Abstract
The objective of this retrospective study was to evaluate the prognostic significance of volume-based metabolic markers of PET/CT along with clinical characteristics in patients with in-operable cervical carcinoma.
Methods
Fifty-eight patients with cervical carcinoma (stage IIB–IVB) underwent FDG PET/CT for pretreatment evaluation and included in this study. Patients were staged according to International Federation of Gynecology and Obstetrics [FIGO] system. After chemoradiation therapy, patients were evaluated for persistent disease (PD) by clinical examinations, smear tests, pelvic MRI and PET/CT. Based upon follow-up results, clinical characteristics (patient age, tumor histology, FIGO stage) and PET/CT findings such as presence of PET-positive pelvic/para-aortic lymph nodes (LN), metabolic tumor volume (MTV), total lesion glycolysis (TLG), maximum standardized uptake values (SUVmax) of tumor and lymph nodes were analyzed for disease persistence. Survival analysis for disease-free survival and overall survival was performed with Kaplan–Meier method using PET findings and clinical characteristics.
Results
At the time of last follow-up (mean: 22 ± 12.6 months, range 6–48), 38 patients (65 %) had PD, 20 patients (35 %) had no evidence of disease (NED). Patient age, tumor histology, MTV, TLG and tumor SUVmax did not differ between groups. The frequency of PET-positive pelvic/para-aortic lymph nodes (84 vs. 60 %, p = 0.03), LN SUVmax (10.2 vs. 6.5, p = 0.02), and FIGO stage differed significantly between PD and NED groups. Cox proportional hazards model demonstrated advanced FIGO stage and the presence of PET-positive para-aortic LN were independent predictors for PD. Both disease-free survival and overall survival curves showed progressive worsening as the disease advanced, p = 0.015. PET LN status was the most important prognostic indicator for disease-free survival and overall survival. The worst outcome curves were detected for patients with PET-positive para-aortic lymph nodes among all patients, p = 0.03.
Conclusion
Advanced FIGO stage and the presence of FDG-avid para-aortic lymph nodes on pretreatment PET/CT are significant prognostic biomarkers for PD and decreased overall survival in patients with in-operable cervical carcinoma independent from MTV, TLG, tumor and lymph node SUVmax.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Cervical carcinoma is the third most frequently diagnosed malignancy in women worldwide and is one of the leading causes of cancer deaths [1, 2]. Traditional prognostic factors in cervical carcinoma include patient age, performance status, clinical tumor stage, lymph node involvement and treatment skills. However, today, many clinical factors, pathological characteristics and/or metabolic parameters of the tumor are proven to correlate with clinical outcome [2–10].
In recent years, the role of F18-fluorodeoxyglucose (FDG) positron emission tomography/computed tomography (PET/CT) in the staging and management of gynecological cancers has been increasing. Strong evidence from a growing number of research support the value of PET/CT in the pretreatment work-up of patients with cervical cancer, as the modality with higher sensitivity and positive predictive value compared to CT and MRI [11–13]. In addition, in our previous report, we observed that PET/CT provides the unique advantage to detect both para-aortic lymphatic involvement and distant metastasis and contributed to patient management plan in a considerable proportion of patients with cervical cancer [11]. Furthermore, beyond accurately determining disease extent, FDG PET/CT provides prognostic information by quantifying FDG uptake in terms of standardized uptake value (SUV) and measuring volume-based metabolic parameters such as metabolic tumor volume (MTV) and total lesion glycolysis (TLG) [14–17].
SUV, as a semi-quantitative measurement of FDG uptake provides intrinsic molecular-biological information of tumors. Such that, high SUVmax of the primary tumor predicts a worse clinical outcome [9, 17–21]. MTV defined as the volume of tumor tissues with increased FDG uptake, is a volumetric and quantitative measurement of tumor cells with high glycolytic activity and reflects the tumor burden. TLG is another volume-based metabolic parameter which represents the metabolic burden of disease, and depends on both tumor volume and glucose utilization rate. Recently, the use of MTV and TLG was suggested as reliable parameters in providing more details about the status of disease in various types of cancers, such as lung tumors, cervical cancers and head-and-neck cancers [21–25].
Up-to-date, several studies are reported concerning the prognostic significance of many metabolic and histopathological parameters in cervical cancer patients. Our group previously evaluated the clinical impact of PET/CT on the pretreatment work-up of a similar patient population with locally advanced cervical cancer and found that the presence of FDG-avid lymph nodes was inversely correlated with prognosis in univariate analysis [11]. Nevertheless, still, there is some controversy and confusion in the literature about which metabolic parameters may be genuinely prognostic when analyzed together with patients’ clinical characteristics for widely varied patient populations [14–17]. For this purpose, we aimed to further evaluate indicators related to poor prognosis in patients with in-operable cervical carcinoma.
In this study, by analyzing volume-based metabolic parameters of PET/CT such as MTV, TLG, tumor SUVmax, lymph node SUVmax, and the presence of FDG-avid lymph nodes in a multivariate analysis together with clinical characteristics such as age at diagnosis and clinical FIGO stage, we aimed to establish a clinical and metabolic imaging profile to identify prognostic factors associated with poor disease-free survival and overall survival in a series of 58 patients treated and followed up in our institution.
Methods
Fifty-eight patients with in-operable cervical carcinoma (FIGO stage IIB–IVB), who underwent FDG PET/CT imaging for pretreatment evaluation, were included in this retrospective study. Patients with early stage cervical cancer (FIGO stage IA–IIA), patients with a prior history of neoadjuvant chemotherapy for cervical cancer, patients with previous diagnosis of another malignant disease, and patients with a follow-up duration less than 3 months were excluded from the study.
This study was approved by the ethics committee of our institution.
Patients were imaged on an integrated PET/CT scanner (Siemens Biograph 6-True Point PET/CT systems). Patients were fasted for at least 6 h prior to injection of 5.3 MBq/kg (144 μCi/kg) 18F-FDG. The blood glucose levels were less than 150 mg/dl in all patients at the time of the FDG injection. Unenhanced CT images were acquired for attenuation correction from the base of the skull to distal thigh using 3 mm slice thickness and calculated effective mAs due to patient weight. The PET and CT images were reviewed on a workstation (Leonardo, Siemens Medical Solutions) in all standard planes along with maximum-intensity-projection images and were analyzed visually and quantitatively by two reviewers experienced in interpreting PET/CT scans. Findings were recorded by consensus.
The maximum standardized uptake value was used to quantify FDG uptake. SUVmax was calculated using the following formula:
where, Cmax is the activity concentration in the voxel of highest tumor activity (Bq/ml), TBW is the total body weight (kg), and IA is the injected activity (kBq).
To provide the most accurate measurement of SUVmax, voxels were created large enough to maintain tumor inside the boundaries, and maximum care was accomplished to avoid “spill-in” from adjacent structures with intense FDG uptake. For this reason, images were acquired immediately after voiding.
Tumor volume was measured from attenuation-corrected FDG PET/CT images using an SUV-based automated contouring program (Syngo MI applications, Volumetric Analysis 6.0.14.4, Siemens Medical Solutions). The boundaries of tumor were drawn large enough to incorporate each target lesion in the axial, coronal, and sagittal FDG PET/CT images. Depending on previous data, a fixed threshold value of 40 % of SUVmax was used [26]. The voxels presenting SUV intensity greater than that of 40 % of SUVmax within the contouring margin were incorporated to define the tumor volumes.
Total lesion glycolysis was calculated as the sum of the product of SUVmean and MTV of primary tumor (TLG = sum of [SUVmean × MTV]).
The size and FDG uptake in pelvic and para-aortic lymph nodes were evaluated by PET/CT. It is known that for tumors smaller than the reconstructed spatial resolution, the partial volume effect may result in a more than 50 % underestimation of the true FDG concentration [27]. Because standard uptake values can be affected by many technical factors, and besides, SUV measurements can be underestimated by partial volume effect, the presence of focal increased FDG uptake in the lymph nodes, regardless of its size and standard uptake value, was considered as “PET positive” and suggestive of nodal involvement.
All patients were staged clinically according to International Federation of Gynecology and Obstetrics [FIGO] staging criteria. All patients were treated with concurrent chemotherapy and radiation therapy following the recommendations in National Comprehensive Cancer Network (NCCN) guidelines [28]. Patients with stage IIB–IVA disease received a total of 45 Gy external beam radiation therapy (EBRT) (in conventional fractionation of 1.8 Gy daily). Starting from the third week of external radiation therapy, a total of 40 Gy intracavitary brachytherapy was administered to Point A. In addition, patients with FDG-avid para-aortic lymph nodes received 45 Gy EBRT to para-aortic region. All patients with stage IIB-stage IVA disease received concomitant cisplatin-based chemotherapy (40 mg/m2 weekly for 6 cycles). Patients with stage IVB disease received systemic chemotherapy and palliative radiation therapy as indicated for system management.
Patients had follow-up examinations approximately every 2 months for the first 6 months and every 3 months for the next 2 years. Pap smear tests, abdominal and pelvic MRI and/or PET/CT were repeated 3 months after the completion of treatment and when warranted by clinical examinations or symptoms. The sites and findings related with malignant disease were recorded. Persistent pelvic disease was defined as residual or recurrent cervical tumor, lymph nodes, or both.
Statistical analysis
Clinical characteristics and PET parameters were analyzed for correlation with outcome data. The association of disease persistence with clinical characteristics and PET parameters were analyzed by univariate analysis using a Chi square test. Tumor SUVmax was compared to MTV using regression analysis. The Cox proportional hazards model was used for multivariate analyses. Disease-free survival and overall survival were calculated using the Kaplan–Meier method. The statistical analyses were performed using commercial software (SPSS 13.0). p values <0.05 were considered to be statistically significant.
Results
Patient and tumor characteristics are listed in Table 1.
The mean age was 57.7 ± 14.5 years in our patient population with a range of 26–86 years. 52 patients had squamous, 1 patient had adenosquamous and 5 had nonsquamous carcinoma. According to FIGO staging criteria, 19 patients had stage IIB, 4 patients had stage IIIA, 6 patients had stage IIIB, 12 patients had stage IVA, and 17 patients had stage IVB disease.
The mean follow-up duration was 22 ± 12.6 months with a range of 6–48 months. At the time of last follow-up, 20 patients (35 %) had no evidence of disease (NED group). Thirty-eight patients (65 %) had persistent disease (PD) and defined as PD group. 13 patients had pelvic disease (either residual/recurrent tumor or lymph nodes) and 25 patients had progressive disease with distant metastases. Among PD group, 8 patients with progressive disease died due to cancer-related causes.
Mean patient age at the time of diagnosis was 61.1 years in NED group and 55.9 years in PD group. Groups were comparable in terms of patient age (p = 0.2). Likewise, tumor histology did not differ significantly between groups (p = 0.5) (Table 2).
We observed that clinical FIGO stage correlated with patient follow-up information; such that, high clinical FIGO stage was associated with disease persistence on follow-up. The number of patients with FIGO stage II–III–IV was 7–6–25 in PD group and 12–4–4 in NED group, respectively. Patients in NED group appeared to have lower FIGO stages than patients with PD group, p = 0.002.
Forty-four patients had PET-positive lymph nodes. Of these, 23 patients had pelvic-only nodes whereas 21 patients had both pelvic and para-aortic nodes. The average size of lymph nodes in our patient population was 0.7 ± 0.2 cm within a range of 0.4–2 cm. The average SUVmax of the lymph node was 7.5 ± 4.1 within a range of 4.1–22.8.
The frequency of FDG-avid pelvic and/or para-aortic lymph nodes was higher in PD group compared to NED (84.2 vs. 60 %, respectively), p = 0.03. For further analysis, the localizations of the PET-positive lymph nodes were evaluated for the association of PD. Patients with FDG-avid para-aortic lymph nodes had higher risk to develop PD than patients with pelvic-only nodes and patients without lymph nodes (p = 0.005).
Univariate analysis revealed significant difference between SUVmax of the most FDG-avid lymph nodes among PD and NED groups. The average of lymph node SUVmax was significantly higher in PD group compared to NED group (10.2 ± 4 vs. 6.5 ± 3.8, respectively, p = 0.02).
We observed that, tumor SUVmax was comparable in between NED and PD groups (Table 2). The average of cervical tumor SUVmax was 17.5 ± 6.1 in PD group and 17.9 ± 6.5 in NED group (p = 0.9). We observed no association between tumor SUVmax and disease persistence. Similarly, we observed no differences in terms of MTV between NED and PD groups (106.9 ± 128 vs. 41.5 ± 22.8 cm3, respectively, p = 0.1). We detected that tumor SUVmax was not correlated with MTV, with the correlation coefficient (R 2) = 0.05. Likewise, we observed that TLG did not differ between PD group and NED group. The average of TLG in PD group was 898 ± 1160, whereas 381 ± 232 in the NED group, p = 0.3.
A Cox proportional hazards model for disease persistence in patients with in-operable cervical cancer was performed including the presence of PET-positive pelvic and para-aortic lymph nodes, the location of lymph nodes and lymph node SUVmax and FIGO stage (Table 3). Cox regression analysis revealed that stage IV disease and the presence of PET-positive para-aortic lymph nodes were independent predictors of disease persistence on follow-up independent from SUVmax. We found that patients with PET-positive para-aortic lymph nodes had 5.3 times greater risk to develop PD [hazard ratio (HR): 5.3, 95 % CI 2.08–12.9] compared to others. Similarly, hazard ratio for developing PD was calculated as 3.3 for patients with stage IV disease compared to stage IIB (95 % CI 1.3–8.5) (Table 3).
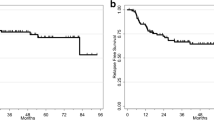
The Kaplan–Meier curves for disease-free survival and overall survival indicated better outcomes for patients without PET-positive lymph nodes compared to patients with pelvic-only nodes and patients with para-aortic nodes. The worst outcome curve was detected for patients with PET-positive para-aortic lymph nodes among all patients, p = 0.03 (Fig. 1). Similarly, disease-free survival and overall survival curves showed progressive worsening as the disease advanced. Better outcome results were demonstrated on Kaplan–Meier curves for stage II patients than stage III patients and stage IV patients, respectively, p = 0.015 (Fig. 2).
Discussion
In this study, we examined the association of PET-based volumetric and metabolic parameters and clinical characteristics with disease persistence in patients with in-operable cervical carcinoma. Our results suggested that advanced FIGO stage and the presence of FDG-avid para-aortic lymph nodes on pretreatment PET/CT were significantly associated with poor disease-free survival and overall survival.
Cervical cancer is the only gynecological cancer clinically staged according to FIGO staging system [29]. Even though there is growing debate in the literature on the prognostic value of FIGO staging system for it does not rely on surgical-pathological findings and lymph node status, the results of this study demonstrate the prognostic value of FIGO staging and its association with disease persistence in patients with in-operable cervical cancer [30–32]. In this study, we observed that patients with stage IV disease had 3.3 times greater risk to develop PD, lower disease-free survival and overall survival compared to patients with earlier stage disease.
In this study, we found that patients with PET-positive para-aortic lymph nodes had 5.3 times higher risk developing PD after chemoradiotherapy. Moreover, our results indicated that, the presence of FDG–avid para-aortic lymph nodes in pretreatment PET/CT was more predictive of clinical outcome than advanced FIGO stage. These findings supported previously published data, in which lower overall survival rates were reported in patients with para-aortic lymph node metastasis [16, 30, 33, 34].
In this regard, the clinical impact of PET/CT imaging in the pretreatment work-up of patients with cervical carcinoma comes into prominence. Because para-aortic lymph nodes are beyond the standard radiation fields, accurate assessment of para-aortic lymph node metastasis is crucial for determining the extent of radiotherapy and improving patient survival [35–37]. Compared to CT and MRI, PET/CT takes a step further in the detection of para-aortic lymph nodes. In our study, the size of FDG-avid lymph nodes ranged within 0.4–2 cm. This means that, lymph nodes smaller than 1 cm in size would have falsely considered as negative on CT and MRI if these nodes were not evaluated with PET/CT.
In our previous research, we observed that PET/CT has led to modifications in the extent of the radiotherapy field in 34 % of patients and to major alterations in treatment plans in 23 % of patients with cervical carcinoma [11]. In addition, as a preliminary result, we observed that the frequency of the presence of FDG-avid para-aortic lymph nodes was significantly higher among patients with poor prognosis in univariate analysis [11]. Based on our previous study, in the present study, we further analyzed all PET-based volumetric and metabolic findings together with clinical characteristics in a detailed multivariate analysis depending on a mean follow-up duration of 22 ± 12.6 months.
Our results indicated that in patients with in-operable cervical carcinoma, factors such as TLG, MTV, and tumor SUVmax, which were previously reported to associate with poor prognosis in patients with early stage disease, fail to associate with patient prognosis on multivariate analysis [5, 9, 32]. In addition, even though the average SUVmax of FDG-avid lymph nodes was significantly higher in patients with PD compared to NED group, the results of the multivariate analysis indicated that there was no association between lymph node SUVmax and overall survival. Even though authors have clarified that tumor volume, high tumor and lymph node SUVmax were predictive of unfavorable prognosis for cervical cancer patients [4, 9, 15–19, 26, 32]; we did not observe any associations of disease persistence with these parameters. However, these studies that demonstrate the association of defined metabolic parameters with poor patient outcome include patients with early stage cervical cancer [5, 7, 15, 16]. We considered that the discrepancy of our results from the former studies derived from the enrollment of in-operable patients and exclusion of patients with early stage disease. In this study, we observed that, having stage IV disease rather than stage II and stage III disease was associated with 3.3 times greater risk to develop PD. We considered that the association of lymph nodes’ SUVmax with disease persistence which was previously reported to be associated with poor prognosis [10] and also seen in our univariate analysis must have been masked both by tumor stage and the location of PET-positive lymph nodes under multivariate Cox regression analysis.
Furthermore, strong evidences supporting our results are available in the literature. Recent data have suggested that, in patients with cervical cancer, the predictors of disease recurrence were clinical stage, lymph node status at the time of the initial diagnosis and tumor response after the completion of therapy [30, 37]. In a recent study, Yoo et al. [14] reported that neither SUVmax, nor MTV of the primary tumor associated with event-free survival in patients with cervical cancer. On the contrary, they found that TLG better predicted prognosis than did other volume-based PET parameters and FIGO staging was not an independent prognostic factor after multivariate analysis [14].
In this regard, we considered that there are controversial results on the prognostic importance of PET-based volumetric and metabolic parameters in the literature. However, all reports agree on the fact that, the impact of PET/CT on the clinical management of patients with cervical cancer is its high sensitivity and accuracy in detecting disease extent in and outside the pelvis. We believe that separate prospective trials with larger homogeneous series of patients with early stage cervical cancer and patients with advanced stage disease are warranted to delineate the prognostic value of these volume-based metabolic parameters obtained by PET/CT.
We considered that when cervical tumors spread to para-aortic lymphatic chain or progress to a stage at which patients cannot benefit from curative surgery, metabolic parameters such as MTV, TLG, FDG uptake in tumor and metastatic lymph nodes which are previously demonstrated to be predictive of patient outcome, may become insignificant. Our results indicated that, regardless of SUVmax, the presence of FDG-avid para-aortic lymph nodes is the most important factor predictive of poor clinical outcome (Fig. 3).
Upper row demonstrates coronal CT (a), PET (b) and fused PET/CT (c) slices in a 48-year-old female patient who underwent FDG PET/CT imaging for the initial staging of cervical carcinoma. The clinical FIGO stage was stage IIB. In PET/CT images, hypermetabolic para-aortic lymph nodes with SUVmax: 7.5 are seen (marked by thin arrows). The lower row demonstrates coronal CT (d), PET (e) and fused PET/CT (f) slices of the follow-up study performed 15 months after the initial PET/CT. The progression of disease is clearly seen with widespread para-aortic lymph node metastasis in addition to liver metastasis (marked by thick arrow) with intense FDG uptake (SUV: 6.47). On follow-up, the patient died due to cancer-specific causes
The primary limitation of our study is that it is a retrospective study of 58 patients from a single center. In addition, the median follow-up duration after PET/CT was relatively short. Therefore, interpretation of the current study must be confined to short-term outcome, which implies a limited period of follow-up. Nevertheless, the results of this study are noteworthy because persistent cervical tumor on clinical examination performed 1–3 months after completion of therapy is previously shown to be associated with an increased risk of recurrence and poor overall survival [38]. Authors have found that posttreatment metabolic response and pretreatment lymph node status (as defined by 18F-FDG PET) were predictive of both cause-specific and progression-free survival after chemoradiation for cervical cancer [30, 39, 40]. However, analyses with larger number of cases and with a longer period of follow-up in prospective trials are required to validate the results of the present study.
In conclusion, we considered that when cervical malignancy advances to a surgically unamenable stage, the presence of para-aortic lymphatic metastasis and advanced FIGO stage appear as two major factors that adversely effect disease-free and overall survival. Regardless of SUVmax, patients with PET-positive para-aortic lymph nodes carry the highest risk to develop PD on follow-up. Thus, by successfully detecting extrapelvic disease, FDG PET/CT provides valuable long-term prognostic information in patients with in-operable cervical carcinoma and, in the future, may be used to guide additional therapy.
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90.
WHO/ICO Information Center of HPV and Cervical Cancer (HPV Information Center). Human papillomavirus and related cancers in the world. Summary report 2010. http://www.who.int/hpvcentre/en/.
Narayan K, Fisher R, Bernshaw D. Significance of tumor volume and corpus uteri invasion in cervical cancer patients treated by radiotherapy. Int J Gynecol Cancer. 2006;16:623–30.
Kim H, Kim W, Lee M, Song E, Loh JJK. Tumor volume and uterine body invasion assessed by MRI for prediction of outcome in cervical carcinoma treated with concurrent chemotherapy and radiotherapy. Jpn J Clin Oncol. 2007;37:858–66.
Chung HH, Kim JW, Han KH, Eo JS, Kang KW, Park NH, et al. Prognostic value of metabolic tumor volume measured by FDG-PET/CT in patients with cervical cancer. Gynecol Oncol. 2011;120:270–4.
Burghardt E, Hofmann HM, Ebner F, Haas J, Tamussino K, Justich E. Magnetic resonance imaging in cervical cancer: a basis for objective classification. Gynecol Oncol. 1989;33:61–7.
Eifel PJ, Winter K, Morris M, Levenback C, Grigsby PW, Cooper J, et al. Pelvic irradiation with concurrent chemotherapy versus pelvic and para-aortic irradiation for high-risk cervical cancer: an update of radiation therapy oncology group trial (RTOG) 90–01. J Clin Oncol. 2004;22:872–80.
Jacobs AJ, Faris C, Perez CA, Kao MS, Galakatos A, Camel HM. Short-term persistence of carcinoma of the uterine cervix after radiation. An indicator of long-term prognosis. Cancer. 1986;57:944–50.
Kidd EA, Siegel BA, Dehdashti F, Grigsby PW. The standardized uptake value for F-18 fluorodeoxyglucose is a sensitive predictive biomarker for cervical cancer treatment response and survival. Cancer. 2007;110:1738–44.
Kidd EA, Siegel BA, Dehdashti F, Rader JS, Mutch DG, Powell MA, et al. Lymph node staging by positron emission tomography in cervical cancer: relationship to prognosis. J Clin Oncol. 2010;28:2108–13.
Akkas BE, Demirel BB, Vural GU. Clinical impact of FDG PET/CT in the pretreatment evaluation of patients with locally advanced cervical carcinoma. Nucl Med Commun. 2012;33:1081–8.
Choi HJ, Roh JW, Seo SS, Lee S, Kim JY, Kim SK, et al. Comparison of the accuracy of magnetic resonance imaging and positron emission tomography/computed tomography in the presurgical detection of lymph node metastases in patients with uterine cervical carcinoma: a prospective study. Cancer. 2006;106:914–22.
Son H, Kositwattanarerk A, Hayes MP, Chuang L, Rahaman J, Heiba S, et al. PET/CT evaluation of cervical cancer: spectrum of disease. Radiographics. 2010;30:1251–68.
Yoo J, Choi JY, Moon SH, Bae DS, Park SB, Choe YS, Lee KH, Kim BT. Prognostic significance of volume-based metabolic parameters in uterine cervical cancer determined using 18F-fluorodeoxyglucose positron emission tomography. Int J Gynecol Cancer. 2012;22:1226–33.
Yen TC, See LC, Lai CH, Tsai CS, Chao A, Hsueh S, Hong JH, Chang TC, Ng KK. Standardized uptake value in para-aortic lymph nodes is a significant prognostic factor in patients with primary advanced squamous cervical cancer. Eur J Nucl Med Mol Imaging. 2008;35:493–501.
Kidd EA, El Naqa I, Siegel BA, Dehdashti F, Grigsby PW. FDG-PET-based prognostic nomograms for locally advanced cervical cancer. Gynecol Oncol. 2012;127:136–40.
Vansteenkiste JF, Stroobants SG, Dupont PJ, De Leyn PR, Verbeken EK, Deneffe GJ, et al. Prognostic importance of the standardized uptake value on (18)F-fluoro-2-deoxy-glucose-positron emission tomography scan in non-small-cell lung cancer: an analysis of 125 cases. Leuven Lung Cancer Group. J Clin Oncol. 1999;17:3201–6.
Schwartz DL, Rajendran J, Yueh B, Coltrera MD, Leblanc M, Eary J, et al. FDG-PET prediction of head and neck squamous cell cancer outcomes. Arch Otolaryngol Head Neck Surg. 2004;130:1361–7.
Hanin FX, Lonneux M, Cornet J, Noirhomme P, Coulon C, Distexhe J, et al. Prognostic value of FDG uptake in early stage non-small cell lung cancer. Eur J Cardiothorac Surg. 2008;33:819–23.
Nair VS, Barnett PG, Ananth L, Gould MK. Veterans affairs solitary nodule accuracy project cooperative studies group PET scan 18F-fluorodeoxyglucose uptake and prognosis in patients with resected clinical stage IA non-small cell lung cancer. Chest. 2010;137:1150–6.
Nakamura K, Okumura Y, Kodama J, Hongo A, Kanazawa S, Hiramatsu Y. The predictive value of measurement of SUVmax and SCC-antigen in patients with pretreatment of primary squamous cell carcinoma of cervix. Gynecol Oncol. 2010;119:81–6.
Lee P, Weerasuriya DK, Lavori PW, Quon A, Hara W, Maxim PG, et al. Metabolic tumor burden predicts for disease progression and death in lung cancer. Int J Radiat Oncol Biol Phys. 2007;69:328–33.
Chung MK, Jeong HS, Park SG, Jang JY, Son YI, Choi JY, et al. Metabolic tumor volume of [18F]-fluorodeoxyglucose positron emission tomography/computed tomography predicts short-term outcome to radiotherapy with or without chemotherapy in pharyngeal cancer. Clin Cancer Res. 2009;15:5861–8.
Chen HH, Chiu NT, Su WC, Guo HR, Lee BF. Prognostic value of whole-body total lesion glycolysis at pretreatment FDG PET/CT in non- small cell lung cancer. Radiology. 2012;264:559–66.
Lim R, Eaton A, Lee NY, Setton J, Ohri N, Rao S, Wong R, Fury M, Schoder H. 18F-FDG PET/CT metabolic tumor volume and total lesion glycolysis predict outcome in oropharyngeal squamous cell carcinoma. J Nucl Med. 2012;53:1506–13.
Miller TR, Grigsby PW. Measurement of tumor volume by PET to evaluate prognosis in patients with advanced cervical cancer treated by radiation therapy. Int J Radiat Oncol Biol Phys. 2002;53:353–9.
Geworski L, Knoop BO, Cabrejas ML, Knapp WH, Munz DL. Recovery correction for quantitation in emission tomography: a feasibility study. Eur J Nucl Med. 2000;27:161–9.
National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: cervical cancer, 06/02/09 update. Available at: http://www.nccn.org/professionals/physician_gls/f_guidelines.asp.
Pecorelli S. Revised FIGO staging for carcinoma of the vulva, cervix, and endometrium. Int J Gynaecol Obstet. 2009;105:103–4.
Grigsby PW, Siegel BA, Dehdashti F, Rader J, Zoberi I. Post therapy [18F] fluorodeoxyglucose positron emission tomography in carcinoma of the cervix: response and outcome. J Clin Oncol. 2004;22:2167–71.
Petignat P, Loubeyre P. Should we modify the current FIGO staging system for early-stage cervical cancer? Expert Rev Anticancer Ther. 2008;8:1015–7.
Kidd EA, Siegel BA, Dehdashti F, Grigsby PW. Pelvic lymph node F-18 fluorodeoxyglucose uptake as a prognostic biomarker in newly diagnosed patients with locally advanced cervical cancer. Cancer. 2010;116:1469–75.
Berman ML, Keys H, Creasman W, DiSaia P, Bundy B, Blessing J. Survival and patterns of recurrence in cervical cancer metastatic to periaortic lymph nodes (a Gynecologic Oncology Group study). Gynecol Oncol. 1984;19:8–16.
Heller PB, Maletano JH, Bundy BN, Barnhill DR, Okagaki T. Clinical-pathologic study of stage IIB, III, and IVA carcinoma of the cervix: extended diagnostic evaluation for paraaortic node metastasis—a Gynecologic Oncology Group study. Gynecol Oncol. 1990;38:425–30.
Varia MA, Bundy BN, Deppe G, Mannel R, Averette HE, Rose PG, et al. Cervical carcinoma metastatic to paraaortic nodes: extended field radiation therapy with concomitant 5-fluorouracil and cisplatin chemotherapy: a Gynecologic Oncology Group study. Int J Radiat Oncol Biol Phys. 1998;42:1015–23.
Kang S, Kim SK, Chung DC, Seo SS, Kim JY, Nam BH, et al. Diagnostic value of (18)F-FDG PET for evaluation of paraaortic nodal metastasis in patients with cervical carcinoma: a metaanalysis. J Nucl Med. 2010;51:360–7.
Schwarz JK, Siegel BA, Dehdashti F, Grigsby PW. Association of post therapy positron emission tomography with tumor response and survival in cervical carcinoma. JAMA. 2007;298:2289–95.
Jacobs AJ, Faris C, Perez CA, Kao MS, Galakatos A, Camel HM. Short-term persistence of carcinoma of the uterine cervix after radiation. An indicator of long-term prognosis. Cancer. 1986;57:944–50.
Schwarz JK, Grigsby PW, Dehdashti F, Delbeke D. The role of 18F-FDG PET in assessing therapy response in cancer of the cervix and ovaries. J Nucl Med. 2009;50(Suppl 1):64S–73S.
Lin LL, Yang Z, Mutic S, Miller TR, Grigsby PW. FDG-PET imaging for the assessment of physiologic volume response during radiotherapy in cervix cancer. Int J Radiat Oncol Biol Phys. 2006;65:177–81.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Akkas, B.E., Demirel, B.B., Dizman, A. et al. Do clinical characteristics and metabolic markers detected on positron emission tomography/computerized tomography associate with persistent disease in patients with in-operable cervical cancer?. Ann Nucl Med 27, 756–763 (2013). https://doi.org/10.1007/s12149-013-0745-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-013-0745-1