Abstract
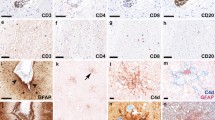
Immunohistochemical techniques have been used to investigate microglial reaction in Creutzfeldt-Jakob diseased (CJD) brains. Autopsy cases of six patients with CJD and age-matched controls were studied. Formalin-fixed, paraffin-embedded brain tissue samples were stained with antibodies against major histocompatibility complex (MHC) class II antigen (Ag), leukocyte common antigen (LCA), CDw75, CD68 and glial fibrillary acidic protein. Of the patients with CJD, two with a subacute spongiform encephalopathic type and short-survival periods after onset of the disease showed an increased number of reactive microglia labeled with anti-MHC class II Ag or LCA in the affected cerebral cortex. In advanced cases of the panencephalopathic type of CJD, in which both cerebral atrophy and astrocytosis were marked, the increase of reactive microglia was small. Some vacuoles developing in the neuropil of the CJD patients were surrounded by MHC class II Ag- or LCA-immunoreactive microglial cells. The number of ramified microglia in the affected lesions was decreased, although their number in the hippocampus was not affected. These results indicate that microglia can frequently be involved in the process of CJD and may be activated at the early stage of the disease.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adrian EK, Williams MG, George FC (1978) Fine structure of reactive cells in injured nervous tissue labeled with [3H]thymidine injected before injury. J Comp Neurol 180:815–840
Akiyama H, McGeer PL (1990) Brain microglia constitutively express beta-2 integrins. J Neuroimmunol 30:81–93
Brosnan CF, Selmaj K, Raine CS (1988) Hypothesis: a role for tumor necrosis factor in immune-mediated demyelination and its relevance to multiple sclerosis. J Neuroimmunol 18:87–94
DelRio-Hortega P (1919) El tercer elemento de los centros neviosos. I. La microglia en estado normal. II. Intervencion de la microglia en los processos patologicos. III. Naturaleza probable de la microglia. Bol Soc Exp Biol 9:68–120
Dickson DW, Mattiace LA (1989) Astrocytes and microglia in human brain share an epitope recognized by a B-lymphocyte-specific monoclonal antibody (LN-1). Am J Pathol 135:135–147
Dickson DW, Mattiace LA, Kure K, Hutchins K, Lyman WD, Brosnan CF (1991) Microglia in human disease, with an emphasis on acquired immune deficiency syndrome. Lab Invest 64:135–156
Fraser H, Dickinson AG (1978) Studies of the lymphoreticular system in the pathogenesis of scrapie: the role of spleen and thymus. J Comp Pathol 88:563–573
Frei K, Malipiero UV, Leist TP, Zinkernagel RM, Schwab ME, Fontana A (1989) On the cellular source and function of interleukin 6 produced in the central nervous system in viral diseases. Eur J Immunol 19:689–694
Gibbs CJ Jr, Gadjusek DC, Asher DM, Alpers MP, Beck E, Daniel PM, Matthews WB (1968) Creutzfeldt-Jakob disease (spongiform encephalopathy): transmission to the chimpanzee. Science 161:388–389
Graeber MB, Streit WJ (1990) Microglia: immune network in the CNS. Brain Pathol 1:2–5
Hayes GW, Woodroofe MN, Cuzner ML (1987) Microglia are the major cell type expressing MHC class II in human white matter. J Neurol Sci 80:25–37
Hulette CM, Downey BT, Burger PC (1992) Macrophage markers in diagnostic neuropathology. Am J Surg Pathol 16:493–499
Kaneko Y, Kitamoto T, Tateishi J, Yamaguchi K (1989) Ferritin immunohistochemistry as a marker for microglia. Acta Neuropathol 79:129–136
Kitamura T, Hattori H, Fujita S (1972) Autoradiographic studies on histogenesis of brain macrophages in the mouse. J Neuropathol Exp Neurol 31:502–518
Lampert PW, Gajdusek DC, Gibbs CJ Jr (1971) Experimental spongiform encephalopathy (Creutzfeldt-Jakob disease) in chimpanzees. Electron microscopic observations. J Neuropathol Exp Neurol 30:20–32
Lampert PW, Gadjusek DC, Gibbs CJ Jr (1972) Subacute spongiform virus encephalopathies. Am J Pathol 68:626–652
Mallat M, Houlgatte R, Brachet P, Prochiantz A (1989) Lipopolysaccharide-stimulated rat brain macrophages release NGF in vitro. Dev Biol 133:309–311
Masters CL, Richardson EP Jr (1978) Subacute spongiform encephalopathy (Creutzfeldt-Jakob disease): the nature and progression of spongiform change. Brain 101:333–344
Mattiace LA, Davies P, Dickson DW (1990) Detection of HLA-DR on microglia in the human brain is a function of both clinical and technical factors. Am J Pathol 136:1101–1114
McGeer PL, Itagaki S, McGeer EG (1988) Expression of the histocompatibility glycoprotein HLA-DR in neurological disease. Acta Neuropathol 76:550–557
Miyazono M, Iwaki T, Kitamoto T, Kaneko Y, Doh-ura K, Tateishi J (1991) A comparative immunohistochemical study of Kuru and senile plaques with a special reference to glial reactions at various stages of amyloid plaque formation. Am J Pathol 139:589–598
Mizutani T (1981) Neuropathology of Creutzfeldt-Jakob disease in Japan: with special reference to the panencephalopathic type. Acta Pathol Jpn 31:903–922
Mohri S, Handa S, Tateishi J (1987) Lack of effect of thymus and spleen on the incubation period of Creutzfeldt-Jakob disease in mice. J Gen Virol 68:1187–1189
Nakazato Y, Ishizeki J, Takahashi K, Yamaguchi H, Kamei T, Mori T (1982) Localization of S-100 protein and glial fibrillary acidic protein-related antigen in pleomorphic adenoma of the salivary glands. Lab Invest 46:621–626
Nakazato Y, Hirato J, Ishida Y, Hoshi S, Hasegawa M, Fukuda T (1990) Swollen cortical neurons in Creutzfeldt-Jakob disease contain a phosphorylated neurofilament epitope. J Neuropathol Exp Neurol 49:197–205
Neumann MA, Cohn R (1987) Long duration Jakob-Creutzfeldt disease. Arch Gerontol Geriatr 6:279–287
Nevin S, McMenemey WH, Behrman S, Jones DP (1960) Subacute spongiform encephalopathy — A subacute form of encephalopathy attributable to vascular dysfunction (Spongiform cerebral atrophy). Brain 83:519–564
Paulus W, Roggendorf W, Kirchner T (1992) Ki-M1P as a marker for microglia and brain macrophages in routinely processed human tissues. Acta Neuropathol 84:538–544
Sasaki A, Nakazato Y (1992) The identity of cells expressing MHC class II antigens in normal and pathological human brain. Neuropathol Appl Neurobiol 18:13–26
Sasaki A, Nakanishi Y, Nakazato Y, Yamaguchi H (1991) Application of lectin and B-lymphocyte-specific monoclonal antibodies for the demonstration of human microglia in formalin-fixed, paraffin-embedded brain tissue. Virchows Arch [A] 419:291–299
Siedler H, Malamud N (1963) Creutzfeldt-Jakob's disease. Clinicopathologic report of 15 cases and review of the literature (with special reference to a related disorder designated as subacute spongiform encephalopathy). J Neuropathol Exp Neurol 22:381–402
Streit WJ, Graeber MB, Kreutzberg GW (1988) Functional plasticity of microglia: a review. Glia 1:301–307
Suzuki H, Franz H, Yamamoto T, Iwasaki Y, Konno H (1988) Identification of the normal microglial population in human and rodent nervous tissue using lectin-histochemistry. Neuropathol Appl Neurobiol 14:221–227
Tomlinson BE (1992) Creutzfeldt-Jakob disease. In: Adams JH Duchen LW (eds) Greenfield's neuropathology, 5th edn. Edward Arnold, London, pp 1366–1375
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sasaki, A., Hirato, J. & Nakazato, Y. Immunohistochemical study of microglia in the Creutzfeldt-Jakob diseased brain. Acta Neuropathol 86, 337–344 (1993). https://doi.org/10.1007/BF00369445
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00369445