Abstract
Many plants accumulate proline in response to salt stress. Δ-pyrroline-5-carboxylate synthetase (P5CS) is the rate-limiting enzyme in proline biosynthesis in plants. Plasmid DNA (pCHF3-PvP5CS1 and pCHF3-PvP5CS2) containing the selectable neomycin phosphotransferase gene for kanamycin resistance and Phaseolus vulgaris P5CS (PvP5CS1 and PvP5CS2) cDNA was introduced into Arabidopsis plants using Agrobacterium-mediated gene transfer. Southern blot, northern blot and RT-PCR analyses demonstrated that the foreign genes were integrated into Arabidopsis chromosomal DNA and expressed. Single-gene transformants were analysed in this study. Transgenic plants expressed higher levels of PvP5CS1 and PvP5CS2 transcripts under salt stress conditions than under normal conditions. When treated with 0, 100 and 200 mM NaCl, the average proline content in leaves of transgenic plants was significantly higher (P < 0.01) than control plants. The average relative electrical conductivity (REC) of transgenic lines was significantly lower (P < 0.01) than control plants under salt stress condition. Biomass production of transgenic lines was significantly higher (P < 0.05) than control plants under 200 mM NaCl stress treatment. These results indicated that introducing PvP5CS1 and PvP5CS2 cDNA into transgenic Arabidopsis caused proline overproduction, increasing salt tolerance. Although the expression of PvP5CS1 in L4 lines and PvP5CS2 in S4 lines was the same under salt stress condition, the S4 lines accumulated 1.6 and 1.9 times more proline than the L4 lines under 100 and 200 mM NaCl treatments, respectively. The REC of S4 plants was 0.5 (100 mM NaCl) and 0.6 times (200 mM NaCl) that of L4 plants. The biomass production of S4 plants was 1.6 times (200 mM NaCl) more than in L4 plants. Total P5CS enzyme activity of S4 was significantly higher than that of L4. These results implied that the PvP5CS2 protein had stronger capacity to catalyze proline synthesis than PvP5CS1 under salt stress condition.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Salt is an important environmental challenge that causes osmotic stress, negatively impacting plant growth and crop productivity (Qin et al. 2011). Plant adaptation to salt stress involves the accumulation of low molecular mass osmolytes, such as proline and glycine betaine (Djilianov et al. 2005; Hossain and Fujita 2010). Proline accumulation has been reported in response to drought, high salinity, high light, heavy metals, oxidative stress and biotic stresses (Kavi Kishor et al. 2005; Wang et al. 2007). In many species, this accumulation of proline has been correlated with stress tolerance, and many researchers have reported higher levels of proline accumulation in salt-tolerant genotypes than in their salt-sensitive counterparts. For example, Igarashi et al. (1997) found that the salt-tolerant rice cultivar DGWG steadily increased its proline level under salt-stressed conditions, whereas level in the salt-sensitive cultivar IR 28 increased only slightly. Compared with a control, the salt-tolerant wheat cultivar KRL-19 exhibited 93% and 148% more proline, three and six days after salt treatment (DAT) with 100 mM NaCl, respectively, whereas a salt-sensitive cultivar, WH-542, had only 86.3% and 114% more, respectively (Mandhania et al. 2010). An increase in proline concentration under salt stress has been observed in crops as diverse as mulberry (Kumar et al. 2003), green gram (Misra and Gupta 2005) and sorghum (Jogeswar et al. 2006). Proline accumulation in plant cytosol not only increases cell osmotic potential, stabilizes membrane proteins, protects cells against oxidative damage and maintains appropriate NAD/NADH ratios (Szabados and Savouré 2009), but can also induce expression of salt-stress responsive genes (Chinnusamy et al. 2005).
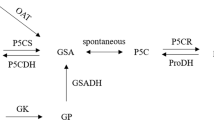
In higher plants, the main proline synthesis is glutamate pathway, which has two reaction steps. The first step is catalyzed by a bifunctional enzyme, pyrroline-5-carboxylate synthetase (P5CS), which is a rate-limiting enzyme in the pathway. Recently, the P5CS gene was isolated from Vigna aconitifolia (Hu et al. 1992), Arabidopsis thaliana (Strizhov et al. 1997), Lycopersicon esculentum (Fujita et al. 1998), Oryza sativa (Igarashi et al. 1997), Medicago truncatula (Armengaud et al. 2004), and other species. Expression assays indicated that P5CS from plants could be induced by various abiotic stresses. For example, the AtP5CS1 gene was upregulated by dehydration, high salinity and abscisic acid (ABA) treatments in most plant organs but silent in dividing cells (Strizhov et al. 1997). Rice P5CS1 was induced by salt, drought, ABA and cold treatment but not by heat treatment (Igarashi et al. 1997). In response to NaCl stress, mRNA of tomPRO2 increased more than three-fold, whereas transcripts of tomPRO1 were undetectable (Fujita et al. 1998). Manipulation of these P5CS genes has demonstrated that their overexpression increases proline production and confers salt tolerance in transgenic plants, including rice (Kumar et al. 2010), wheat (Vendruscolo et al. 2007), potato (Hmida-Sayari et al. 2005), tobacco (Yamchi et al. 2007) and chickpea (Ghanti et al. 2011).
We previously cloned two full-length cDNAs, PvP5CS1 (GenBank: EU340347) and PvP5CS2 (GenBank: EU407263), for Δ1-pyrroline-5-carboxylatesynthetase, an enzyme involved in the biosynthesis of proline, from common bean, Phaseolus vulgaris (Chen et al. 2009). Nucleotide sequence analysis showed that PvP5CS1 and PvP5CS2 shared 73.1% and 73.2% homology with that of AtP5CS1, respectively, and shared 74.5% and 74.5% homology with that of AtP5CS2, respectively. The present study had two aims: (i) to introduce PvP5CS1 and PvP5CS2 cDNAs into Arabidopsis via Agrobacterium tumefaciens and (ii) to evaluate differences in expression of the transgenes in Arabidopsis and the tolerance of transgenic plants under salt stress.
Materials and methods
Plasmid construction
To transfer PvP5CS1 and PvP5CS2 into Arabidopsis, we first PCR-amplified the open reading frames of the two genes from common bean using specific primers. The primer pairs P1 (5′-CCC GGG ATG GAG AAC ACA GAT CCT TGTA GAC-3′ and 5′-GGA TCC TAA ACA TCT CTC CCC TCT ATT G-3′) and P2 (5′-GGT ACC GCT ATT GCT CGT ATC AGT GCT CAG-3′ and 5′-GTC GAC CAA AAG GAA TCA AAG CAC C-3′) were designed for PvP5CS1 and PvP5CS2, respectively. PCR was performed for P5CS gene in a 20 μL reaction volume containing 2 μL common bean leaf cDNA template, 1× PCR buffer, 2.0 mM of MgCl2, 0.25 μmol of each primers, 1.2 mM dNTPs and 1.6 U of Taq polymerase (Promega, Shanghai, China), respectively. The PCR was carried out using PTC-100TM Programmable Thermal Controller (MJ Research, Watertown, USA) as follows: initial denaturation at 94°C for 3 min; 30 cycles of 94°C for 0.5 min, 55°C for 50 s, 72°C for 1.5 min; and a final extension at 72°C for 10 min. The PCR products were digested with SmaI/BamHI and KpnI/SalI, and subcloned into the expression vector pCHF3, which contains a CaMV 35S promoter, the nopaline synthase terminator and the selectable neomycin phosphotransferase gene for kanamycin resistance, to create recombinant plasmids pCHF3-PvP5CS1 and pCHF3-PvP5CS2.
Generation of transgenic Arabidopsis plants
To transform A. thaliana ecotype Landsberg, the recombinant plasmids (pCHF3-PvP5CS1 and pCHF3-PvP5CS2) was transferred into Agrobacterium tumefaciens (GV3101) by the flower-dip electroporation transformation method (Clough and Bent 1998). The pCHF3 vector was transformed using the same procedure. Seeds from transformed Arabidopsis plants were harvested and termed T1 generation transformants.
Southern blot analysis
Genomic DNA was isolated from leaves of transgenic Arabidopsis plants by the hexadecyltrimethylammonium bromide (CTAB) method (Murray and Thompson 1980). Approximately 10 μg of DNA was digested with KpnI and subjected to electrophoresis on a 0.8% agarose gel. The DNA was then blotted onto a nylon membrane (DuPont/NEN, Wilmington, USA). The membrane was probed with [32P]dATP-labelled PvP5CS1 and PvP5CS2 coding regions at 55°C. All the other procedures were carried out according to the manufacturer’s instructions.
Salt stress treatment of transgenic T3 generation plants
Wild type, pCHF3 and T3 transgenic Arabidopsis seeds were germinated in MS medium. After seven days, the young plants were transferred to four new MS medium plates containing 0, 100, 200 and 300 mM NaCl and grown in the greenhouse under a 16/8 light/dark cycle at 25°C for 5, 10 and 15 days. In salt stress assay, four Arabidopsis plants of each Arabidopsis type: wild type, pCHF3, L4 (transgenic with PvP5CS1) and S4 (transgenic with PvP5CS2) were transplanted in a plate. A total of 60 plates were treated in each salt stress condition.
RT-PCR
To determine the expression patterns of PvP5CS1 and PvP5CS2 in Arabidopsis, semiquantitative RT-PCR was carried out. Total RNA was extracted using Trizol reagent (Invitrogen, Carlsbad, USA) from leaves of Arabidopsis plants treated with salt for 10 days, and cDNAs were synthesized using ImPromIITM Reverse Transcriptase (Promega, Fitchburg, USA). The primers were P1 for PvP5CS1 and P2 for PvP5CS2.
Northern RNA hybridizations
Total RNA (15 μg) of transformed and wild-type Arabidopsis plants treated with salt for 10 days were subjected to electrophoresis in 1.2% agarose gel, transferred to nylon filter (DuPont/NEN), and hybridized to PvP5CS1 and PvP5CS2 cDNA labelled with [32P]dATP. Hybridization was carried out at 60°C and filters were exposed to X-ray film.
Determination of physiological parameters
Free proline content in fresh leaves of single transformed and wild-type Arabidopsis plants treated with salt for 10 days was determined according to Bates et al. (1973) and expressed as μg proline per gram fresh weight (μg/g, fresh weight).
Leaves from transformed and wild-type Arabidopsis plants treated with salt for 10 days were used for an electrolyte leakage assay as follows. First, leaves from plants were placed in glass tubes containing 10 mL of distilled water and the excess of air was removed from the tubes by vacuum pressure. Closed tubes were incubated in a shaker for 1 h to ensure the leakage of ions from the cells into water phase. The electrical conductivity of water extracts was measured with a conductivity meter (DDS-11A, Shanghai Precision and Scientific Instruments, Shanghai, China). Then, the tubes were incubated in a boiling water bath for 5 min, shaken for additional 20 min and the electric conductivity was measured again. The relative electrical conductivity (REC) was calculated according to the equation I = (L1/L2) ×100%, where L1 is the conductivity of a sample after salt treatment and L2 is the conductivity of a sample after boiling.
All leaves of a plant were used in the measurement of free proline content and REC. The whole plant was used in the measurement of biomass production of seedlings. For each physiological parameter, a total of 60 plants, which come from each stress-treated plate, were used.
P5CS activity assay
The activity of P5CS was assayed following the method described by Špoljarević et al. (2011). Frozen leaf tissue samples (0.5 g) were ground to a fine powder with liquid nitrogen and homogenized in the appropriate extraction buffer. The ratio of buffer volume:g tissue was 2:1. The extraction buffer used for P5CS assays was 50 mM Tris-HCl (pH 7.5) containing 10 mM MgCl2, 10 mM β-mercaptoethanol, 4 mM DTT, 2 mM PMSF, 1 mM EDTA and 2% PVPP. The extracts were centrifuged at 4°C for 15 min at 20,000 g and the resulting supernatant was used as the enzyme source. The assay mixture contained 50 mM Tris-HCl buffer (pH 7.0), 20 mM MgCl2, 50 mM L-glutamate, 100 mM hydroxylamine-HCl and 10 mM ATP. The reaction was initiated by the addition of enzyme extract. After incubation at 15 min for 37°C, the reaction was stopped by adding 1 mL of stop buffer (2.5 g FeCl3 and 6 g trichloroacetic acid in a final volume of 100 mL of 2.5 M HCl). The precipitated proteins were removed by centrifugation (4°C for 15 min at 10,000 g) and the absorbance recorded (UV-VIS spectrophotometer Carry 50, Varian Medical Systems Inc., Palo Alto, USA) at 535 nm against a blank identical to the above but lacking ATP. The amount of γ-glutamyl hydroxamate complex produced was estimated from the molar extinction coefficient of 250 M − 1 cm − 1 reported for the Fe3 + hydroxamate complex of the compound (Hayzer and Leisinger 1980). One unit of enzyme activity was defined as the amount of enzyme required to release 1 μmol of γ-glutamyl hydroxamate per minute. Total enzyme activity was expressed per g of frozen tissue (fresh weight).
Statistical analysis
Statistical analyses were performed using SAS statistical software (North Carolina State University, USA). The Fisher’s protected LSD test was used to determine the differences among the treatments. Significance was declared at P < 0.05.
Results
Production of transgenic Arabidopsis plants
The full-length cDNA sequences of PvP5CS1 and PvP5CS2 were separately subcloned into pCHF3 vector and transformed into wild-type Arabidopsis via A. tumefaciens. Arabidopsis seeds (T1) produced by the transformed plants were screened on plates (MS medium containing 50 μg/mL kanamycin). Transformed seeds could germinate and grow. Five T1 positive transgenic individuals transformed with PvP5CS1 (denominated L1 to L5) and with PvP5CS2 (denominated S1 to S5) were transplanted from screening plates to pots filled with soil and cultured in the greenhouse. RT-PCR using leaf cDNA from each positive T1 seedling as template showed that PvP5CS1 and PvP5CS2 had been inserted into the Arabidopsis genomes and were expressed normally in the transgenic plants (figure 1). The T2 seeds obtained from selfpollinated T1 seedlings were again germinated on screening plates, transplanted to pots in the greenhouse and used to generate seeds (T3). Positive transformants with the pCHF3 vector were screened on plates and used as control plants in this study.
Expression of two PvP5CS genes in transgenic T1 plants. (A) RT-PCR analysis PvP5CS1 gene using primer pair P1. (B) RT-PCR analysis PvP5CS2 gene using primer pair P2. L1, L2, L3, L4 and L5 represent the individual of PvP5CS1-transgenic T1 plant. S1, S2, S3, S4 and S5 represent the individual of PvP5CS2-transgenic T1 plant. ck1, ck2, Wt and pC represent pCHF3-PvP5CS1 vector, pCHF3-PvP5CS2 vector, nontransformed plant and plant transformed with vector pCHF3. 500 bp marker (M) was used in this assay.
Southern blot analysis of transgenic plants
Genomic Southern blot analysis of the 10 T3-generation transgenic plants showed that one to four copies of the PvP5CS1 and PvP5CS2 cDNAs were inserted into their genomes. Each line had a specific hybridization pattern, with different higher molecular-weight bands, suggesting that these transgenic lines were derived from independent transformation events (figure 2). The single-gene transformant L4 and S4 lines were selected for further analysis.
Southern blotting of transgenic T3 Arabidopsis lines. (A) The blot was probed by 32P[dATP]-labelled PvP5CS1 cDNA. (B) The blot was probed by 32P[dATP]-labelled PvP5CS2 cDNA. (C) Structure of the vector map of pCHF3::PvP5CS1/2. L1, L2, L3, L4 and L5 represent the individual of PvP5CS1-transgenic T1 plant. S1, S2, S3, S4 and S5 represent the individual of PvP5CS2-transgenic T1 plant. Wt represents nontransformed plant and pC represents plant transformed with vector pCHF3. Genomic DNA was digested by kpnI and resolved on 0.8% agarose gel.
Salt stress tolerance of PvP5CS1 and PvP5CS2 transgenic Arabidopsis plants
To investigate the functional roles of PvP5CS1 and PvP5CS2 in salt stress tolerance, the L4 and S4 lines were analysed. Wild type and pCHF3 plants were used as controls in the salt stress experiment. Result showed that there was no apparent phenotype difference among the transgenic Arabidopsis seedlings after five days salt treatment, and that all the transgenic Arabidopsis seedlings were dead after 15 days salt treatment. Seedlings after being treated for 10 days, exhibited obvious phenotype differences. After treatment for 10 d, 100 mM NaCl inhibited plant growth and 300 mM NaCl had a more severe inhibitory effect (figure 3). In the 100 mM NaCl treatment, growth of the control plants (Wt and pCHF3) was inhibited, but the transgenic plants (L4 and S4) grew normally; the control plant leaves were also more yellow than those of transgenic plants. Although 200 mM NaCl treatment inhibited the growth of both control and transgenic plants, the latter grew better. The S4 plants had stronger growth than L4 plants. In the 300 mM NaCl treatment, all of the control plants died. The transgenic plants survived, but their growth was severely inhibited. These results indicated that PvP5CS1 and PvP5CS2 overexpression could confer strong salt tolerance, and S4 plants exhibited stronger salt tolerance than L4 plants.
Salt tolerance of PvP5CS1 and PvP5CS2 overexpressed Arabidopsis plants under different concentration NaCl treatment. A, B, C and D represent Arabidopsis plants cultured on MS + 0 mM NaCl, MS + 100 mM NaCl, MS + 200 mM NaCl and MS + 300 mM NaCl plates for 10 days, respectively. a, b, c and d represent PvP5CS1-transgenic line (L4), PvP5CS2-transgenic line (S4), wild type and pCHF3 plant, respectively.
REC and biomass production of T3 transgenic plants under salt stress
The REC in transgenic lines was lower than that of the control plants (table 1). Under normal culture conditions (MS + 0 mM NaCl), the REC was not significantly different among wild type, pCHF3, transgenic L4 line and transgenic S4 line plants. The average REC values of S4 lines decreased to 77.8% (100 mM NaCl) and 58.6% (200 mM NaCl) of those of Wt plants. The average REC values of L4 lines decreased to 55.4% (100 mM NaCl) and 28.9% (200 mM NaCl) of those of Wt plants. Biomass production by T3 transgenic plants under salt stress in terms of seedling fresh weight was measured (table 2). Seedling growth did not differ between the untreated control and transgenic plants. However, growth of the transgenic lines was less inhibited than growth of the control plants under salt stress. Biomass production of S4 lines was 36.1% and 192.6% higher than that of Wt plants in the 100 and 200 mM NaCl treatments, respectively. Biomass production of L4 lines was 24.4% and 81.8% higher than that of Wt plants in the 100 and 200 mM NaCl treatments, respectively. These results indicate that PvP5CS1 and PvP5CS2 overexpression can confer strong salt tolerance.
The REC did not differ between L4 and S4 plants in the 0 mM NaCl treatments. However, a significant difference (P < 0.01) occurred under stress condition. The REC of S4 plants was 0.5 times (100 mM NaCl) and 0.6 times (200 mM NaCl) that of L4 plants. The biomass production of seedlings did not differ between L4 and S4 plants in the 0 and 100 mM NaCl treatments. A significant difference (P < 0.05) occurred under 200 mM NaCl stress condition. The biomass production of S4 plants was 1.6 times that of L4 plants. The result implied that S4 plants exhibited stronger salt tolerance than L4 plants under stress conditions.
Proline accumulation in T3 transgenic plants
A significant difference (P < 0.01) in proline content was seen between the control and transformed plants under different culture conditions (table 3). Overall, in the 0, 100 and 200 mM NaCl stresses, the average proline content in leaves of S4 plants increased by 175.7%, 402.9% and 598.1%, respectively, compared with Wt plants. The average proline content in leaves of L4 plants increased by 180.5%, 223.1% and 268.9% compared with Wt plants under 0, 100 and 200 mM NaCl stresses, respectively. There was no obvious difference in proline content between the L4 and S4 lines under normal (0 mM NaCl) conditions. However, significant differences in proline content (P < 0.01) between L4 and S4 lines occurred under salt-stress conditions (table 3). In summary, overexpression of one copy of PvP5CS2 resulted in 1.6 and 1.9 times more proline accumulating than did overexpression of one copy of PvP5CS1 in transgenic Arabidopsis plants under 100 and 200 mM NaCl treatments, respectively.
Expression of the PvP5CS2 cDNA in transgenic Arabidopsis plants
RT-PCR analysis was carried out on L4 and S4 transgenic lines (figure 4A). Although transgenic plants expressed higher levels of PvP5CS1 (and PvP5CS2) transcripts under salt stress conditions (lanes 2 and 3) than under normal culture condition (lane 1), no notable difference was seen between the 100 and 200 mM salt stress conditions. The expression of PvP5CS1 in L4 lines was the same as the expression of PvP5CS2 in S4 lines under normal (lane 1) and identical salt stress (lanes 2 and 3) conditions. The results were supported by the Northern blot assay (figure 4B).
RT-PCR (A) and Northern blot (B) analysis of the expressions of PvP5CS1 and PvP5CS2 in transgenic Arabidopsis L4 and S4 plants, respectively. Total RNA used for RT-PCR and Northern blot analysis was extracted from transgenic lines (L4 and S4) cultured on MS + 0 mM NaCl, MS + 100 mM NaCl, MS + 200 mM NaCl plates for 10 days.
P5CS enzyme activity in transgenic Arabidopsis plants
The result of enzyme activity assay showed that both under 100 mM NaCl salt stress condition and 200 mM NaCl salt stress conditions, total P5CS enzyme activity of S4 was significantly higher than that of L4, and there was no obvious difference between S4 an L4 under normal conditions (table 3).
Discussion
In plants, key enzymes of metabolic pathways are generally encoded by redundant genes, which may have been generated by historical gene duplication events. Afterwards, duplicated genes often incur sequence alterations, causing changes in transcriptional regulation (Turchetto-Zolet et al. 2009). Thus far, several duplicate P5CS genes have been found in Arabidopsis (Strizhov et al. 1997), rice (Igarashi et al. 1997), tomato (Fujita et al. 1998) and common bean (Chen et al. 2010). These duplicate P5CS genes originated from a single common ancestral gene (Turchetto-Zolet et al. 2009). Though many authors reported that transcriptional control of the duplicate P5CS genes in plants had been shown to be different in different organs and under different osmotic stresses, no previous report had compared the capacity of duplicate P5CS proteins in catalyze proline synthesis. We previously reported that the duplicate PvP5CS genes presented 15.2% divergence in amino acid sequences. Especially, there were three amino acids divergence in Glu-5-kinase catalytic domain and two amino acids divergence in GSA-DH catalytic domain between PvP5CS1 and PvP5CS2 (Chen et al. 2010). Amino acids divergence probably changes catalytic ability of PvP5CS1 and PvP5CS2 in proline synthesis. Here, we found that under same stress conditions, although no obvious differences existed between the expression of PvP5CS1 and PvP5CS2 in transgenic plants, the PvP5CS2 transgenic plants accumulated more proline than the PvP5CS1 transgenic plants. And total P5CS enzyme activity of S4 was significantly higher than that of L4. These results indicated that the PvP5CS2 protein had stronger capacity to catalyze proline synthesis than the PvP5CS1 protein under salt stress condition.
In this study, the induction of PvP5CS1 and PvP5CS2 transcripts also occurred in transgenic plants under salt stress. The mRNA levels of PvP5CS1 and PvP5CS2 in transgenic lines were higher when plants were salt stressed than when they were not. This phenomenon was also found in transgenic potato plants transformed with A. thaliana P5CS cDNA triggered by the cauliflower mosaic virus 35S RNA promoter (Hmida-Sayari et al. 2005). More AtP5CS transcripts accumulated in potato transgenic lines cultivated in the presence of 100 mM NaCl than in those cultivated without NaCl. Hong et al. (2000) comparison of P5CS protein levels between control and salt-treated plants indicated an increase of about 50% under salt stress conditions (200 mM NaCl). Recently, real-time quantitative PCR analysis indicated that PvP5CS1 and PvP5CS2 mRNA transcript levels in common bean seedling leaves steadily increased under cold, drought and salt stress (Chen et al. 2009). These results imply that some molecular mechanism in Arabidopsis upregulates PvP5CS1 and PvP5CS2.
Genetic engineering strategies for abiotic stress tolerance in plants depend on the expression of genes involved in signalling and regulatory pathways, that encode proteins conferring stress tolerance, or that encode enzymes in pathways that synthesize structural metabolites and osmolytes (Goel et al. 2010). Various earlier researchers have reported higher proline accumulation and subsequent abiotic stress tolerance of transgenic plants overexpressing P5CS; the genes transformed were mainly from Vigna aconitifolia P5CS (Su and Wu 2004; Kumar et al. 2010; Karthikeyan et al. 2011). This study verified the function of two P5CS genes from common bean to improve plant stress tolerance by genetic engineering. Our results demonstrated that the successful insertion and functional expression of PvP5CS1 and PvP5CS2 in the genome of Arabidopsis. That the average proline content in transgenic plants increased by 177.1% relative to control plants under normal culture conditions demonstrated that exogenous PvP5CS1 and PvP5CS2 functioned in Arabidopsis.
Under adversity, plants produce large amounts of oxygen free radicals that directly or indirectly peroxidize membrane lipids, damaging or destroying the membrane, reducing the cell membrane’s selectivity, and increasing electrical conductivity. Electrical conductivity in transgenic plants was lower than in control plants, implying that transgenic plants suffered less membrane destruction than control plants under salt stress and that increased proline accumulation in the transgenic plants protected the cell membrane. The alleviation of salt stress in transgenic Arabidopsis plants was evidenced by phenotypic characters, relative conductivity rate and biomass production. The transgenic approach to improve plant salt tolerance via overproducing proline has shown promise. For example, Su and Wu (2004) observed significantly higher tolerance to NaCl or drought stress by P5CS-containing transgenic rice with higher proline accumulation. Karthikeyan et al. (2011) also reported that almost four times more proline content in P5CS-transgenic rice lines under salt stress; the transgenic plants grew well in the presence of up to 200 mM NaCl, while control plants died within 10 days under these conditions. All growth characters of canola plants decreased with increasing salinity stress, but applying proline (200 mg/L) and glycine betaine (400 mg/L) alleviated these negative effects (Sakr et al. 2012). Thus, the increase in proline content contributed to alleviating the adverse effects of salt stress on plants (Ashraf and Foolad 2007). Our results with free proline-containing transgenic lines under saline conditions also supported the hypothesis of a positive correlation between proline accumulation and salt stress tolerance of plants.
References
Armengaud P., Thiery L., Buhot N., Grenie-De March G. and Savoure A. 2004 Transcriptional regulation of proline biosynthesis in Medicago truncatula reveals developmental and environmental specific features. Physiol. Plant. 120, 442–450.
Ashraf M. and Foolad M. R. 2007 Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ. Exp. Bot. 59, 206–216.
Bates L. S., Waldren R. P. and Teare I. D. 1973 Rapid determination of free proline for water studies. Plant Soil 39, 205–208.
Chen J. B., Wang S. M., Jing R. L. and Mao X. G. 2009 Cloning of PvP5CS gene from common bean (Phaseolus vulgaris) and its response to abiotic stresses. J. Plant Physiol. 166, 12–16.
Chen J. B., Zhang X., Jing R., Blair M. W., Mao X. and Wang S. M. 2010 Cloning and genetic diversity analysis of a new P5CS gene from common bean (Phaseolus vulgaris L). Theor. Appl. Genet. 120, 1393–1404.
Chinnusamy V., Jagendorf A. and Zhu J. K. 2005 Understanding and improving salt tolerance in plants. Crop Sci. 45, 437–448.
Clough S. J. and Bent A. F. 1998 Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 16, 735–743.
Djilianov D., Georgieva T., Moyankova D., Atanassov A., Shinozaki K., Smeeken S. C. M. et al. 2005 Improved abiotic stress tolerance in plants by accumulation of osmoprotectants-gene transfer approach. Biotechnol. Biotechnol. 19, 63–71.
Fujita T., Maggio A., Garcia-Rios M., Bressan R. A. and Csonka L. N. 1998 Comparative analysis of the regulation of expression and structures of two evolutionarily divergent genes for delta 1-pyrroline-5-carboxylate synthetase from tomato. Plant Physiol. 118, 661–674.
Ghanti K. K. S., Sujata K. G., Vijay Kumar B. M., Natraja K. N., Janardhan R. K., Srinath R. M. et al. 2011 Heterologous expression of P5CS gene in chickpea enhances salt tolerance without affecting yield. Biol. Plant. 55, 634–640.
Goel D., Singh A. K., Yadav V., Babbar S. B. and Bansal K. C. 2010 Overexpression of osmotin gene confers tolerance to salt and drought stresses in transgenic tomato (Solanum lycopersicum L.). Protoplasma 245, 133–141.
Hayzer B. D. J. and Leisinger T. 1980 The gene–enzyme relationships of proline riosynthesis in Escherichia coli. J. Gen. Microbiol. 11, 287–293.
Hmida-Sayari A., Gargouri-Bouzid R., Bidani A., Jaoua L., Savouré A. and Jaoua S. 2005 Overexpression of Δ-pyrroline-5-carboxylate synthetase increases proline production and confers salt tolerance in transgenic potato plants. Plant Sci. 169, 746–752.
Hong Z., Lakkineni K., Zhang Z. and Verma D. P. S. 2000 Removal of feedback inhibition of Δ-pyrroline-5-carboxylate synthetase results in increased proline accumulation and protection of plants from osmotic stress. Plant Physiol. 122, 1129–1136.
Hossain M. A. and Fujita M. 2010 Evidence for a role of exogenous glycinebetaine and proline in antioxidant defense and methylglyoxal detoxification systems in mung bean seedlings under salt stress. Physiol. Mol. Biol. Plants 16, 19–29.
Hu C. A. A., Delauney A. J. and Verma D. P. S. 1992 A bifunctional enzyme (Δ1-pyrroline-5-carboxylate synthetase) catalyzes the first two steps in proline biosynthesis in plants. Proc. Natl. Acad. Sci. USA 89, 9354–9358.
Igarashi Y., Yoshika Y., Sanada Y., Yamaguchi-Shinozaki K., Wada K. and Shinozaki K. 1997 Characterization of the gene for Δ1-pyrroline-5-carboxylate synthetase and correlation between the expression of the gene and salt tolerance in Oryza sativa. Plant Mol. Biol. 33, 857–865.
Jogeswar G., Pallela R., Jakka N. M., Reddy P. S., Rao J. V., Sreeniwasulu N. et al. 2006 Antioxidative response in different sorghum species under short term salinity stress. Acta Physiol. Plant 28, 465–475.
Karthikeyan A., Pandian S. K. and Ramesh M. 2011 Transgenic indica rice cv. ADT 43 expressing a Δ1-pyrroline-5-carboxylate synthetase (P5CS) gene from Vigna aconitifolia demonstrates salt tolerance. Plant Cell Tiss. Organ Cult. 107, 383–395.
Kavi Kishor P. B., Sangam S., Amrutha R. N., Laxmi P. S., Naidu K. R., Rao K. R. S. S. et al. 2005 Regulation of proline biosynthesis, degradation, uptake and transport in higher plants: its implications in plant growth and abiotic stress tolerance. Curr. Sci. 88, 424–438.
Kumar S. G., Reddy A. M. and Sudhakar C. 2003 NaCl effects on proline metabolism in two high yielding genotypes of mulberry (Morus alba L.) with contrasting salt tolerance. Plant Sci. 165, 1245–1251.
Kumar V., Shriram V., Kavi Kishor P. B., Jawali N. and Shitole M. G. 2010 Enhanced proline accumulation and salt stress tolerance of transgenic Indica rice by over-expressing P5CSF129A gene. Plant Biotechnol. Rep. 4, 37–48.
Mandhania S., Madan S. and Sheokand S. 2010 Differential response in salt tolerant and sensitive genotypes of wheat in terms of ascorbate, carotenoids proline and plant water relations. Asian J. Exp. Biol. Sci. 1, 792–797.
Misra N. and Gupta A. K. 2005 Effect of salt stress on proline metabolism in two high yielding genotypes of green gram. Plant Sci. 169, 331–339.
Murray M. G. and Thompson W. F. 1980 Rapid isolation of higher molecular weight plant DNA. Nucleic Acids Res. 8, 4321–4325.
Qin F., Shinozaki K. and Yamaguchi-Shinozaki K. 2011 Achievements and challenges in understanding plant abiotic stress responses and tolerance. Plant Cell Physiol. 52, 1569–1582.
Sakr M. T., El-Sarkassy N. M. and Fuller M. P. 2012 Osmoregulators proline and glycine betaine counteract salinity stress in canola. Agron. Sustain. Dev. 32, 747–754.
Špoljarević M., Agić D., Lisjak M., Gumze A., Wilson I. D., Hancock J. T. et al. 2011 The relationship of proline content and metabolism on the productivity of maize plants. Plant Signal. Behav. 6, 251–257.
Strizhov N., Abraham E., Okresz L., Blichling S., Zilberstein A. and Schell J. 1997 Differential expression of two P5CS genes controlling proline accumulation during salt stress requires ABA and is regulated by ABA1, ABI1 and AXR2 in Arabidopsis. Plant J. 12, 557–569.
Su J. and Wu R. 2004 Stress-inducible synthesis of proline in transgenic rice confers faster growth under stress conditions than that with constitutive synthesis. Plant Sci. 166, 941–948.
Szabados L. and Savouré A. 2009 Proline: a multifunctional amino acid. Trends Plant Sci. 15, 89–97.
Turchetto-Zolet A. C., Margis-Pinheiro M. and Margis R. 2009 The evolution of pyrroline-5-carboxylate synthetase in plants: a key enzyme in proline synthesis. Mol. Genet. Genomics 281, 87–97.
Vendruscolo E. C. G., Schuster I., Pileggi M., Scapim C. A., Molinari H. B. C., Marur C. J. et al. 2007 Stress-induced synthesis of proline confers tolerance to water deficit in transgenic wheat. J. Plant Physiol. 164, 1367–1376.
Wang Z. Q., Yuan Y. Z., Ou J. Q., Lin Q. H. and Zhang C. F. 2007 Glutamine synthetase and glutamate dehydrogenase contribute differentially to proline accumulation in leaves of wheat (Triticum aestivum) seedlings exposed to different salinity. J. Plant Physiol. 164, 695–701.
Yamchi A., Rastgar Jazii F., Mousavi A., Karkhane A. A. and Renu 2007 Proline accumulation in transgenic tobacco as a result of expression of Arabidopsis Δ1-pyrroline-5-carboxylate synthetase (P5CS) during osmotic stress. J. Plant Biochem. Biotechnol. 16, 9–15.
Acknowledgements
This work was an integral part of research project nos: 112102110159 and 2010A180016 supported by the Science and Technology Department, Henan Province of China and the Education Department, Henan Province of China, respectively.
Author information
Authors and Affiliations
Corresponding author
Additional information
[Chen J. B., Yang J. W., Zhang Z. Y., Feng X. F. and Wang S. M. 2013 Two P5CS genes from common bean exhibiting different tolerance to salt stress in transgenic Arabidopsis. J. Genet. 92, xx–xx]
Rights and permissions
About this article
Cite this article
CHEN, J.B., YANG, J.W., ZHANG, Z.Y. et al. Two P5CS genes from common bean exhibiting different tolerance to salt stress in transgenic Arabidopsis . J Genet 92, 461–469 (2013). https://doi.org/10.1007/s12041-013-0292-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12041-013-0292-5