Abstract
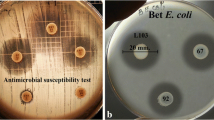
Fifty-five strains of enterococci isolated from the piglet intestine were characterized in vitro for probiotic activity. Identification of the isolates revealed Enterococcus faecium as the predominant species (84 %). Forty strains (73 %) were found to produce bacteriocin-like substances (only into solid media) with activity almost only toward Gram-positive genera. Thirty-eight % of strains were resistant to tetracycline, 27 % to chloramphenicol, 18 % to erythromycin and 16 % to vancomycin. In addition to control of strain safety, 6 % of isolates were β-hemolytic and 16 % produced gelatinase. Seven strains selected for further probiotic assays exhibited sufficient survival rate at pH 3.0 after 3 h, in the presence of 1 % ox-bile and lysozyme after 1 d (over 107 CFU/mL in all tests). The adhesion of tested strains to porcine and human intestinal mucus was found in a similar range (1.4–14.0 % and 1.4–17.6 %, respectively). In accordance with current research effort to use and/or to combine various health promoting substances, the sensitivity of all isolates toward plant extracts and toward bacteriocins produced by animal and environmental strains was determined. All enterococci were sensitive toward oregano and sage extracts and toward one (E. faecium EF55 — chicken isolate, activity of 25 600 AU/mL) of ten bacteriocin substances. It means that a similar anti-enterococcal potential of some bacteriocin substances may be observed as for certain plant extracts.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- BHI:
-
brain heart infusion (plate)
- CFU:
-
colony-forming unit
- MRS:
-
de Man-Rogosa-Sharpe (medium)
- PPB:
-
partially purified bacteriocin
- Amp:
-
ampicillin
- Cmp:
-
chloramphenicol
- Ery:
-
erythromycin
- Rif:
-
rifampicin
- Tet:
-
tetracycline
- Van:
-
vancomycin
References
Baele M., Baele P., Vaneechoutte M., Storms V., Butaye P., Devriese L.A., Verchraegen G., Gillis M., Haesebrouck F.: Application of tRNA intergenic spacer PCR for identification of Enterococcus species. J.Clin.Microbiol.38, 4201–4207 (2000).
Bernbom N., Licht T.R., Brogren C.-H., Jelle B., Johansen A.H., Badiola I., Vogensen F.K., Norrung B.: Effect of Lactococcus lactis on the composition of intestinal microbiota: role of nisin. Appl.Environ.Microbiol.72, 239–244 (2006).
Bomba A., Jonecová Z., Koščová J., Nemcová R., Gancarčíková S., Mudroňová D., Sciranková Ľ., Buleca V., Lazar G., Pošivák J., Kašteľ R., Mareková M.: The improvement of probiotics efficacy by synergistically acting components of natural origin: a review. Biologia61, 729–734 (2006).
Butaye P., Devriese L.A., Haesebrouck F.: Differences in antibiotic resistance patterns of Enterococcus faecalis and Enterococcus faecium strains isolated from farm and pet animals. Antimicrob.Agents Chemother.45, 1374–1378 (2001).
De Vuyst L., Callewaer R., Pot B.: Characterization of antagonistic activity of Lactobacillus amylovorus DCE471 and large-scale isolation of its bacteriocin amylovorin L471. Syst.Appl.Microbiol.19, 9–20 (1996).
Drahovská H., Slobodníková L., Kocíncová D., Seman M., Končeková R., Trupl J., Turňa J.: Antibiotic resistance and virulence factors among clinical and food enterococci isolated in Slovakia. Folia Microbiol.49, 763–768 (2004).
Eaton T.J., Gasson M.J.: Molecular screening of Enterococcus virulence determinants and potential for genetic exchange between food and medical isolates. Appl.Environ.Microbiol.67, 1628–1635 (2001).
European Commission: List of authorized additives in feedingstuffs published in application of Article 9t(b) of Council Directive 70/524/EEC concerning additives in feedingstuffs. Official Journal of the European Union of 25.2.2004, C50 (2004).
Franz C.M.A.P., Van Belkum M.J., Holzapfel W.H., Abriouel H., Gálvez A.: Diversity of enterococcal bacteriocins and their grouping in a new classification scheme. FEMS Microbiol.Rev.31, 293–310 (2007).
Fricourt B.V., Barefoot S.F., Testin R.F., Hayasaka S.S.: Detection and activity of plantaricin F, an antibacterial substance from Lactobacillus plantarum BF001 isolated from processed channel catfish. J.Food Prot.57, 698–702 (1994).
Gilliland S.E., Walker D.K.: Factors to consider when selecting a culture of Lactobacillus acidophilus as a dietary adjunct to produce a hypocholesterolemic effect in humans. J.Dairy Sci.73, 905–911 (1990).
Kročko M., Čanigová M., Ducková V.: Occurence, isolation and antibiotic resistance of Enterococcus species isolated from raw pork, beef and poultry. J.Food Nutr.Res.46, 91–95 (2007).
Lauková A., Mareková M.: Production of bacteriocins by different Enterococcal isolates. Folia Microbiol.6, 49–52 (2001).
Lauková A., Czikková S., Burdová O.: Anti-staphylococcal effect of enterocin in Sunar® and yogurt. Folia Microbiol.44, 707–711 (1999).
Lauková A., Vlaemynck G., Czikková S.: Effetct of enterocin CCM 4231 on Listeria monocytogenes in Saint-Paulin cheese. Folia Microbiol.46, 157–160 (2001).
Lauková A., Guba P., Nemcová R., Vasilková Z.: Reduction of Salmonella in gnotobiotic Japanese quails caused by the enterocin A-producing EK13 strain of Enterococcus faecium. Vet.Res.Commun.27, 275–280 (2003).
Lauková A., Strompfová V., Ouwehand A.C.: Adhesion properties of enterococci to intestinal mucus of different hosts. Vet.Res. Commun.28, 647–655 (2004).
Ogunbanwo S.T., Sanni A.I., Onilude A.A.: Influence of bacteriocin in the control of Escherichia coli infection of broiler chickens in Nigeria. World J.Microbiol.Biotechnol.20, 51–56 (2004).
Ouwehand A.C., Kirjavainen P.V., Grönlund M.M., Isolauri E., Salminen S.J.: Adhesion of probiotic microorganisms to intestinal mucus. Internat.J.Food Microbiol.64, 119–126 (1999).
Ouwehand A.C., Salminen S., Tölkkö S., Roberts P., Ovaska J., Salminen E.: Resected human colonic tissue: new model for characterizing adhesion on lactic acid bacteria. Clin.Diagn.Lab.Immunol.9, 184–186 (2002).
Pantev A., Kabadjova P., Dalgalarrondo M., Haertlé T., Ivanova I., Dousset X., Prévost H., Chobert J.-M.: Isolation and partial characterization of an antibacterial substance produced by Enterococcus faecium. Folia Microbiol.47, 391–400 (2002).
Park S.C., Hwang M.H., Kim Y.H., Kim J.C., Song J.C., Lee K.W., Jeong K.S., Rhee M.H., Kim K.S., Kim T.W.: Comparison of pH and bile resistance of Lactobacillus acidophilus strains isolated form rat, pig, chicken, and human sources. World J.Microbiol. Biotechnol.22, 35–37 (2006).
Pollmann M., Nordhoff M., Pospischil A., Tedin K., Wieler L.H.: Effects of a probiotic strain of Enterococcus faecium on the rate of natural chlamydia infection in swine. Infect.Immun.73, 4346–4353 (2005).
Scharek L., Guth J., Reiter K., Weyrauch K.D., Taras D., Schwerk P., Schierack P., Schmidt M.F.G., Wieler L.H., Tedin K.: Influence of a probiotic Enterococcus faecium strain on development of the immune system of sows and piglets. Vet. Immunol.Immunopathol.105, 151–161 (2005).
Si W., Gong J., Tsao R., Zhou T., Yu H., Poppe C., Johnson R., Du Z.: Antimicrobial activity of essential oils and structurally related synthetic food additives towards selected pathogenic and beneficial gut bacteria. J.Appl.Microbiol.100, 296–305 (2006).
Skalka B., Pillich J., Pospíšil L.: Further observation on Corynebacterium renale as an indicator organism in the detection of exfoliation-positive strains Staphylococcus aureus. Zbl.Bakteriol.Hyg.A256, 168–174 (1983).
Stein H.H.: Feeding the pigs’ immune system and alternatives to antibiotics, pp. 65–82 in Proc. London Swine Conference — Today’s Challenges-Tomorrow’s Opportunities (2007).
Strompfová V., Lauková A., Mudroňová D.: Effect of bacteriocin-like substance produced by Enterococcus faecium EF55 on the composition of avian gastrointestinal microflora. Acta Vet.Brno72, 559–564 (2003).
Strompfová V., Lauková A., Ouwehand A.C.: Lactobacilli and enterococci — potential probiotics for dogs. Folia Microbiol.49, 203–207 (2004).
Strompfová V., Marciňáková M., Simonová M., Gancarčíková S., Jonecová Z., Sciranková Ľ., Koščová J., Buleca V., Čobanová K., Lauková A.: Enterococcus faecium EK13 — an enterocin A-producing strain with probiotic character and its effect in piglets. Anaerobe12, 242–248 (2006).
Strompfová V., Lauková A., Simonová M., Marciňaková M.: Occurence of the structural enterocin A, P, B, L50B genes in enterococci of different origin. Vet.Microbiol.132, 293–301 (2008).
Šustáčková A., Nápravníková E., Schlegelová J.: Antimicrobial resistance of Enterococcus spp. isolates from raw beef and meat products. Folia Microbiol.49, 411–417 (2004).
Taras D., Vahjen W., Macha M., Simon O.: Performance, diarrhea incidence, and occurrence of Escherichia coli virulence genes during long-term administration of a probiotic Enterococcus faecium strain to sows and piglets. J.Anim.Sci.84, 608–617 (2006).
Trojanová I., Vlková E., Rada V., Marounek M.: Different utilization of glucose and raffinose in Bifidobacterium breve and Bifidobacterium animalis. Folia Microbiol.51, 320–324 (2006).
Welsh J., Mcclelland M.: Genomic fingerprints produced by PCR with consensus tRNA gene primers. Nucl.Acids Res.19, 861–866 (1991).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Strompfová, V., Lauková, A. Enterococci from piglets — Probiotic properties and responsiveness to natural antibacterial substances. Folia Microbiol 54, 538–544 (2009). https://doi.org/10.1007/s12223-009-0078-6
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-009-0078-6