Abstract
Inflammasomes are intracellular protein complexes, members of the innate immune system, and their activation and regulation play an essential role in maintaining homeostatic conditions against exogenous and endogenous stimuli. Inflammasomes occur as cytosolic proteins and assemble into a complex during the recognition of pathogen-associated or danger-associated molecular patterns by pattern-recognition receptors in host cells. The formation of the inflammasome complex elicits signaling molecules of proinflammatory cytokines such as interleukin-1β and interleukin 18 via activation of caspase-1 in the canonical inflammasome pathway whereas caspase-11 in the case of a mouse and caspase-4 and caspase-5 in the case of humans in the non-canonical inflammasome pathway, resulting in pyroptotic or inflammatory cell death which ultimately leads to neuroinflammation and neurodegenerative diseases. Inflammasome activation, particularly in microglial cells and macrophages, has been linked to aging as well as age-related neurodegenerative diseases. The accumulation of abnormal/ misfolded proteins acts as a ligand for inflammasome activation in neurodegenerative diseases. Although recent studies have revealed the inflammasomes’ functionality in both in vitro and in vivo models, many inflammasome signaling cascade activations during biological aging, neuroinflammation, and neurodegeneration are still ambiguous. In this review, we comprehensively unveil the cellular and molecular mechanisms of inflammasome activation during neuronal aging and age-related neurodegenerative disorders such as Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, multiple sclerosis, prion disease, and amyotrophic lateral sclerosis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Biological aging of the brain is the progressive loss of neurons and neuroglial cells and increased pigmentation in the remaining neural tissues due to the accumulation of lipofuscin [1] and neuromelanin [2] as aging progresses. Apart from pigmentation, neuronal tissues also accumulate potentially harmful substances such as abnormal/misfolded proteins, iron, aluminum, and free radicals along with the inflammatory markers [3, 4]. Aging is also linked with the lower production of many neurotransmitters, including noradrenaline, glutamate, dopamine, and serotonin. Recently, several studies have shown that immunological inflammation contributes to the aging process and immunosenescence, a phenomenon where the functioning of the adaptive immunity declines as the biological system ages, which results in the senescent with a low-grade inflammatory process known as inflammaging [5,6,7,8]. Inflammaging results in the progressive increase in the levels of proinflammatory cytokines produced by the activated inflammasomes that contribute to the development of various age-related chronic and neurodegenerative diseases. The alteration of structures and functions of the aging brain at cellular and molecular levels has been considered predisposing to neurodegenerative disorders; however, the mechanism that causes the transformation from biological aging to neurodegeneration remains unclear.
Neurodegenerative diseases affect millions of people worldwide, especially, Alzheimer’s and Parkinson’s disease are the most common neurodegenerative disorders among aged populations. Age-related neurodegenerative diseases are characterized by the progressive and irreversible loss of neuronal cells or their functions [9] resulting in an impairment of movement, cognition, and memory. Several environmental factors, the aggregation of misfolded protein, genetic and hereditary disorders, oxidative stress, mitochondrial dysfunction, inflammation, and even biological aging are considered high-risk factors for neurodegenerative diseases. In addition to the above risk factors, activation of inflammasomes either directly or indirectly plays a critical role in causing neuroinflammation and subsequently neurodegeneration [10]. This review article summarizes the current knowledge of the role of inflammasome activation and the mechanism of cell death on various age-related neurodegenerative diseases.
Inflammasomes
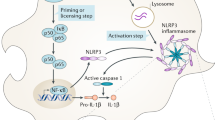
Inflammasomes are intracellular proteins predominantly occurring in the cytosol in an inactive form, and upon activation, monomers of inflammasome form an oligomeric complex in association with three major components such as sensor protein, inflammatory caspase, and in some cases an adaptor protein connecting the two. The interaction between the sensor and adaptor domains initiates oligomerization, and it clubs with interleukin-1 converting enzyme (ICE), resulting in inflammasome signaling. Interestingly, the nucleotide-binding oligomerization domain-like receptors (NLRs) containing pyrin domain (PYD) and apoptosis-associated speck-like protein containing a caspase recruitment domain (ASC) comprising the caspase recruitment domain (CARD) bind to form a complex that recruits the procaspase-1. The procaspase-1 containing CARD directly interacts with the CARD domain of ASC to form the oligomerization. ASC ablation shows a decreased level of caspase-1 activation as well as proinflammatory cytokine secretion [11]. Similarly, sensors such as absent in melanoma 2 (AIM2), NLRPs, Pyrin, NAIPs domain bind to adaptor molecule ASC, and zymogen procaspase-1 undergo autoproteolytic cleavage to produce the active caspase-1. IL-1β converting enzyme is crucial for the release of proinflammatory cytokines such as IL-1β and IL-18 that mediate the pyroptotic cell death [12] during inflammasome activation. Activated caspase-1 cleaves the pore-forming protein, gasdermin D, which is one of the crucial mechanisms that is required to release proinflammatory cytokines. Released cytokines and chemokines recruit the myeloid cells to the target site by the activation of adhesion, and co-stimulation in the neighboring cells [13]. Proinflammatory cytokines have been shown to bind to the IL-1 receptors and undergo the signaling cascade to neutralize the inflammation. However, the excessive production of proinflammatory cytokines is responsible for several neuroinflammatory diseases including Alzheimer’s and Parkinson’s disease [14].
A sensor is a protein domain that recognizes specific stimuli through its target site called pattern-recognition receptor (PRR), which is further classified into two major groups based on their localization: the toll-like receptors (TLR) and C-type lectins (CLR). Both are membrane-bound receptors that trap the pathogen-associated molecular patterns (PAMPs) and bring them into the cells via the endosomal pathway. The retinoic acid-inducible gene-1 (RIG-1)-like receptors (RLR) [15], NOD-like receptors (NLR) [16], and AIM2-like receptor (ALR) [17] belong to the cytoplasmic receptors. To date, several inflammasomes have been identified, but very few NLR families are correlated with neurodegenerative diseases. NLR is composed of three parts namely the N-terminal binding domain (PYD, CARD, and BIR), nucleotide-binding oligomerization domain (NBD), and a C-terminal domain containing leucine-rich repeat (LRR), which recognizes the stimuli effectively.
Another important component of the inflammasome is an adaptor protein, ASC dimer, which is arranged into large speck-like protein and interacts with the NLRP3 and procaspase via their death fold domains, PYD and CARD [18]. Most of the inflammasomes depend on the adaptor molecule because ASC recruits the procaspase-1 as well as it provides efficiency for the activation of the inflammasome; however, few inflammasomes are activated independently of adaptor molecules. Consequently, the conformational changes and autocatalytic cleavage of the procaspase-1 forming caspase-1 undergo the canonical pathway, whereas some inflammasomes help in mediating pyroptosis or inflammatory cell death via caspase-4 or caspase-5 activation, which is considered a non-canonical pathway. Figure 1A and B show an overview of various components of inflammasome such as sensor and adaptor proteins, inflammatory caspases, as well as the formation of inflammasome complex by oligomerization.
Inflammasomes and Neuronal Aging
Inflammasomes are closely associated with the aging process, and their activation accelerates the progression of biological aging and age-related disorders [19]. Microglia and macrophages are the two major cell types in the brain in which the inflammasomes are assembled and activated during the aging processes as well as they are the critical effectors and regulators in several neurodegenerative diseases [20]. Microglia are the major innate immune cells abundant in the CNS as the resident macrophages of the brain. Apart from being involved in the development of brain, maturation, and maintaining homeostasis, microglia are also involved in the recognition and elimination of infections in the CNS. During aging processes, the number of microglia increases, and their morphology changes from having multi-branched, long processes to a more spherical amoeboid shape [21]. Inflammasome activation is also observed in other cells in the brain, such as dendritic cells, neutrophils as in the case of traumatic brain injury, neurons, astrocytes, and oligodendrocytes. Inflammaging is a persistent low-grade inflammation in various organs as a result of the biological aging process and neurodegenerative diseases and can be attributed to inflammasome activation. Due to the drastic decrease in the functionality of the immune system during biological aging, the role of inflammasomes becomes more significant. The accumulated damage-associated molecular patterns (DAMPs) are recognized by the PRRs in macrophages, which results in endogenous stimulation of NLRP3 inflammasomes. Hence, chronic, low-level inflammation, which occurs during inflammaging, is triggered by the proinflammatory cytokines released by inflammasomes. The chronic low-level inflammation also contributes to the onset of numerous chronic and neurodegenerative diseases [22]. The components of inflammasomes, such as NLRC4, caspase-1, caspase-11, ASC, and GSDMD, and proinflammatory cytokine levels are elevated in the hippocampal region of the aged-mice [23]. The NLRP3 is increased in the senile microglia, further activating caspase-1 in the aged brain. The accumulation of numerous age-related DAMPs, such as extracellular ATP, urate, palmitate, oxidative stress, cholesterol, and ceramides, activates the NLRP3 inflammasomes and downstream caspases, consequently the proinflammatory cytokines. The cognitive and motor functions of the brain significantly increase in IL-1 dependent manner when the NLRP3 inflammasome is inhibited [24]. A major characteristic feature of the aging brain is the persistently increased inflammation and has been supported by the transcriptomic analysis that about 50% of the genes involved in inflammation and oxidative stress are upregulated in aged rodent brains [19]. Microglia show defective phagocytic abilities and the release of proinflammatory compounds during aging [25]. Aging also causes the microglial cells to become hyperactive, which is reflected in the increased production of cytokines and cell surface receptors. Microglial priming has a heightened response to the second stimulus compared to the naïve microglia [26]. IL-1β is a potent inflammatory cytokine that is elevated in the blood of aged people [27] and is also associated with several degenerative diseases [28, 29]. In the brains of patients with neurodegenerative disorders, high levels of tumor necrosis factor (TNF), IL-1β, and IL-6 are expressed in the brains, cerebrospinal fluid, and serum, the source of which is the microglial cells [30]. Increased levels of IL-1β in the brains of senescence-accelerated mouse-prone 8 models have also been observed along with the alteration of learning and memory [31]. Therefore, aging is considered a major risk factor that shifts biological aging to degenerative diseases.
Inflammasome Signaling in Neurodegenerative Diseases
Activation of inflammasomes is highly seen in the cells of the CNS in response to the aggregation of abnormal/misfolded proteins; virulent components of bacteria, viruses, and fungi; mitochondrial dysfunction; free radicals; and uric acid crystals [32]. In the CNS, inflammasome activations are predominantly observed in microglial cells and have also been shown to be expressed in astrocytes, oligodendrocytes, and endothelial cells [33, 34]. Inflammasomes play a diverse role in inflammation-associated neurodegenerative diseases; and undeniably, the innate immune response of inflammasome activation is well-known to be a defensive mechanism against several pathogenic stimuli. The assembly of the inflammasome is initiated by the sensor that recognizes internal or external insults and mediates pyroptotic cell death. Furthermore, the activated microglia that exert proinflammatory cytokines recruit phagocytic cells such as macrophages and monocytes to the injured or damaged sites [35]. Aggregated abnormal protein/misfolded proteins acts as ligands for the inflammasome activating receptors and mediate-pyroptotic cell death through the inflammasome either via canonical or non-canonical signaling pathways. Hence, excessive deposition of unwanted proteins and DAMPs leads to hyperactivation of the inflammasome. Accumulation of abnormal proteins such as amyloid-β in case of AD and α-synuclein in PD, aggregation of misfolded protein in Prion disease and mHTT-encoded protein in HD, immigration of immune cells in the CNS in case of multiple sclerosis and alteration of the cellular microenvironment by the fluctuation of the ions, the accumulation of reactive oxygen species due to mitochondrial dysfunction are some of the major risk factors of inflammasome activation and pathogenesis of the neurodegenerative disease. Even though several mechanisms have been proposed to neuronal aging and age-related neurodegenerative diseases, inflammasome signaling is critical for neuroinflammation and neurodegeneration. Regardless of the mechanism of the disease, activation of inflammasome components aggravates either directly or indirectly the symptoms of several neurodegenerative disorders by a positive feedback inflammatory cascade. Silencing and knocking out of inflammasome signaling components have been shown to ameliorate the adverse effects of the disease [36, 37] suggesting that inflammasomes play a critical role in neuroinflammation and neurodegeneration. The detailed role of inflammasome signaling in various neurodegenerative diseases is discussed below.
The Role of Inflammasomes in Alzheimer’s Disease
Alzheimer’s disease is one of the most common and progressive neurodegenerative disorders that typically impairs memory and cognitive function in the elderly population [38]. To date, there is no cure for this devastating disease, but a few therapeutic drugs are available to reduce its symptomatic progression [39]. Aggregation of amyloid-β (Aβ) peptides and abnormal phosphorylation of tau (p-tau) protein, which forms neurofibrillary tangles in the neurons, are the hallmark pathological features of AD [40]. The mechanism of pathogenesis and the role of inflammasomes and cytokines in Alzheimer’s disease is shown in Fig. 2A (inset).
A The mechanistic pathway shows the cause and pathological changes in Alzheimer’s disease as well as the central role of inflammasomes in causing neuroinflammation and neurodegenerations. B The mechanistic pathway shows the cause and pathological changes in Parkinson’s disease and the role of inflammasome in mediating neuroinflammation and neurodegeneration that lead to selective degeneration of dopaminergic neurons in the area of substantia nigra pars compacta. C The molecular pathway depicts the etiology and pathogenic alterations in Huntington disease, as well as the involvement of the inflammasome in mediating neuroinflammation and neurodegeneration, which results in degeneration of GABA producing neurons. D The pathway illustrates the cause, pathogenic abnormalities, and major involvement of inflammasomes in neuroinflammation, neurodegeneration, and demyelination in multiple sclerosis. E This pathway depicts the cause, pathological abnormalities, and key role of inflammasomes in triggering neuroinflammation and neurodegeneration in prion’s disease. F The involvement of inflammasomes and cytokines in the mechanistic pathway of amyotrophic lateral sclerosis is depicted in the flowchart
The senile plaque or Aβ fibril oligomers accumulate in the extracellular matrix of the neurons and are morphologically similar to the curli-extracellular components produced by bacteria such as Salmonella typhimurium and Escherichia coli [41]. Aβ is considered the damage-associated molecular patterns (DAMPs) and is predominantly recognized by pathogen recognition receptors (PRRs) in microglia, astrocytes, and oligodendrocytes that further initiate the activation of the inflammasome. NLRP3 associated with ASC involves the proteolytic cleavage of caspase-1 for the production of proinflammatory cytokines, IL-1β, and IL-18 [42]. To implicate the production of cytokines, the inflammasome undergoes two signaling pathways: one via the TLR-MyD88-NFκB in the innate immune cells of the CNS and the other through the NLRP3 activation [43]. These two pathways are required for the NLRP3 canonical pathway, but in the non-canonical pathway, the stimuli itself can activate the inflammasome pathway. NLRP10 is less associated with ASC in Aβ stimulated glial culture compared to untreated [43]. NLRP,1NLRP,3PYCARD,caspase-1, caspase-5, and caspase-, 8 and proinflammatory cytokines IL-18 and IL-1β are elevated in patients with AD [44].
Microglia, the resident macrophages of the brain, are activated and recruited by senile plaques that initiate phagocytosis of Aβ, along with the secretion of cytokines [45, 46]. Excessive production of caspase-1 has been reported in the hippocampal and frontal cortex regions of patients with AD [47]. P2X7R, a purinergic receptor, plays a significant role in the cleavage of Aβ peptides and NLRP3 activation. The lack of P2X7R decreases Aβ production and also reduces cognitive impairment by increasing the synaptic plasticity in transgenic AD mice [48] without altering the production of IL-1β [48]. The continuous synthesis of Aβ and the neurofibrillary tangles worsen the neurodegeneration and thereby cause the death of the patients. Upon stimulation with Aβ, IL-1β is released by activated microglia and higher expression of IL-1β is detected in microglial cells surrounding Aβ plaques in patients with AD as well as in AD animal models, and the activated microglia surrounding Aβ plaques also secrete chemotactic and proinflammatory molecules that recruit microglia and further amplify the neurotoxic effects of Aβ [46]. IL-1β, IL-6, and TNFα undergo a feedback loop, where they activate more astrocytes and microglia to further release the inflammatory cytokines and affect memory and cognition [49, 50]. Inflammasomes are required for the activation of interleukins as they are synthesized in their inactive forms as pro-IL-1β and pro-IL-18. Saturated fatty acids are considered to increase the production of cytokines in astrocytes and microglia [51, 52]. Recently, palmitate has been identified to stimulate the NLRC4 inflammasome in the astrocytes to produce IL-1β [53]. The levels of NLRC4 inflammasomes are also elevated in the brains of patients with AD [53]. Microglia are also stimulated to release IL-1β when stimulated by the fatty acids, but the exact inflammasome that is responsible for the release is yet to be identified. NLRP3 inflammasome activation has also been shown in the transgenic APP/PS1 AD mouse model, and deficiency in NLRP3 significantly reduces Aβ deposition in hippocampal and cortical regions of the brain, along with a decrease in the levels of proinflammatory cytokines and improved microglial phagocytic ability [47]. Similar changes have also been reported in caspase-1 deficient APP/PS1 mice [47]. Hence, as shown in Fig. 3A, the accumulated Aβ undergoes phagocytosis, which results in lysosomal rupture and releases cathepsin B. Cathepsin B activates the NLRP3 inflammasomes, thereby activating caspases which converts inactive cytokines to active form such as IL-18 and IL-1β. Collectively, all these findings signify that misfolded Aβ activates the microglial inflammasome components, especially NLRP3, which triggers the release of proinflammatory cytokines that perpetuate a chronic neuroinflammatory microenvironment and promote AD pathology.
Schematic representation of inflammasome activation pathways in various neurodegenerative disorders. The assembly of the NLRP3 inflammasome in microglial cells can be induced by proteins such as α-synuclein, Aβ, abnormal huntingtin and PrPSC in Parkinson’s disease, Alzheimer’s disease, Huntington disease, and prions disease respectively. The abnormal proteins Aβ and α-synuclein are phagocytized by the microglial cells, and subsequently release cathepsin B which further activates the inflammasomes, whereas the huntingtin and PrPSC proteins activate the microglial cells to assemble the NLRP3 inflammasome. In amyotrophic lateral sclerosis, TDP-43 protein binds to CD14 receptors, resulting in the activation of the transcription factor, NFκB, thereby resulting in the NLRP3 formation. The TDP-43 protein binding also upregulates the NADPH oxidase 2 (NOX2), and there is an increase in the ROS, leading to the activation of inflammasomes. The inflammasome assembly ultimately results in the release of the various cytokines which promotes the neuroinflammation and neurodegeneration of the neurons
The Role of Inflammasomes in Parkinson’s Disease
Idiopathic Parkinson’s disease is the second most common life-limiting neurodegenerative disease and primarily affects the extrapyramidal system of the brain. The pathological characterization of PD includes the deposition of aggregated fibrillar α-synuclein or Lewy bodies leading to the progressive loss of dopaminergic neurons in the substantia nigra pars compacta in the basal ganglia and axonal terminals of the striatum [54, 55]. The hallmark symptoms of PD are characterized by bradykinesia, tremors, rigidity, and postural impairment, and symptoms start to appear at the late stage when more than 50–80% of the dopaminergic neurons have been lost.
Exposure to environmental toxicants is one of the risk factors by augmentation of misfolded protein that triggers the inflammatory response in PD [56]. Inflammasome activation plays a vital role in neuroinflammation and neurodegenerative diseases [57], and similar to Aβ plaque in AD, endogenous stimuli and fibrillar α-synuclein triggers the resident macrophages, and microglia that predominantly surrounds the α-synuclein, and mediates pyroptosis by secreting proinflammatory cytokines and reactive oxygen species [58]. Figure 2B (inset) depicts the pathway and involvement of microglia and proinflammatory cytokines in the development of Parkinson disease.
The α-synuclein, when phagocytised, stimulates the release of cathepsin B, activation of NLRP3, and subsequently the release of inflammatory cytokines as shown in Fig. 3B. The expression of toll-like receptor-2 (TLR) and toll-like receptor-4 is elevated in the brain and blood samples of patients with PD due to the deposition of α-synuclein [59]. When sporadic PD is stimulated via toxic contents, microglia primarily undergoes morphological change to modify its phenotypes [60] and its expression of various inflammatory cytokines and chemokines such as TNFα, INFγ, iNOS, and COX2 [61, 62], and interestingly, microglial cell-mediated neuroinflammation has been reported in the postmortem brain of the patients with PD [63]. NLRP3 is involved in the PD as it is required for the maturation and secretion of caspase-1 and other proinflammatory cytokines within the endoplasmic reticulum. Microglia releases ROS, which activates NLRP3 inflammasomes via upstream regulators such as cdk5 and c-Abl kinase [64, 65]. The interaction of monomeric or fibrillary α-synuclein with TLR-2 has been shown to induce cytokine production in monocytes of patients with Parkinson’s disease [66], and the levels of IL-1β and IL-6 have been shown to increase in the cerebrospinal fluid and in the plasma of sporadic AD and de novo PD patients compared to control without changes in the level of IL-2 [67]. Increased levels of inflammatory cytokines [68]; increased protein levels of NLRP3, caspase-1, and IL-1β in peripheral blood mononuclear cells [69]; and elevated levels of IL-1β and TNFα in the striata have been observed in PD patients [70]. NLRP3 null mice are resistant to dopaminergic neurodegeneration in the 1-methyl-4-phenyl 1,2,3,6-tetrahydropyridine (MPTP)-induced PD model, along with the reduction of caspase-1 activation and IL-1β and IL-18 secretion [71]. All these studies signify the role of inflammasomes and their activation in the pathogenesis of PD.
The Role of Inflammasomes in Huntington’s Disease
Huntington’s disease (HD) is a progressive form of the neurodegenerative disorder that primarily damages the striatum at the early stage but causes extensive neurodegeneration in the brain throughout life [72]. The hallmark symptoms include impaired functionality of the motor system and psychiatric and cognitive disorders [73]. A well-known etiology of the disease is characterized by the mutation in the huntingtin (Htt) gene that is essential for brain development and the survival of neuronal cells. Loss of Htt in mice shows embryonic lethality, and mutant Htt causes abnormal protein deposition, which leads to central nervous system damage [74]. Indeed, the mutant Htt gene forms an aggregation of proteins in neuronal cells, which leads to cell death. As shown in Fig. 3C, the abnormal huntingtin protein activates the astrocytes and microglial cells to increase the expression of NLRP3 inflammasome, which leads to the elevation in the activity of caspases and ultimately results in the release of cytokines such as IL-18, TNFα, and IL-1β. Caspase-3, a proteolytic enzyme, and caspase-1, a downstream target of inflammasome activation, are also responsible for cleavage of pro-IL-1β to produce the mature and active form of the cytokine, which is elevated and activated in the animal model of HD brain [75] and brains of humans with HD [76]. The caspase-1 inhibitor has the ability to reduce the progression of HD [76], suggesting that caspase-1 plays a critical role in the pathogenesis of HD and that caspase-1 inhibitor could be a potential drug for the treatment of cortical and striatal neuronal cell death. Similarly, minocycline, a derivative of tetracycline, has been shown to attenuate the progression of neurodegeneration in an HD mouse model by inhibiting both caspase-1 and caspase-3, additionally reducing the upregulation of inducible nitric oxide synthase [76, 77]. Figure 2C (inset) shows the pathway that link the pathophysiology of Huntington disease and inflammasome activation in microglia.
The Role of Inflammasomes in Multiple Sclerosis
Multiple sclerosis (MS) is a chronic autoimmune disorder that affects both the central nervous system and the spinal cord due to a compromised blood–brain barrier that results in the infiltration of immune cells from the periphery as well as activation of microglia and astrocytes, thereby promoting neuroinflammation and neurodegeneration. The hallmark pathology of MS is characterized by infiltration of mononuclear cell-mediated inflammatory injury, gliosis, and demyelination of neuronal cells [78]. The patient with MS manifests symptoms like muscle weakening, impaired vision, and cognitive decline. Figure 2D (inset) highlights how microglial activation and cytokine release connect to the progression of multiple sclerosis. Viral or bacterial infections are major factors for accumulating myelin basic protein in the target site that can be recognized by the phagocytic cells in the CNS, which subsequently recruit T cells that release chemokines and cytokines to clear the myelin basic protein [79]. Genes encoding HMGB1, TLR-2, and TLR4 are highly expressed in MS and experimental autoimmune encephalomyelitis (EAE) [80]. It is well-known that inflammasomes are involved in multiple sclerosis and other autoimmune diseases, and furthermore, NLRP3 inflammasome activation contributes to disease progression in MS [81]. Upregulation of caspase-1, IL-18, and IL-1β has been observed in the peripheral blood mononuclear cells as well as in the cerebrospinal fluid (CSF) of MS patients [82, 83]. Caspase-1 activating NLRP3 inflammasomes are actively recognized by the DAMPs and secrete proinflammatory cytokines such as IL-18 and IL-1β, which infiltrate the T cells and granulocyte–macrophage colony-stimulating factor in EAE [84]. Elevated levels of caspase-1 expression are noted in acute and chronic demyelinating lesions [85] as well as in the serum of MS patients, and serum levels of caspase-1 and ASC can be considered a potential biomarker for MS onset [86]. MCC950, an NLRP3 inflammasome activation inhibitor, which is known to be effectively blocking caspase-1 and -11 activation, IL-1β processing, and IL-1β and IL-6 secretion, has been shown to reduce the EAE [87]. Osteopontin (OPN) is highly expressed in demyelinating lesions in MS and EAE and has been shown to play an important role in lymphocyte recruitment into the lesion [88].
The Role of Inflammasomes in Prion Disease
Prion disease, also known as transmissible spongiform encephalopathy or TSE, is a rare group of fatal neurodegenerative disorders that affect animals and humans. Prion disease causes a progressive decline in brain function due to the conversion of the normal prion protein, PrPc, into an abnormal prion protein, PrPsc [89]. Subsequent transmission of cellular prion protein and accumulation of the prion protein in the CNS are well-defined causative agents for both sporadic and familial prion diseases and are characterized as one of the rare forms of progressive neurodegenerative fatal disorders. Creutzfeldt-Jakob disease is considered one of the prion diseases; those patients express excessive secretion of IL-1β [90]. Indeed, the deposition of misfolded or abnormal prion proteins is crucial for neuroinflammation following phagocytosis. To ensure that phagocytic cell activation in prion pathogenesis, in vitro analysis on ganciclovir-treated microglia in tga20/CD11b-HSVTK (thymidine kinase of Herpes simplex virus) mice, ablated microglial processes and increased neurotoxicity, which eventually causes fatal conditions, suggesting that microglial involvement is mandatory to fight against prion disease [91]. The NLRP3 inflammasome, activated in microglia, are induced by amyloidogenic PrP peptides and secrete the cytokines via the activation of procaspase-1 [92], signifying the role of the inflammasome and inflammatory cytokines in prion disease (Fig. 3D). Activation and proliferation of microglia are present predominantly in regions where that the PrPsc accumulation occurs in animal and postmortem human brain tissue [93]. PrPSc accumulation that triggers the microglia and depletion of sialic acid in PrPsc is responsible for the secretion of proinflammatory cytokines [94]. When the microglial cells are activated, NALP3, the intracellular PRR, is triggered to form the inflammasome complex that promotes the proteolytic cleavage of caspase-1, resulting in the secretion of proinflammatory cytokines that mediate neuroinflammation in prion disease. Prion protein 106–126 in humans is a prion peptide fragment, forming amyloid fibrils that activate NALP3, causing the secretion of proinflammatory cytokines from microglia. Silencing of NALP3 and ASC decreases the mRNA expression of TNF-α and CCL3. Further, the inhibitors of NFκB largely downregulate the maturation and secretion of IL-1β [92]. The stimuli, prion fibrils, are considered crucial activators for the release of IL-1β in LPS-induced microglia and macrophages with lower concentrations of intracellular potassium, thus providing toxicity to neurons [95]. Accumulation of scrapie prion proteins PrPsc acts as one of the DAMPs for microglia, but specific epitope aiding its acceleration of inflammatory response is ambiguous. To identify its binding region, BV2 microglial cells treated with 22L PrPsc showed modification in the carbohydrate of the PrPsc surface, and also the depletion of sialic acid in PrPsc, which is the major reason behind the secretion of proinflammatory cytokines [96]. Treatment may reduce neuroinflammation in patients with prion protein-mediated neurodegenerative disease, but although it could reduce the progression rate, the complexity of the prion disease should be studied in the future to halt it at the onset stage of the disease. The pathway of microglial activation and the role of inflammasome-mediated cytokine release in the course of prion disease is represented in Fig. 2E (inset).
The Role of Inflammasomes in Amyotrophic Lateral Sclerosis
Amyotrophic lateral sclerosis (ALS) is a rare form of progressive neurological disease that primarily affects the nerve cells in the cerebral cortex, brain stem, and spinal cord, which are involved in controlling voluntary muscle movement. Drastic progression of inflammasome-mediated neuronal cell death causes the characteristic features of ALS such as muscle dystrophy, impairment of voluntary movement, and paralysis [97] that eventually cause lethality. Sporadic (sALS) and familial (fALS) cases of ALS have been reported, but there is no marked evidence for its etiology. However, it is considered one of the multifactorial diseases, and a mutation in the homo-dimeric enzyme Cu/Zn superoxide dismutase 1 encoding gene (SOD1) is significant to rely on familial ALS, which causes impairment of the motor neuron and its function [98]. The expression of NLRP3, caspase-1, proinflammatory cytokines such as IL-1β and IL-18, and transcription factor NFκB are elevated in the SOD1G93A transgenic rat, where NLRP3 is predominant [99]. The expression of NLRP3, NLRC4, AIM2, and caspase-1 activation has been reported in the neural tissue of mutant SOD1 transgenic animals [99,100,101], and mutant SOD1 activates caspase-1 and IL-1β in microglia [102, 103]. Further, the TLR4 antagonist inhibits motor neuron lethality by effectively abrogating the NLRP3 activation and IL-1β secretion in LPS-induced microglial cells from the ALS mouse model [104]. Figure 2F (inset) displays the impact of microglial activation and cytokine release in the pathophysiology of amyotrophic lateral sclerosis.
Postmortem spinal cord tissues of patients with ALS support the major role of NLRP3 along with ASC, IL-18, and active caspase-1, in mediating neuroinflammation by releasing the inflammatory cytokines [101]. In the spinal cord, astrocytes are the largest reservoir of the NLRP3 inflammasome [101]. Infiltration of T cell induces M1 microglia to produce proinflammatory cytokines, which is a major risk factor for the neuropathology in the animal model of inherited ALS [105, 106]. Unfortunately, the enhanced secretion of cytokines and ROS inhibits glutamate transporters. As a result, the cerebrospinal fluid glutamate increases to induce neurotoxicity of the motor neurons and, subsequently, causes the progression of ALS [107]. The TDP-43 protein binds to CD14 receptors found in microglia, macrophages, and monocytes, causing NFκB to be activated and thereby stimulating the NLRP3 inflammasome, as shown in the Fig. 3E. TDP-43 protein binding also activates inflammasomes by upregulating NADPH oxidase 2 (NOX2) and elevating the production of ROS (Fig. 3E).
The Role of Inflammasomes in Traumatic Brain Injury
The traumatic disorder is one of the highly correlated neurodegenerative diseases and develops as a secondary injury following traumatic brain injury. The progression of neurodegenerative disease happens at an older age, but, in contrast, secondary injury or damage in the brain leads to progression and chronic conditions in patients with TBI. The loss of cognition, ischemia, and neuronal death are hallmark characteristic features of TBI. Primary injury is considered physical damage to the brain, and secondary injury refers to internal damage in the CNS following primary injury, depending on the damage and time course. Interestingly, NLRP3, ASC, caspase-1, and proinflammatory cytokines are involved in the TBI-induced cerebral inflammatory responses [108], and inflammasomes act as an excellent biomarker of TBI [109]. NLRP3-deficient mice, as well as pharmacological blockade of NLRP3 activation, improves recovery from TBI [110]. Interestingly, inflammasomes upstream and downstream activators such as ASC and caspase-1 are elevated in the CSF of patients with TBI [109]. An increase in the level of intracellular calcium also activates caspases that result in programmed apoptotic cell death in TBI [111]. Similar to Alzheimer’s and Parkinson’s diseases, the NLRP3 inflammasome may also be a potential therapeutic target for treating neurodegenerative diseases in TBI. Early growth response protein 1 (EGR1) is essential for cell proliferation and regulation of apoptosis, and its expression level is reduced after TBI [112]. Neutralization of proinflammatory cytokine, IL-1β, with their antibody, reverses the impaired cognitive function in mice with TBI [113]. Higher levels of inflammasome markers such as ASC, caspase-1, NLRP1, and NLRP3 have been observed in the CSF of patients with TBI [114, 115] suggesting that the activation of inflammasomes plays a crucial role in TBI.
Therapeutic Potentials of Inflammasomes
The role of inflammasomes in the above-mentioned pathways of neurodegenerative diseases makes them a potential drug target for neuroinflammation and neurodegeneration. The inhibitors of NLRP3 can be classified into two groups: inhibitors that directly inhibit NLRP3 and inhibitors that inactivates NLRP3 by inhibiting their components or blocking their signaling pathways. Anakinra, canakinumab, and rilonacept are a few of the clinically approved drugs by the US Food and Drug Administration (FDA) that inhibit the downstream effector cytokines, released due to the NLRP3 inflammasome activation. Anakinra is an IL-1 receptor antagonist, canakinumab is an IL1β neutralizing antibody, and rilonacept is a decoy receptor which binds with IL-1β and IL-1α. These drugs are found to be effective in treating autoimmune diseases, but in the case of neuroinflammation, the specific inhibitors of NLRP3 inflammasomes are preferred [116, 117]. Under the category of inhibitors which target the NLRP3 inflammasomes directly, some of the compounds are found to be effective in vitro: MCC950, β-hydroxybutyrate, parthenolide, glyburide, auranofin, isoliquiritigenin, 3,4-methylenedioxy-β-nitrostyrene, cyclopentenone prostaglandin 15d-PGJ2 and 25-hydroxycholesterol (25-HC), Bay 11–7082, dimethyl sulfoxide (DMSO), and interferon-β [87, 118,119,120,121,122,123,124,125,126]. But the efficacy of most of these drugs was not found to be satisfactory, and among these compounds, MCC950 was more specific and potent, as it showed efficacy against EAE, AD and PD models. MCC950 is considered to block both canonical and non-canonical pathway. [87, 127, 128]. But the FDA has so far not approved this compound for the treatment of neurodegenerative diseases. In an in vitro AD model, stavudine, an inhibitor designed to target HIV was shown to have the ability to decrease NLRP3 activation along with a reduction in the production of IL-18 and caspase-1 [129]. Glyburide, a commonly used drug for treating diabetes type 2, inhibits caspase-1 activation, IL-1β secretion, and neuronal cell death [83]. Sulphophrane, a naturally occurring phytochemical, inhibits caspase-1 activation and IL-1β maturation in the inflammasome pathway. It is also considered a possible drug for AD, as it has the ability to inhibit NLRP3 activation, which is induced by the Aβ protein aggregates. This prospective application has yet to be subjected to clinical studies; however, treatments for inflammasome-mediated inflammation have not yet been thoroughly investigated, although it is a promising field for future research.
Concluding Remarks
Inflammasomes are intracellular proteins that maintain homeostatic conditions in the cell, but unfortunately, dysregulation of inflammasome activation tends to cause several diseases, including neuroinflammatory and neurodegenerative diseases (Table 1). Several activators such as ATP, ROS, HAMPs, DAMPs, and PAMPs play a crucial role in triggering the formation of inflammasome complexes. Inflammation and cellular degradation are elevating via enormous secretion of proinflammatory cytokines that recruit immune cells to the appropriate target site. In several neurodegenerative diseases, the NLRP3 inflammasome activation plays an immense role in neuroinflammation against protective mechanisms. Deletion of caspases and adaptor molecules inhibits adverse conditions in many inflammatory and neurodegenerative diseases. The anti-inflammatory drugs and natural anti-oxidants are reducing the severity. MCC950 is the direct NLRP3 inhibitor used against the inflammasome, for experimental purposes. To date, treatment for inflammation has only been to control the symptomatic progression of the disease. To completely cease the deregulatory action of inflammasome activation, we should explore more in-depth to identify the intertwined signaling mechanisms of the cell. The well-identified inflammasomes are NLRP1, NLRP3, NLRP6, NLRP12, NLRC4, NLRP9, NLRP7, AIM2, and Pyrin, and among these, the NLRP3 inflammasome is involved in many diseases. Based on the review, we conclude that the NLRP3 inflammasome activation leads to the production of proinflammatory cytokines that recruit immune and non-immune cells to the target site and damage the cells that eventually cause the shift from biological aging to neuroinflammation-mediated neurodegeneration. Several neurodegenerative diseases, apart from tremendously affecting the socio-economic status of the patients, unfortunately, also reduce their life expectancy. Finding a drug that mechanistically targets inflammasome signaling could save neurons from degeneration due to biological aging, inflammaging, and neurodegenerative diseases.
Data Availability
Not applicable.
Code Availability
Not applicable.
Abbreviations
- AIM2:
-
Absent-in-melanoma 2
- AD:
-
Alzheimer’s disease
- APP:
-
Amyloid-beta precursor protein
- ASC:
-
Apoptosis-associated speck-like protein containing a caspase-recruitment domain
- CCL3:
-
C-C motif chemokine ligand 3
- PrPc :
-
Cellular prion protein
- CSF:
-
Cerebrospinal fluid
- DAMP:
-
Damage-associated molecular pattern
- GSDMD:
-
Gasdermin D
- IFN:
-
Interferon
- IL:
-
Interleukin
- IL-1R:
-
IL-1 receptor
- LRR:
-
Leucine-rich repeat
- NFκB:
-
Nuclear factor kappa-light-chain-enhancer of activated B
- NLR:
-
Nod-like receptor
- NLRP:
-
Nod-like receptor protein
- NLRP3:
-
NLR family pyrin domain containing 3
- NLRC4:
-
NLR family CARD domain-containing protein 4
- NOD:
-
Nucleotide-binding oligomerization domain
- PAMP:
-
Pathogen-associated molecular pattern
- PRR:
-
Pattern-recognition receptor
- P2RX7:
-
P2X purinoceptor 7
- PYD:
-
Pyrin domain
- PrPsc :
-
Scrapie isoform of the prion protein
- TLR:
-
Toll-like receptor
- TLR4:
-
Toll-like receptor 4
- TNF:
-
Tumor necrosis factor
References
Ottis P, Koppe K, Onisko B, Dynin I, Arzberger T, Kretzschmar H, Requena JR, Silva CJ et al (2012) Human and rat brain lipofuscin proteome. Proteomics 12(15–16):2445–2454. https://doi.org/10.1002/pmic.201100668
Clewett DV, Lee TH, Greening S, Ponzio A, Margalit E, Mather M (2016) Neuromelanin marks the spot: identifying a locus coeruleus biomarker of cognitive reserve in healthy aging. Neurobiol Aging 37:117–126. https://doi.org/10.1016/j.neurobiolaging.2015.09.019
Latz E, Duewell P (2018) NLRP3 inflammasome activation in inflammaging. Semin Immunol 40:61–73. https://doi.org/10.1016/j.smim.2018.09.001
Singh T, Newman AB (2011) Inflammatory markers in population studies of aging. Ageing Res Rev 10(3):319–329. https://doi.org/10.1016/j.arr.2010.11.002
Franceschi C, Garagnani P, Parini P, Giuliani C, Santoro A (2018) Inflammaging: a new immune-metabolic viewpoint for age-related diseases. Nat Rev Endocrinol 14(10):576–590. https://doi.org/10.1038/s41574-018-0059-4
Franceschi C, Zaikin A, Gordleeva S, Ivanchenko M, Bonifazi F, Storci G, Bonafe M (2018) Inflammaging 2018: an update and a model. Semin Immunol 40:1–5. https://doi.org/10.1016/j.smim.2018.10.008
Calabrese V, Santoro A, Monti D, Crupi R, Di Paola R, Latteri S, Cuzzocrea S, Zappia M et al (2018) Aging and Parkinson’s disease: inflammaging, neuroinflammation and biological remodeling as key factors in pathogenesis. Free Radical Biol Med 115:80–91. https://doi.org/10.1016/j.freeradbiomed.2017.10.379
Cicolari S, Catapano AL, Magni P (2021) Inflammaging and neurodegenerative diseases: role of NLRP3 inflammasome activation in brain atherosclerotic vascular disease. Mech Ageing Dev 195:111467. https://doi.org/10.1016/j.mad.2021.111467
Dugger BN, Dickson DW (2017) Pathology of neurodegenerative diseases. Cold Spring Harbor perspectives in biology 9(7):a028035. https://doi.org/10.1101/cshperspect.a028035
Heneka MT, McManus RM, Latz E (2018) Inflammasome signalling in brain function and neurodegenerative disease. Nat Rev Neurosci 19(10):610–621. https://doi.org/10.1038/s41583-018-0055-7
Kumar M, Roe K, Orillo B, Muruve DA, Nerurkar VR, Gale M Jr, Verma S (2013) Inflammasome adaptor protein apoptosis-associated speck-like protein containing CARD (ASC) is critical for the immune response and survival in west Nile virus encephalitis. J Virol 87(7):3655–3667. https://doi.org/10.1128/JVI.02667-12
Fink SL, Bergsbaken T, Cookson BT (2008) Anthrax lethal toxin and Salmonella elicit the common cell death pathway of caspase-1-dependent pyroptosis via distinct mechanisms. Proc Natl Acad Sci U S A 105(11):4312–4317. https://doi.org/10.1073/pnas.0707370105
Khare S, Luc N, Dorfleutner A, Stehlik C (2010) Inflammasomes and their activation. Crit Rev Immunol 30(5):463–487. https://doi.org/10.1615/critrevimmunol.v30.i5.50
Guzman-Martinez L, Maccioni RB, Andrade V, Navarrete LP, Pastor MG, Ramos-Escobar N (2019) Neuroinflammation as a common feature of neurodegenerative disorders. Front Pharmacol 10:1008. https://doi.org/10.3389/fphar.2019.01008
Yoneyama M, Fujita T (2008) Structural mechanism of RNA recognition by the RIG-I-like receptors. Immunity 29(2):178–181. https://doi.org/10.1016/j.immuni.2008.07.009
Benko S, Philpott DJ, Girardin SE (2008) The microbial and danger signals that activate Nod-like receptors. Cytokine 43(3):368–373. https://doi.org/10.1016/j.cyto.2008.07.013
Hornung V, Ablasser A, Charrel-Dennis M, Bauernfeind F, Horvath G, Caffrey DR, Latz E, Fitzgerald KA (2009) AIM2 recognizes cytosolic dsDNA and forms a caspase-1-activating inflammasome with ASC. Nature 458(7237):514–518. https://doi.org/10.1038/nature07725
Latz E, Xiao TS, Stutz A (2013) Activation and regulation of the inflammasomes. Nat Rev Immunol 13(6):397–411. https://doi.org/10.1038/nri3452
Hu MY, Lin YY, Zhang BJ, Lu DL, Lu ZQ, Cai W (2019) Update of inflammasome activation in microglia/macrophage in aging and aging-related disease. CNS Neurosci Ther 25(12):1299–1307. https://doi.org/10.1111/cns.13262
Mammana S, Fagone P, Cavalli E, Basile MS, Petralia MC, Nicoletti F, Bramanti P, Mazzon E (2018) The role of macrophages in neuroinflammatory and neurodegenerative pathways of Alzheimer's disease, amyotrophic lateral sclerosis, and multiple sclerosis: pathogenetic cellular effectors and potential therapeutic targets. Int J Mol Sci 19(3): 831. https://doi.org/10.3390/ijms19030831
Davis EJ, Foster TD, Thomas WE (1994) Cellular forms and functions of brain microglia. Brain Res Bull 34(1):73–78. https://doi.org/10.1016/0361-9230(94)90189-9
Marin-Aguilar F, Ruiz-Cabello J, Cordero MD (2018) Aging and the inflammasomes. Exp Suppl 108:303–320. https://doi.org/10.1007/978-3-319-89390-7_13
Mejias NH, Martinez CC, Stephens ME, de Rivero Vaccari JP (2018) Contribution of the inflammasome to inflammaging. J Inflamm (Lond) 15:23. https://doi.org/10.1186/s12950-018-0198-3
Youm YH, Grant RW, McCabe LR, Albarado DC, Nguyen KY, Ravussin A, Pistell P, Newman S et al (2013) Canonical Nlrp3 inflammasome links systemic low-grade inflammation to functional decline in aging. Cell Metab 18(4):519–532. https://doi.org/10.1016/j.cmet.2013.09.010
Sebastian-Valverde M, Pasinetti GM (2020) The NLRP3 inflammasome as a critical actor in the inflammaging process. Cells 9(6):1552. https://doi.org/10.3390/cells9061552
Labzin LI, Heneka MT, Latz E (2018) Innate immunity and neurodegeneration. Annu Rev Med 69:437–449. https://doi.org/10.1146/annurev-med-050715-104343
Furman D, Jojic V, Kidd B, Shen-Orr S, Price J, Jarrell J, Tse T, Huang H et al (2013) Apoptosis and other immune biomarkers predict influenza vaccine responsiveness. Mol Syst Biol 9:659. https://doi.org/10.1038/msb.2013.15
Sardi F, Fassina L, Venturini L, Inguscio M, Guerriero F, Rolfo E, Ricevuti G (2011) Alzheimer’s disease, autoimmunity and inflammation. The good, the bad and the ugly. Autoimmun Rev 11(2):149–153. https://doi.org/10.1016/j.autrev.2011.09.005
Scrivo R, Vasile M, Bartosiewicz I, Valesini G (2011) Inflammation as “common soil” of the multifactorial diseases. Autoimmun Rev 10(7):369–374. https://doi.org/10.1016/j.autrev.2010.12.006
Wang WY, Tan MS, Yu JT, Tan L (2015) Role of pro-inflammatory cytokines released from microglia in Alzheimer’s disease. Ann Transl Med 3(10):136. https://doi.org/10.3978/j.issn.2305-5839.2015.03.49
Tha KK, Okuma Y, Miyazaki H, Murayama T, Uehara T, Hatakeyama R, Hayashi Y, Nomura Y (2000) Changes in expressions of proinflammatory cytokines IL-1beta, TNF-alpha and IL-6 in the brain of senescence accelerated mouse (SAM) P8. Brain Res 885(1):25–31. https://doi.org/10.1016/s0006-8993(00)02883-3
Lamkanfi M, Dixit VM (2012) Inflammasomes and their roles in health and disease. Annu Rev Cell Dev Biol 28:137–161. https://doi.org/10.1146/annurev-cellbio-101011-155745
Hanisch UK (2002) Microglia as a source and target of cytokines. Glia 40(2):140–155. https://doi.org/10.1002/glia.10161
Freeman L, Guo H, David CN, Brickey WJ, Jha S, Ting JP (2017) NLR members NLRC4 and NLRP3 mediate sterile inflammasome activation in microglia and astrocytes. J Exp Med 214(5):1351–1370. https://doi.org/10.1084/jem.20150237
Theriault P, ElAli A, Rivest S (2015) The dynamics of monocytes and microglia in Alzheimer’s disease. Alzheimers Res Ther 7(1):41. https://doi.org/10.1186/s13195-015-0125-2
Ou Z, Zhou Y, Wang L, Xue L, Zheng J, Chen L, Tong Q (2021) NLRP3 inflammasome inhibition prevents alpha-synuclein pathology by relieving autophagy dysfunction in chronic MPTP-treated NLRP3 knockout mice. Mol Neurobiol 58(4):1303–1311. https://doi.org/10.1007/s12035-020-02198-5
Han C, Yang Y, Guan Q, Zhang X, Shen H, Sheng Y, Wang J, Zhou X, Li W, Guo L, Jiao Q (2020) New mechanism of nerve injury in Alzheimer’s disease: beta-amyloid-induced neuronal pyroptosis. J Cell Mol Med 24(14):8078–8090. https://doi.org/10.1111/jcmm.15439
Lindenberg R (1990) Neuropathological diagnosis of Alzheimer disease. J Neuropathol Exp Neurol 49(5):535–537. https://doi.org/10.1097/00005072-199009000-00010
Yiannopoulou KG, Papageorgiou SG (2020) Current and future treatments in Alzheimer disease: an update. J Cent Nerv Syst Dis 12:1179573520907397. https://doi.org/10.1177/1179573520907397
Weiner HL, Frenkel D (2006) Immunology and immunotherapy of Alzheimer’s disease. Nat Rev Immunol 6(5):404–416. https://doi.org/10.1038/nri1843
Chapman MR, Robinson LS, Pinkner JS, Roth R, Heuser J, Hammar M, Normark S, Hultgren SJ (2002) Role of Escherichia coli curli operons in directing amyloid fiber formation. Science 295(5556):851–855. https://doi.org/10.1126/science.1067484
Halle A, Hornung V, Petzold GC, Stewart CR, Monks BG, Reinheckel T, Fitzgerald KA, Latz E, Moore KJ, Golenbock DT (2008) The NALP3 inflammasome is involved in the innate immune response to amyloid-beta. Nat Immunol 9(8):857–865. https://doi.org/10.1038/ni.1636
Murphy N, Grehan B, Lynch MA (2014) Glial uptake of amyloid beta induces NLRP3 inflammasome formation via cathepsin-dependent degradation of NLRP10. NeuroMol Med 16(1):205–215. https://doi.org/10.1007/s12017-013-8274-6
Saresella M, La Rosa F, Piancone F, Zoppis M, Marventano I, Calabrese E, Rainone V, Nemni R et al (2016) The NLRP3 and NLRP1 inflammasomes are activated in Alzheimer’s disease. Mol Neurodegener 11:23. https://doi.org/10.1186/s13024-016-0088-1
Simard AR, Rivest S (2006) Neuroprotective properties of the innate immune system and bone marrow stem cells in Alzheimer’s disease. Mol Psychiatry 11(4):327–335. https://doi.org/10.1038/sj.mp.4001809
Itagaki S, McGeer PL, Akiyama H, Zhu S, Selkoe D (1989) Relationship of microglia and astrocytes to amyloid deposits of Alzheimer disease. J Neuroimmunol 24(3):173–182. https://doi.org/10.1016/0165-5728(89)90115-x
Heneka MT, Kummer MP, Stutz A, Delekate A, Schwartz S, Vieira-Saecker A, Griep A, Axt D et al (2013) NLRP3 is activated in Alzheimer’s disease and contributes to pathology in APP/PS1 mice. Nature 493(7434):674–678. https://doi.org/10.1038/nature11729
Martin E, Amar M, Dalle C, Youssef I, Boucher C, Le Duigou C, Bruckner M, Prigent A et al (2019) New role of P2X7 receptor in an Alzheimer’s disease mouse model. Mol Psychiatry 24(1):108–125. https://doi.org/10.1038/s41380-018-0108-3
Gemma C, Bickford PC (2007) Interleukin-1beta and caspase-1: players in the regulation of age-related cognitive dysfunction. Rev Neurosci 18(2):137–148. https://doi.org/10.1515/revneuro.2007.18.2.137
Bourgognon JM, Cavanagh J (2020) The role of cytokines in modulating learning and memory and brain plasticity. Brain Neurosci Adv 4:2398212820979802. https://doi.org/10.1177/2398212820979802
Wang Z, Liu D, Wang F, Liu S, Zhao S, Ling EA, Hao A (2012) Saturated fatty acids activate microglia via Toll-like receptor 4/NF-kappaB signalling. Br J Nutr 107(2):229–241. https://doi.org/10.1017/S0007114511002868
Gupta S, Knight AG, Gupta S, Keller JN, Bruce-Keller AJ (2012) Saturated long-chain fatty acids activate inflammatory signaling in astrocytes. J Neurochem 120(6):1060–1071. https://doi.org/10.1111/j.1471-4159.2012.07660.x
Liu L, Chan C (2014) IPAF inflammasome is involved in interleukin-1beta production from astrocytes, induced by palmitate; implications for Alzheimer’s Disease. Neurobiol Aging 35(2):309–321. https://doi.org/10.1016/j.neurobiolaging.2013.08.016
Gcwensa NZ, Russell DL, Cowell RM, Volpicelli-Daley LA (2021) Molecular mechanisms underlying synaptic and axon degeneration in Parkinson’s disease. Front Cell Neurosci 15:626128. https://doi.org/10.3389/fncel.2021.626128
Burke RE, O’Malley K (2013) Axon degeneration in Parkinson’s disease. Exp Neurol 246:72–83. https://doi.org/10.1016/j.expneurol.2012.01.011
Anderson FL, Coffey MM, Berwin BL, Havrda MC (2018) Inflammasomes: an emerging mechanism translating environmental toxicant exposure into neuroinflammation in Parkinson’s disease. Toxicol Sci 166(1):3–15. https://doi.org/10.1093/toxsci/kfy219
Voet S, Srinivasan S, Lamkanfi M, van Loo G (2019) Inflammasomes in neuroinflammatory and neurodegenerative diseases. EMBO Mol Med 11(6):e10248. https://doi.org/10.15252/emmm.201810248
Whitton PS (2007) Inflammation as a causative factor in the aetiology of Parkinson’s disease. Br J Pharmacol 150(8):963–976. https://doi.org/10.1038/sj.bjp.0707167
Drouin-Ouellet J, St-Amour I, Saint-Pierre M, Lamontagne-Proulx J, Kriz J, Barker RA, Cicchetti F (2014) Toll-like receptor expression in the blood and brain of patients and a mouse model of Parkinson's disease. Int J Neuropsychopharmacol 18(6):pyu103. https://doi.org/10.1093/ijnp/pyu103
Nayak D, Roth TL, McGavern DB (2014) Microglia development and function. Annu Rev Immunol 32:367–402. https://doi.org/10.1146/annurev-immunol-032713-120240
Litteljohn D, Cummings A, Brennan A, Gill A, Chunduri S, Anisman H, Hayley S (2010) Interferon-gamma deficiency modifies the effects of a chronic stressor in mice: implications for psychological pathology. Brain Behav Immun 24(3):462–473. https://doi.org/10.1016/j.bbi.2009.12.001
Knott C, Stern G, Wilkin GP (2000) Inflammatory regulators in Parkinson’s disease: iNOS, lipocortin-1, and cyclooxygenases-1 and -2. Mol Cell Neurosci 16(6):724–739. https://doi.org/10.1006/mcne.2000.0914
McGeer PL, Itagaki S, Boyes BE, McGeer EG (1988) Reactive microglia are positive for HLA-DR in the substantia nigra of Parkinson’s and Alzheimer’s disease brains. Neurology 38(8):1285–1291. https://doi.org/10.1212/wnl.38.8.1285
Lawana V, Singh N, Sarkar S, Charli A, Jin H, Anantharam V, Kanthasamy AG, Kanthasamy A (2017) Involvement of c-Abl kinase in microglial activation of NLRP3 inflammasome and impairment in autolysosomal system. J Neuroimmune Pharmacol 12(4):624–660. https://doi.org/10.1007/s11481-017-9746-5
Zhang P, Shao XY, Qi GJ, Chen Q, Bu LL, Chen LJ, Shi J, Ming J et al (2016) Cdk5-dependent activation of neuronal inflammasomes in Parkinson’s disease. Mov Disord 31(3):366–376. https://doi.org/10.1002/mds.26488
Codolo G, Plotegher N, Pozzobon T, Brucale M, Tessari I, Bubacco L, de Bernard M (2013) Triggering of inflammasome by aggregated alpha-synuclein, an inflammatory response in synucleinopathies. PLoS ONE 8(1):e55375. https://doi.org/10.1371/journal.pone.0055375
Blum-Degen D, Muller T, Kuhn W, Gerlach M, Przuntek H, Riederer P (1995) Interleukin-1 beta and interleukin-6 are elevated in the cerebrospinal fluid of Alzheimer’s and de novo Parkinson’s disease patients. Neurosci Lett 202(1–2):17–20. https://doi.org/10.1016/0304-3940(95)12192-7
Qin XY, Zhang SP, Cao C, Loh YP, Cheng Y (2016) Aberrations in peripheral inflammatory cytokine levels in Parkinson disease: a systematic review and meta-analysis. JAMA Neurol 73(11):1316–1324. https://doi.org/10.1001/jamaneurol.2016.2742
Fan Z, Pan YT, Zhang ZY, Yang H, Yu SY, Zheng Y, Ma JH, Wang XM (2020) Systemic activation of NLRP3 inflammasome and plasma alpha-synuclein levels are correlated with motor severity and progression in Parkinson’s disease. J Neuroinflammation 17(1):11. https://doi.org/10.1186/s12974-019-1670-6
Mogi M, Harada M, Kondo T, Riederer P, Inagaki H, Minami M, Nagatsu T (1994) Interleukin-1 beta, interleukin-6, epidermal growth factor and transforming growth factor-alpha are elevated in the brain from parkinsonian patients. Neurosci Lett 180(2):147–150. https://doi.org/10.1016/0304-3940(94)90508-8
Yan Y, Jiang W, Liu L, Wang X, Ding C, Tian Z, Zhou R (2015) Dopamine controls systemic inflammation through inhibition of NLRP3 inflammasome. Cell 160(1–2):62–73. https://doi.org/10.1016/j.cell.2014.11.047
Nopoulos PC (2016) Huntington disease: a single-gene degenerative disorder of the striatum. Dialogues Clin Neurosci 18(1):91–98
Roos RA (2010) Huntington’s disease: a clinical review. Orphanet J Rare Dis 5:40. https://doi.org/10.1186/1750-1172-5-40
Davies SW, Turmaine M, Cozens BA, DiFiglia M, Sharp AH, Ross CA, Scherzinger E, Wanker EE et al (1997) Formation of neuronal intranuclear inclusions underlies the neurological dysfunction in mice transgenic for the HD mutation. Cell 90(3):537–548. https://doi.org/10.1016/s0092-8674(00)80513-9
Sanchez Mejia RO, Friedlander RM (2001) Caspases in Huntington’s disease. Neuroscientist 7(6):480–489. https://doi.org/10.1177/107385840100700604
Ona VO, Li M, Vonsattel JP, Andrews LJ, Khan SQ, Chung WM, Frey AS, Menon AS et al (1999) Inhibition of caspase-1 slows disease progression in a mouse model of Huntington’s disease. Nature 399(6733):263–267. https://doi.org/10.1038/20446
Chen M, Ona VO, Li M, Ferrante RJ, Fink KB, Zhu S, Bian J, Guo L et al (2000) Minocycline inhibits caspase-1 and caspase-3 expression and delays mortality in a transgenic mouse model of Huntington disease. Nat Med 6(7):797–801. https://doi.org/10.1038/77528
Compston A (2004) The pathogenesis and basis for treatment in multiple sclerosis. Clin Neurol Neurosurg 106(3):246–248. https://doi.org/10.1016/j.clineuro.2004.02.007
Goverman J (2009) Autoimmune T cell responses in the central nervous system. Nat Rev Immunol 9(6):393–407. https://doi.org/10.1038/nri2550
Andersson A, Covacu R, Sunnemark D, Danilov AI, Dal Bianco A, Khademi M, Wallstrom E, Lobell A et al (2008) Pivotal advance: HMGB1 expression in active lesions of human and experimental multiple sclerosis. J Leukoc Biol 84(5):1248–1255. https://doi.org/10.1189/jlb.1207844
Malhotra S, Costa C, Eixarch H, Keller CW, Amman L, Martinez-Banaclocha H, Midaglia L, Sarro E et al (2020) NLRP3 inflammasome as prognostic factor and therapeutic target in primary progressive multiple sclerosis patients. Brain 143(5):1414–1430. https://doi.org/10.1093/brain/awaa084
Inoue M, Shinohara ML (2013) NLRP3 Inflammasome and MS/EAE. Autoimmune Dis 2013:859145. https://doi.org/10.1155/2013/859145
Mamik MK, Power C (2017) Inflammasomes in neurological diseases: emerging pathogenic and therapeutic concepts. Brain 140(9):2273–2285. https://doi.org/10.1093/brain/awx133
Russi AE, Walker-Caulfield ME, Guo Y, Lucchinetti CF, Brown MA (2016) Meningeal mast cell-T cell crosstalk regulates T cell encephalitogenicity. J Autoimmun 73:100–110. https://doi.org/10.1016/j.jaut.2016.06.015
Voet S, Mc Guire C, Hagemeyer N, Martens A, Schroeder A, Wieghofer P, Daems C, Staszewski O et al (2018) A20 critically controls microglia activation and inhibits inflammasome-dependent neuroinflammation. Nat Commun 9(1):2036. https://doi.org/10.1038/s41467-018-04376-5
Keane RW, Dietrich WD, de Rivero Vaccari JP (2018) Inflammasome proteins as biomarkers of multiple sclerosis. Front Neurol 9:135. https://doi.org/10.3389/fneur.2018.00135
Coll RC, Robertson AA, Chae JJ, Higgins SC, Munoz-Planillo R, Inserra MC, Vetter I, Dungan LS et al (2015) A small-molecule inhibitor of the NLRP3 inflammasome for the treatment of inflammatory diseases. Nat Med 21(3):248–255. https://doi.org/10.1038/nm.3806
Hur EM, Youssef S, Haws ME, Zhang SY, Sobel RA, Steinman L (2007) Osteopontin-induced relapse and progression of autoimmune brain disease through enhanced survival of activated T cells. Nat Immunol 8(1):74–83. https://doi.org/10.1038/ni1415
Prusiner SB, Hsiao KK (1994) Human prion diseases. Ann Neurol 35(4):385–395. https://doi.org/10.1002/ana.410350404
Van Everbroeck B, Dobbeleir I, De Waele M, De Deyn P, Martin JJ, Cras P (2004) Differential diagnosis of 201 possible Creutzfeldt-Jakob disease patients. J Neurol 251(3):298–304. https://doi.org/10.1007/s00415-004-0311-9
Zhu C, Herrmann US, Falsig J, Abakumova I, Nuvolone M, Schwarz P, Frauenknecht K, Rushing EJ et al (2016) A neuroprotective role for microglia in prion diseases. J Exp Med 213(6):1047–1059. https://doi.org/10.1084/jem.20151000
Shi F, Yang L, Kouadir M, Yang Y, Wang J, Zhou X, Yin X, Zhao D (2012) The NALP3 inflammasome is involved in neurotoxic prion peptide-induced microglial activation. J Neuroinflammation 9:73. https://doi.org/10.1186/1742-2094-9-73
Sasaki A, Hirato J, Nakazato Y (1993) Immunohistochemical study of microglia in the Creutzfeldt-Jakob diseased brain. Acta Neuropathol 86(4):337–344. https://doi.org/10.1007/BF00369445
Giese A, Brown DR, Groschup MH, Feldmann C, Haist I, Kretzschmar HA (1998) Role of microglia in neuronal cell death in prion disease. Brain Pathol 8(3):449–457. https://doi.org/10.1111/j.1750-3639.1998.tb00167.x
Hafner-Bratkovic I, Bencina M, Fitzgerald KA, Golenbock D, Jerala R (2012) NLRP3 inflammasome activation in macrophage cell lines by prion protein fibrils as the source of IL-1beta and neuronal toxicity. Cell Mol Life Sci 69(24):4215–4228. https://doi.org/10.1007/s00018-012-1140-0
Srivastava S, Katorcha E, Makarava N, Barrett JP, Loane DJ, Baskakov IV (2018) Inflammatory response of microglia to prions is controlled by sialylation of PrP(Sc). Sci Rep 8(1):11326. https://doi.org/10.1038/s41598-018-29720-z
Bellezza I, Grottelli S, Costanzi E, Scarpelli P, Pigna E, Morozzi G, Mezzasoma L, Peirce MJ et al (2018) Peroxynitrite activates the NLRP3 inflammasome cascade in SOD1(G93A) mouse model of amyotrophic lateral sclerosis. Mol Neurobiol 55(3):2350–2361. https://doi.org/10.1007/s12035-017-0502-x
Borchelt DR, Lee MK, Slunt HS, Guarnieri M, Xu ZS, Wong PC, Brown RH Jr, Price DL et al (1994) Superoxide dismutase 1 with mutations linked to familial amyotrophic lateral sclerosis possesses significant activity. Proc Natl Acad Sci U S A 91(17):8292–8296. https://doi.org/10.1073/pnas.91.17.8292
Gugliandolo A, Giacoppo S, Bramanti P, Mazzon E (2018) NLRP3 inflammasome activation in a transgenic amyotrophic lateral sclerosis model. Inflammation 41(1):93–103. https://doi.org/10.1007/s10753-017-0667-5
Pasinelli P, Borchelt DR, Houseweart MK, Cleveland DW, Brown RH Jr (1998) Caspase-1 is activated in neural cells and tissue with amyotrophic lateral sclerosis-associated mutations in copper-zinc superoxide dismutase. Proc Natl Acad Sci U S A 95(26):15763–15768. https://doi.org/10.1073/pnas.95.26.15763
Johann S, Heitzer M, Kanagaratnam M, Goswami A, Rizo T, Weis J, Troost D, Beyer C (2015) NLRP3 inflammasome is expressed by astrocytes in the SOD1 mouse model of ALS and in human sporadic ALS patients. Glia 63(12):2260–2273. https://doi.org/10.1002/glia.22891
Meissner F, Molawi K, Zychlinsky A (2010) Mutant superoxide dismutase 1-induced IL-1beta accelerates ALS pathogenesis. Proc Natl Acad Sci U S A 107(29):13046–13050. https://doi.org/10.1073/pnas.1002396107
Lehmann S, Esch E, Hartmann P, Goswami A, Nikolin S, Weis J, Beyer C, Johann S (2018) Expression profile of pattern recognition receptors in skeletal muscle of SOD1((G93A)) amyotrophic lateral sclerosis (ALS) mice and sporadic ALS patients. Neuropathol Appl Neurobiol 44(6):606–627. https://doi.org/10.1111/nan.12483
De Paola M, Sestito SE, Mariani A, Memo C, Fanelli R, Freschi M, Bendotti C, Calabrese V et al (2016) Synthetic and natural small molecule TLR4 antagonists inhibit motoneuron death in cultures from ALS mouse model. Pharmacol Res 103:180–187. https://doi.org/10.1016/j.phrs.2015.11.020
Beers DR, Henkel JS, Zhao W, Wang J, Appel SH (2008) CD4+ T cells support glial neuroprotection, slow disease progression, and modify glial morphology in an animal model of inherited ALS. Proc Natl Acad Sci U S A 105(40):15558–15563. https://doi.org/10.1073/pnas.0807419105
Henkel JS, Beers DR, Zhao W, Appel SH (2009) Microglia in ALS: the good, the bad, and the resting. J Neuroimmune Pharmacol 4(4):389–398. https://doi.org/10.1007/s11481-009-9171-5
Rothstein JD, Tsai G, Kuncl RW, Clawson L, Cornblath DR, Drachman DB, Pestronk A, Stauch BL et al (1990) Abnormal excitatory amino acid metabolism in amyotrophic lateral sclerosis. Ann Neurol 28(1):18–25. https://doi.org/10.1002/ana.410280106
Zhang R, Liu Y, Yan K, Chen L, Chen XR, Li P, Chen FF, Jiang XD (2013) Anti-inflammatory and immunomodulatory mechanisms of mesenchymal stem cell transplantation in experimental traumatic brain injury. J Neuroinflammation 10:106. https://doi.org/10.1186/1742-2094-10-106
Kerr N, Lee SW, Perez-Barcena J, Crespi C, Ibanez J, Bullock MR, Dietrich WD, Keane RW et al (2018) Inflammasome proteins as biomarkers of traumatic brain injury. PLoS ONE 13(12):e0210128. https://doi.org/10.1371/journal.pone.0210128
Irrera N, Pizzino G, Calo M, Pallio G, Mannino F, Fama F, Arcoraci V, Fodale V et al (2017) Lack of the Nlrp3 inflammasome improves mice recovery following traumatic brain injury. Front Pharmacol 8:459. https://doi.org/10.3389/fphar.2017.00459
Weber JT (2012) Altered calcium signaling following traumatic brain injury. Front Pharmacol 3:60. https://doi.org/10.3389/fphar.2012.00060
Awasthi D, Kutz SC, Beuerman R, Nguyen D, Carey ME, Zeiller S (2003) Early gene expression in the rat cortex after experimental traumatic brain injury and hypotension. Neurosci Lett 345(1):29–32. https://doi.org/10.1016/s0304-3940(03)00497-x
Clausen F, Hanell A, Bjork M, Hillered L, Mir AK, Gram H, Marklund N (2009) Neutralization of interleukin-1beta modifies the inflammatory response and improves histological and cognitive outcome following traumatic brain injury in mice. Eur J Neurosci 30(3):385–396. https://doi.org/10.1111/j.1460-9568.2009.06820.x
Adamczak S, Dale G, de Rivero Vaccari JP, Bullock MR, Dietrich WD, Keane RW (2012) Inflammasome proteins in cerebrospinal fluid of brain-injured patients as biomarkers of functional outcome: clinical article. J Neurosurg 117(6):1119–1125. https://doi.org/10.3171/2012.9.JNS12815
Wallisch JS, Simon DW, Bayir H, Bell MJ, Kochanek PM, Clark RSB (2017) Cerebrospinal fluid NLRP3 is increased after severe traumatic brain injury in infants and children. Neurocrit Care 27(1):44–50. https://doi.org/10.1007/s12028-017-0378-7
Piancone F, La Rosa F, Marventano I, Saresella M, Clerici M (2021) The role of the inflammasome in neurodegenerative diseases. Molecules 26(4):953. https://doi.org/10.3390/molecules26040953
Duan Y, Kelley N, He Y (2020) Role of the NLRP3 inflammasome in neurodegenerative diseases and therapeutic implications. Neural Regen Res 15(7):1249–1250. https://doi.org/10.4103/1673-5374.272576
Youm YH, Nguyen KY, Grant RW, Goldberg EL, Bodogai M, Kim D, D’Agostino D, Planavsky N et al (2015) The ketone metabolite beta-hydroxybutyrate blocks NLRP3 inflammasome-mediated inflammatory disease. Nat Med 21(3):263–269. https://doi.org/10.1038/nm.3804
Juliana C, Fernandes-Alnemri T, Wu J, Datta P, Solorzano L, Yu JW, Meng R, Quong AA et al (2010) Anti-inflammatory compounds parthenolide and Bay 11–7082 are direct inhibitors of the inflammasome. J Biol Chem 285(13):9792–9802. https://doi.org/10.1074/jbc.M109.082305
Ahn H, Kim J, Jeung EB, Lee GS (2014) Dimethyl sulfoxide inhibits NLRP3 inflammasome activation. Immunobiology 219(4):315–322. https://doi.org/10.1016/j.imbio.2013.11.003
Inoue M, Williams KL, Oliver T, Vandenabeele P, Rajan JV, Miao EA, Shinohara ML (2012) Interferon-beta therapy against EAE is effective only when development of the disease depends on the NLRP3 inflammasome. Sci Signal 5(225):ra38. https://doi.org/10.1126/scisignal.2002767
Isakov E, Weisman-Shomer P (1840) Benhar M (2014) Suppression of the pro-inflammatory NLRP3/interleukin-1beta pathway in macrophages by the thioredoxin reductase inhibitor auranofin. Biochim Biophys Acta 10:3153–3161. https://doi.org/10.1016/j.bbagen.2014.07.012
Honda H, Nagai Y, Matsunaga T, Okamoto N, Watanabe Y, Tsuneyama K, Hayashi H, Fujii I et al (2014) Isoliquiritigenin is a potent inhibitor of NLRP3 inflammasome activation and diet-induced adipose tissue inflammation. J Leukoc Biol 96(6):1087–1100. https://doi.org/10.1189/jlb.3A0114-005RR
He Y, Varadarajan S, Munoz-Planillo R, Burberry A, Nakamura Y, Nunez G (2014) 3,4-methylenedioxy-beta-nitrostyrene inhibits NLRP3 inflammasome activation by blocking assembly of the inflammasome. J Biol Chem 289(2):1142–1150. https://doi.org/10.1074/jbc.M113.515080
Maier NK, Leppla SH, Moayeri M (2015) The cyclopentenone prostaglandin 15d-PGJ2 inhibits the NLRP1 and NLRP3 inflammasomes. J Immunol 194(6):2776–2785. https://doi.org/10.4049/jimmunol.1401611
Reboldi A, Dang EV, McDonald JG, Liang G, Russell DW, Cyster JG (2014) Inflammation. 25-Hydroxycholesterol suppresses interleukin-1-driven inflammation downstream of type I interferon. Science 345(6197):679–684. https://doi.org/10.1126/science.1254790
Gordon R, Albornoz EA, Christie DC, Langley MR, Kumar V, Mantovani S, Robertson AAB, Butler MS, Rowe DB, O'Neill LA, Kanthasamy AG, Schroder K, Cooper MA, Woodruff TM (2018) Inflammasome inhibition prevents alpha-synuclein pathology and dopaminergic neurodegeneration in mice. Sci Transl Med 10(465):eaah4066. https://doi.org/10.1126/scitranslmed.aah4066
Dempsey C, Rubio Araiz A, Bryson KJ, Finucane O, Larkin C, Mills EL, Robertson AAB, Cooper MA, O’Neill LAJ, Lynch MA (2017) Inhibiting the NLRP3 inflammasome with MCC950 promotes non-phlogistic clearance of amyloid-beta and cognitive function in APP/PS1 mice. Brain Behav Immun 61:306–316. https://doi.org/10.1016/j.bbi.2016.12.014
La Rosa F, Saresella M, Marventano I, Piancone F, Ripamonti E, Al-Daghri N, Bazzini C, Zoia CP, Conti E, Ferrarese C, Clerici M (2019) Stavudine reduces NLRP3 inflammasome activation and modulates amyloid-beta autophagy. J Alzheimers Dis 72(2):401–412. https://doi.org/10.3233/JAD-181259
Acknowledgements
The authors gratefully acknowledge the Department of Science and Technology, Government of India, for their support in the form of FIST Grant.
Funding
This work was supported by the Science & Engineering Research Board, Government of India (EMR/2017/002793); the University Grants Commission, Government of India (Startup Research Grant); and the Indian Council of Medical Research, Government of India (IRIS ID: 2020–5687).
Author information
Authors and Affiliations
Contributions
LC outlined and structured the review; SB, NS and LC wrote the manuscript; NS and LC created the figures and tables; SB, NS, and LC proofed the text and edited the tables and figures. LC and NS revised the manuscript. All authors read and approve the final version of the manuscript at the time of submission.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
All contributing authors agree to the publication of this article.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Brahadeeswaran, S., Sivagurunathan, N. & Calivarathan, L. Inflammasome Signaling in the Aging Brain and Age-Related Neurodegenerative Diseases. Mol Neurobiol 59, 2288–2304 (2022). https://doi.org/10.1007/s12035-021-02683-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-021-02683-5