.
Neumann, P.M. 2008. Coping mechanisms for crop plants in drought-prone environments. Annals of Botany 101: 901–907.
Nimah, N.M., and R. Bashour. 2010. Modeling deficit irrigation water productivity as a function of crop root depth, Paper No. 1008942. 2010 American Society of Agricultural and Biological Engineers (ASABE) Annual International Meeting, ASABE, Pittsburgh, Pennsylvania
Oosterhuis, D.M., and S.D. Wullschleger. 1987. Osmotic adjustment in cotton (Gossypium hirsutum L.) leaves and roots in response to water stress. Plant Physiology 84: 1154–1157.
Padhi, J., R.K. Misra, and J.O. Payero. 2012. Estimation of soil water deficit in an irrigated cotton field with infrared thermography. Field Crops Research 126: 45–55.
Parker, C.J., M.K.V. Carr, N.J. Jarvis, M.T.B. Evans, and V.H. Lee. 1989. Effect of subsoil loosening and irrigation on soil physical properties, root distribution and water uptake of potatoes (Solanum tuberosum). Soil & Tillage Research 13: 267–285.
Parsons, L.R., B. Sheikh, R. Holden, and D.W. York. 2010. Reclaimed water as an alternative water source for crop irrigation. HortScience 45: 1626–1629.
Pehrson, L., R.L. Mahler, E.J. Bechinski, and C. Williams. 2010. Water management practices used in potato production in Idaho. American Journal of Potato Research 87: 253–260.
Persson, A., P. Pilesjö, and L. Eklundh. 2005. Spatial influence of topographical factors on yield of potato (Solanum tuberosum L.) in Central Sweden. Precision Agriculture 6: 341–357.
Pescod, M.B. 1992. Wastewater treatment and use in agriculture. FAO Irrigation and Drainage Paper (FAO) No 47, FAO, Rome, 139 pp.
Pierce, F.J., and P. Nowak. 1999. Aspects of precision agriculture. Advances in Agronomy 67: 1–85.
Porter, J.R., and M.A. Semenov. 2005. Crop responses to climatic variation. Philosophical Transactions of the Royal Society of London, Series B: Biological Sciences 360: 2021–2035.
Qadir, M., D. Wichelns, L. Raschid-Sally, P.G. McCornick, P. Drechsel, A. Bahri, and P.S. Minhas. 2010. The challenges of wastewater irrigation in developing countries. Agricultural Water Management 97: 561–568.
Qi, S.Z., and F. Luo. 2005. Water environmental degradation of the Heihe River Basin in arid northwestern China. Environmental Monitoring and Assessment 108: 205–215.
Rensink, W., A. Hart, J. Liu, O.Y. Shu, V. Zismann, and C.R. Buell. 2005a. Analyzing the potato abiotic stress transcriptome using expressed sequence tags. Genome 48: 598–605.
Rensink, W.A., S. Iobst, A. Hart, S. Stegalkina, J. Liu, and C.R. Buell. 2005b. Gene expression profiling of potato responses to cold, heat, and salt stress. Funct Integr Genomics 5: 201–207.
Reynolds, M.P., and R. Ortiz. 2010. Adapting crops to climate change: A summary. In Climate change and crop production, ed. M.P. Reynolds, 1–8. Wallingford: CABI.
Reynolds, M., F. Dreccer, and R. Trethowan. 2007. Drought-adaptive traits derived from wheat wild relatives and landraces. Journal of Experimental Botany 58: 177–186.
Rhoades, J.D. 1999. Use of saline drainage water for irrigation. In Agricultural drainage, monograph no 38, ed. R.W. Skaggs and van Schilfgaarde, 619–657. Madison: American Society of Agronomy.
Rhoades, J.D., F.T. Bingham, J. Letey, G.J. Hoffman, A.R. Dedrick, P.J. Pinter, and J.A. Replogle. 1989. Use of saline drainage water for irrigation: Imperial Valley study. Agricultural Water Management 16: 26–36.
Rhoades, J.D., A. Kandiah, and A.M. Mashali. 1992. The use of saline waters for crop production,. FAO irrigation and drainage paper 48, Rome, Italy.
Ribaut, J.M., and M. Ragot. 2007. Marker-assisted selection to improve drought adaptation in maize: The backcross approach, perspectives, limitations, and alternatives. Journal of Experimental Botany 58: 351–360.
Richards, R.A. 2006. Physiological traits used in the breeding of new cultivars for water-scarce environments. Agricultural Water Management 80: 197–211.
Rizhsky, L., H.J. Liang, J. Shuman, V. Shulaev, S. Davletova, and R. Mittler. 2004. When defense pathways collide. The response of Arabidopsis to a combination of drought and heat stress. Plant Physiology 134: 1683–1696.
Rockström, J. 2003. Water for food and nature in drought-prone tropics: Vapour shift in rain-fed agriculture. Philosophical Transactions of the Royal Society of London, Series B: Biological Sciences 358: 1997–2009.
Rodda, N., L. Salukazana, S.A.F. Jackson, and M.T. Smith. 2011. Use of domestic greywater for small-scale irrigation of food crops: Effects on plants and soil. Physics and Chemistry of the Earth 36: 1051–1062.
Sanz, M.A., V. Bonnélye, and G. Cremer. 2007. Fujairah reverse osmosis plant: 2 years of operation. Desalination 203: 91–99.
Schafleitner, R. 2009. Growing more potatoes with less water. Tropical Plant Biology 2: 111–121.
Schittenhelm, S., H. Sourell, and F.J. Löpmeier. 2006. Drought resistance of potato cultivars with contrasting canopy architecture. European Journal of Agronomy 24: 193–202.
Scholze, M., W. Knorr, N.W. Arnell, and I.C. Prentice. 2006. A climate-change risk analysis for world ecosystems. Proceedings of the National Academy of Sciences of the United States of America 103: 13116–13120.
Scoffoni, C., M. Rawls, A. McKown, H. Cochard, and L. Sack. 2011. Decline of leaf hydraulic conductance with dehydration: Relationship to leaf size and venation architecture. Plant Physiology 156: 832–843.
Sepaskhah, A.R., and S.H. Ahmadi. 2010. A review on partial root-zone drying irrigation. International Journal of Plant Production 4: 241–258.
Shalhevet, J. 1994. Using water of marginal quality for crop production: Major issues. Agricultural Water Management 25: 233–269.
Sharma, B.R., and P.S. Minhas. 2005. Strategies for managing saline/alkali waters for sustainable agricultural production in South Asia. Agricultural Water Management 78: 136–151.
Sharma, N., P. Kumar, M.S. Kadian, S.K. Pandey, S.V. Singh, and S.K. Luthra. 2011. Performance of potato (Solanum tuberosum) clones under water stress. Indian Journal of Agricultural Sciences 81: 825–829.
Shimshi, D., J. Shalhevet, and T. Meir. 1983. Irrigation regime effects on some physiological responses of potatoes. Agronomy Journal 75: 262–267.
Shin, D., S.J. Moon, S. Han, B.G. Kim, S.R. Park, S.K. Lee, H.J. Yoon, H.E. Lee, H.B. Kwon, D. Baek, B.Y. Yi, and M.O. Byun. 2011. Expression of StMYB1R-1, a novel potato single MYB-like domain transcription factor, increases drought tolerance. Plant Physiology 155: 421–432.
Shinghal, K., A. Noor, N. Srivastava, and R. Singh. 2010. Wireless sensor networks in agriculture: For potato farming. International Journal of Engineering Science and Technology 2: 3955–3963.
Shock, C.C., and E.B.G. Feibert. 2002. Deficit irrigation of potato, FAO Corporate Document Repository. Natural Resources Management and Environment Department. http://www.fao.org/docrep/004/Y3655E/y3655e08.htm#TopOfPage, Accessed May1, 2012.
Shock, C.C., A.B. Pereira, and E.P. Eldredge. 2007. Irrigation best management practices for potato. American Journal of Potato Research 84: 29–37.
Sinclair, T.R. 2011. Challenges in breeding for yield increase for drought. Trends in Plant Science 16: 289–293.
Smith, R.J., J.N. Baillie, A.C. McCarthy, S.R. Raine, and C.P. Baillie. 2010. Review of Precision Irrigation Technologies and their Application. NCEA Publication 1003017/1. National Centre for Engineering in Agriculture (NCEA), University of Southern Queensland, Toowoomba.
Spitters, C.J.T., and A.H.C.M. Schapendonk. 1990. Evaluation of breeding strategies for drought tolerance in potato by means of crop growth simulation. Plant and Soil 123: 193–203.
Spooner, D.M., and A. Salas. 2006. Chapter 1: Structure, biosystematics, and genetic resources. In Handbook of potato production, improvement, and postharvest management, ed. J. Gopal and S.M.P. Khurana, 1–40. New York: The Haworth Press.
Stalham, M.A., and E.J. Allen. 2001. Effect of variety, irrigation regime and planting date on depth, rate, duration and density of root growth in the potato (Solanum tuberosum) crop. Journal of Agricultural Science 137: 251–270.
Stalham, M.A., and E.J. Allen. 2004. Water uptake in the potato (Solanum tuberosum) crop. Journal of Agricultural Science 142: 373–393.
Tanner, C.B., G.G. Weis, and D. Curwen. 1982. Russet Burbank rooting in sandy soils with pans following deep plowing. American Potato Journal 59: 107–112.
Tardieu, F. 2012. Any trait or trait-related allele can confer drought tolerance: Just design the right drought scenario. Journal of Experimental Botany 63: 25–31.
Taylor, H.M. 1983. Managing root systems for efficient water use: An overview. In Limitations to efficient water use in crop production, ed. H.W. Taylor, W.R. Jordan, and T.R. Sinclair, 87–113. Wisconsin: American Society of Agronomy.
The Potato Genome Sequencing Consortium. 2011. Genome sequence and analysis of the tuber crop potato. Nature 475: 189–195.
Turner, N.C. 1982. The role of shoot characteristics in drought resistance of crop plants. In Drought resistance in crops with emphasis on rice, ed. The International Rice Research Institute, 115–134. Manila: IRRI.
Turral, H., J. Burke, and J.M. Faures. 2011. Climate change, water and food security. FAO Water Reports 36 FAO, Rome p 174
UN Water. 2006. Coping with water scarcity. UN-Water Thematic Initiatives A strategic issue and priority for system-wide action. http://www.unwater.org, Accessed May 25, 2012.
Upadhyaya, C.P., J. Venkatesh, M.A. Gururani, L. Asnin, K. Sharma, H. Ajappala, and S.W. Park. 2011. Transgenic potato overproducing L-ascorbic acid resisted an increase in methylglyoxal under salinity stress via maintaining higher reduced glutathione level and glyoxalase enzyme activity. Biotechnology Letters 33: 2297–2307.
Valliyodan, B., and H.T. Nguyen. 2006. Understanding regulatory networks and engineering for enhanced drought tolerance in plants. Current Opinion in Plant Biology 9: 189–195.
van Bergeijk, J., D. Goense, and L. Speelman. 2001. Soil tillage resistance as a tool to map soil type differences. Journal of Agricultural Engineering Research 79: 371–387.
van der Zaag, D.E. 1991. The potato crop in Saudi Arabia. Ministry of Agriculture and Water. Riyadh: Saudi Potato Development Programme.
van Loon, C.D. 1981. The effect of water stress on potato growth, development, and yield. American Potato Journal 58: 51–69.
Vasquez-Robinet, C., S.P. Mane, A.V. Ulanov, J.I. Watkinson, V.K. Stromberg, D. De Koeyer, R. Schafleitner, D.B. Willmot, M. Bonierbale, H.J. Bohnert, and R. Grene. 2008. Physiological and molecular adaptations to drought in Andean potato genotypes. Journal of Experimental Botany 59: 2109–2123.
Vellidis, G., M. Tucker, C. Perry, C. Wen, and C. Bednarz. 2008. A real-time wireless smart sensor array for scheduling irrigation. Computers and Electronics in Agriculture 61: 44–50.
Verslues, P.E., and E.A. Bray. 2004. LWR1 and LWR2 are required for osmoregulation and osmotic adjustment in Arabidopsis. Plant Physiology 136: 2831–2842.
Vigneswaran, S., and M. Sundaravadivel. 2004. Recycle and reuse of domestic wastewater in Wastewater: Recycle, Reuse and Reclamation. In Encyclopedia of Life Support Systems (EOLSS), Developed under the auspices of UNESCO, ed. Saravanamuthu (Vigi) Vigneswaran. Oxford: Eolss.
Vos, J., and J. Groenwold. 1989a. Genetic differences in water-use efficiency, stomatal conductance and carbon isotope fractionation in potato. Potato Research 32: 113–121.
Vos, J., and J. Groenwold. 1989b. Characteristics of photosynthesis and conductance of potato canopies and the effects of cultivar and transient drought. Field Crops Research 20: 237–250.
Vos, J., and A.J. Haverkort. 2007. Water availability and potato crop performance. In Potato biology and biotechnology: Advances and perspectives, ed. D. Vreugdenhil, J. Bradshaw, C. Gebhardt, F. Govers, M.A. Taylor, D.K.L. MacKerron, and H.A. Ross, 333–351. Amsterdam: Elsevier.
Walworth, J.L., and D.E. Carling. 2002. Tuber initiation and development in irrigated and non-irrigated potatoes. American Journal of Potato Research 79: 387–395.
Wang, F.X., Y.H. Kang, S.P. Liu, and X.Y. Hou. 2007. Effects of soil matric potential on potato growth under drip irrigation in the North China Plain. Agricultural Water Management 88: 34–42.
Wang, X.Z., W.P. Yang, A. Wheaton, N. Cooley, and B. Moran. 2010. Efficient registration of optical and IR images for automatic plant water stress assessment. Computers and Electronics in Agriculture 74: 230–237.
Watanabe, K.N., A. Kikuchi, T. Shimazaki, and M. Asahina. 2011. Salt and drought stress tolerances in transgenic potatoes and wild species. Potato Research 54: 319–324.
WATEC. 2007. Wastewater Treatment and Reclamation. Mekerot, Israel National Water Co. www.mekorot.co.il/Eng/Activities/Pages/WastewaterTreatmentandReclamation.aspx, Accessed May 29, 2012.
Waterer, D., N.T. Benning, G.H. Wu, X.M. Luo, X.J. Liu, M. Gusta, A. McHughen, and L.V. Gusta. 2010. Evaluation of abiotic stress tolerance of genetically modified potatoes (Solanum tuberosum cv. Desiree). Molecular Breeding 25: 527–540.
Watkinson, J.I., L. Hendricks, A.A. Sioson, C. Vasquez-Robinet, V. Stromberg, L.S. Heath, M. Schuler, H.J. Bohnert, M. Bonierbale, and R. Grene. 2006. Accessions of Solanum tuberosum ssp. andigena show differences in photosynthetic recovery after drought stress as reflected in gene expression profiles. Plant Science 171: 745–758.
Weisz, R., J. Kaminski, and Z. Smilowitz. 1994. Water deficit effects on potato leaf growth and transpiration: Utilizing fraction extractable soil water for comparison with other crops. American Potato Journal 71: 829–840.
White, J.W., P. Andrade-Sanchez, M.A. Gore, K.F. Bronson, T.A. Coffelt, M.M. Conley, K.A. Feldmann, A.N. French, J.T. Heun, D.J. Hunsaker, M.A. Jenks, B.A. Kimball, R.L. Roth, R.J. Strand, K.R. Thorp, G.W. Wall, and G. Wang. 2012. Field-based phenomics for plant genetics research. Field Crops Research 133: 101–112.
Whitmore, A.P., and W.R. Whalley. 2009. Physical effects of soil drying on roots and crop growth. Journal of Experimental Botany 60: 2845–2857.
Witcombe, J.R., P.A. Hollington, C.J. Howarth, S. Reader, and K.A. Steele. 2008. Breeding for abiotic stresses for sustainable agriculture. Philosophical Transactions of the Royal Society of London, Series B: Biological Sciences 363: 703–716.
Wolf, J., and M. van Oijen. 2002. Modelling the dependence of European potato yields on changes in climate and CO2. Agricultural and Forest Meteorology 112: 217–231.
Xie, K.Y., X.X. Wang, R.F. Zhang, X.F. Gong, S.B. Zhang, V. Mares, C. Gavilán, A. Posadas, and R. Quiroz. 2012. Partial root-zone drying irrigation and water utilization efficiency by the potato crop in semi-arid regions in China. Scientia Horticulturae 134: 20–25.
Yoo, C.Y., H.E. Pence, P.M. Hasegawa, and M.V. Mickelbart. 2009. Regulation of transpiration to improve crop water use. Critical Reviews in Plant Sciences 28: 410–431.
Zakaluk, R., and R. Sri Ranjan. 2006. Artificial neural network modelling of leaf water potential for potatoes using RGB digital Images: A greenhouse study. Potato Research 49: 255–272.
Zhang, N., H.J. Si, G. Wen, H.H. Du, B.L. Liu, and D. Wang. 2011. Enhanced drought and salinity tolerance in transgenic potato plants with a BADH gene from spinach. Plant Biotechnology Reports 5: 71–77.
Zhu, X.G., G.L. Zhang, D. Tholen, Y. Wang, C.P. Xin, and Q.F. Song. 2011. The next generation models for crops and agro-ecosystems. Science China-Information Sciences 54: 589–597.
Acknowledgments
We thank Drs Yafit Cohen and V. Alchnatis from the Volcani Center, Israel, for providing and allowing using unpublished data of their research (Fig. 2) and for reviewing the PI section and contributing to its content.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Levy, D., Coleman, W.K. & Veilleux, R.E. Adaptation of Potato to Water Shortage: Irrigation Management and Enhancement of Tolerance to Drought and Salinity. Am. J. Potato Res. 90, 186–206 (2013). https://doi.org/10.1007/s12230-012-9291-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12230-012-9291-y