Abstract
Effect of anti-predator defense due to fear of predator felt by prey and effect of toxic substance released by external sources on prey–predator system is a serious mater of concern in mathematical biology. In the proposed model we have discussed a prey–predator system in which both the species are infected by environmental toxicant. In our consideration prey species is directly infected by environmental toxicant and predator gets infected by consuming infected prey. Prey’s growth rate is assumed to be affected by fear of predator. In this work the proposed predator–prey model is analyzed in presence of environmental fluctuation, i.e., stochastic analysis of this model is discussed. Using Itô formula: positivity, boundedness, uniform continuity criterion and global attractivity of solutions of this system have been established. Conditions for which the prey as well as the predator goes extinct have been derived. Conditions for persistence of the system have also been discussed. Mathematical findings have been validated in numerical simulation by MATLAB. Different effects of different levels of toxicant and different levels of fear have been demonstrated by depicting figures in numerical simulation using MATLAB.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The history of the study to gain knowledge about the interactions among the individuals of the prey–predator species dates back long. Many ecologists and mathematicians [6, 8, 14, 38] have made a significant contributions to analyze prey–predator interactions over the years. Analysis of the persistence and extinction scenario has been one of the most important objectives, though there are several thematic areas of interest. Analyzing these models in rapidly fluctuating environment make these findings more meaningful and interesting.
From field experiments, some theoretical ecologists and biologists have realised that a prey–predator model should involve not only direct killing but also the fear (felt by prey)-factor as they have observed a change in the behaviour and psychology of the prey population in presence of the predator, sometimes more powerful than direct killing [4, 5]. However, the intensity of the impact left by fear factor on the ecosystem is not totally clear till now. There are some controversies and some believe that the physiological impacts of fear on young populations may lead to a lower survival rate of adults, although so far such an impact has not yet been shown by any direct experimental results.
Fear makes different animals respond differently [5, 31, 33, 34]: some find a different place to live, some change their foraging behaviour, some become circumspect, whereas others go through psychological changes. Under fear, a prey species may find a comparatively low risk (with respect to fear) area to live but this effort may get nullified in case the new habitat is not suitable for the prey to live as well - the prey may starve out of fear rather than searching for food [4, 5, 43]. In an experiment made on the snowshoe hare, it has been found that high fear factor played a bigger role in decreasing their reproduction rate than high hare density or poor food level [2]. Therefore, it would not be appropriate to neglect the fear factor prevailing in the minds of the prey species particularly when the reproduction rate is reduced by it [2, 3, 32]. The estimation of Pangle et al. [30] upon direct killings and fear effects on three species of zoo-plankton on Lake Michigan and Lake Erie by the predation of water fleas (Bythotrephes longimanus) over six combinations of location and depth is that the effect of the fear factor on the growth rates were seven times more than the effect of direct killing of them. Altendorf et al. [1] found that mule deer, under the fear of mountain lions, refrained themselves from searching for food to a large extent. Creel et al. [3] found that elk (one of the largest species of the deer family) faces changes in reproductive physiology due to the predation fear of wolves. It has also been found that birds flee from their nests as an anti-predator behaviour when they discover the sound of predator (first sign of danger) [5]. A recent experiment, performed by Zanette et al. [45], on song sparrows during an entire breeding season, found their reproduction rate to have fallen down by 40% only by the fear of predator (sound of predator). A study of Krapivsky et al. [22] shows that different positions and different numbers of the predator (lion) compel lambs to behave differently.
Recently, an observation has brought it into notice that prey population is reduced by the fear of large carnivores and thereby the number of species that the prey population feeds on or competes with has increased. Das and Samanta [7] has found that predator population dominates prey population because of high cost of fear. It emerged out in a field experiment along the shoreline of British Columbia that the racoon’s predation rate and its hunting of intertidal crabs, subtidal red rock crabs and intertidal fish decreased for fear of carnivorous animals and the abundance of intertidal crabs, subtidal red rock crabs and intertidal fish went up by 97%, 61% and 81% respectively [40]. Again, the findings in the cases of Siberian Jay by Eggers et al. [12], dugongs by Wirsing et al. [44] and birds by Ghalambor et al. [13] and Hua et al. [18, 19] have led mathematicians to believe that fear factor plays a key role in population dynamics. Many researchers have studied prey–predator models, but mathematical models considering fear factor have only been successfully developed by Wang et al. [41, 42]. Despite having a lot of field experimental results, there is little evidence which upholds the fact that population dynamics is affected by fear [9, 10, 27,28,29].
Existence of toxic substance in environment is a major concern for bioeconomic modeling. Involving toxic substance in mathematical model is started with the studies of Hallam and Clark [15], Hallam and De Luna [16], Dubey and Hussain [11], Kar and Chaudhuri [21], Samanta [36], etc. General single species or two species communities without any special emphasis on aquatic environments are considered in most of the models. The toxin released by one species or by environment not only affects that species but may affect the growth of the other species also (such as predator of that species). Day by day industries are producing a huge amount of toxicants in to the environment because of growing human needs. These toxicants mostly affect the species which are living there. Maynard Smith [39] incorporated the effects of toxic substances in a two species Lotka–Volterra competitive system by considering that in presence of other species each species produces a toxic substance.
To evaluate the effect of environmental noises on dynamical systems some researches [23, 26, 35] have introduced Gaussian white noise as a model of environmental variations. May in 1973 [26] pointed out that all the parameters such as birth rates, death rates, competition coefficient, carrying capacity etc. involved in a dynamical system can be randomly fluctuated to a great lesser extent for the cause of continuous fluctuation in the environment. Das and Samanta [6] have shown that the environmental noise effects on extinction and persistence of the system.
In the present work, we have considered a prey–predator system in a randomly fluctuating environment incorporating fear of predator (felt by prey) and prey is directly affected by toxic substance and predator gets affected by consuming infected prey.
We have divided the rest of this work into some sections and subsections. Section 2 contains formulation of the model which is divided into four subsections. In Sect. 2.1 functional response is described and Sect. 2.2 deals with fear function. Deterministic model is formulated in Sect. 2.3 followed by stochastic model in Sect. 2.4. Positivity of solutions of both the deterministic and stochastic model is analyzed in Sect. 3 followed by boundedness of solutions in Sect. 4. Section 5 contains uniform continuity criterion of solutions of the stochastic system. In Sect. 6 extinction scenario in presence of environmental noise is discussed. Most important theorem of persistence of the system is discussed in Sect. 7. In Sect. 8 numerical simulation justify the mathematical findings numerically using MATLAB. In Sect. 9, we have given some conclusion of our findings.
2 Model formulation
We shall discuss a predator prey model with the effect of anti-predator defense because of fear (felt by prey) in presence of predator and effect of toxic substance present in the environment (released by some other external sources). It is assumed that the infection among the individuals of the prey species is caused directly by some external toxic substance and the predator gets affected by consuming infected prey.
Let us consider x(t) and y(t) to represent the biomass of prey and predator respectively at any time \(t>0.\) Let us model the effect of fear (felt by prey) through a function \(f(\zeta , \beta , y)\) called fear function that accounts for the cost of anti-predator defence. Here \(\zeta\) represents the cost of minimum fear and \(\displaystyle \frac{1}{\beta }\) stands for the level of fear which causes the anti-predator behavior of the prey. We consider the following system:
The term h(x) is the prey density dependent function which defines the intake rate for predator. Here, g is the coefficient of birth rate of the prey, r is the coefficient of the rate of intraspecific competition among the individuals of the prey species, \(d_{1}\) and \(d_{2}\) are the natural mortality rates of prey and predator respectively, \(a_{1}\in (0,1]\) is the conversion rate.
Now we introduce the term \(c_{1}x^{3}\) that accounts for direct transmission of the infection among the individuals of the prey species by external toxic substances and the term \(c_{2}y^{2}\) which comes indirectly through the infection of the predator by consuming infected prey. So, system (2.1) becomes:
2.1 Functional response
The consuming rate per predator towards prey is called functional response in ecology. Ecologist C.S. Holling [17] described functional response into three types: Holling type I, II and III. Holling type II is the mostly used functional response among them and we also consider Holling type II as the functional response in our model. We have considered the following particular form of h(x) in our proposed model:
Here, \(\displaystyle \frac{\alpha }{\eta }\) is the maximum consumption rate and \(\displaystyle \frac{1}{\eta }\) is the half saturation constant which are non negative.
2.2 Fear function
As per the experimental evidences, the prey population will be diminished due to the fear effect, so it is reasonable to assume that \(f(\zeta ,\beta ,y)\) has the following properties, where \(\zeta \in [0,1]\) represents the cost of minimum fear and \(\displaystyle \frac{1}{\beta }\) stands for the level of fear:
Now we consider the following function as the fear function for our model:
which satisfies all the conditions stated in (2.4). We can consider the fear function in many ways satisfying the conditions described in (2.4) and \(0\le \zeta \le 1.\)
2.3 Deterministic model
We take the functions \(f(\zeta , \beta , y)\) and h(x) as described in (2.5) and (2.3) respectively in system (2.2) and obtain the following system:
Let us consider \(\beta _{1} = g \beta\) and \(\theta = a_{1} \alpha\), then system (2.6) becomes:
with initial conditions \(x_0 > 0\) and \(y_0 > 0\).
2.4 Stochastic model
Introducing Gaussian white noise on birth rate and death rate of prey and predator respectively, the following stochastic system arises:
The parameters g and \(d_{2}\) have been perturbed by independent Gaussian white noise terms \(\gamma _{1}\) and \(\gamma _{2}\) in system (2.7) because these are the vital parameters subject to coupling the environment where the species live [37]. The terms \(\gamma _{1}\) and \(\gamma _{2}\) are independent Gaussian white noises characterised by:
\(\langle \gamma _{j}(t)\rangle = 0\) and \(\langle \gamma _{j}(t_{1})\gamma _{j}(t_{2})\rangle = \sigma ^{2}_{j}\delta _{j}(t_{1}-t_{2}),\) for \(j= 1,2.\)
having the respective intensities \(\sigma _{1}> 0 , \sigma _2 > 0\). The functions \(\delta _{j}(x)\) are the Dirac delta function defined as follows:
and \(\langle \cdot \rangle\) represents the ensemble average. System (2.7) becomes:
with initial conditions \(x_0 > 0\) and \(y_0 > 0\), where \(\displaystyle \gamma _{1} = \sigma _1 \frac{dw_1}{dt}\), \(\displaystyle \gamma _{2} = \sigma _2 \frac{dw_2}{dt}\) and two-dimensional standard Brownian motion is expressed as \(w = \left\{ w_1 , w_2 , t \ge 0\right\}\).
3 Positivity
Theorem 3.1
If \((x_0, y_0) \in {\mathbb {R}}_{+}^{2}\) be any initial value, then system (2.7) has global positive solution (x(t), y(t)) which is unique for all \(t \ge 0.\)
Proof
The right hand side of system (2.7) is continuous and locally Lipschitz on \({\mathbb {R}}_{+}^{2}\), the solution (x(t), y(t)) of system (2.7) exists and unique on \([0, \tau ]\), \(\tau \in (0,\infty )\). From (2.7):
Hence the theorem. \(\square\)
Lemma 3.1
[8] \(z \le 2(z + 1 - \log _{e} (z)) - 2(2 - \log _{e} (2)),\ \forall z> 0.\)
Theorem 3.2
For system (2.8): \((x(t), y(t)) \in {\mathbb {R}}_{+}^{2},\ \forall t> 0\), almost surely.
Proof
Since coefficients of system (2.8) satisfy local Lipschitz condition, for any \((x_0, y_0) \in {\mathbb {R}}_{+}^{2}\) there exists a unique local solution \(x(t), y(t) \in [0, \tau _{e})\), where \(\tau _e\) is the explosion time. To prove that it is a global positive solution, we have to show that \(\tau _{e} = \infty\). Let \(s_0 \ge 0\) be sufficiently large so that both \(x_0\) and \(y_0\) lie in the interval \(\displaystyle \left[ \frac{1}{s_0} , s_0 \right]\). We define stopping time \((\tau _s)\) for each integer \(s \ge s_0\) such that
with \(\inf \phi = \infty\) ( \(\phi\) denotes the empty set). It is easy to observe that \(\tau _s\) increases as \(t \rightarrow \infty\). Here we set \(\displaystyle \tau _{\infty } = \lim _{r \rightarrow \infty } \tau _s\), whence \(\tau _\infty \le \tau _e\) a.s. If it can be proved that \(\tau _\infty = \infty ,\) then it is easy to conclude that \(\tau _e = \infty\) and \((x(t), y(t)) \in {\mathbb {R}}^{2}_{+}\) for all \(t \ge 0\) almost surely. So, to complete the proof, we need to show that \(\tau _\infty = \infty\). It can be proved by contradiction. If possible, suppose the statement is false, then there exists a pair of constants \(T > 0\) and \(\epsilon \in (0, 1)\) such that
So, there exists an integer \(s_1 \ge s_0\) such that
Now we define a \({\mathcal {C}}^2\)-function \(G : {\mathbb {R}}^{2}_{+} \longrightarrow \mathbb {R_{+}}\) by
Since \((z + 1 - \log _{e}(z)) \ge 0 , \ \forall z >0\), so G(x, y) is positive.
Using Itô formula:
where, \(b_{1} = \left( g \zeta - d_{1} + \frac{\beta _{1} (1-\zeta )}{\beta } + r \right) , b_{2}= \left( \frac{\theta }{\eta } - d_{2} + c_{2} \right) \ \mathrm {and}\) \(b_{3}= - \left( g \zeta - d_{1} + \frac{\beta _{1} (1-\zeta )}{\beta } + \frac{\theta }{\eta } - d_{2} - \frac{\sigma _{1}^{2} \zeta ^{2} + \sigma _{2}^{2}}{2} \right)\)
Lemma 3.1 leads to the following result:
Let \(b_{4} = \max \left\{ 2b_{1}, 2b_{2}, b_{3} \right\}\). We define \(v_{1} \bigwedge v_{2} = \min \{ v_{1}, v_{2} \}\). Hence for \(t_1 \le T\),
Taking expectation on both sides, we get
By Gronwall inequality [24]:
where \(\displaystyle b_{5} = \left( G(x_0, y_0) + b_{4} T \right) e ^{b_{4}T}.\)
Define \(\displaystyle \Omega _{s} = \left\{ \tau _s \le T \right\}\) for \(\displaystyle s \ge s_{1}\) and by (3.1), \(P(\Omega _{s}) \ge \epsilon .\) Note that for each \(\tau ^{\prime } \in \Omega _{s}\), there exists at least one of \(x(\tau _s,\tau ^{\prime }), y(\tau _s, \tau ^{\prime } )\) which is equal either s or \(\displaystyle \frac{1}{s}\). So \(F\left( x\left( \tau _s, \tau ^{\prime }\right) , y\left( \tau _s, \tau ^{\prime }\right) \right)\) is not less than the smallest of
Consequently,
where \(1_{\Omega _{s}}\) is the indicator function of \(\Omega _{s}\). Therefore, \(s \rightarrow \infty\) leads to the contradiction \(\infty > b_{5} = \infty\). Hence \(\tau _{\infty } = \infty\).
\(\square\)
4 Boundedness
Let us discuss about boundedness of solutions of (2.8) and derive the conditions under which the solutions are bounded.
We define \(\displaystyle M_{1}(t) = \int _{0}^{t} \sigma _{1} dw_{1}, \ M_{2}(t) = \int _{0}^{t} \sigma _{2} dw_{2}\) are real valued continuous local martingales. Applying strong law of large numbers: \(\displaystyle \lim _{t \rightarrow \infty } \frac{M_{1}(t)}{t} = \lim _{t \rightarrow \infty } \frac{M_{2}(t)}{t} = 0.\)
Theorem 4.1
Let (x(t), y(t)) be a solution of system (2.8) with \((x_{0}, y_{0}) \in {\mathbb {R}}_{+}^{2}\), then \(E(x^{p}(t)) \le M(p), \forall p \ge 1\), where
and for \(\displaystyle \frac{\theta }{\eta } + \frac{p-1}{2}\sigma _{2}^{2} \le d_{2}\), \(\displaystyle E\left( y^{p}(t) \right) \le y_{0}^{p}.\)
Proof
From system (2.8), we have
We take \(V(x,t) = e^{t}x^{p}\) and apply Itô formula:
Let \(\displaystyle h(x) = x^{p} \left( \frac{1}{p} + (g \zeta -d_{1}) + \frac{\beta _{1}(1-\zeta ) }{\beta } - r x + \frac{(p-1)}{2} \zeta ^{2} \sigma _{1}^{2} \right)\)
Hence, \(\displaystyle h_{max} = \left( \frac{p}{r}\right) ^{p} \left[ \frac{ \frac{1}{p} + (g \zeta -d_{1}) + \frac{\beta _{1}(1-\zeta ) }{\beta } + \frac{(p-1)}{2} \zeta ^{2} \sigma _{1}^{2} }{p+1}\right] ^{p+1}\)
\(\therefore\) \(\displaystyle E(v(t)) \le x_{0}^{p} + p \left( \frac{p}{r}\right) ^{p} \left[ \frac{ \frac{1}{p} + (g \zeta -d_{1}) + \frac{\beta _{1}(1-\zeta ) }{\beta } + \frac{(p-1)}{2} \zeta ^{2} \sigma _{1}^{2} }{p+1}\right] ^{p+1} (e^{t} - 1)\)
So, for \(t=0\), \(E(x^{p}) \le x_{0}^{p}\) and for \(\displaystyle t \rightarrow \infty\)
\(E(x^{p}) \le p \left( \frac{p}{r}\right) ^{p} \left( \frac{ \frac{1}{p} + (g \zeta -d_{1}) + \frac{\beta _{1}(1-\zeta ) }{\beta } + \frac{(p-1)}{2} \zeta ^{2} \sigma _{1}^{2} }{p+1}\right) ^{p+1}\).
Now let
Hence, \(E(x^{p}) \le M(p).\)
From second equation of (2.8), we have
Let \(G(y)= \log _{e} (y)\) and applying Itô formula:
Now for \(\displaystyle \frac{\theta }{\eta } + \frac{p-1}{2}\sigma _{2}^{2} \le d_{2}\), \(\displaystyle E\left( y^{p}(t) \right) \le y_{0}^{p}.\)
Hence the theorem.
\(\square\)
5 Some groundworks
Theorem 5.1
Let (x(t), y(t)) be the solution of (2.8) with \((x_0, y_0) \in {\mathbb {R}}_{+}^{2},\) then
-
(i)
\(\ \phi (t) \le x(t) \le \Phi (t)\)
-
(ii)
\(\ \psi (t) \le y(t) \le \Psi (t)\),
where
Proof
From system (2.8), we have
Let us consider \(\Phi (t)\) be the unique solution of the following equation:
Considering \(\displaystyle H_{1}(t) = \frac{1}{\Phi (t)} \ \mathrm {with} \ H_{1}(0) = \frac{1}{x_{0}}\) and applying Itô formula, we get
After solving this stochastic differential equation, we get
Hence,
From system (2.8) we also have
Let us consider \(\Psi (t)\) be the unique solution of the following equation:
Considering \(\displaystyle H_{2}(t) = \frac{1}{\Psi (t)} \ \mathrm {with} \ H_{2}(0) = \frac{1}{y_{0}}\) and applying Itô formula, we get
After solving this stochastic differential equation, we get
Hence,
Again from (2.8), we have
Let us consider \(\psi (t)\) be the unique solution of the following equation:
Considering \(\displaystyle H_{3}(t) = \frac{1}{\psi (t)} \ \mathrm {with} \ H_{3}(0) = \frac{1}{y_{0}}\) and applying Itô formula, we get
After solving this stochastic differential equation, we get
Hence,
From first equation of system (2.8) and using (5.1), we also get
Let us consider \(\phi (t)\) be the unique solution of the following equation:
Considering \(\displaystyle H_{4}(t) = \frac{1}{\phi (t)} \ \mathrm {with} \ H_{4}(0) = \frac{1}{x_{0}}\) and applying Itô formula, we get
After solving this stochastic differential equation, we get
Now, from (5.1), (5.2), (5.3) and (5.4) we can conclude that
\(\ \phi (t) \le x(t) \le \Phi (t)\) and \(\ \psi (t) \le y(t) \le \Psi (t)\) Hence the theorem.
\(\square\)
Theorem 5.2
Let (x(t), y(t)) be a solution of system (2.8), then almost every sample path of (x(t), y(t)) is uniformly continuous on \(t\ge 0\) for any initial value \((x_0, y_0) \in {\mathbb {R}}_{+}^{2}\)if \(\displaystyle \frac{\theta }{\eta } + \frac{p-1}{2}\sigma _{2}^{2} \le d_{2}.\)
Proof
From system (2.8) we have:
We take \(\displaystyle \Gamma _{1}(x(t),y(t)) := x \left[ (g \zeta -d_{1}) + \frac{\beta _{1}(1-\zeta ) }{\beta + y} - r x - \frac{\alpha y}{1+ \eta x} - c_{1} x^{2} \right]\) and \(\displaystyle \Gamma _{2}(x(t),y(t)) := \zeta \sigma _{1} x.\)
Hence, \(\displaystyle dx = \Gamma _{1}(x(t),y(t)) dt + \Gamma _{2}(x(t),y(t)) dw_{1}.\)
Using Theorem 5.1 and \(A.M \ge G.M.\):
Also we have, \(\displaystyle E\left| \Gamma _{2}(x(t),y(t))\right| ^{p} \le \zeta ^{P} \sigma _{1}^{p} M(p) = H_{2}(p) \ (\mathrm {say})\)
Let us write the SDE in it’s stochastic integration form as follows:
From moment inequality [25] of Itô for \(0 \le t_1< t_2 < \infty\) and \(p \ge 2\), we have
Now we apply Hölder’s inequality for \(t_{2} - t_{1} \le 1\) and (5.5),
Hence it can be concluded that every sample path of x(t) is locally but uniformly Hölder continuous with exponent \(\gamma \in \left( 0 , \frac{p-2}{2p} \right)\). So, it can be concluded that every sample path of x(t) is uniformly continuous on \(t\ge 0\). Similarly, it can be shown that every sample path of y(t) is uniformly continuous on \(t\ge 0\).
\(\square\)
6 Extinction
A species is said to be extinct if there exists no member which can reproduce or create a new generation in the habitat in long run. That can happen for various environmental and artificial reasons. In the next theorem we shall show that extinction of prey population propel predator population towards extinction.
Definition 6.1
Population x(t) is said to be going extinct with probability one if
Theorem 6.1
Let (x(t), y(t)) be the solution of system (2.8). Then the prey and predator both population go extinct in long run if \(\displaystyle g\zeta + \beta _{1}(1-\zeta ) < d_{1} + \frac{\sigma _{1}^{2} \zeta ^{2}}{2}\), that is,
Proof
We take \(u(x) = \log (x)\) and apply Itô formula on the first equation of system (2.8):
Let \(\displaystyle h(x) = (g \zeta -d_{1}) + b_{1}(1-\zeta ) - rx - c_{1}x^{2} - \frac{\zeta ^{2}\sigma _{1}^{2}}{2}\)
So, \(\displaystyle h^\prime (x) = -r - 2c_{1} x < 0 .\) i.e., h(x) is a deceasing function. Hence, the supremum occurs at \(x=0\).
\(\displaystyle \sup _{x\ge 0} h(x) = (g \zeta -d_{1}) + b_{1}(1-\zeta ) - \frac{\zeta ^{2}\sigma _{1}^{2}}{2}.\)
Hence,
Therefore, for every \(\epsilon > 0\) there exists \(t_{0} ( > 0 )\) and set \(\Omega _{\epsilon }\) such that \(P(\Omega _{\epsilon }) \ge 1- \epsilon\) and \(x < \epsilon\) for every \(t \ge t_{0}\) and \(x \in \Omega _{\epsilon }.\)
Hence \(\displaystyle \lim _{t\rightarrow \infty } x(t) = 0 \ \mathrm {a.s.}\)
From the second equation of (2.8):
Consider \(v(x) = \log _{e}(y)\) and use Itô formula:
Since, \(\epsilon (>0)\) is arbitrarily small, therefore \(\displaystyle \limsup _{t \rightarrow \infty } \frac{\log _{e} y(t)}{t} < 0 .\) So, we can conclude that \(\displaystyle \lim _{t\rightarrow \infty } y(t) = 0 \ \mathrm {a.s.}\) \(\square\)
7 Persistent
In this section the persistence of system (2.8) will be discussed. First we shall define persistence in mathematical terms.
Definition 7.1
System (2.8) is said to be persistent in mean if \(\ \displaystyle \liminf _{ t \rightarrow \infty } \left\langle y \right\rangle _{t} > 0\), where \(\displaystyle \left\langle y \right\rangle _{t} = \frac{1}{t} \int _{0}^{t} y(s) ds.\)
Lemma 7.1
[7] Suppose \(Z(t) \in {\mathbb {C}} ( \Omega \times [0,\infty ), {\mathbb {R}}_{+}).\)
- (a):
-
If there exist \(T, \delta , \delta _{0} \in {\mathbb {R}}_{+}\) such that \(\displaystyle \log _{e} Z(t) \le \delta t - \delta _{0} \int _{0}^{t} Z(s) ds + \sum \limits _{i=1}^{n} \alpha _{i} W(t) \ \ \mathrm {a.s.} \ \forall t \ge T,\) where \(\alpha _{i}\) are constants for \(i = 1,2, ... ,n,\) then
$$\begin{aligned} \left\{ \begin{array}{ll}\displaystyle {\limsup _{t\rightarrow \infty } \left\langle Z \right\rangle _{t} \le \frac{\delta }{\delta _{0}} },\ \mathrm {a.s.}\ \ \ \ \ \mathrm {if} \ \ \delta > 0,\\ \displaystyle \lim _{t\rightarrow \infty } \left\langle Z \right\rangle _{t} = 0, \ \mathrm {a.s.}\ \ \ \ \ \ \ \ \ \ \mathrm {if} \ \ \delta < 0. \end{array}\right. \end{aligned}$$ - (b):
-
If there exist \(T, \delta , \delta _{0} \in {\mathbb {R}}_{+}\) such that
$$\begin{aligned} \displaystyle \log _{e} Z(t) \ge \delta t- \delta _{0} \int _{0}^{t} Z(s) ds+ \sum \limits _{i=1}^{n} \alpha _{i} W_{i}(t) \ \ \ \ a.s. \ \ \forall \ t \ge T, \end{aligned}$$where \(\alpha _{i}\) are constants for \(i = 1,2,...,n,\) then
$$\begin{aligned} \liminf _{t\rightarrow \infty }\left\langle Z \right\rangle _{t} \ge \frac{\delta }{\delta _{0}} \ \ \ \mathrm {a.s.} \end{aligned}$$
Lemma 7.2
[20] Consider the following one dimensional stochastic system:
- (a):
-
If \(\displaystyle \left( g\zeta + \frac{\beta _{1}(1-\zeta )}{\beta }- d_{1} - \frac{\zeta ^{2}\sigma _{1}^{2}}{2} \right) < 0,\) then \(\displaystyle \lim _{ t \rightarrow \infty } z(t) = 0\) a.s.
- (b):
-
If \(\displaystyle \left( g\zeta + \frac{\beta _{1}(1-\zeta )}{\beta }- d_{1} - \frac{\zeta ^{2}\sigma _{1}^{2}}{2} \right) > 0,\) then \(\displaystyle \lim _{ t \rightarrow \infty } \frac{1}{t} \int _{0}^{t} z(s) ds = g\zeta + \frac{\beta _{1}(1-\zeta )}{\beta }- d_{1} - \frac{\zeta ^{2}\sigma _{1}^{2}}{2}\) and \(\displaystyle {\mathbb {P}} \left\{ \lim _{t\rightarrow \infty }\frac{1}{t} \int _{0}^{t} f(z(s))ds = \int _{{\mathbb {R}}_{+}} f(x) \mu (x) dx \right\} = 1\), where \(\mu (x)\) is the stationary density.
Theorem 7.1
Let us consider \(\displaystyle \left( g\zeta + \frac{\beta _{1}(1-\zeta )}{\beta }- d_{1} - \frac{\zeta ^{2}\sigma _{1}^{2}}{2} \right) > 0\) and \(\displaystyle \Lambda = \theta \int _{0}^{\infty } \frac{z}{1+ \eta z}\mu (x) dx - d_{2} - \frac{\sigma _{2}^{2}}{2}.\) If (x(t), y(t))be a solution of system (2.8) for any \((x_0, y_0) \in {\mathbb {R}}_{+}^{2},\) then system (2.8) is persistent in mean provided \(\Lambda > 0\).
Proof
From the first equation of system (2.8), applying Itô formula, we have
Now integrating both sides and dividing by t, we get
Applying Itô formula on (7.1), we have
Now integrating both sides and dividing by t, we get
From, (7.2) and (7.3) it can be easily observed that \(F_{1}(t) \le F_{2}(t)\)
Hence,
From second equation of system (2.8), applying Itô formula, we have
Integrating both side and dividing by t, we get
For sufficiently large t, using Lemma 7.1(b) and arbitrariness of \(\epsilon\):
Therefore, from the definition of persistence of a system, it can be concluded that system (2.8) is persistence if \(\Lambda > 0\). Hence the theorem.
\(\square\)
8 Numerical simulations
This section deals with numerical findings to validate theoretical outcomes that the previous sections offered. We approximate the solution of stochastic system (2.8) by numerical simulation using Euler Maruyama method in MATLAB.
To simulate the system, we set the values of biological and environmental parameters as follows:
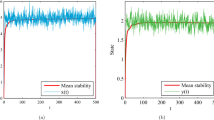
We have started the simulating system (2.8) from the initial point (1.7, 0.72). It is found the effects of environmental noise on prey and predator population and observed that after some initial transients the biomass of prey population varies around 0.6 and predator population varies around 0.3 which are depicted in (1.a) and (1.b) of Fig. 1 respectively.
Extinction of prey and predator population under the stated condition in Theorem 6.1
In theoretical study, it is found that prey extincts if \(\displaystyle g\zeta + \beta _{1}(1-\zeta ) < d_{1} + \frac{\sigma _{1}^{2} \zeta ^{2}}{2}\) and extinction of prey propels predator population towards extinction. To satisfy the condition we take \(g = 0.5\) and \(d_{1} = 0.55\) i.e., \(\beta _{1} = g \beta = 0.5\) and simulate system (2.8) in MATLAB. It is found in numerical simulation also that extinction of prey causes immediate extinction of predator population which is depicted in Fig. 2.
Theorem 7.1 affirms the persistence of system (2.8) under some conditions which are satisfied by the given set of values in Table 1. Figure 3 validates the mathematical result.
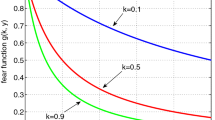
In this work, the fear function has been considered as \(\displaystyle f(\zeta ,\beta ,x)= \zeta +\frac{1-\zeta }{1+\frac{y}{\beta }}\) where \(\zeta\) represents the minimum fear level and \(\frac{1}{\beta }\) is the cost of fear. We have tested the system for high fear level, i.e., for a low value of \(\beta\). Value of \(\beta\) is considered as 0.1, i.e., \(\frac{1}{\beta } =10\) and \(\beta _{1}=g\beta =0.1\) and it can be observed in Fig. 4 that fear affects negatively on predator population.
To test if the cost of fear is very high we have taken \(\frac{1}{\beta } = 100\), i.e., \(\beta =0.01\) and \(\beta _{1}=0.01\). In Fig. 5 we observe that prey population persists but predator population extincts after a certain period of time.
Only for a lower fear level, say, for \(\frac{1}{\beta }=0.0001\) and \(\zeta =0.1\), it is observed that the system persists which is a very normal situation in reality. This behavior is shown in Fig. 6.
System (2.8) persists in presence of minimum fear only
We have tested the effects of toxicity numerically. In all the figures from Figs. 1, 2, 3, 4, 5 and 6, values of \(c_{1}\) and \(c_{2}\) have been considered as 0.2 and 0.15 respectively. Now we consider values of \(c_{1}\) and \(c_{2}\) as 1.5 and 0.8 in Fig. 7 and 3.5 and 2.8 in Fig. 8 respectively. Here it is observed that both prey and predator population decrease but mostly predator population gets affected.
In Fig. 8, it is observed that prey population decreases and predator population is going to extinct after a certain period of time.
If we consider the system in a toxic free environment, i.e., \(c_{1}=0\) and \(c_{2}=0\), then it is observed that prey–predator population persists together which is depicted in Fig. 9.
System (2.8) persists for \(c_{1} = 0\) and \(c_{2}=0\)
9 Conclusion
In this work we have considered a Lotka–Volterra predator–prey model where prey species is directly infected by some external toxic substances while the predator is indirectly affected for feeding on these infected prey. This model also involves fear of predator felt by prey. Birth rate of prey and death rate of predator are considered as stochastic parameters and they are perturbed by introducing Gaussian white noise. Existence of unique global positive solutions are established for both deterministic and stochastic systems. It is found analytically that environmental noise plays an important role in the extinction of both the species. In numerical simulation through Fig. 2 this analytical finding is justified. Boundedness and uniform continuity criteria of the solution of the underlying system (2.8) are also derived. We have also established most useful criteria of persistence of this system under some conditions.
In numerical simulations, it is observed how fear factor affects the system. It is also found that fear factor affects negatively on predator population (see Figs. 4, 5). It is observed in Fig. 5 that predator population is going to extinct for a very high cost of fear. It is shown in Fig. 6 that existence of only a lower fear level makes the system persistent.
Numerically, we have also observed the effects of toxicity. High toxicity affects both the species (see Figs. 7, 8). In Fig. 8 it is found that extremely high toxicity can be a cause of extinction of predator. In Fig. 9 we have found that the system persists if it is free from toxicity.
References
Altendorf, K.B., Laundre, J.W., Gonzalez, C.A.L., Brown, J.S.: Assessing effects of predation risk on foraging behavior of mule deer. J. Mammal. 82(2), 430–439 (2011)
Boonstra, R., Hik, D., Singleton, G.R., Tinnikov, A.: The impact of predator-induced stress on the snowshoe hare cycle. Ecol. Monogr. 68, 371–394 (1998). https://doi.org/10.2307/2657244
Creel, S., Christianson, D., Liley, S., Winnie Jr., J.A.: Predation risk affects reproductive physiology and demography of elk. Science 315(5814), 960 (2007). https://doi.org/10.1126/science.1135918
Creel, S., Christianson, D.: Relationships between direct predation and risk effects. Trends Ecol. Evol. 23, 194–201 (2008)
Cresswell, W.: Predation in bird populations. J. Ornithol. 152, 251–263 (2011)
Das, A., Samanta, G.P.: Stochastic prey–predator model with additional food for predator. Phys. A Stat. Mech. Appl. 512, 121–141 (2018). https://doi.org/10.1016/j.physa.2018.08.138
Das, A., Samanta, G.P.: Modelling the fear effect on a stochastic prey–predator system with additional food for predator. J. Phys. A Math. Theor. 51, 465601 (2018). https://doi.org/10.1088/1751-8121/aae4c6
Das, A., Samanta, G.P.: A prey–predator model with refuge for prey and additional food for predator in a fluctuating environment. Phys. A Stat. Mech. Appl. 538, 122844 (2020). https://doi.org/10.1016/j.physa.2019.122844
Das, M., Samanta, G.P.: A delayed fractional order food chain model with fear effect and prey refuge. Math. Comput. Simul. 178, 218–245 (2020a)
Das, M., Samanta, G.P.: A prey–predator fractional order model with fear effect and group defense. Int. J. Dyn. Control (2020b). https://doi.org/10.1007/s40435-020-00626-x
Dubey, B., Hussain, J.: A model for the allelopathic effect on two competing species. Ecol. Modell. 129, 195–207 (2000)
Eggers, S., Griesser, M., Ekman, J.: Predator-induced plasticity in nest visitation rates in the Siberian jay (Perisoreus infaustus). Behav. Ecol. 16(1), 309–315 (2005). https://doi.org/10.1093/beheco/arh163
Ghalambor, C.K., Peluc, S.I., Marti, T.E.: Plasticity of parental care under the risk of predation: how much should parents reduce care? Biol. Lett. 9(4), 20130154 (2013). https://doi.org/10.1098/rsbl.2013.0154
Ghosh, J., Sahoo, B., Poria, S.: Prey–predator dynamics with prey refuge providing additional food to predator. Chaos Solitons Fract. 96, 110–119 (2017)
Hallam, T.G., Clark, C.W.: Non-autonomous logistic equations as models of populations in a deteriorating environment. J. Theor. Biol. 93, 303–311 (1982)
Hallam, T.G., Luna, T.J.: Effects of toxicants on populations: a qualitative approach III, environmental and food chain pathways. Theor. Biol. 109, 411–429 (1984)
Holling, C.S.: The functional response of predator to prey density and its role in mimicry and population regulation. Mem. Ent. Soc. Can. 45, 5–60 (1965)
Hua, F., Fletcher, R.J., Sieving, K.E., Dorazio, R.M.: Too risky to settle: avian community structure changes in response to perceived predation risk on adults and offspring. Proc. R. Soc. B Biol. Sci. 280(1764), 20130762 (2013). https://doi.org/10.1098/rspb.2013.0762
Hua, F., Sieving, K.E., Fletcher, R.J., Wright, C.A.: Increased perception of predation risk to adults and offspring alters avian reproductive strategy and performance. Behav. Ecol. 25(3), 509–519 (2014). https://doi.org/10.1093/beheco/aru017
Ji, C., Jiang, D.: Dynamics of a stochastic density dependent predator–prey system with Beddington–DeAngelis functional response. J. Math. Anal. Appl. 381, 441–453 (2011)
Kar, T.K., Chaudhuri, K.S.: On non-selective harvesting of two competing fish species in the presence of toxicity. Ecol. Modell. 161, 125–137 (2003)
Krapivsky, P.L., Redner, S.: Kinetics of a diffusive capture process: lamb besieged by a pride of lions. J. Phys. A Math. Gen. 26(17). http://stacks.iop.org/0305-4470/29/i=17/a=011 (1996)
Lande, R.: Risks of population extinction from demographic and environmental stochasticity and random catastrophes. Am. Nat. 142, 911–927 (1993)
Mao, X.: Stochastic Differential Equations and Applications. Horwood, New York (1997)
Mao, X.: Stochastic Differential Equations and Applications. Woodhead Publishing, Oxford (2011)
May, R.M.: Stability in randomly fluctuating deterministic environments. Am. Nat. 107, 621–650 (1973)
Mondal, S., Maiti, A., Samanta, G.P.: Effects of fear and additional food in a delayed predator–prey model. Biophys. Rev. Lett. 13(4), 157–177 (2018)
Mondal, S., Samanta, G.P.: Dynamics of a delayed predator–prey interaction incorporating nonlinear prey refuge under the influence of fear effect and additional food. J. Phys. A Math. Theor. 53, 295601 (2020)
Mondal, S., Samanta, G.P.: Time-delayed predator–prey interaction with the benefit of antipredation response in presence of refuge. Z. Naturforschung A (2020). https://doi.org/10.1515/zna-2020-0195
Pangle, K.L., Peacor, S.D., Johannsson, O.E.: Large nonlethal effects of an invasive invertebrate predator on zooplankton population growth rate. Ecology 88, 402–412 (2007)
Peacor, S.D., Peckarsky, B.L., Trussell, G.C., Vonesh, J.R.: Costs of predator-induced phenotypic plasticity: a graphical model for predicting the contribution of nonconsumptive and consumptive effects of predators on prey. Oecologia 171(1), 1–10 (2013). https://doi.org/10.1007/s00442-012-2394-9
Peckarsky, B.L., Cowan, C.A., Penton, M.A., Anderson, C.: Sublethal consequences of streamdwelling predatory stoneflies on mayfly growth and fecundity. Ecology 74, 1836–1846 (1993). https://doi.org/10.2307/1939941
Pettorelli, N., Coulson, T., Durant, S.M., Gaillard, J.M.: Predation, individual variability and vertebrate population dynamics. Oecologia 167(2), 305–314 (2011). https://doi.org/10.1007/s00442-011-2069-y
Preisser, E.L., Bolnick, D.I.: The many faces of fear: comparing the pathways and impacts of nonconsumptive predator effects on prey populations. PlOS One 3(6), e2465 (2008). https://doi.org/10.1371/journal.pone.0002465
Samanta, G.P.: Influence of environmental noises on the Gomatam model of interacting species. Ecol. Modell. 91(1–3), 283–291 (1996)
Samanta, G.P.: A two-species competitive system under the influence of toxic substances. Appl. Math. Comput. 216(1), 291–299 (2010)
Samanta, G.P., Maiti, A.: Stochastic Gomatam model of interacting species: non-equilibrium fluctuation and stability. Syst. Anal. Modell. Simul. 43, 683–692 (2003)
Schwarzl, M., Godec, A., Oshanin, G., Metzler, R.: A single predator charging a herd of prey: effects of self volume and predator–prey decision-making. J. Phys. A Math. Theor. 49, 225601 (2016)
Smith, M.J.: Models in Ecology, p. 146. Cambridge University Press, Cambridge (1974)
Suraci, J.P., Clinchy, M., Dill, L.M., Roberts, C., Zanette, L.Y.: Fear of large carnivores causes a trophic cascade. Nat. Commun. (2016). https://doi.org/10.1038/ncomms10698
Wang, X., Zanette, L.Y., Zou, X.: Modelling the fear effect in predator–prey interactions. J. Math. Biol. 73(5), 1179–1204 (2016). https://doi.org/10.1007/s00285-016-0989-1
Wang, X., Zou, X.: Modeling the fear effect in predator–prey interactions with adaptive avoidance of predators. Bull. Math. Biol. 79, 1325–1359 (2017). https://doi.org/10.1007/s11538-017-0287-0
Wirsing, A.J., Heithaus, M.R., Dill, L.M.: Living on the edge: dugongs prefer to forage in microhabitats that allow escape from rather than avoidance of predators. Anim. Behav. 74(1), 93–101 (2007). https://doi.org/10.1016/j.anbehav.2006.11.016
Wirsing, A.J., Ripple, W.J.: A comparison of shark and wolf research reveals similar behavioural responses by prey. Front. Ecol. Environ. 9(6), 335–341 (2011). https://doi.org/10.1890/090226
Zanette, L.Y., White, A.F., Allen, M.C., Clinchy, M.: Perceived predation risk reduces the number of offspring songbirds produce per year. Science 334(6061), 1398–1401 (2011). https://doi.org/10.1126/science.1210908
Acknowledgements
The authors are grateful to the anonymous referees, Dr. Pasquale Vetro, Editor-in-Chief, for their careful reading, valuable comments and helpful suggestions, which have helped them to improve the presentation of this work significantly.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Das, A., Samanta, G.P. Modelling the fear effect in a two-species predator–prey system under the influence of toxic substances. Rend. Circ. Mat. Palermo, II. Ser 70, 1501–1526 (2021). https://doi.org/10.1007/s12215-020-00570-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12215-020-00570-x