Abstract
Objective
The purpose of this study is to identify factors which predict adherence in stroke survivors.
Design
This is a longitudinal study where 180 stroke survivors were assessed 1 year after their first ischaemic stroke. The relationship between adherence and illness and medication beliefs was tested at baseline (time 1) and again 5–6 weeks later (time 2).
Main Outcome Measures
The main outcome measures used in this study are Medication Adherence Report Scale and urinary salicylate levels.
Results
Four variables predicted time 1 poor adherence: (1) younger age, (2) increased specific concerns about medications, (3) reduced cognitive functioning and (4) low perceived benefit of medication. Three out of these four variables were again predictive of time 2 adherence and accounted for 24% of the variance: (1) younger age, (2) increased specific concerns about medications and (3) low perceived benefit of medication. The urinary salicylate assay failed to differentiate between patients taking and not taking aspirin.
Conclusion
Interventions to improve adherence should target patients’ beliefs about their medication.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Adherence to therapy is a primary determinant of treatment success. Poor adherence reduces the overall effectiveness of health care interventions, yet it is estimated that in developed countries only 50% of patients who suffer from chronic diseases adhere to treatment recommendations [1]. The current Cochrane review of interventions to improve medication adherence concluded that “Current methods of improving adherence for chronic health problems are mostly complex and not very effective, so that the full benefits of treatment cannot be realized. High priority should be given to fundamental and applied research concerning innovations to assist patients to follow medication prescriptions for long-term medical disorders” [2].
This study investigates determinants of medication adherence in the secondary prevention of stroke. Stroke is the third most common cause of death in the USA and UK, and is the most common cause of severe physical disability amongst adults. The risk of a recurrent stroke is 30–43% within 5 years. Large randomised controlled trials and meta-analyses have identified several drugs which significantly reduce the risk of future vascular events after stroke. Guidelines for secondary prevention after stroke now recommend antiplatelet therapy and reduction of both blood pressure and cholesterol level (e.g. [3, 4]). In the treatment of hypertension, it is estimated that only 30–50% of patients regularly take their drugs as prescribed and that non-adherence may cause half of antihypertensive drug “failures” [5]. Non-adherence to medication is, therefore, a risk factor for recurring vascular events or death. In stroke, limited data are available on patient adherence to medication, but this is unlikely to be better than in other chronic conditions and indeed, given that cognitive impairment is a common consequence of stroke, may be significantly worse.
Poor adherence may be both intentional and non-intentional. Non-intentional poor adherence (e.g. forgetting) is often a consequence of cognitive impairment. Very mild cognitive impairment, even in healthy elderly patients, is associated with poor medication adherence [6]. After a stroke, the impact of cerebrovascular disease on cognitive functioning, particularly memory, may mitigate against adherence, particularly if the patient is elderly and the drug regime complicated [7]. Intentional non-adherence occurs when a patient adopts a deliberate strategy of taking medication in a manner that differs from medical advice. This is likely to depend on the patient’s beliefs concerning their condition (illness perceptions) and their medication, i.e. the perceived benefits versus the perceived risks (or side effects) of the medication.
The theoretical background for the current study draws heavily on Leventhal’s common sense self-regulation model [8, 9]. According to the common sense self-regulation model, there are five discrete attributes (identity, cause, timeline, consequences, cure/control) to the way people think about their condition (their illness perceptions), and these are organised into a mental model or illness representation that predicts health behaviours such as medication adherence. For example, medication adherence in patients with hypercholesterolaemia has been shown to be related to the belief that the disease has severe coronary consequences. Hypertensive patients who believe that their condition is chronic are more likely to adhere to antihypertensive medication than those who believe it to be an acute condition. The latter group ceased taking the medication, perhaps believing themselves “cured” [10]. This suggests that when a patient believes (a) they have a chronic condition and (b) that they are able to control symptoms or prevent future illness using medication, they are more likely to adhere to treatment recommendations. Thus, the specific attributes of timeline and cure/control were incorporated as predictors of adherence in this study.
The valid and reliable measurement of adherence is critical. Participants may respond in what they believe to be a socially desirable manner and over-report their adherence. In order to address this possibility, we fulfilled four fundamental criteria: first, the researcher/interviewer was fully independent from the clinical team; second, we reinforced to patients that their responses were confidential and would not influence their treatment; third, questions regarding adherence were framed in a manner so as to make non-adherent responses appear entirely acceptable, reasonable and understandable. The wording of questions in the main outcome measure used in this study, the Medication Adherence Report Scale (MARS) [11], is designed to take these factors into account. Finally, we directly measured socially desirable responding and tested whether this was correlated with scores on the MARS.
Trewby et al. [12] found that in order to accept taking a hypothetical, completely safe, new drug for 5 years to prevent another myocardial infarction (MI), patients, on average, wanted a 20% (1 in 5) chance of being benefited. This is far higher than the benefit accrued from any current treatment available for MI or stroke. Our study also included measures of patients’ beliefs about their medication in order to determine, for the first time: (a) the risk each patient believes they are at of suffering a further stroke, (b) the size of benefit each patient believes they are accruing from their current medication and (c) whether these measures are predictive of adherence.
In summary, this study was designed to facilitate a realistic assessment of adherence and to examine the predictive value of known risk factors and the additional contribution of relevant illness and medication beliefs. We also included an objective measure of adherence using urinary assay of salicylate levels.
Methods
Participants
One hundred and eighty first-time ischaemic stroke patients, approximately 1-year post-stroke were recruited. Ethical approval for the study was obtained from NHS Lothian Research Ethics Committee, and the study was conducted according to the guidelines laid down in the Declaration of Helsinki. Eligible patients were identified from an ongoing audit of all patients admitted to or seen in outpatient clinics at the Western General Hospital in Edinburgh. Study inclusion criteria were diagnosis with an ischaemic stroke in the past 15 months, no stroke prior to the index event, not resident in a nursing or residential home, living within approximately a 50-mile radius of the hospital, the participant was responsible for taking their own medication and having a permanent residential address. Only patients scoring over 13 on the Frenchay Comprehension screening test [13] were eventually included in the study because of level of the language ability required for completion of questionnaires and interviews.
Screening (via patients’ general practitioner physicians) was carried out to exclude (a) any deceased patients prior to sending out letters of invitation and (b) any participants whom the family physician felt did not have capacity to give informed consent to participate. Telephone follow-up interviews were conducted to ascertain interest in participating. Interviews were arranged according to patients’ preference either in their own home or in a clinical research facility. All participants gave informed consent. Participants were taking a variety of medications, and most were taking an antihypertensive, a statin and aspirin. Participants were taking a mean of 5.48 (SD = 2.2, range 1–10), different medications per day, and a mean of 7.31 (SD = 4.4, range 1–22), number of tablets per day, and took their medications 2.66 (SD = 0.9, range 1–5) times per day.
Procedure
A psychologist trained in the procedures interviewed and assessed patients in a single 60–90-min session. Patients first completed the questionnaires and then participated in a brief semi-structured, digitally recorded interview. Finally, a urine sample was collected from all participants for assessment of urinary salicylate levels. We decided to focus on salicylate as: (a) over 80% of the samples were prescribed aspirin 75 mg and (b) an assay to measure low levels of urinary salicylate in urine was available [14].
Measures
Outcome Measures
The MARS was used as the primary outcome measure. The MARS is a brief self-report instrument, assessing five separate non-adherent behaviours, and provides a sensitive assessment of drug adherence behaviours [11]. It is specifically worded so as to reduce social desirability effects. This five-item scale asks respondents to rate the frequency with which they engage in five aspects of non-adherent behaviour (“I forget to take them”, “I alter the dose”, “I stop taking them for a while”, “I decide to miss out a dose” and “I take less than instructed”) rated on a five-point scale, where 5 = never and 1 = very often. Scores for each of the five items are summed to give a total score of 5–25, with higher scores representing higher levels of self-reported adherence. The MARS is internally reliable (Cronbach’s alpha across four studies, 0.68–0.86), with excellent test–retest reliability (r = 0.97) [11]. We also directly tested for socially desirable responding, and in an attempt to avoid participant overload, we asked participants to complete the brief five-item Brief Social Desirability Scale [15].
Second, an opportunistic urinary sample was provided by all but one patient. Since samples were not continuous over a 24-h period, assays for salicylic acid [14] and creatinine levels [16] were carried out to calculate an overall urinary salicylic acid/creatinine ratio. The salicylic acid/creatinine ratio is used to control for volume of urine produced.
Predictor Variables
Demographic factors of age, sex and Carstairs social deprivation index (based on postal code using the following census indicators: low social class, lack of car ownership, overcrowding and male unemployment [17]) were collected for each participant. The National Institute of Health Stroke Scale (NIHSS) score (a measure of stroke severity) was recorded at entry to the register. Cognitive functioning was assessed using two measures. The Mini-Mental State Examination (MMSE) is a brief, valid and reliable assessment of various components of cognitive function, widely used in stroke research [18]. The Rivermead Behavioural Memory Test (RBMT) [19] is an ecologically valid measure of memory function. Scores from the Belongings and Appointment sub-scales (two of the 11 sub-scales in the full scale) were combined to form an overall prospective memory functioning score, following O'Carroll et al. [20].
Patients’ perceptions and beliefs about their illness and medication were explored using several measures. Patients’ perception of their risk of further stroke in the next 5 years was assessed by presentation of a visual analogue 0–100 scale (score calculated as a percentage). In addition, similar scales were used to assess the extent to which patients desired secondary stroke preventive medication and their perception of benefits of their current preventive medication, following Trewby et al. [12].
The Illness Perception Questionnaire (IPQ) was developed to provide quantitative measurement of the main components of illness representations in Leventhal’s self-regulation model [8]. The revised version of the measure, the IPQ-R, provides a comprehensive and psychometrically robust measure of the major components [21]. For the present study, only two scores were selected for analysis, as it was hypothesised that adherence would be particularly related to whether patients regarded the timeline of their condition as chronic or acute, and the perceived treatment control patients felt regarding their medication.
The Beliefs about Medicines Questionnaire (BMQ) was designed to assess cognitive representations regarding medication and measures four domains: specific necessity, specific concerns, general harm and general overuse [22]. For this study, it was hypothesised that adherence would be predominantly related to patients specific concerns regarding their secondary preventative stroke medication, and their views as to the specific necessity of that medication.
The Hospital Anxiety and Depression Scale (HADS) [23] is a 14-item scale, with seven items assessing each of general anxiety and depression; for this study, the overall emotional distress score was computed.
Statistical Power and Analysis
In hierarchical multiple regression analyses predicting adherence, with 14 predictors (age, gender, social deprivation category, stroke severity, perceived risk of further stroke, perception of benefit, desire for medication, BMQ necessity, BMQ concerns, IPQ-R chronic timeline, IPQ-R treatment control, HADS emotional distress, MMSE and prospective memory scores), G-Power calculation specified a minimum sample size of 166. This would allow us to detect a medium effect size (0.15) with a power of 0.90, with alpha set at 0.05.
Data were entered into an SPSS database for analysis. The primary outcome measure was the measure of adherence to prescribed medication using the MARS score. Participants were asked to complete the MARS in relation to their adherence to all medication they were taking to reduce the risk of a further stroke. A secondary outcome measure was the urinary salicylic acid/creatinine ratio, as an objective measure of adherence to aspirin.
The primary analysis was a hierarchical multiple regression to evaluate the ability of independent variables in six broad categories to predict medication adherence (MARS score). The rational for the order of entry of our hierarchical regression was as follows. Our ultimate aim was to test how much (if any) additional variance in self-reported medication adherence could be explained by illness- and medication-related beliefs, over and above that explained by traditional predictors. We, thus, first accounted for variance associated with demographic factors. We then tested whether more severe stroke/acquired cognitive impairment would predict poor adherence. We then tested whether higher perceived risk would be associated with poorer adherence. After these “traditional” factors had been accounted for, we tested whether first illness- and then medication-related beliefs would add any predictive power. Finally, we tested whether affective state would make any further contribution. At the first step, basic demographic variables were entered (age, gender, social deprivation index). Next, factors relevant to stroke severity were entered (stroke severity, cognitive impairment (MMSE) and prospective memory functioning (RBMT)). At the third step, the patient’s perception of their risk of having a further stroke was added. Next, the two illness belief variables from the IPQ-R were entered, assessing patients’ beliefs about the timeline (acute/chronic) and treatment control over their illness. The fifth step entered variables relating to patients beliefs and concerns about their medications (specific concerns, specific necessity, desire for medication and perception of benefit) and the final step, the measure of emotional distress (HADS total). This allowed us to determine the additional variance in self-reported medication adherence explained at each stage of analysis, and in particular, the additional predictive role that illness and medication beliefs had over and above demographic and stroke severity. This analysis was conducted twice, first to test the ability of the predictor variables to account for contemporaneous variation at baseline (time 1 adherence), and then to truly test prediction by using the same baseline predictor variables to account for variation in future (time 2) adherence, measured 5–6 weeks later. To first assess the ability of the assay to discriminate aspirin takers, urinary salicylate levels were compared between those taking and not taking aspirin using Student’s t test.
Results
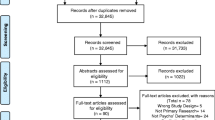
Of 321 patients identified for possible participation, 45 were not contactable, 84 declined and 12 failed to meet inclusion criteria, leaving 180 participants. Mean age was 69 years (SD = 11.4), with slightly more males (54%) than females.
Table 1 summarises the patient characteristics. The reliability (Cronbach’s alpha) of the scales used in the study was as follows: MARS = 0.67, Social Desirability Scale = 0.66, IPQ-R timeline acute/chronic = 0.87, treatment control = 0.73, BMQ specific concerns = 0.77, specific necessity = 0.77, HADS = 0.86. The mean (SD) value of the MARS score, the primary outcome measure, was high (i.e. time 1 = 23.9 (1.62), range 13–25; time 2 = 24.1 (1.49), range 17–25), suggesting that patients generally reported high levels of medication adherence. Importantly, the MARS total score was not significantly correlated with the Brief Social Desirability Scale (p > 0.10).
Predictors of Medication Adherence
Time 1 Prediction
Hierarchical multiple regression examined the ability of the independent variables to contemporaneously predict the primary outcome of MARS self-reported adherence (see Table 2). Overall, the final model explained 34.3% of variance in medication adherence scores (F(14,159) = 5.93, p < 0.001). Four independent variables made a statistically significant contribution. These were age (β = 0.371, p < 0.001), specific medication concerns (β = −0.355, p < 0.001), MMSE score (β = 0.201, p = 0.010) and perceived benefit of medication score (β = 0.159, p = 0.043).
Preliminary analyses confirmed the absence of multi-collinearity; however, MARS scores were skewed and not normally distributed (Kolmogrov–Smirnov statistic, p < 0.001) showing ceiling effects. An attempt was therefore made to transform the raw MARS total scores, following the recommendations of Field [24] and Tabachnick and Fidell [25]. However, log, square root, reciprocal and reflect and inverse transformations all failed to produce a normally distributed dependent variable. Therefore, a confirmatory logistic regression analysis was conducted after the MARS score was transformed into a dichotomous variable. Following the recommendation by Horne [11], all those participants who scored 23 and under were categorised into a “sub-optimal adherence group”, and those who scored 24 or 25, into an “optimal adherence group”. A logistic regression was run using a forced entry method to assess the predictive value of the same variables on the likelihood of (self-reported) adherence to medication; all independent variables were entered in one block to assess their predictive value while controlling for the effects of other predictors in the model. The full model containing all the predictors was statistically significant (χ 2 (14, N=174) = 31.2, p = 0.005) and correctly classified 81% of cases. Only one of the predictor variables made a unique and statistically significant contribution to the model: patient’s specific concerns about the medication, with an odds ratio of .835.
Time 2 Prediction
In order to test the ability of the model to predict future medication adherence, the participants again completed the MARS adherence scale, 5–6 weeks following baseline assessment of the predictor variables, mean (SD) time interval 36.9 days (7.2 days). The same hierarchical multiple regression examined the ability of the independent variables to predict time 2 MARS self-reported adherence (see Table 3). Overall, the final model explained 24.3% of variance in MARS scores (F(14,157) = 3.61, p < 0.001). Three out of the four variables which were significant at time 1 again made a significant contribution to the final time 2 MARS model (MMSE was no longer significant). They were specific medication concerns (β = −0.254, p = 0.007), age (β = 0.241, p = 0.004) and perceived benefit of medication score (β = 0.273, p = 0.001). As was conducted with the baseline data, a confirmatory logistic regression was run on the time 2 data where MARS scores were divided into optimal and sub-optimal categories. The full model containing all the predictors was statistically significant (χ 2 (14, N=172) = 28.55, p = 0.012) and correctly classified 82% of cases. Only two predictor variables made a statistically significant contribution to the model: again patient’s specific concerns about the medication, with an odds ratio of .837, and prospective memory functioning, odds ratio 1.25.
Objective Measure of Adherence: Urinary Salicylate Levels
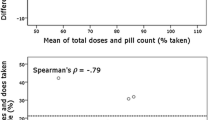
No significant difference in urinary salicylic acid/creatinine ratio was found between aspirin takers (n = 145) and non-takers (n = 31) (mean (SD) 13.06 (26.41) versus 26.59 (47.05); t(174) = 1.55, p = 0.13, not significant (ns)). This raises concerns regarding the sensitivity of the assay for our participants who were prescribed low doses (75mg) of aspirin; hence, no further analyses were conducted using the salicylic acid/creatinine ratio.
Supplementary Analysis
We also tested the contribution that a spouse/partner may have had in relation to the participants’ self-reported medication adherence. We, thus, compared the 65 participants who lived alone versus the 115 who lived with a partner/relative on time 1 and time 2 MARS scores. At time 1, mean (SD) MARS total for those living alone was 24.1 (1.2) versus 24.0 (1.8) for those living with a partner/relative, t(178) = 0.32, p = 0.75, ns. At time 2, the scores were 23.9 (1.7) versus 24.2 (1.3) respectively, t(176) = 1.48, p = 0.14, ns.
Discussion
There is a dearth of literature on medication adherence in stroke patients. The current study provides the first evidence of psychological factors predictive of poor medication adherence following stroke, which may be important targets for intervention.
The main finding of this study is that stroke survivors generally report good adherence to their stroke medication. Since the MARS score was not significantly correlated with scores on a measure of social desirability, socially desirable responding (faking better adherence) would not appear to be responsible. Clearly framing is an important issue, in attempting to accurately assess medication adherence. Patients rarely reported deliberate, intentional non-adherence, but did report forgetting. It is possible that some patients were using the terminology of “forgetting” as a way of communicating deliberate non-adherence in what they perceive to be a more socially acceptable manner.
The variables predictive of adherence are important. Younger age was predictive of poor adherence to medication. Younger participants reported forgetting some medication doses due to not having established a set medication-taking routine, coupled with conflicting demands, e.g. rushing to leave for work in the morning. This is an important finding in targeting any interventions to improve medication adherence in stroke survivors, not least because economic opportunity costs arising from illness will be greater in younger populations. At baseline only, reduced level of general cognitive functioning (MMSE score) was also a significant predictor of poor medication adherence. Contrary to our hypothesis, measures of illness perceptions were not significant predictors of adherence. However, at both time 1 and time 2, patients’ specific concerns and perceived benefits of their medication were strongly associated with poor adherence. This finding is consistent with Leventhal’s self-regulatory theory, as other studies have also found that treatment beliefs are more strongly related to behaviour than illness beliefs [26]. At interview, adherent participants tended to have faith in their medicines and the prescribers’ competence. Participants displaying poorer adherence tended to believe that they were prescribed too many medicines and that they derived no symptomatic benefit. Participants who were less adherent did not appear to believe that there may be significant consequences of missing/stopping their medication, and they had poorer knowledge regarding the proposed mechanism of action of their preventative medication, e.g. via lowering blood pressure and cholesterol. A focus on patients’ specific concerns about their medication may be a key area to target when attempting to improve adherence.
Limitations
We had intended to use opportunistic urine sampling (for assay of salicylate levels) to provide an objective biological measure of adherence to prescribed aspirin. The results were disappointing, in that the assay was not able to differentiate between those taking and not taking aspirin. It may be that the level of non-adherence to aspirin was not sufficiently high or variable to be detected by the assay. Twenty-four hour urine sampling may provide a more reliable measure, and future studies should evaluate the appropriateness of this method before concluding that the assay is unable to detect differences in low-dose prescribing of aspirin. We also acknowledge that the internal reliabilities of both the MARS and the SDS measure of social desirability were slightly below the recommended Cronbach’s alpha level of 0.7. Given our inclusion/exclusion criteria, our participants represented the milder end of the spectrum of stroke patients with regard to cognitive impairment and disability. We also did not recruit participants at extreme socio-demographic disadvantage (e.g. homeless, illiterate). This study also focused on the beliefs and cognitive abilities of the participants, and did not fully assess the potential role of significant others (e.g. partners); however, we found no differences in self-reported adherence at either time point when we compared those participants who lived alone with those who lived with a partner/relative. Participants were asked to report their adherence to all medication that they were taking to reduce the risk of a further stroke. It is possible that some participants were selective in their intentional non-adherence, i.e. deciding to take some medications and not others. Finally, we assessed the predictive ability of our variables to account for variation in adherence over a short (6 weeks) time interval. Longer-term follow-up studies are required to test these relationships over extended periods of time.
Conclusions
We found questionnaire self-reported adherence to medication in stroke survivors to be generally high. Our multivariate model predicted one third of the variance in self-reported medication adherence at baseline and one quarter of the variance in future medication adherence. Three variables made a consistent contribution to the prediction of poor adherence: (1) younger age, (2) increased specific concerns about medications and (3) low perceived benefit of medication. Optimal medication adherence amongst stroke survivors is vital in the prevention of recurrent strokes and other cardiovascular events. Our results suggest that targeting younger stroke patients may be important, and that for all patients, interventions which elicit and challenge patients’ specific concerns regarding the negative consequences versus the benefits of taking medication may be helpful in improving rates of medication adherence and are worthy of controlled evaluation. We are now embarking on just such an intervention [27]. Finally, our results provide strong support for recent medicines adherence guidelines (e.g. 3), namely that patients should be routinely asked if they have any specific concerns about their medication and that these concerns should be addressed whenever medicines are prescribed, dispensed or reviewed.
References
World Health Organisation. Adherence to long-term therapies: Evidence for action. Geneva: WHO; 2003.
Haynes RB, Ackloo E, Sahota N, McDonald HP, Yao X. Interventions for enhancing medication adherence (Review). The Cochrane Collaboration, 4; 2008.
National Institute for Clinical Excellence, UK. Medicines adherence, Clinical Guideline, 76; 2009.
Scottish Intercollegiate Guidelines Network. Management of patients with stroke or TIA: assessment, investigation, immediate management and secondary prevention—A national guideline. Guideline 108. Edinburgh: SIGN; 2008
Stephenson J. Noncompliance may cause half of antihypertensive drug "failures". JAMA. 1999; 282[4]: 313-314.
Hayes TL, Larimer N, Adami A, Kaye JA. Medication adherence in healthy elders: small cognitive changes make a big difference. J Aging Health. 2009; 21[4]: 567-580.
Fischer B, Lehrl S, Weber E, Gundert-Remy U, Fischer, U. [Cerebrovascular insufficiency and compliance with drug therapy]. Z Gerontol. 1981; 14[2]: 145-152.
Leventhal H, Diefenbach M. Illness cognition: using common sense to understand treatment adherence and affect cognition interactions. Cognit Ther and Res. 1992; 16[2]: 143-163.
Meyer D, Leventhal H, Gutmann M. Common-sense models of illness: the example of hypertension. Health Psychol. 1985; 4: 5-135.
Brewer NT, Chapman GB, Brownlee S, Leventhal EA. Cholesterol control medication adherence and illness cognition. Br J Health Psychol. 2002; 7(Part 4): 433-447.
Horne R. Measuring adherence: the case for self-report. Intl J Behavioral Med. 2004; 11:75.
Trewby PN, Reddy AV, Trewby CS, Ashton VJ, Brennan G, Inglis J. Are preventive drugs preventive enough? A study of patients' expectation of benefit from preventive drugs. Clin Med. 2002; 2[6]: 527-533.
Enderby PM, Wood VA, Wade DT, Hewer RL. The Frenchay Aphasia Screening Test: a short simple test for aphasia appropriate for non-specialists. Int Rehabil Med. 1987; 8[4]: 166-170.
Baxter GJ, Lawrence JR, Graham AB, Wiles D, Paterson JR. Identification and determination of salicylic acid and salicyluric acid in urine of people not taking salicylate drugs. Ann Clin Biochem. 2002; 39(Pt 1): 50-55.
Hays RD, Corporation TR. A five-item measure of socially desirable response set. Educ Psychol Meas. 1989; 49: 629-636.
Bartels H, Bohmer M, Heierli C. Serum creatinine determination without protein precipitation. Clin Chim Acta. 1972; 37: 193-197.
McLoone P. Carstairs scores for Scottish Postcode Sectors from the 2001 census. MRC Social and Public Health Sciences Unit. Glasgow: MRC; 2004
Folstein MF, Folstein SE, McHugh PR. Mini-Mental State: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975; 12: 189-198.
Wilson B, Cockburn J, Baddeley AD. Rivermead Behavioural Memory Test, Titchfield, Fareham: Thames Valley Test Co; 1985.
O'Carroll RE, McGregor LM, Swanson V, Masterton G, Hayes PC. Adherence to medication after liver transplantation in Scotland: a pilot study. Liver Transpl. 2006; 12[12]: 1862-1868.
Moss-Morris R, Weinman J, Petrie KJ, Horne R, Cameron LD, Buick D. The Revised Illness Perception Questionnaire (IPQ-R). Psychol Health. 2002; 16: 1-15.
Horne R, Weinman J, Hankins M. The beliefs about medicines questionnaire: the development and evaluation of a new method for assessing the cognitive representation of medication. Psychol Health. 1999; 14: 1-24.
Zigmond AS, Snaith RP. The Hospital Anxiety and Depression Scale. Acta Psychiatr Scand. 1983; 67:361-370.
Field A. Discovering statistics using SPSS (2nd ed). London: Sage; 2005.
Tabachnick BG, Fidell LS. Using multivariate statistics (4th ed). Needham Heights, MA: Allyn Bacon; 2001.
Leventhal H, Weinman J, Leventhal EA, Phillips LA. Health psychology: The search for pathways between behavior and health. Annu Rev Psychol. 2008; 59: 477-505.
O'Carroll RE, Dennis M, Johnston M, Sudlow C. Improving adherence to medication in stroke survivors (IAMSS): A randomised controlled trial: Study protocol BMC Neurol. 2010; 10:15.
Acknowledgements
We would also like to thank Dr. Julie Chambers, Dr Carolyn Choudhary and three anonymous reviewers for their helpful comments on a previous version of this paper.
We would also like to thank the patients and partners for their participation. Finally, we acknowledge the grant support provided by the Chief Scientist Office-Scotland, Health Services Research Committee, grant reference CZH/4/297.
Conflict of Interest Statement
The authors have no conflict of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
O’Carroll, R., Whittaker, J., Hamilton, B. et al. Predictors of Adherence to Secondary Preventive Medication in Stroke Patients. ann. behav. med. 41, 383–390 (2011). https://doi.org/10.1007/s12160-010-9257-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12160-010-9257-6