Abstract
In this study, a central composite design and response surface methodology were used to study the effect of various hydrolysis variables (temperature, pH, and time) on the acid hydrolysis of Jerusalem artichoke-derived inulin using three different mineral acids (HCl, H2SO4, and H3PO4). Numerical optimization was used to maximize the sugar yield of Jerusalem artichoke powder within the experimental range for each of the mentioned acid. The influence of each acid on the formation of hydroxymethylfurfural (HMF; a known by-product and inhibitor for fermentative organisms) was also investigated. H2SO4 was found to have a better potential for sugar yields compared to two other acids (HCl and H3PO4) since it can hydrolyze the highest amount of inulin (98.5 %) under optimal conditions (temperature of 97 °C, pH of 2.0, and time period of 35 min) without producing inhibiting HMF concentrations. The sulfuric hydrolysate of Jerusalem artichoke was fermented via solventogenic clostridia to acetone-butanol-ethanol (ABE). An ABE yield of 0.31 g g−1 and an overall fermentation productivity of 0.25 g l−1 h−1 were obtained, indicating the suitability of this feedstock for fermentative ABE production.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In recent years, the academic and industrial biofuel sectors are increasingly investigating options beyond grain-based ethanol. Alternative biofuels, such as biomass-derived long-chain alcohols, are of growing importance [1–3]. Butanol (n-butanol) is a very promising biofuel exhibiting several advantages over ethanol and represents also an important bulk chemical for industrial purposes. It is more hydrophobic than ethanol (due to its two additional methyl groups), possesses less volatility, has a higher energy density, and is fully miscible with gasoline [4]. One of the major obstacles to commercial acetone-butanol-ethanol (ABE) fermentation is the high cost and availability concerns of conventional substrates (corn, molasses) [5]. Substrate cost account for over 50 % of the total production costs; therefore, it is crucially important, from a process economics perspective, to identify inexpensive biomass feedstocks that can be fermented by Clostridium species [4, 6–8]. While a number of low-cost fermentation substrates have previously been evaluated [9, 10], Jerusalem artichokes (Helianthus tuberosus L.) as an alternative carbon source have a good potential to be fermented to butanol. Jerusalem artichoke can grow well in non-fertile land and is resistant to plant diseases, not competing with grain crops for arable land [4–11]. Unlike typical crops that use starch, a glucose polymer, as energy storage, Jerusalem artichoke (as all member of the Asteraceae family) stored excess carbon as inulin, linear chains of β (2→1)-linked d-fructose units terminated by a d-glucose linked to fructose by α (1→2) bond [11]. Though the principal storage carbohydrate of Jerusalem artichoke is inulin (15 to 20 %), monomeric sucrose, glucose, and fructose are also present [12].
Most microorganisms cannot directly ferment inulin; therefore, inulin first needs to be hydrolyzed into fructose and glucose monomers. Hydrolysis can be achieved via an acid catalyst or enzymes. Acid hydrolysis as a simple pretreatment for inulin feedstock has a number of important advantages including a low-cost easily available acid catalyst and a short hydrolysis time [13]. However, acidic pretreatment strategies are also known for the irreversible production of growth and fermentation inhibitors, such as 5-hydroxymethylfurfural (HMF) [14]. At high concentrations, these inhibitors can substantially affect the fermenting organism [15, 16].
Various acids can be used as catalyst for inulin hydrolysis, but mineral acids were shown to be more effective compared to organic acids. Among mineral acids, hydrochloric acid, sulphuric acid, and phosphoric acid have been used in many studies for inulin hydrolysis; however, the available literature provides little information about the optimum condition and yield of inulin hydrolysis, nor a controlled comparison of different acids [13, 17–20]. Also the information about potentially fermentation-inhibiting hydrolysis by-product (HMF) is limited in the current literature.
The purpose of this study was therefore threefold: (1) to optimize acid hydrolysis of inulin to maximize its corresponding fermentable sugar yield using three different mineral acids (HCl, H2SO4, and H3PO4), (2) to examine the influence of each acid (HCl, H2SO4, and H3PO4) on HMF formation, and (3) to study the feasibility of butanol production from the hydrolysate of Jerusalem artichoke’s tuber.
Materials and Methods
Acid Hydrolysis
Preparation of Jerusalem Artichoke Flour
Jerusalem artichoke tubers, white flesh, were obtained from the Institute for Chemicals and Fuels from Alternative Resources (ICFAR), University of Western Ontario. The entire Jerusalem artichoke tubers were washed and sliced to approximately 2-cm cubes. The obtained slices were transferred directly to a drying oven and dried at 105 °C for 72 h, ground to fine particles using a coffee grinder, and passed through a 250-μm mesh. The prepared sample with approximately 3 % moisture content was stored in a dry container at 4 °C for further use.
Inulin Extraction
Inulin extraction was performed based on a method by Bekers et al. (2007). Extracts were obtained by adding 100 ml of water to 5 g of Jerusalem artichoke powder. The slurry was put into a water bath at 25 °C and agitated using a magnetic stirrer at 300 rpm for 1 h. The samples were then centrifuged for 20 min at 12,000 × g [21]. The supernatant contained the extractable carbohydrate fraction of Jerusalem artichoke tubers including 0.52 g g−1 inulin, 0.16 g g−1 fructose, 0.1 g g−1 glucose, and 0.05 g g−1 sucrose (Table 1). The precipitate contained the non-extractable fraction of Jerusalem artichoke tubers including 0.03 g g−1 cellulose and 0.02 g g−1 hemicellulose [25]. The cellulose and hemicellulose fraction in Jerusalem artichoke tubers is relatively small; therefore, only the supernatant was removed for HPLC analysis and acid hydrolysis.
Chemicals
Hydrochloric acid (12.2 M), sulphuric acid (18.0 M), and phosphoric acid (14.8 M) were obtained from Caledon (Ontario, Canada).
Experimental Design
A central composite design (CCD) with three factors was selected to evaluate the response pattern and to determine the optimal combination of temperature, pH, and time for maximizing inulin hydrolysis to fermentable sugars using three different mineral acids (HCl, H2SO4, and H3PO4). An initial full factorial design had shown significant curvature and confirmed the significance of all three parameters (data not shown), and the design was expanded to a CCD. The uncoded values for each parameter were as follows [low star point, low central point, center point, high central point, high star point]: temperature in degree Celsius [77.31, 80, 88.5, 97, 99.69], pH [1.84, 2.0, 2.5, 3.0, 3.16], and time in minutes [1.78, 7.0, 23.5, 40, 45.22]. The experimental design was developed using Design Expert 8.0.7.1 (Stat-Ease, Inc., Minneapolis, MS, USA) and resulted in 14 conditions for each acid. All conditions were tested in triplicates, including three center points. The resulting 51 conditions (8 × 3 factorial + 6 × 3 augmented + 3 × 3 center points) were fully randomized.
Acid Hydrolysis of Inulin
Batch acid hydrolysis was performed in 20-ml scintillation vials using the above selected experimental conditions for each of the mentioned acids (hydrochloric acid, sulphuric acid, and phosphoric acid). Each vial contained 10 ml of water-extracted inulin from Jerusalem artichoke tubers obtained in “Inulin Extraction” section, and the pH was adjusted using the respective acid. The concentrations of acid added to adjust the pH of extract to [2.0, 2.5, and 3.0] were as follows: HCl [4.4, 3.2, and 2.5 μl l−1], H2SO4 [1.7, 1.2, and 0.8 μl l−1], and H3PO4 [4.3, 2.5, and 1.6 μl l−1]. The vials were hermetically covered with Parafilm and aluminum foil to avoid evaporative loss, and the mixture was heated at the required temperature for the selected reaction time while shaking at 300 rpm. All hydrolysis assays were conducted in triplicate.
Statistical Analysis
Linear regression analysis was used to fit the experimental data with a second-order model as given in Eq. (1):
The experimental data was analyzed using Design Expert 8.0.7.1. The significance of each term was verified via analysis of the variance (ANOVA). The significance of each main effect, quadratic effects, and effects were determined based on an α of 0.05 using the F test. The fitted model was evaluated by normal probability plots, R 2 and adjusted R 2, and lack of fit coefficient for determining the adequacy. Numerical optimization via Design Expert 8.0.7.1 determined the optimal conditions for maximizing sugar yields. The model and optimization results were validated by performing experiments around the predicted optimum.
Analytical Methods
Concentration of sugars and HMF in Jerusalem artichoke juice, as well as hydrolysate, was determined via high performance liquid chromatography (HPLC) on an Agilent 1260 infinity (Agilent USA, Santa Clara) using an Agilent Hi-Plex H (7.7 × 300 mm) column and Cation H+ guard column (Agilent USA, Santa Clara) operating at 60 °C. A refractive index detector (RID) was used for sugar detection. Also a diode array detector (DAD) was used for HMF detection with spectral absorbance at 276 nm. Water was used as the isocratic mobile phase at a constant flow rate of 0.6 ml min−1. Before injection, samples were diluted to the appropriate concentration with deionized water and filtered through a 0.2-μm membrane filter. The analytes were quantified using pure inulin, HMF (Sigma Aldrich Co.), fructose, glucose, and sucrose (VWR Co.) as standards.
The total sugar yield was evaluated based on fructose and glucose production. The fructose to glucose ratio after complete conversion was 4 ± 0.45 (Table 1). Complete hydrolysis resulted in 0.15 and 0.60 g g−1 Jerusalem artichoke glucose and fructose, respectively. This was achieved by acid hydrolysis developed for analytical purposes at a temperature of 100 °C, pH of 2.0, and reaction time of 60 min using H2SO4 [22].
Fermentation
Chemicals
Yeast extract and peptone were obtained from BD—Becton, Dickinson and Company (New Jersey, USA). Soluble starch was purchased from Alfa Aesar (Massachusetts, USA). Glucose was from AMRESCO (Ohio, USA) and MgSO4 was from EMD Millipore (Massachusetts, USA). Ammonium acetate, KH2PO4, and K2HPO4 were purchased from Caledon (Ontario, Canada). FeSO4 and NaCl were obtained from BDH (Georgia, USA).
General Microbiological Conditions
All microbiological work was performed in an aseptic anaerobic chamber (Model 855-ACB, Plas Labs, Lansing, MI).
Strain and Maintenance
Clostridium saccharobutylicum DSM 13864 was purchased from Leibniz Institute DSMZ-German Collection of Microorganisms and Cell Cultures. Cultures of this strain were routinely maintained as spore suspensions in a seed medium containing (per liter) 3 g of yeast extract, 5 g of peptone, 5 g of soluble starch, 5 g of glucose, 2 g of ammonium acetate, 2 g of NaCl, 3 g of MgSO4, 1 g of KH2PO4, 1 g of K2HPO4, and 0.1 g of FeSO4, with pH 6.0 at 4 °C. Spores in the seed medium were heat shocked for 2 min at 90 °C and transferred to a fresh seed medium. Three milliliters of actively growing cells was inoculated into 50 ml of inoculum development P2 medium, prepared in a 100-ml screw-capped bottle. The P2 medium contained 30 g l−1 glucose, 1 g l−1 yeast extract, and stock solutions (minerals, buffer, and vitamins) [7]. The solution containing glucose and yeast extract was sterilized at 121 °C for 20 min, and 0.5 ml of each of the filter-sterilized stock solutions were added to 50 ml glucose-yeast extract solution. Subsequently the bottles were placed in an anaerobic chamber for 24 h. The culture (inoculum) was allowed to grow for approximately 10 h at 37 °C before it was inoculated into the ABE production medium.
ABE Fermentation
All fermentation studies were conducted in 150-ml flasks containing 100 ml of fermentation medium. Control fermentation medium contained 60 g l−1 glucose, fructose, or mixed sugars (fructose and glucose in the ratio of 3:1), 1 g l−1 yeast extract, and stock solutions (minerals, buffer, and vitamins) [7]. The solution containing glucose and yeast extract was sterilized at 121 °C for 20 min, and 1 ml of each of the filter-sterilized stock solutions was added to 100 ml glucose-yeast extract solution. Ten milliliters of actively growing cells was inoculated into 100 ml of fermentation medium and kept in an anaerobic chamber at 200 rpm for 72 h.
For the hydrolysate fermentation, the pH was adjusted to 6.0 using 1 M NaOH solution. One milliliter of sterile yeast extract solution (1 g l−1) and 1 ml of each stock solution were added to each bottle to reach the same nutrient concentration level as in P2 medium. Subsequently the bottles were inoculated with 10 ml of actively growing culture followed by incubation at 37 °C. Following this, the bottles were kept in an anaerobic chamber and placed on a shaker running at 200 rpm for 72 h. Samples were taken intermittently and filtered using 0.2-μm grade filters. Clear liquid was stored at −20 °C for ABE and sugar analysis (HPLC, see section 2.1.7 for conditions).
Analytical Methods
Bacterial growth was monitored by measuring the optical density at 600 nm using a 200 pro infinite series microplate reader (Tecan, Switzerland) using 96-well microplates at 200 μl per well. Concentration of solvents produced in the fermentation was determined via HPLC as described in “Analytical Methods” section. Total solvents were quantified using pure butanol, acetone, and ethanol (Caledon, Ontario, Canada) as standards.
Productivity was calculated as the maximum ABE concentration achieved (g l−1) divided by the fermentation time at a fixed time of 60 h and is expressed as gram per liter per hour. Product yield was calculated as the total amount of solvents produced divided by the amount of fermentable sugar utilized and is expressed as gsolvent g−1 sugar. At least three parallel samples were used in all analytical determinations, and data are presented as the means of three replicates.
Results and Discussion
The total solid content of Jerusalem artichoke tuber used in this study was about 30 % of the fresh weight. Inulin, fructose, glucose, and sucrose composition of the material are shown in Table 1. Samples for analysis were randomly taken from the available material and the small standard deviation indicates the compositional homogeneity of the tubers. The measured values are in agreement with values typically found for Jerusalem artichoke [12–24].
Acid Hydrolysis
Experimental conditions were chosen based on a central composite design, and the actual values of the independent variables and the measured responses are shown in Table 2. Acid hydrolysis was performed on water-extracted inulin. Hydrolyzing the complete tuber could increase the total sugar yield, as the selected condition would also favor hydrolysis of the cellulose faction, releasing additional glucose monomers. However, the typical cellulose content in Jerusalem artichoke tuber is <0.03 g g−1 while also 0.02 g g−1 hemicellulose is present [25]. The potential increase in fermentable sugar is negligible considering 0.83 g g−1 of extractable carbohydrates (Table 1), and the hemicellulose fraction would likely result in the formation of fermentation-inhibiting by-products under the employed acid hydrolysis conditions.
Response Surface Model Validation
The total sugar yield was chosen as the only response factor when evaluating the results of the CCD experiments. The HMF selectivity was not a suitable response as it was below the detection limit for multiple experimental conditions. Other factors such as rate constants or pseudo-rate constants were also not suitable, as the goal of the CCD was to establish a simple empirical correlation between the final sugar yield and multiple parameters, including the hydrolysis time. The hydrolysis would not affect the rate constant, unless hydrolysis follows a more complex mechanism as in the case of hemicellulose [26, 27]. A detailed mechanistic study of acid hydrolysis was not the aim of this study, hence the choice of a CCD followed by response surface methodology.
As can be seen from the experimental results in Table 2, the hydrolysis using any of the three mentioned acids (HCl, H2SO4, and H3PO4) was successful in converting inulin to monomeric sugars within the ranges of the input variables.
The complete dataset could be fitted with a quadratic model as describe in Eq. (1) for each acid. The resulting model parameters are shown in Table 3. The F values of the models are 73.4, 304.2, and 49.7 for HCl, H2SO4, and H3PO4, respectively. The F values are very high compared to the critical values, indicating that all three models are highly significant. The significance of each parameter coefficient was determined by p values. In this case, all factors have great effect on acid hydrolysis using any of the three acids. The quadratic effects of pH, time, as well as temperature and interaction effect of every two variables on sugar yields for each acid are shown in Table 3. The goodness of fit of each model was confirmed by the coefficient of determination R 2 and adjusted determination coefficient Adj. R 2 (Table 3).
Based on the selected significant variables, the quadratic model for the sugar yield in terms of actual factors is shown as follows:
HCl
H2SO4
H3PO4
The residuals can be judged as normally distributed based on a normal probability (data not shown).
Combined effect of Temperature, pH, and Time
Response surface methodology was used to study the interaction effects of the three factors using any of the mentioned mineral acids. Surface plots of the combined effects of pH and time on sugar yield using HCl, H2SO4, and H3PO4 at a constant temperature of 88.5 are shown in Fig. 1a, b, c, respectively. The sugar yield is a function of both the pH and time using any of the mentioned mineral acids. The plots clearly indicate that an optimum exists within the observed design space with respect to pH and time, and increasing the temperature appears to increase the sugar yield over the observed design space. The sugar yield is also a function of both the pH and temperature only when H2SO4 was used as catalyst.
Response Optimization and Model Validation
Based on the model, numerical optimization was used to determine the optimal combination of process parameters for the maximum sugar yield. The optimal conditions were a temperature of 96 °C, pH of 2.0, and time period of 36 min using HCl; temperature of 97 °C, pH of 2.0, and time period of 35 min using H2SO4; and temperature of 95 °C, pH of 2.0, and 39-min time period using H3PO4. The best result was observed using H2SO4 which can hydrolyze up to 98.5 % of inulin under optimal conditions compared to 95.5 and 94 % using HCl and H3PO4, respectively. To the best knowledge of the authors, this is the first attempt to optimize the conditions of acid hydrolysis of water-extracted inulin from Jerusalem artichoke tubers using different mineral acids and to compare the effectiveness of each acid on hydrolysis at optimal conditions.
In a closely related study, the hydrolysis of Jerusalem artichoke tubers was carried out using HCl at a temperature of 120 °C, pH of 2.5, for 60 min resulting in 68 % hydrolysis of inulin [17], which is lower than the results obtained in this study. It has been reported that the sugar yield, evaluated as fructose and glucose production, is very heat sensitive in acidic conditions as the products easily degrade at temperature higher than 97 °C [19], as was the case for the work of Razmovski et al. [20], resulting in a lower hydrolysis yield. Szambelan et al. reported complete inulin hydrolysis in 1 h using H2SO4 at temperature of 100 °C, and pH of 2.0 [22], which is close to the results obtained in this study in terms of temperature and pH. The longer hydrolysis time can be potentially explained by different methods of preparation of Jerusalem artichoke tubers for hydrolysis. The Jerusalem artichoke powder used in this study appears to have positive effects on hydrolysis time compare to mashed tubers used by Szambelan et al. [22]. In another study, H3PO4 was used to hydrolyze 90 % of inulin in 7 h at temperature of 80 °C, and pH of 2.0 [18], which is close to the result of this study at temperature of 80 °C, pH of 2.0, and time period of only 40 min.
To validate the applicability of this RSM model, some confirming experiments were carried out around the estimated optimal conditions. The measured and predicted sugar yields of three conditions around the optimum are listed in Table 4. The predicted results were compared with the actual values obtained experimentally. T test at 95 % confidence showed no significant difference between the predicted and actual values. In summary, the proposed RSM model could be a useful model for the prediction of maximum sugar yield.
HMF Contents of Jerusalem Artichoke Hydrolysate
The data on HMF formed during acid hydrolysis of Jerusalem artichoke using three different mineral acids (HCl, H2SO4, and H3PO4) are shown in Table 2. An increase in temperature, decrease in the pH, and increase in hydrolysis time are expected to result in an increase of HMF during dilute acid hydrolysis [17–20] which is also the result of this study. However, the use of less-intense conditions, temperature of 80 to 97 °C, and time of 7 to 40 min resulted in a low amount of HMF (0.0–6.4 mgHMF g−1 reducing sugar). Accordingly, the highest HMF concentration in a batch fermentation of the Jerusalem artichoke hydrolysate with an initial sugar concentration of 50 g l−1 would be 0.32 g l−1. This worst-case value was achieved in the Jerusalem artichoke hydrolysate using H3PO4 as catalyst at temperature of 97 °C, pH of 2.0, and time period of 40 min (Table 2). In a related study, the effect of HMF on Clostridium beijerinckii growth and fermentation was assessed which shows no negative effect on growth and butanol fermentation up to 2 g l−1 HMF concentration [28]. A similar result was obtained from the work of Zhang et al. [29] and Qureshi et al. [30]. By comparison, the highest HMF concentration in this study (6.4 mgHMF g−1 reducing sugar) is noticeably lower than the HMF concentration typically considered inhibitory to the growth and fermentation.
Within the current design space, phosphoric acid produced the highest HMF concentration, followed by hydrochloric acid and sulfuric acid. It is clear that the nature of the acid can influence the HMF formation. Taking into consideration the efficiency of inulin hydrolysis, expressed as maximum yield of fermentable sugars (fructose and glucose) and non-inhibiting HMF concentration, it can be concluded that H2SO4 seems to have a better potential as a catalyst for inulin hydrolysis compared to two other acids (HCl and H3PO4).
Comparison of Acid and Enzymatic Hydrolysis of Water-Extracted Inulin from Jerusalem Artichoke Tubers
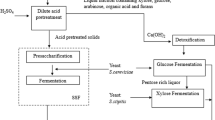
Hydrolysis of water-extracted inulin from Jerusalem artichoke tubers can be achieved via an acid catalyst or enzymes. Acid hydrolysis can lead to fermentation-inhibiting by-products, while enzymatic hydrolysis is dependent on potentially high-cost enzymes and has a longer hydrolysis time. Results typically achieved through enzymatic hydrolysis are summarized in Table 5 with hydrolysis times varying between 2 and 72 h and yields between 70 and 95 %. By comparison, sulphuric acid in this study can hydrolyze up to 98.5 % of inulin within 35 min with a non-inhibiting HMF concentrations, while the same Jerusalem artichoke extract required 24 h to achieve similar numbers enzymatically [4]. The shorter reaction times and lower catalyst costs would imply acid hydrolysis to be favorable over enzymatic conversion if conducted as separate process steps.
Alternatively consolidated bioprocessing can be used allowing enzymatic hydrolysis to occur simultaneously with the fermentation step. Yeast strains expressing high levels of inulases have been developed and successfully used for ethanol production from Jerusalem artichoke [31]. However, not very biotransformation can be achieved by inulase-producing strains. In such cases, simultaneous saccharification and fermentation (SSF) can still be realized by adding commercial enzyme during the fermentation stage [25], which might also be possible for the butanol process due to the relatively low pH value during the butanol fermentation.
ABE Fermentation from Mixed Sugars and Acid Hydrolysate of Jerusalem Artichoke by C. saccharobutylicum DSM 13864
Prior to carrying out butanol fermentation on Jerusalem artichoke-derived carbohydrates, control experiments with synthetic media simulating the hydrolysate were carried out with C. saccharobutylicum DSM 13864. The initial total sugar level was 55 g l−1, including 14 g l−1 glucose and 41 g l−1 fructose. As was shown in Fig. 2a, the culture started to use glucose and fructose directly after the inoculation. Almost all the glucose was utilized by the culture within 24 h. In contrast, 79.6 % fructose was consumed at 60 h, leaving behind 8.4 g l−1 unused fructose in the medium. It was anticipated that the glucose utilization rate was greater than the fructose rate, as glucose is the preferred carbon source [32]. In a closely related study, batch fermentation of the glucose/fructose mixture by Clostridium acetobutylicum L7 on a complex medium showed that this bacterium metabolizes glucose first and rapidly before utilizing fructose for ABE production [33]. Results presented in Fig. 2a suggest that the C. saccharobutylicum DSM 13864 was able to utilize glucose and fructose simultaneously. A solvent concentration of 15.1 g l−1 was achieved after 60-h fermentation with 3.1 g l−1 acetone, 2.3 g l−1 ethanol, and 9.7 g l−1 butanol. Yield and productivity of the solvent were 0.32 ± 0.008 gsolvent g−1 sugar and 0.25 ± 0.002 g l−1 h−1, respectively. The yields obtained in this work are of similar values reported for ABE fermentation with Clostridium (0.25–0.37 gsolvent g−1 sugar) [34].
Profiles of solvent production and sugar utilization in a mixed sugar and b hydrolysate of Jerusalem artichoke by Clostridium saccharobutylicum DSM 13864. Residual glucose content (filled square), residual fructose content (filled circle), ethanol concentration (filled diamond), acetone concentration (star), butanol concentration (filled triangle), and combined solvent concentration (filled inverted triangle). A biomass increased was observed during the fermentation through an increase of turbidity from 0.185 to 1.654 OD units
Hydrolysate of Jerusalem artichoke was obtained using H2SO4 under optimal conditions. It was subsequently used for ABE fermentation by C. saccharobutylicum DSM 13864 under similar condition as in the control experiment. For the hydrolysate fermentation, the pH was adjusted to 6.0 using 1 M NaOH solution. At the beginning, 59.4 g l−1 sugars were present, of which glucose and fructose were 16.8 and 42.6 g l−1, respectively. After 24 h of fermentation, glucose was completely utilized, as was shown in Fig. 2b. When the fermentation stopped at 60 h, 73.6 % fructose was used, leaving behind 11.2 g l−1 fructose unused, compared to 8.4 g l−1 fructose when mixed sugar was used. At the end of the fermentation, the culture produced 15.1 g l−1 ABE, resulting in a productivity of 0.25 ± 0.008 g l−1 h−1. The individual levels of solvents were acetone 3.0 g l−1, ethanol 2.3 g l−1, and butanol 9.8 g l−1 (Fig. 2b). The culture used 48.2 g l−1 sugar to produce 15.1 g l−1 ABE, thus resulting in a yield of 0.31 ± 0.004 gsolvent g−1 sugar. Based on the amount of sugars present in the medium, the maximum theoretical yield is 0.39 gsolvent g−1 sugar [35] corresponding to a percent yield of 80 %. In a comparable study, C. acetobutylicum L7 was used for hydrolysate fermentation of Jerusalem artichoke with 62.9 g l−1 sugars, resulting in a solvent concentration of 17.2 g l−1, corresponding to a yield of 0.29 gsolvent g−1 sugar [33], which appears to be lower than the results obtained in this study; however, a larger amount of sugars could be converted. It has been reported that raising the initial carbohydrate concentration above 60 g l−1, as was the case for the work of Chen et al., will reduce the fermentation efficiency [34]. Additional deviation can be potentially explained by strain characteristics of the Clostridia (L7 vs. DSM 13864).
Fermentation with water-extracted inulin from Jerusalem artichoke tubers showed identical yields (within error) with mixture of glucose and fructose as a control fermentation. This indicates that acid hydrolysate of Jerusalem artichoke is a reliable feedstock for ABE production.
The experimental work in the study largely focuses on the acid hydrolysis of water-extracted inulin from Jerusalem artichoke tubers as a potential feedstock for butanol production. The fermentation process was not optimized and the setup used in this study is not intended to represent a potential industrial process. More advanced fermentation process design, possibly including continuous fermentation and/or in situ product removal, would likely have to be used in an industrial process, as evaluated for different feedstocks elsewhere [36, 37].
Conclusions
The optimal conditions for inulin hydrolysis to glucose and fructose are temperature of 97 °C, pH of 2.0, for 35 min using H2SO4, which was the most suitable acid tested. Phosphoric acid resulted in the highest amount of HMF followed by hydrochloric acid, whereas sulfuric acid results in the lowest HMF concentration. C. saccharobutylicum DSM 13864 is able to ferment acid hydrolysate of Jerusalem artichoke similarly to fermenting synthetic medium with an equivalent carbohydrate composition. The culture converts 48.2 g l−1 sugar to 15.1 g l−1 ABE, resulting in a yield of 0.31 gsolvent g−1 sugar. Therefore, the inulin of Jerusalem artichoke can be seen as a good raw material for butanol production after simple acid hydrolysis.
References
Schiel-Bengelsdorf B, Montoya J, Linder S, Dürre P (2013) Butanol fermentation. Environ Technol 34:1691–1710. doi:10.1080/09593330.2013.827746
Alvira P, Tomás-Pejó E, Ballesteros M, Negro MJ (2010) Pretreatment technologies for an efficient bioethanol production process based on enzymatic hydrolysis: a review. Bioresour Technol 101:4851–4861. doi:10.1016/j.biortech.2009.11.093
Sánchez OJ, Cardona CA (2008) Trends in biotechnological production of fuel ethanol from different feedstocks. Bioresour Technol 99:5270–5295. doi:10.1016/j.biortech.2007.11.013
Sarchami T, Rehmann L (2014) Optimizing enzymatic hydrolysis of inulin from Jerusalem artichoke tubers for fermentative butanol production. Biomass Bioenergy 69:175–182. doi:10.1016/j.biombioe.2014.07.018
Jones DT, Woods DR (1986) Acetone-butanol fermentation revisited. Microbiol Rev 50:484–524
Dürre P (2007) Biobutanol: an attractive biofuel. Biotechnol J 2:1525–1534. doi:10.1002/biot.200700168
Qureshi N, Ezeji TC, Ebener J et al (2008) Butanol production by Clostridium beijerinckii. Part I: use of acid and enzyme hydrolyzed corn fiber. Bioresour Technol 99:5915–5922. doi:10.1016/j.biortech.2007.09.087
García V, Päkkilä J, Ojamo H et al (2011) Challenges in biobutanol production: how to improve the efficiency? Renew Sustain Energy Rev 15:964–980. doi:10.1016/j.rser.2010.11.008
Raganati F, Olivieri G, Procentese A et al (2013) Butanol production by bioconversion of cheese whey in a continuous packed bed reactor. Bioresour Technol 138:259–265
Gao K, Rehmann L (2014) ABE fermentation from enzymatic hydrolysate of NaOH-pretreated corncobs. Biomass Bioenergy 66:110–115. doi:10.1016/j.biombioe.2014.03.002
Szambelan K, Nowak J, Jelen H (2005) The composition of Jerusalem artichoke (Helianthus tuberosus L.) spirits obtained from fermentation with bacteria and yeasts. Eng Life Sci 5:68–71. doi:10.1002/elsc.200400052
Matías J, González J, Royano L, Barrena R (2011) Analysis of sugars by liquid chromatography-mass spectrometry in Jerusalem artichoke tubers for bioethanol production optimization. Biomass Bioenergy 35:2006–2012. doi:10.1016/j.biombioe.2011.01.056
Tasić MB, Konstantinović BV, Lazić ML, Veljković VB (2009) The acid hydrolysis of potato tuber mash in bioethanol production. Biochem Eng J 43:208–211. doi:10.1016/j.bej.2008.09.019
Pedersen M, Viksø-Nielsen A, Meyer AS (2010) Monosaccharide yields and lignin removal from wheat straw in response to catalyst type and pH during mild thermal pretreatment. Process Biochem 45:1181–1186. doi:10.1016/j.procbio.2010.03.020
Almeida RM, Modig T, Petersson A et al (2007) Increased tolerance and conversion of inhibitors in lignocellulosic hydrolysates by Saccharomyces cerevisiae. Journal Chem Technol Biotechnol 349:340–349. doi:10.1002/jctb
Schwab K, Wood JA, Rehmann L (2013) Pyrolysis byproducts as feedstocks for fermentative biofuel production: an evaluation of inhibitory compounds through a synthetic aqueous phase. Ind Eng Chem Res 52:18234–18240. doi:10.1021/ie403354k
Razmovski R, Vucurovic V, Miljic U, Puskas V (2013) Effect of temperature on acid hydrolysis of Jerusalem artichoke as raw material for ethanol production. Acta Period Technol 44:279–287. doi:10.2298/APT1344279R
Jain VK, Baratti J (1985) Effect of acid or enzymatic hydrolysis on ethanol production by Zymomonaz mobilis growing on Jerusalem artichoke juice. Biotechnol Lett 530:527–530
Kim K, Hamdy MK (1986) Acid hydrolysis of Jerusalem artichoke for ethanol fermentation. Biotechnol Bioeng XXVIII:138–141
Razmovski RN, Šćiban MB, Vučurović VM (2011) Bioethanol production from Jerusalem artichoke by acid hydrolysis. Rom Biotechnol Lett 16:6497–6503
Bekers M, Grube M, Upite D et al (2007) Carbohydrates in Jerusalem artichoke powder suspension. Nutr Food Sci 37:42–49. doi:10.1108/00346650710726940
Szambelan K, Nowak J (2006) Acid and enzymatic hydrolysis of Jerusalem artichoke (Helianthus tuberosus) tubers for further ethanol production. Electron J Polish Agric Univ 9:36
Bekers M, Grube M, Upite D et al (2008) Inulin syrup from dried Jerusalem artichoke. LLU Raksti 21:116–121
Böhm A, Kaiser I, Trebstein A, Henle T (2004) Heat-induced degradation of inulin. Eur Food Res Technol 220:466–471. doi:10.1007/s00217-004-1098-8
Dao TH, Zhang J, Bao J (2013) Characterization of inulin hydrolyzing enzyme(s) in commercial glucoamylases and its application in lactic acid production from Jerusalem artichoke tubers (Jat). Bioresour Technol 148:157–162. doi:10.1016/j.biortech.2013.08.123
Zhang C, Houtman CJ, Zhu JY (2014) Using low temperature to balance enzymatic saccharification and furan formation during SPORL pretreatment of Douglas-fir. Process Biochem 49:466–473. doi:10.1016/j.procbio.2013.12.017
Zhu W, Houtman CJ, Zhu JY et al (2012) Quantitative predictions of bioconversion of aspen by dilute acid and SPORL pretreatments using a unified combined hydrolysis factor (CHF). Process Biochem 47:785–791. doi:10.1016/j.procbio.2012.02.012
Ezeji T, Qureshi N, Blaschek HP (2007) Butanol production from agricultural residues: impact of degradation products on Clostridium beijerinckii growth and butanol fermentation. 97:1460–1469. doi: 10.1002/bit
Zhang Y, Han B, Ezeji TC (2012) Biotransformation of furfural and 5-hydroxymethyl furfural (HMF) by Clostridium acetobutylicum ATCC 824 during butanol fermentation. N Biotechnol 29:345–351. doi:10.1016/j.nbt.2011.09.001
Qureshi N, Bowman MJ, Saha BC et al (2012) Effect of cellulosic sugar degradation products (furfural and hydroxymethyl furfural) on acetone–butanol–ethanol (ABE) fermentation using Clostridium beijerinckii P260. Food Bioprod Process 90:533–540. doi:10.1016/j.fbp.2011.09.002
Guo L, Zhang J, Hu F et al (2013) Consolidated bioprocessing of highly concentrated Jerusalem artichoke tubers for simultaneous saccharification and ethanol fermentation. Biotechnol Bioeng 110:2606–2615. doi:10.1002/bit.24929
Gao K, Li Y, Tian S, Yang X (2012) Screening and characteristics of a butanol-tolerant strain and butanol production from enzymatic hydrolysate of NaOH-pretreated corn stover. World J Microbiol Biotechnol 28:2963–2971. doi:10.1007/s11274-012-1107-1
Chen L, Xin C, Deng P, Ren J, Liang HBF (2010) Butanol production from hydrolysate of Jerusalem artichoke juice by Clostridium acetobutylicum L7. Chin J Biotechnol 26:991–996
Shaheen R, Shirley M, Jones DT (2000) Comparative fermentation studies of industrial strains belonging to four species of solvent-producing clostridia. J Mol Microbiol Biotechnol 2:115–124
Yerushalmi L, Volesky B, Leung WK, Neufeld RJ (1983) Variations of solvent yield in acetone-butanol fermentation. Eur J Appl Microbiol Biotechnol 18:279–286
Napoli F, Olivieri G, Russo ME et al (2011) Continuous lactose fermentation by Clostridium acetobutylicum—assessment of acidogenesis kinetics. Bioresour Technol 102:1608–1614. doi:10.1016/j.biortech.2010.09.004
Lee SY, Park JH, Jang SH et al (2008) Fermentative butanol production by Clostridia. Biotechnol Bioeng 101:209–228. doi:10.1002/bit.22003
Acknowledgments
The authors wish to thank the Institute for Chemicals and Fuels from Alternative Resources (ICFAR) for providing Jerusalem artichoke tubers as well as the Sand Plains Community Development Fund, Agriculture Canada, Natural Sciences and Engineering Research Council of Canada (NSERC), and the Canada Foundation for Innovation (CFI) for the financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sarchami, T., Rehmann, L. Optimizing Acid Hydrolysis of Jerusalem Artichoke-Derived Inulin for Fermentative Butanol Production. Bioenerg. Res. 8, 1148–1157 (2015). https://doi.org/10.1007/s12155-014-9568-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12155-014-9568-8