Abstract
In this paper, a new version of liquid–liquid microextraction based on heat-induced homogeneous liquid–liquid microextraction has been developed for the extraction and preconcentration of some neonicotinoid insecticides from fruit juice and vegetable samples followed by their determination by high-performance liquid chromatography–diode array detection. In this method, cyclohexyl amine is dissolved in an aqueous solution thermostated at 0 °C. Then, the solution is transferred into a heated water bath. During heating, the solubility of cyclohexyl amine in water is decreased and the produced tiny droplets of the extraction solvent (cyclohexyl amine) are dispersed in whole parts of the solution. The obtained cloudy solution is centrifuged. Finally, an aliquot of the extractant is taken and injected into the separation system for analysis. Various parameters affecting the extraction efficiency such as volume of extraction solvent, temperature, salt addition, and centrifuging rate and time were evaluated. Under the optimum conditions, enrichment factors were obtained in the range of 180–235. The linear ranges of the calibration curves were wide. Limits of detection were in the ranges of 0.38–1.31 and 0.19–0.69 ng mL−1, in vegetable samples and fruit juices, respectively. Limits of quantification were in the ranges of 1.23–4.09 and 0.64–1.15 ng mL−1 in the mentioned samples. Finally, the proposed method was applied to analyze the target analytes in different fruit juice and vegetable samples.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In recent decades, food production quality and the consciousness of health and safety in world have increased. On the other hand, use of insecticides in agriculture is necessary to prevent or control of fruit diseases or weeds (Wang et al. 2012; Amadeo et al. 1996). Neonicotinoid insecticides are one of the most growing insecticides which are active against numerous sucking and biting pests and insects (Shuaihua et al. 2012). These insecticides have developed rapidly since the first advent of imidacloprid in 1991 (Farajzadeh et al. 2015). Neonicotinoids include cyano-substituted (acetamiprid and thiacloprid) and nitro-substituted (dinotefuran, nitenpyram, thiamethoxam, imidacloprid, and clothianidin) compounds (Pavle et al. 2015). Due to wide spreading of neonicotinoids on agricultural land, there are of great notifications to serious risks for the health and safety of the consumers. The amended European Union legislation has set the maximum residue limits (MRLs) for neonicotinoid insecticides in different agricultural products. Their MRLs for fruits, vegetables, and cereals are between 0.1 and 1.0 mg kg−1 (Commission Directive 2007). Neonicotinoid insecticides are not suitable for direct analysis using gas chromatography (GC) due to their high polarity and low volatility (Wang et al. 2012). Therefore, high-performance liquid chromatography (HPLC) equipped with different detectors such as diode array (Serenella et al. 2008; Garrido Frenich et al. 2000), ultraviolet (Rosales-Conrado et al. 2002; Carabias Martinez et al. 1992), mass spectrometry (Gbylik-Sikorska et al. 2015), and fluorimetry (Antonio et al. 1999; Garcia et al. 2007) is preferred for them. Although HPLC is a selective and sensitive method, due to low concentration of neonicotinoids in different samples and the complexity of sample matrices, a sample preparation is still required. Sample preparation is an essential step to separate the target analytes from complex matrices and concentrate them to obtain high selectivity and sensitivity (Huahua et al. 2009). Liquid–liquid extraction (LLE) (Liu et al. 2010) and solid phase extraction (SPE) (Fang et al. 2012) are the most commonly used sample preparation techniques. These procedures are expensive and time-consuming and require large volumes of hazardous organic solvents. To avoid these difficulties, solid phase microextraction (SPME) (Tsoutsi et al. 2006; Xianlei et al. 2012; Dimitra and Triantafyllos 2001) has been developed. Although SPME does not use an extraction solvent, it is time-consuming and labor-intensive. Recently, several improved liquid phase microextraction (LPME) techniques having high extraction efficiencies were reported. LPME is a miniaturized sample preparation method which was firstly introduced in 1996 (Liu and Dasgupta 1996). This method is a preconcentration method in which the analytes are extracted from an aqueous solution into microliter level of an organic solvent. Different modes of LPME including single drop microextraction (SDME) (Yogesh and Dhananjay Kumar 2010), hollow fiber-liquid phase microextraction (HF-LPME) (Cai et al. 2016), dispersive liquid–liquid microextraction (DLLME) (Farajzadeh et al. 2009a, b), solidification of floating organic drop microextraction (SFODME) (Pelit and Yengin 2014) air-assisted liquid-liquid microextraction (AALLME) (Farajzadeh and Afshar Mogaddam 2012), etc. have been reported. A major development in sample preparation involves quick, easy, cheap, effective, rugged, and safe approach (QuEChERS) for insecticide residue analysis from different samples. This method is a simple and straightforward sample preparation technique suitable for neonicotinoid insecticides from food and agricultural products (Dankyi et al. 2015; Jiao et al. 2016). However, its major disadvantage is the low enrichment factor (EF) that can be achieved (Koesukwiwat et al. 2010). Another extraction procedure, namely homogeneous liquid–liquid extraction (HLLE), extracts the desired analyte existing in the homogeneous aqueous solution into a water-miscible organic phase which then is separated by one phase separation phenomenon. Temperature or salt addition has been already studied for phase separation (Murata et al. 1972; Farajzadeh et al. 2016). However, the HLLE method mentioned above usually require a large amount of hazardous and volatile organic solvents. In addition, a large amount of sample is often required and its handling can be time-consuming besides being tedious, although the methods are often effective. Therefore, there is a trend towards less organic solvent consumption, rapidity, simplicity, and miniaturization of the extraction methods. Subsequently, homogeneous liquid–liquid microextraction (HLLME) has been developed in recent years (Yazdanfar et al. 2014).
The main goal of this study was to develop a new sample preparation method by performing a heat-induced HLLME method. The extraction capability of the developed method is evaluated using neonicotinoid insecticides as model analytes from different samples. It is noted that the mentioned insecticides are frequently used in agricultural activities in Iran. Therefore, their residues may be found in vegetables and fruit juices. In this method, initially, an organic solvent is dissolved in an aqueous sample solution to achieve a homogeneous solution at low temperature. Then, the solution is heated in a water bath. By this action, the extraction solvent is regenerated and dispersed in whole parts of the solution. During this stage, the target analytes are extracted into the extraction solvent. To the best of our knowledge, this is the first report on the performing of a heat-induced HLLME technique. It is noted that the former temperature-induced HLLME methods have been carried out by cooling. To achieve high extraction efficiency, different parameters will be studied and optimized.
Experimental
Chemicals and Reagents
The studied insecticides including acetamiprid, imidacloprid, and thiamethoxam with purity higher than 98% were kindly provided by GYAH Corporation (Karadj, Iran). Analytical-grade sodium chloride, 1,2-dibromoethane (1,2-DBE), chloroform, and cyclohexyl amine were obtained from Merck (Darmstadt, Germany). HPLC-grade water and acetonitrile (ACN) were purchased from Chemlab (Zedelgem, Belgium). A stock solution of insecticides (100 mg L−1 of each analyte) was prepared by dissolving a specific amount of each insecticide in ACN. Working solutions were prepared daily by appropriate dilutions of the stock solution with HPLC-grade water.
Samples
The vegetable samples including beet, cucumber, potato, and tomato were purchased from a local store (Tabriz, Iran). The samples were squeezed by a juice extractor (JE600T, Kenwood, England). The obtained juices were centrifuged at 2191×g for 6 min, and then, the supernatants were diluted at a ratio of 1:4 with HPLC-grade water and then subjected to the proposed procedure. The fruit juice samples including apple, grape, and sour cherry from different brands were purchased from local supermarkets (Tabriz, Iran). All fruit juices were diluted at a ratio of 1:2 with HPLC-grade water and exposed to the proposed microextraction method.
Apparatus
Quantitative analysis of the selected insecticides was performed on a Hewlett–Packard 1090–II liquid chromatograph (Palo Alto, CA, USA) equipped with a diode array detector (DAD). An Alltech Alltima analytical C18 column (150 × 4.6 mm id, 5 μm particle size) (Fisher Scientific, Massachusetts, USA) was used for the separation. The selectivity and sensitivity of HPLC-DAD depend on mobile phase composition and its flow rate (Meyer 1994). Optimization of these parameters was performed by direct injection of the target analytes at a concentration of 100 mg L−1 (each analyte). For this purpose, ACN and water compositions were altered at the ratios of 35:65, 30:70, 25:75, and 20:80, v/v. The obtained results showed that better resolution factor was obtained at the ratio of 25:75, v/v, ACN/water. Flow rate of the selected mobile phase was studied at the range of 0.7–1.3 mL min−1, and suitable result was obtained at 1 mL min−1. Therefore, the mobile phase was ACN/water (25:75, v/v) mixture delivered at a flow rate of 1 mL min−1. Monitoring of the analytes was done at 271 nm for imidacloprid, 253 nm for thiamethoxam, and 244 nm for acetamiprid. Data acquisition and processing were done using ChemStation software. A D-7200 centrifuge from Hettich (Kirchlengern, Germany) was used in the sample preparation.
Microextraction Procedure
In 5 mL HPLC-grade water spiked with 100 ng mL−1 of each analyte or sample solution (see “Samples” Section), 1.00 g NaCl (20%, w/v) was dissolved into a 15-mL glass test tube with conical bottom. The solution was placed into an ice bath for 2 min, and then 89 μL cyclohexyl amine (as the extraction solvent) was added. By manual shaking, a homogeneous solution was formed. After that, the tube was transferred into a water bath thermostated at 70 °C for 3 min. During this step, the solubility of cyclohexyl amine into water was decreased and a cloudy solution due to formation of the fine droplets of cyclohexyl amine was obtained. Subsequently, the cloudy solution was centrifuged at 1610×g for 5 min, and the aqueous phase was removed by a 5-mL syringe. The organic phase collected at the bottom of the tube was 20 ± 1 μL. Finally, 10 μL of the extractant was taken and injected into the HPLC–DAD system for analysis.
Calculation of EF and ER
EF was calculated as the ratio of the target analyte concentration in the collected organic phase (C coll) to its initial concentration in aqueous solution (C 0) (Farajzadeh et al. 2016):
Extraction recovery (ER) is defined as the percentage of the total amount of analyte that is extracted into the collected organic phase. The following equation was used for ER calculation (Zhang et al. 2012).
where V coll and V aq represent the volumes of the collected organic phase and the aqueous solution, respectively. Also, n coll and n 0 are the amount of analyte in the collected organic phase and the initial aqueous solution, respectively.
Results and Discussion
To obtain the optimum experimental conditions of the proposed method, some important variables that can affect efficiency of the method including type and volume of extraction solvent, ionic strength, and extraction temperature and time should be optimized. The peak areas were employed as the responses in the optimization of the procedure.
Selection of Extraction Solvent Type
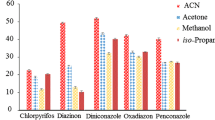
The selection of an extraction solvent is an important parameter in the extraction efficiency of the developed method. The properties of extraction solvent should meet the following requirements: (1) immiscible with water, (2) low toxicity, (3) good solubility for the analytes, and (4) good chromatographic behavior. On the other hand, considering this fact that the developed method is a heat-induced HLLE method and dispersion of the extraction solvent is performed in an elevated temperature, the main parameter in the selection of the extraction solvent is decreasing extraction solvent solubility in the aqueous phase by temperature increasing. Therefore, based on the mentioned requirements and preliminary experiments, three different extractants including chloroform, 1,2-DBE, and cyclohexyl amine were tested as the extraction solvents. In all experiments, the temperature of the aqueous solution containing 25 ng mL−1 of each analyte was adjusted at 0 °C, and then 26 μL of 1,2-DBE, 70 μL of chloroform, and 145 μL of cyclohexyl amine were added, separately. Different volumes for various solvents were used to obtain a similar volume (20 ± 1) for the collected phase. It is mentioned that the solvents have different solubilities in water, and it is necessary that different initial volumes of them are used to obtain the same volume for the collected phase. By manual shaking, the homogenous solutions were obtained. Then, the solutions were heated at 40 °C in a water bath for 3 min. By this action, the solubility of the used extraction solvents was decreased and the extraction solvents were dispersed as tiny droplets in whole parts of the solution. Figure 1 shows the variations of ER vs. extraction solvent type. According to the obtained data, cyclohexyl amine is the most effective extraction solvent and gives the highest extraction efficiencies for the target analytes among the tested solvents. Then, it was selected as the extraction solvent in the subsequent experiments.
Selection of extraction solvent type. Extraction conditions: aqueous sample volume, 5 mL deionized water spiked with 25 ng mL−1 of each analyte adjusted at 0 °C; extraction solvent, 1,2 DBE (26 μL), chloroform (70 μL), and cyclohexyl amine (145 μL); water bath temperature, 40 °C; heating time, 3 min; without salt addition; centrifuge rate, 1610×g; and centrifuge time, 5 min. The error bars show the minimum and maximum of three repeated determinations
Study of Extraction Solvent Volume
The volume of extraction solvent is an essential parameter in all LPME methods which is effective on the analytical signals. Generally, the extraction solvent volume is selected as small as possible in order to attain the high EFs and the least toxicity hazards for the environment. On the other hand, the collected organic phase volume after performing the method should be enough for the subsequent analysis. In order to select the extraction solvent volume, different volumes of cyclohexyl amine (120, 135, 145, 160, and 170 μL) were subjected to the same extraction procedure. The obtained results indicated that the analytical signals increased with decreasing the extraction solvent volume in the tested range. However, the volume of the collected organic phase decreased from 45 to 13 μL with reducing the volume of the extraction solvent from 170 to 120 μL. As a result, at low volumes of the extraction solvent, high analytical signals are attainable. But, it is remarkable that in the case of 120 μL cyclohexyl amine, the volume of the collected organic phase was low and the repeatability of the method was not suitable. Thereby, in the following studies, the volume of the extraction solvent was selected 135 μL in which 20 ± 1 μL organic phase was collected after performing the method.
Heating Temperature and Time
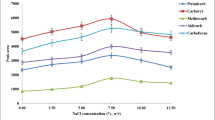
The main parameter in the performing of this method is the temperature of aqueous phase. In this method, the solubility of the selected extraction solvent is decreased at high temperatures and the extractant is dispersed in the whole parts of the solution as tiny droplets. On the other hand, the aqueous solution temperature can affect the diffusion and distribution coefficients of the selected analytes, and it is a driving force for better dispersion of the extraction solvent into the aqueous solution. High temperatures increase the contact area between the extractant and the sample increases, and hence mass transfer rates of the analytes are improved. The optimization of the aqueous phase temperature was carried out using a series of experiments performed at the temperature range of 0–90 °C at a constant extraction time (3 min). It is noted that at the temperatures less than 30 °C, the solution was not turbid and the method became useless. Based on the results in Fig. 2 at temperatures higher than 30 °C, the extraction efficiency of the method increases until 70 °C and then remains constant. So, 70 °C was selected in the subsequent experiments.
Effect of extraction temperature on the extraction efficiency. Extraction conditions are the same as used in Fig. 1, except that (135 μL) cyclohexyl amine was used as the extraction solvent. The error bars show the minimum and maximum of three repeated determinations
On the other hand, the heating time is another important parameter in the efficiency of the developed method. To evaluate the effect of heating time on the performance of the method, the aqueous solution temperature was adjusted at 70 °C by its placing into the water bath for 0.5, 1.0, 3.0, 5.0, 7.0, and 10 min. The results indicated that by increasing the heating time until 3 min, the analytical signals increased and then remained constant. Consequently, 3 min was selected as the suitable heating time for the next experiments.
Salt Addition Effect
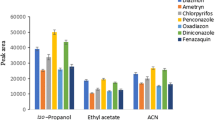
The influence of ionic strength was investigated by adding different concentrations of NaCl (0–30%, w/v). The addition of a salt to the sample solution increases the ionic strength of the aqueous phase which leads to decreasing the solubility of the analytes in the sample solution. This promotes their transferring into the organic phase because of the salting out effect. In order to obtain a constant volume ratio of the aqueous phase to the organic phase, the experiments were performed using different volumes of the extraction solvent to achieve 20 μL of the collected organic phase volume after performing the developed method (135, 124, 113, 103, 89, 78, and 68 μL for 0.0, 5.0, 10, 15, 20, 25, and 30%, w/v, NaCl, respectively). The results in Fig. 3 show that ERs of the analytes increase with increasing NaCl concentration up to 20%, w/v, and then decrease gradually in the cases of higher concentrations of NaCl. This is due to increase in the aqueous solution viscosity which leading to mass transfer resistance into the aqueous phase. Therefore, 20%, w/v, NaCl was used in the subsequent experiments.
Study of salt addition. Extraction conditions are the same as used in Fig. 2, except the extraction temperature which was adjusted at 70 °C. The extraction solvent volume ranged from 135 to 68 μL by increasing NaCl concentration from 0 to 30%, w/v. The error bars show the minimum and maximum of three repeated determinations
Optimization of Centrifuging Speed and Time
Centrifugation is an important process to achieve a rapid separation of the extractant droplets from the aqueous phase. Centrifugation speed and time were studied in the ranges of 402–2191×g and 2–6 min, respectively. The obtained results indicated that the efficiency of the method increased with increasing centrifugation speed and time up to 1610×g and 5 min, respectively, and then the analytical signals were nearly constant. Therefore, centrifugation speed and time were chosen 1610×g and 5 min, respectively, in the following experiments.
Method Validation
The analytical figures of merit for the proposed method were calculated under the optimized experimental conditions established above. The analytical performance of the proposed method was evaluated in terms of linear range (LR), correlation coefficient, repeatability, EF, ER, and limits of detection (LOD) and quantification (LOQ) according to DG SANCO Guidelines 296 (European Commission 2011). The obtained results are summarized in Table 1. The wide LRs were obtained with correlation coefficients ranging from 0.994 to 0.999. The LODs, calculated on the base of signal to noise ratio (S/N) of 3, ranged from 0.09 to 0.32, 0.68 to 13.1, and 0.34 to 0.69 ng mL−1 in de-ionized water, vegetable samples (beet, potato, cucumber, and tomato), and fruit juices (apple, grape, and sour cherry), respectively. The LOQs (S/N = 10) ranged from 0.30 to 1.0, 2.23–4.09, 1.12–2.05 ng mL−1 in de-ionized water, vegetable samples (beet, potato, cucumber, and tomato), and fruit juices (apple, grape, and sour cherry), respectively. The EFs and ERs for the selected insecticides ranged from 180 to 235 and 72 to 94%, respectively. The relative standard deviations (RSDs) at a concentration of 5 ng mL−1 of each analyte were in the ranges of 4–5 and 5–7% for intra-day (n = 6) and inter-day (n = 4) precisions, respectively. The accuracy of an analytical method describes the closeness of the mean experimental results obtained by the method to the true value (concentration) of the analyte. The best way to evaluate the accuracy of a method is its application to Certified Reference Materials (CRMs) analysis. Unfortunately, there is no CRM for the studied analytes. Furthermore, no other analytical method was reported in literature for determination of all analytes used in the studied samples. Therefore, the accuracy of the method was determined by added-found method using five replicate determinations at 5 and 25 ng mL−1 levels (each analyte), and the obtained deviations were less than 6% for all analytes. These results show that the method is sensitive and suitable for the analysis of the selected insecticides in aqueous samples.
Real Samples Analysis
The method applicability to real samples was investigated by analyzing different fruit juices including apple, grape, and sour cherry and vegetables including fresh tomato, cucumber, potato, and beet under the optimized conditions. Figure 4 shows the typical HPLC–DAD chromatograms for a standard solution of the analytes (direct injection), the potato sample, and the spiked potato sample in those the developed method was performed prior to chromatographic separation. As it can be seen, there is no peak in the retention times of the analytes in the potato sample chromatogram. According to the obtained results, analytes’ contents of all samples were lower than LODs of the method. To assess the matrix effect, the samples and HPLC-grade water were spiked with the analytes at different concentrations (10, 25, and 50 ng mL−1 of each analyte) and the method was performed on them. As mentioned in “Samples” section, fruit juices and vegetable juices were diluted with HPLC-grade water at ratios of 1:2 and 1:4, respectively. It is noted that the undiluted samples showed relatively high matrices effects. Comparison of the analytes peak areas in the samples and the HPLC-grade water spiked with the related levels gave relative recoveries between 86 and 97% (Table 2). According to the results, matrices of the real samples had no significant effect on the performance of the developed method. The obtained results show that the proposed method can be used as an applicable and adequate method for the analysis of the selected neonicotinoid insecticides in fruit juice and vegetable samples.
Typical HPLC–DAD chromatograms of direct injection of a standard solution of the analytes prepared in ACN (10 mg L−1 of each analyte) (I), the potato juice spiked with 25 ng mL−1 of each analyte (II), and the unspiked potato juice after performing the proposed method (III). Detection wavelength was 244 nm. For more chromatographic conditions, see the “Experimental” Section. Peaks identification: acetamiprid (1), imidacloprid (2), and thiamethoxam (3)
Comparison of the Developed Method with Other Methods
Table 3 indicates the values of LOD, LR, RSD, extraction time, and ER of the proposed method and the other methods for the extraction and determination of the selected insecticides from different samples. As it can be seen, the RSDs of the proposed method are better than or comparable with those reported for the other methods. The LODs and LOQs for the presented method are lower than those of the mentioned methods. On the other hand, the developed method has wider linear range compared to the other methods. The extraction time and ER of the proposed method are comparable with those of the mentioned methods. These results show that the presented method proves to be a rapid, sensitive, efficient, and reliable technique for the extraction and preconcentration of the selected neonicotinoid insecticides from different samples.
Conclusion
A simple, sensitive, and reliable analytical method based on heat-induced HLLME combined with HPLC–DAD was developed for the extraction and determination of low concentrations of the selected neonicotinoid insecticides in fruit juice and vegetable samples. The experimental results demonstrate that this technique exhibits many merits such as high ERs and EFs, low LODs, and LOQs short extraction time, and good repeatability. In views of the simplicity, speed, and efficiency that were observed, this method is recommended for analyzing the target analytes in other matrices.
Abbreviations
- AALLME:
-
Air-assisted liquid–liquid microextraction
- DES:
-
Deep eutectic solvent
- DLLME:
-
Dispersive liquid–liquid microextraction
- HF–LPME:
-
Hollow fiber liquid-phase microextraction
- IL:
-
Ionic liquid
- LLE:
-
Liquid–liquid extraction
- SPE:
-
Solid phase extraction
- SDME:
-
Single drop microextraction
References
Amadeo R, Fernandez A, Antonio V, Ana A, Mariano C, Serge C (1996) Determination of imidacloprid in vegetables by high-performance liquid chromatography with diode-array detection. J Chromatogr A 721:97–105
Antonio A, Moreno M, Rosa P, Isabel M, Adolfo S, Miguel G, Angel M (1999) Determination of carbaryl, carbofuran and methiocarb in cucumbers and strawberries by monoclonal enzyme immune assays and high-performance liquid chromatography with fluorescence detection: an analytical comparison. J Chromatogr A 833:3–12
Cai J, Chen G, Qiu J, Zeng F, Zhu F, Ouyang G (2016) Hollow fiber based liquid phase microextraction for the determination of organochlorine pesticides in ecological textiles by gas chromatography–mass spectrometry. Talanta 146:375–380
Carabias Martinez R, Rodriguez Gonzalo E, Amigo Moran MJ, Hernandez Mendez J (1992) Sensitive method for the determination of organophosphorus pesticides in fruits and surface waters by high-performance liquid chromatography with ultraviolet detection. J Chromatogr A 607:37–45
Commission Directive 2007/11/EC on 21 February 2007. Amending certain Annexes to Council Directives 86/362/EEC, 86/363/EEC and 90/642/EEC O. J. E.U. L63, 26–37
Dankyi E, Carboo D, Gordon C, Fomsgaard IS (2015) Application of the QuEChERS procedure and LC-MS/MS for the assessment of neonicotinoid insecticide residues in cocoa beans and shells. J Food Com Anal 44:149–157
Dimitra A, Triantafyllos A (2001) Optimization of headspace solid-phase microextraction conditions for the determination of organophosphorus insecticides in natural waters. J Chromatogr A 922:243–255
European Commission 2011 Method validation and quality control procedures for pesticide residues 426 analysis in food and feed. Document No SANCO/12495/2011. Available from: 427 http://ec.europa.eu/food/plant/protection/pesticides/docs/qualcontrol_en.pdf (cited 13.01.14)
Fang G, Chen W, Yao Y, Wang J, Qin J, Wang S (2012) Multi-residue determination of organophosphorus and organochlorine pesticides in environmental samples using solid-phase extraction with cigarette filter followed by gas chromatography–mass spectrometry. J Sep Sci 35:534–540
Farajzadeh MA, Afshar Mogaddam MR (2012) Air-assisted liquid–liquid microextraction method as a novel microextraction technique; application in extraction and preconcentration of phthalate esters in aqueous sample followed by gas chromatography–flame ionization detection. Anal Chim Acta 728:31–38
Farajzadeh MA, Bahram M, Zorita S, Mehr BG (2009a) Optimization and application of homogeneous liquid–liquid extraction in preconcentration of copper (II) in a ternary solvent system. J Hazard Mater 161:1535–1543
Farajzadeh MA, Seyedi SE, Safi Shalamzari M, Bamorowat M (2009b) Dispersive liquid–liquid microextraction using extraction solvent lighter than water. J Sep Sci 32:3191–3200
Farajzadeh MA, Mohebbi A, Feriduni B (2016) Development of continuous dispersive liquid-liquid microextraction performed in home-made device for extraction and preconcentration of aryloxyphenoxy-propionate herbicides from aqueous samples followed by gas chromatography-flame ionization detection. Anal Chim Acta 920:1–9
Garcia G, Galera MM, Valverde RS, Galanti A, Girotti S (2007) Column switching liquid chromatography and post-column photochemically fluorescence detection to determine imidacloprid and 6-chloronicotinic acid in honeybees. J Chromatogr A 1147:17–23
Garrido Frenich A, Egea Gonzalez FJ, Martanez Vidal JL, Parrilla P, Mateu Sanchez M (2000) Determination of imidacloprid and its metabolite 6-chloronicotinic acid in greenhouse air by high-performance liquid chromatography with diode-array detection. J Chromatogr A 869:497–504
Gbylik-Sikorska M, Sniegocki T, Posyniak A (2015) Determination of neonicotinoid insecticides and their metabolites in honey bee and honey by liquid chromatography tandem mass spectrometry. J Chromatogr B 990:32–140
Huahua B, Qing Xiang Z, Guohong X, Junping X (2009) Enrichment and sensitive determination of dichlorodiphenyltrichloroethane and its metabolites with temperature controlled ionic liquid dispersive liquid phase microextraction prior to high performance liquid phase chromatography. Anal Chim Acta 651:64–68
Jiao W, Xiao Y, Qian X, Tong M, Hu Y, Hou R, Hua R (2016) Optimized combination of dilution and refined QuEChERS to overcome matrix effects of six types of tea for determination eight neonicotinoid insecticides by ultra performance liquid chromatography-electrospray tandem mass spectrometry. Food Chem 210:26–34
Koesukwiwat U, Lehotay S, Miao S, Leepipatpiboon N (2010) High throughput analysis of 150 pesticides in fruits and vegetables using QuEChERS and low-pressure gas chromatography–time-of-flight mass spectrometry. J Chromatogr A 1217:6692–6703
Liu H, Dasgupta PK (1996) Analytical chemistry in a drop. Solvent extraction in a microdrop. Anal Chem 68:1817–1821
Liu S, Zheng Z, Wei F, Ren Y, Gui W, Wu H, Zhu G (2010) Simultaneous determination of seven neonicotinoid pesticide residues in food by ultraperformance liquid chromatography tandem mass spectrometry. J Agric Food Chem 58:3271–3278
Meyer V. R (1994) Practical high-performance liquid chromatography, 5th edn. A John Wiley and Sons Publication, Switzerland
Murata K, Yokoyama Y, Ikeda S (1972) Homogeneous liquid-liquid extraction method, extraction of iron(III) thenoyltrifluoroacetonate by propylene carbonate. Anal Chem 44:805–810
Obana H, Okihashi M, Akutsu K, Kitagawa Y, Hori S (2003) Determination of neonicotinoid pesticide residues in vegetables and fruits with solid phase extraction and liquid chromatography mass spectrometry. J Agric Food Chem 51:2501–2505
Pavle J, Vale G, Sanja L, Mladen F, Marijana S, Ljubis A, Jovana K (2015) Development of HPLC-DAD method for determination of neonicotinoids in honey. J Food Comp Anal 40:106–113
Pelit F, Yengin A (2014) Application of solidified floating organic drop microextraction method for biomonitoring of chlorpyrifos and its oxon metabolite in urine samples. J Chromatogr B 949-950:109–114
Pirsaheb M, Fattahi N, Pourhaghighat S, Shamsipur M, Sharafi K (2015) Simultaneous determination of imidacloprid and diazinon in apple and pear samples using sonication and dispersive liquid-liquid microextraction. LWT-Food Sci and Technol 60:825–831
Qiao F, Zhang X, Wang M, Kang Y (2010) Rapid extraction of imidacloprid in tomatoes by ultrasonic dispersion liquid-liquid microextraction coupled with LC determination. Chromatographia 72(3–4):331–335
Rosales-Conrado N, Leon-Gonzalez ME, Perez-Arribas LV, Polo-Diez LM (2002) Determination of chlorophenoxy acid herbicides and their esters in soil by capillary high performance liquid chromatography with ultraviolet detection, using large volume injection and temperature gradient. Anal Chim Acta 470:147–154
Serenella S, Paola F, Domenico M, Patrizia M (2008) Determination of neonicotinoid insecticides residues in bovine milk samples by solid-phase extraction clean-up and liquid chromatography with diode-array detection. J Chromatogr A 1214:115–120
Shuaihua Z, Xiumin Y, Xiaofang Y, Chun W, Zhi W (2012) Dispersive liquid–liquid microextraction combined with sweeping micellar electro kinetic chromatography for the determination of some neonicotinoid insecticides in cucumber samples. Food Chem 133:544–550
Tsoutsi C, Konstantinou I, Hela D, Albanis T (2006) Screening method for organophosphorus insecticides and their metabolites in olive oil samples based on headspace solid-phase microextraction coupled with gas chromatography. Anal Chim Acta 573-574:216–222
Wang P, Yang X, Wang J, Cui J, Dong AJ, Zhao HT, Zhang LW, Wang ZY, Xu RB, Li WJ, Zhang YC, Zhang H, Jing J (2012) Multi-residue method for determination of seven neonicotinoid insecticides in grains using dispersive solid-phase extraction and dispersive liquid–liquid micro-extraction by high performance liquid chromatography. Food Chem 134:1691–1698
Xianlei H, Mingqiu Z, Wenhong R, Fang Z, Gangfeng O (2012) Determination of organophosphorus pesticides in ecological textiles by solid-phase microextraction with a siloxane-modified polyurethane acrylic resin fiber. Anal Chim Acta 736:62–68
Yazdanfar N, Yamini Y, Ghambarian M (2014) Homogeneous liquid–liquid microextraction for determination of organochlorine pesticides in water and fruit samples. Chromatographia 77:329–336
Yogesh B, Dhananjay Kumar T (2010) Development and applications of single-drop microextraction for pesticide residue analysis: a review. J Sep Sci 33:3683–3691
Zhang J, Liang Z, Li S, Li Y, Peng B, Zhou W, Gao H (2012) In-situ metathesis reaction combined with ultrasound-assisted ionic liquid dispersive liquid–liquid microextraction method for the determination of phenylurea pesticides in water samples. Talanta 98:145–151
Acknowledgments
Authors are grateful to the Research Council of the University of Tabriz for financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Mir Ali Farajzadeh has received research grants from University of Tabriz.
Conflict of Interest
Mir Ali Farajzadeh declares that he has no conflict of interest. Ali Shahedi Hojaghan declares that he has no conflict of interest. Mohammad Reza Afshar Mogaddam declares that he has no conflict of interest.
Ethical Approval
This article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable.
Rights and permissions
About this article
Cite this article
Farajzadeh, M.A., Hojaghan, A.S. & Afshar Mogaddam, M.R. Development of Heat-Induced Homogeneous Liquid–Liquid Microextraction for Extraction and Preconcentration of Neonicotinoid Insecticides from Fruit Juice and Vegetable Samples. Food Anal. Methods 10, 3738–3746 (2017). https://doi.org/10.1007/s12161-017-0942-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-017-0942-y