Abstract
Glycogen synthase kinase-3β (GSK-3β) is a key element to phosphorylate tau and form neurofibrillary tangles (NFTs) found in tauopathies including Alzheimer’s disease (AD). A current topic for AD therapy is focused upon how to prevent tau phosphorylation. In the present study, PKCε activated Akt and inactivated GSK-3β by directly interacting with each protein. Inhibition of protein tyrosine phosphatase 1B (PTP1B), alternatively, caused an enhancement in the tyrosine phosphorylation of insulin receptor substrate 1 (IRS-1), allowing activation of Akt through a pathway along an IRS-1/phosphatidylinositol 3 kinase (PI3K)/3-phosphoinositide-dependent protein kinase-1 (PDK1)/Akt axis, to phosphorylate and inactivate GSK-3β. Combination of PKCε activation and PTP1B inhibition more sufficiently activated Akt and inactivated GSK-3β than each independent treatment, to suppress amyloid β (Aβ)-induced tau phosphorylation and ameliorate spatial learning and memory impairment in 5xFAD transgenic mice, an animal model of AD. This may represent an innovative strategy for AD therapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Alzheimer’s disease (AD) is a really tragic disease that a human being loses human dignity. AD is characterized by extensive deposition of amyloid β (Aβ) and formation of neurofibrillary tangles (NFTs) consisting of hyperphosphorylated tau. One thinks that Aβ and tau serve as an initiator and an executor of AD dementia, respectively. The pathogenic mechanisms of AD, however, are not fully understood as yet. So far, the major focus of the AD research has been Aβ, and a variety of Aβ-targeted drugs have been developed for AD therapy but ended in failure. Accordingly, the focus of the AD research is currently turned to tau.
Tau includes six isoforms referred to as τ3L, τ3S, τ3, τ4L, τ4S, and τ4 [1]. Tau is abundantly expressed in neurons of the central nervous system and stabilizes microtubules by interacting with tubulin. Tau also interacts with other cytoskeletal components spectrin and actin filaments or the isomerase Pin 1. Microtubles are the tracks for motor proteins bearing intracellular transport of vesicles, organelles, and protein complexes [1, 2], and tau modulates microtubule dynamics including axonal transport [3–7]. Moreover, expression of tau is upregulated during neuronal development to promote generation of cell processes and establish cell polarity [8].
In AD, Tau is hyperphosphorylated, is missorted into the somatodendritic compartment, and aggregates into NFT [9]. Aβ induces tau phosphorylation, enhances tau pathology, and accelerates NFT formation [10–15]. Tau is phosphorylated by a variety of serine/threonine protein kinases such as GSK-3β, cyclin-dependent kinase 5 (CDK5), extracellular-signal-regulated kinase 2 (ERK2), S6 kinase, microtubule-affinity-regulating kinase (MARK), SAD kinase, and PKA or Src family kinases such as fyn and c-Abl [16–18]. Of them, the essential kinase is GSK-3β, which is inactivated and activated each through its phosphorylation at Ser9 and Tyr216 [17]. Akt, a serine/threonine protein kinase, is activated through a pathway along a receptor tyrosine kinase (RTK)/IRS-1/PI3K/PDK1/Akt axis, to phosphorylate and inactivate GSK-3β [19]. Tau phosphorylation, accordingly, should be suppressed by activating Akt through a RTK pathway or directly interacting with Akt and GSK-3β.
We show here that combination of PKCε activation and PTP1B inhibition effectively suppresses Aβ-induced tau phosphorylation by targeting Akt and GSK-3β, leading to improvement of spatial learning and memory impairment in 5xFAD AD model mice.
Methods
Animal Care
All procedures have been approved by the Animal Care and Use Committee at Hyogo College of Medicine and were in compliance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Cell Culture
PC-12 cells, obtained from RIKEN Cell Bank (Tsukuba, Japan), were cultured in DMEM with 10 % (v/v) heat-inactivated FBS and 10 % (v/v) heat-inactivated horse serum supplemented with penicillin (100 U/ml), and streptomycin (0.1 mg/mL), in a humidified atmosphere of 5 % CO2 and 95 % air at 37 °C.
Construction and Transfection of siRNA
The small interfering RNAs (siRNAs) to silence the PI3K-, PDK1-, Akt1/2-, PKCε-, mammalian target of rapamycin (mTOR)-, or PTP1B-targeted gene and the negative control siRNA (NC siRNA) were obtained from Santa Cruz Biotechnology (Santa Cruz, CA, USA) and Ambion (Carlsbad, CA, USA). siRNAs were transfected into PC-12 cells using a Lipofectamine reagent (Invitrogen, Carlsbad, CA, USA), and cells were used for experiments 48 h after transfection. Knockdown of the relevant proteins was confirmed in the Western blot analysis (Supplementary Fig. S1a–f).
Plasmid Construction and Transfection of Plasmid PKCε
Nucleotide sequence coding for PKCε was cloned into pcDNA6/V5-His A vector (Invitrogen) at the KpnI-XhoI site, and the PKCε plasmid was constructed. The PKCε plasmid was transfected into PC-12 cells using a Lipofectamine reagent (Invitrogen), and cells were used for experiments 48 h after transfection. Knocking-in of PKCε was confirmed in the Western blot analysis (Supplementary Fig. S1g).
Western Blotting
Western blotting was carried out using antibodies against Akt1/2 (Cell Signaling, Beverly, MA, USA), phospho-Thr308/309-Akt1/2 (pT308/309; Cell Signaling), phospho-Ser473/474-Akt1/2 (pS473/474; Cell Signaling), GSK-3β (Cell Signaling), phospho-Ser9-GSK-3β (pS9; Cell Signaling), phospho-Tyr216-GSK-3β (pY216; BD Biosciences, San Jose, CA, USA), PKCε (BD Biosciences), tau (Cell Signaling), phospho-Ser202/Thr205-Tau (pS202/pT205; Thermo Fisher Scientific, Rockford, IL, USA), phospho-Ser396-Tau (pS396; Cell Signaling), β-catenin (Cell Signaling), and phospho-Ser33/Ser37/Thr41-β-catenin (pS22/S37/T41; Cell Signaling).
Immunoprecipitation
Rat hippocampal slices (male Wistar, 6 weeks old; 400 μm) were incubated in a standard artificial cerebrospinal fluid oxygenated with 95 % O2 and 5 % CO2 in the presence and absence of 8-[2-(2-pentyl-cyclopropylmethyl)-cyclopropyl]-octanoic acid (DCP-LA; 100 nM) for 3 min at 34 °C. Then, slices were homogenized by sonication in TBS-T [150 mM NaCl, 0.1 % (v/v) Tween-20 and 20 mM Tris, pH 7.5] containing 1 % (v/v) protease inhibitor cocktail and 1 % (v/v) phosphatase inhibitor cocktail and, subsequently, homogenates were centrifuged at 800g for 5 min at 4 °C. Supernatants (200 μg of protein) were incubated with an anti-PKCε antibody overnight at 4 °C. Then, 20 μl of protein G sepharose (GE healthcare, Piscataway, NJ, USA) was added to the extracts and incubated for 60 min at 4 °C. Pellets were washed three times with TBS-T and dissolved in an SDS sample buffer [0.2 mM Tris, 0.05 % (w/v) SDS, 25 % (v/v) 2-mercaptoethanol, and 20 % (v/v) glycerol, pH 6.8] for Western blotting.
Cell-Free Kinase Assay
In the cell-free Akt assay, human recombinant Akt1 (Active Motif, Carlsbad, CA, USA), Akt2 (Active Motif), or Akt3 (Active Motif) was reacted with human recombinant PKCε (Calbiochem, San Diego, CA, USA) or human recombinant PKCγ (Calbiochem) in medium A (20 mM Tris–HCl, 5 mM Mg acetate, and 1 mM ATP, pH 7.5) containing DCP-LA or diDCP-LA-PE or with PI3K (p110β/p85α; Sigma, St. Louis, MO, USA) or PDK1 (SignalChem, Richmond, Canada) in medium B (25 mM 3-morpholinopropanesulfonic acid, 25 mM MgCl2, 12.5 mM glycerol 2-phosphate, 5 mM EGTA, 2 mM EDTA, 0.25 mM dithiothreitol, and 0.25 mM ATP, pH 7.2) at 30 °C for 20 min. In the cell-free GSK-3β assay, human recombinant GSK-3β (Sigma) was reacted with human recombinant PKCε in the medium A containing DCP-LA or human recombinant Akt1, Akt2, or Akt3 in a medium C (20 mM Tris–HCl, 5 mM Mg acetate, 12.5 mM glycerol 2-phosphate, and 250 μM ATP, pH 7.5) at 30 °C for 20 min. In the cell-free β-catenin assay, human recombinant β-catenin (Abcam, Cambridge, UK) was reacted with human recombinant GSK-3β in the medium B containing DCP-LA at 30 °C for 20 min. Then, phosphorylation of each protein was monitored in the Western blot analysis.
Water Maze Test
5xFAD mice were purchased from The Jackson Laboratory (Bar Harbor, ME, USA) and maintained by crossing heterozygous transgenic mice with B6/SJL F1 breeders. Non-transgenic wild-type littermate mice were used as controls. It is recognized that suppression of long-term potentiation, a cellular mode of learning and memory, in the hippocampal CA1 region or impairment of hippocampal-dependent fear memory formation is found in 5xFAD mice at 6 months of age, but not at <4 months of age. We, therefore, carried out water maze tests using male mice at 5.5–6.5 months of age.
A circular plastic water tank 90 cm in diameter and 36 cm deep was used for a water maze test. The entire inside of the pool was white, and the pool was filled up to 20 cm from the bottom with murky water containing white Indian ink at 22–25 °C. A platform (11 cm in diameter) painted white was placed in the water, the top sinking 1 cm below the water surface. The pool was put in a test room, where there were several marks that mice were able to see from the pool. The position of the marks remained unchanged throughout testing. A platform was located in the constant position, i.e., in the middle of one quadrant, equidistant from the center and edge of the pool. Mice facing the wall of the pool were placed into water at one of three positions selected at random, and time from start to escape onto the platform (escape latency) was measured. When they had succeeded, mice were allowed to stay on the platform for 10 s. When mice failed to find the platform within 90 s, the trial was stopped and mice were put on the platform for 10 s. Two trials were carried out for a day, and the second trial began 30 s after the end of the first trial. Mice received the task for 8 consecutive days, and the mean latency from 2 consecutive days was calculated. Then, we tested two trials again 7 days later, and time from start to escape onto the platform was monitored as retention latency. Mice were killed after water maze test, and the hippocampus was isolated from the brain, followed by Western blotting.
8-[2-(2-Pentyl-cyclopropylmethyl)-cyclopropyl]-octanoic acid (DCP-LA) was dissolved with polyethylene glycol (PEG). DCP-LA or PEG was administered daily using a feeding needle from 7 days before to the end of water maze test.
Statistical Analysis
Statistical analysis was carried out using unpaired t- test, analysis of variance (ANOVA) followed by a Bonferroni correction, and ANOVA followed by Fisher’s protected least significant difference (PLSD) test.
Results
PTP1B Inhibition and PKCε Are Implicated in the Activation of Akt
The linoleic acid derivative DCP-LA serves as a potent inhibitor of PTP1B and a selective activator of the novel PKC isozyme PKCε [20–22]. We have found that DCP-LA activates PKC in a bell-shaped concentration (10 nM to 100 μM)-dependent manner in PC-12 cells, with the maximum at 100 nM [21]. We, therefore, used 100 nM of DCP-LA for the present experiments using hippocampal slices and PC-12 cells. DCP-LA enhanced phosphorylation of IRS-1 at Tyr1222 (Fig. 1a) and Akt1/2 at Thr308/309 and Ser473/474 in rat hippocampal slices (Fig. 1b–d). DCP-LA-induced Akt1/2 phosphorylation at both the sites was inhibited by the PI3K inhibitor wortmannin (WM, Fig. 1b), while the phosphorylation at Thr308/309 alone was abrogated by the PDK1 inhibitor BX912 (BX, Fig. 1c). Similarly, DCP-LA enhanced Akt1/2 phosphorylation in PC-12 cells, and the phosphorylation at Thr308/309 and Ser473/474 or only Thr308/309 is inhibited by knocking-down PI3K (Fig. 2a) or PDK1 (Fig. 2b), respectively. Collectively, these results suggest that Akt is activated through an IRS-1/PI3K/PDK1 pathway stimulated in association with PTP1B inhibition.
Hippocampal slices were untreated and treated with DCP-LA (100 nM) in the absence and presence of WM (1 μM), BX (100 nM), GF (100 nM), or MK (5 μM) for 3 min, and phosphorylation of IRS-1 (a), Akt1/2 (b–d), and GSK-3β (e–h) was monitored in the Western blotting. The arrow shown in (h) indicates the signal band at 46 kDa for pY216-GSK-3β. In the graphs, each column represents the mean (±SEM) signal intensity for phosphorylation relative to that for each protein (n = 4 independent experiments). P value in (a), unpaired t-test. P values in (b–h), ANOVA followed by a Bonferroni correction. NS, not significant
PC-12 cells, transfected with the siRNA for negative control (NC), PI3K, PDK1, Akt1/2, or PKCε, were untreated and treated with DCP-LA (100 nM) for 3 min, and phosphorylation of Akt1/2 (a–c) and GSK-3β (d–g) was monitored in the Western blotting. KD, knockdown. In the graphs, each column represents the mean (±SEM) signal intensity for phosphorylation relative to that for each protein (n = 4–8 independent experiments). P values, ANOVA followed by a Bonferroni correction. NS, not significant
Notably, the PKC inhibitor GF109203X (GF) cancelled DCP-LA-induced Akt1/2 phosphorylation at Ser473/474 in rat hippocampal slices (Fig. 1d). Moreover, the phosphorylation at Ser473/474 in PC-12 cells was prevented by knocking-down PKCε (Fig. 2c). These results indicate that PKCε is implicated in Akt1/2 activation, regardless of PTP1B inhibition.
PTP1B Inhibition and PKCε Are Implicated in the Inactivation of GSK-3β
If DCP-LA activates Akt, the drug should phosphorylate and inactivate GSK-3β. To address this issue, we monitored phosphorylation of GSK-3β. DCP-LA significantly enhanced GSK-3β phosphorylation at Ser9 in rat hippocampal slices, although Tyr216 phosphorylation was not affected (Fig. 1e–h). DCP-LA-induced Ser9 phosphorylation was abolished by WM (Fig. 1e) and the Akt inhibitor MK2206 (MK, Fig. 1g), although it was not significantly inhibited by BX (Fig. 1f). DCP-LA-induced Ser9 phosphorylation in PC-12 cells was clearly inhibited by knocking-down PI3K (Fig. 2d), PDK1 (Fig. 2e), or Akt1/2 (Fig. 2f). These results suggest that Akt, activated through an IRS-1/PI3K/PDK1 pathway stimulated in association with PTP1B inhibition, phosphorylates and inactivates GSK-3β.
DCP-LA-induced GSK-3β phosphorylation at Ser9, on the other hand, was apparently inhibited by GF in rat hippocampal slices (Fig. 1h) or knocking-down PKCε in PC-12 cells (Fig. 2g). This indicates that PKCε is also implicated in the inactivation of GSK-3β.
β-Catenin is recognized to be a substrate of GSK-3β [23]. DCP-LA significantly reduced phosphorylation of β-catenin at Ser33/Ser37/Thr41 in rat hippocampal slices (Supplementary Fig. S2a). This provides further evidence that DCP-LA-induced PTP1B inhibition and PKCε activation cause GSK-3β inactivation.
PKCε Activates Akt and Inactivate GSK-3β Through Its Direct Interaction
We have earlier found that DCP-LA activates PKCε in a concentration (0.01–100 μM)-dependent manner under the cell-free conditions, reaching maximum at 100 μM [21, 22]. We, therefore, used 100 μM of DCP-LA for the present cell-free experiments. In the cell-free kinase assay, DCP-LA phosphorylated Akt1, Akt2, and Akt3 at Ser473, Ser474, and Ser472, respectively, in the presence of PKCε in a concentration (1–100 μM)-dependent manner, and the effect was abolished by deleting ATP from extracellular solution or GF (Fig. 3a–c). This implies that PKCε, activated by DCP-LA, directly phosphorylates and activates Akt.
In the cell-free systems, Akt1 (1 μg/mL) (a), Akt2 (1 μg/mL) (b), Akt3 (1 μg/mL) (c), or GSK-3β (1 μg/mL) (g) was reacted with PKCε (1 μg/mL) in the absence and presence of DCP-LA at concentrations as indicated for 20 min, followed by Western blotting. In a different set of experiments, GSK-3β (1 μg/mL) was reacted with Akt1 (d), Akt2 (e), or Akt3 (f) at concentrations as indicated for 20 min, followed by Western blotting. -ATP, ATP-free extracellular solution; GF, 100 nM; MK, 5 μM. In the graphs, each column represents the mean (±SEM) signal intensity for phosphorylation relative to that for each protein (n = 4 independent experiments). P values, ANOVA followed by a Bonferroni correction. h Immunoprecipitation using an anti-PKCε antibody was carried out in rat hippocampal slices untreated and treated with DCP-LA (100 nM) for 3 min, followed by Western blotting. In the graphs, each column represents the mean (±SEM) signal intensity for Akt1/2 or GSK-3β relative to that for PKCε (n = 4 independent experiments). P values, unpaired t-test. Input, 10 % of the total lysates used for immunoprecipitation
Akt1, Akt2, or Akt3 phosphorylated GSK-3β at Ser9 in a concentration (1–10 μg/mL)-dependent manner, which was cancelled by deleting ATP from extracellular solution or MK (Fig. 3d–f), confirming Akt-mediated GSK-3β inactivation. This suggests that PKCε, activated by DCP-LA, activates Akt directly, to phosphorylate and inactivate GSK-3β indirectly. Amazingly, DCP-LA also phosphorylated GSK-3β at Ser9 in the presence of PKCε in a concentration (1–100 μM)-dependent manner, and the effect was abrogated by deleting ATP from extracellular solution or GF (Fig. 3g). This implies that PKCε, activated by DCP-LA, directly phosphorylates and inactivates GSK-3β.
In the cell-free assay, GSK-3β phosphorylated β-catenin at Ser33/Ser37/Thr41, and the GSK-3β inhibitor lithium actually inhibited the phosphorylation (Supplementary Fig. S2b). Likewise, DCP-LA markedly reduced GSK-3β-mediated β-catenin phosphorylation in the presence of PKCε (Supplementary Fig. S2b). β-Catenin was not phosphorylated in the absence of GSK-3β, and DCP-LA had no effect on β-catenin phosphorylation in the absence of GSK-3β and in the presence of PKCε (Supplementary Fig. S2c). These results support the notion that PKCε, activated by DCP-LA, directly inactivates GSK-3β, to inhibit phosphorylation of β-catenin, and that PKCε by itself is not engaged in the phosphorylation of β-catenin.
In the Western blot analysis using immunoprecipitants from lysates of rat hippocampal slices with an anti-PKCε antibody, the immunoreactive signals for Akt1/2 and GSK-3β were detected, and DCP-LA significantly enhanced the signals (Fig. 3h). This implies that PKCε associates with Akt1/2 or GSK-3β and DCP-LA significantly increased the association. PKCε thus could activate Akt and inactivate GSK-3β by directly interacting with each protein.
Combination of PKCε Activation and PTP1B Inhibition Suppresses Tau Phosphorylation More Effectively than Each Independent Treatment
The PTP1B inhibitor orthovanadate (Na3VO4) enhanced Akt1/2 phosphorylation at Ser473/474 in rat hippocampal slices, but phosphorylation of GSK-3β and tau was not affected (Fig. 4a–c). The PKC activator 1-oleoyl-2-acetyl-sn-glycerol (OA-DAG), which activates PKCε to an extent similar to that for DCP-LA as well as other PKC isozymes [21, 24] but has lesser/no potential to inhibit PTP1B (Supplementary Fig. S3a-c), did not enhance phosphorylation of Akt1/2, GSK-3β, tau, and β-catenin in rat hippocampal slices, but conversely, Akt1/2 phosphorylation at Ser473/474 and GSK-3β phosphorylation at Tyr216 were significantly attenuated (Fig. 4d–g). Consequently, activation of Akt1/2, inactivation of GSK-3β, and suppression of tau phosphorylation are not effectively induced by sole PTP1B inhibition or non-selective PKC activation.
Rat hippocampal slices were untreated and treated with Na3VO4 (1 μM) (a–c) or OA-DAG (30 μM) (d–g) for 3 min, followed by Western blotting. In the graphs, each column represents the mean (±SEM) signal intensity for phosphorylation relative to that for each protein (n = 4 independent experiments). P values, unpaired t-test. NS, not significant. pβ-Ca, phospho-Ser33/Ser37/Thr41-β-catenin. P values, unpaired t-test. NS, not significant
In contrast, both knocking-down PTP1B and knocking-in PKCε significantly enhanced phosphorylation of Akt1/2 at Ser473/474 and GSK-3β at Ser9 in PC-12 cells as compared with the phosphorylation induced by each independent procedure (Fig. 5a, b). Phosphorylation of tau at Ser396 and β-catenin at Ser33/ Ser37/Thr41 was significantly inhibited only by knocking-down PKCε, and no further inhibition was obtained by additionally knocking-down PTP1B (Fig. 5c, d). Taken together, these results indicate that combination of PKCε activation and PTP1B inhibition is a valid tool to activate Akt, inactivate GSK-3β, and inhibit tau phosphorylation more sufficiently than each independent treatment and that PKCε dominantly governs this machinery.
The PTP1B siRNA and/or the PKCε plasmid was transfected into PC-12 cells, and phosphorylation of Akt1/2 (a), GSK-3β (b), tau (c), and β-Catenin (d) was monitored in the Western blotting. KD, knockdown; KI, knock-in. In the graphs, each column represents the mean (±SEM) signal intensity for phosphorylation relative to that for each protein (n = 4 independent experiments). P values, ANOVA followed by a Bonferroni correction. NS, not significant
DCP-LA Suppresses Aβ-Induced GSK-3β Activation and Tau Phosphorylation
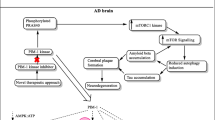
DCP-LA enhanced phosphorylation of IRS-1 at Tyr1222 and Akt1/2 at Ser473/474 in mouse hippocampal slices untreated and treated with Aβ1–42 (Fig. 6a, b). Aβ1–42 significantly reduced phosphorylation of GSK-3β at Ser9 and enhanced phosphorylation of Tau at Ser202/Thr205/Ser396 and β-catenin at Ser33/ Ser37/Thr41, which were neutralized by DCP-LA (Fig. 6c–e). Overall, these results lead to a conclusion that DCP-LA suppresses Aβ1–42-induced GSK-3β activation and tau phosphorylation by combination of PKCε activation and PTP1B inhibition—the former bearing activation of Akt and inactivation of GSK-3β through its direct interaction and the latter involving indirect inactivation of GSK-3β through an IRS-1/PI3K/PDK1/Akt pathway (Fig. 6f).
Mouse hippocampal slices were treated with Aβ1–42 (1 μM) in the absence and presence of DCP-LA (100 nM) for 3 h, followed by Western blotting (a–e). The arrow shown in (c) indicates the signal band at 46 kDa for pY216-GSK-3β. In the graphs, each column represents the mean (±SEM) signal intensity for phosphorylation relative to that for each protein (n = 4 independent experiments). P values, ANOVA followed by a Bonferroni correction. NS, not significant. f A schematic diagram for suppression of tau phosphorylation due to PTP1B inhibition and PKCε activation
DCP-LA Ameliorates Spatial Learning and Memory Impairment in 5xFAD Mice
We have found that the beneficial effect in behavioral tests is obtained with 1 mg/kg of DCP-LA [25–27], and therefore, the same dose of DCP-LA was used in the present experiments. In the water maze test, the escape and retention latencies in 5xFAD mice at 6 months of age was prolonged as compared with those in wild-type mice (Fig. 7a, b). This, taken together with the finding that the escape latency in the visible platform water maze test was not different between wild-type and 5xFAD mice (Supplementary Fig. S4a), confirms that the prolonged escape latency in 5xFAD mice is not due to disturbed visual acuity but to learning impairment. Oral administration with DCP-LA for 7 days significantly shortened the prolonged escape and retention latencies in 5xFAD mice as compared with those in PEG-administered mice (Fig. 7a, b). There was no significant difference in the swim speed between PEG- and DCP-LA-administered groups in wild-type and 5xFAD mice (Supplementary Fig. S4b). These results indicate that the effect of DCP-LA on the prolonged latencies in 5xFAD mice is not due to change in the motor activity but to improvement of spatial learning and memory deficits in 5xFAD mice.
After oral administration with DCP-LA (DCP) (1 mg/kg) or PEG for 7 days, wild-type and 5xFAD mice at 6 months of age underwent water maze task and then the hippocampus isolated from the brain for Western blotting. a The escape latency in the water maze test. In the graph, each point represents the mean (±SEM) latency from 2 consecutive days (n = 11–12 independent mice). ***, ### P < 0.0001, ANOVA followed by Fisher’s PLSD test. b The retention latency in the water maze test. In the graph, each column represents the mean (±SEM) latency (n = 11–12 independent mice). P values, ANOVA followed by a Bonferroni correction. c, d Western blot analysis. The arrow shown in (c) indicates the signal band at 46 kDa for pY216-GSK-3β. In the graphs, each column represents the mean (±SEM) signal intensity for phosphorylation relative to that for each protein (n = 11 independent experiments). P values, ANOVA followed by a Bonferroni correction. NS, not significant
Aβ1–42 levels in the brain of 5xFAD mice elevate in an age-dependent manner, and marked spatial memory deficits occur from 4–5 months of age [28]. GSK-3β phosphorylation at Ser9 and Tau phosphorylation at Ser396 in the hippocampus from 5xFAD mice at 6 months of age were significantly decreased and enhanced, respectively, as compared with those in wild-type mice at the same months of age, which were restored to the basal conditions by DCP-LA (Fig. 7c, d). DCP-LA thus appears to ameliorate spatial learning and memory impairment in 5xFAD mice by neutralizing Aβ-induced GSK-3β activation and tau phosphorylation.
Discussion
Tauopathies are a class of neurodegenerative diseases associated with aggregation of hyperphosphorylated tau in an insoluble form in the brain, referred to as NFT, which include AD, frontotemporal dementia, and parkinsonism linked to chromosome 17, progressive supranuclear palsy, Pick’s disease, and corticobasal degeneration [29]. Several lines of evidence have pointed to the relation between Aβ and tau in the pathogenesis of AD; Aβ induces tau phosphorylation, accelerates NFT formation, and enhances tau pathology, or misfolded Aβ induces prion-like misfolded tau oligomers [13, 14, 30]. Tau aggregation is shown to precede β-amyloid deposits by about 30 years [31, 32]. To date, none of Aβ-directed drugs for AD therapy have been successful and, therefore, a current target for AD therapy focuses on tau.
The most significant finding in the present study is that PKCε, activated by DCP-LA, phosphorylated Akt1/-2/-3 at Ser473/-474/-472 and GSK-3β at Ser9 by directly interacting with each protein. This implies that PKCε is capable of directly inactivating GSK-3β and indirectly through direct Akt activation. In contrast, the conventional PKC isozyme PKCγ, activated by the phosphatidylethanolamine derivative 1,2-O-bis-[8-{2-(2-pentyl-cyclopropylmethyl)-cyclopropyl}-octanoyl]-sn-glycero-3-phosphatidylethanolamine (diDCP-LA-PE)[33], had no effect on phosphorylation of Akt1/-2/-3 or GSK-3β (Supplementary Fig. S5a–d). This indicates that the effect of PKCε on Akt and GSK-3β is not due to the non-specific action of PKCs. Strangely, neither activation of Akt nor inactivation of GSK-3β was obtained with the OA-DAG, which activates a variety of PKC isozymes including PKCε. In plausible explanation of this, a certain PKC isozyme might act on PKCε negatively.
When activated, RTK phosphorylates its own receptor and IRS-1 at Tyr1185 and Tyr1222, respectively. PTP1B dephosphorylates tyrosine phosphorylation and, therefore, PTP1B inhibition could enhance tyrosine phosphorylation and its relevant signaling. Indeed, DCP-LA-induced PTP1B inhibition enhanced IRS-1 phosphorylation at Tyr1222 in rat and mouse hippocampal slices. Phosphorylated IRS-1 recruits and activates PI3K, which produces phosphatidylinositol 3,4,5-triphosphate [PI(3,4,5)P3] by phosphorylating phosphatidylinositol 4,5-bisphosphate. PI(3,4,5)P3 binds to and activates PDK1. In the cell-free kinase assay, PI3K phosphorylated Akt1 at Thr308 and Ser473, Akt2 at Ser474, and Akt3 at Ser472, which were definitely inhibited by deleting ATP from extracellular solution or WM (Supplementary Fig. S6a, c, e). PDK1, on the other hand, phosphorylated Akt1 at Thr308, Akt2 at Thr309, and Akt3 at Thr305, which were also inhibited by deleting ATP from extracellular solution or BX (Supplementary Fig. S6b, d, f). These data confirm that Akt is activated by PI3K together with and without PDK1. DCP-LA-induced Akt1/2 phosphorylation was fully/partially prevented by inhibitors of PI3K and PDK1 or knocking-down each protein. This indicates that DCP-LA-induced PTP1B inhibition could activate Akt by enhancing an IRS1/PI3K/PDK1 pathway.
Mammalian target of rapamycin (mTOR), which is activated by PI3K, is recognized to phosphorylate Akt1/2 at Ser473/474 [34–36]. DCP-LA-induced Akt1/2 phosphorylation was not inhibited by knocking-down mTOR (Supplementary Fig. S7a). In addition, DCP-LA-induced GSK-3β phosphorylation at Ser9 was not affected by knocking-down mTOR (Supplementary Fig. S7b). These findings exclude participation of mTOR in DCP-LA-induced Akt1/2 activation and GSK-3β inactivation.
Combination of PKCε activation and PTP1B inhibition activated Akt1/2, inactivated GSK-3β, and decreased tau phosphorylation more effectively than each independent treatment. Of particular interest are the findings that Aβ induces GSK-3β activation and tau phosphorylation and that the Aβ effects were neutralized by PKCε activation and PTP1B inhibition under the control of DCP-LA. In addition, DCP-LA ameliorated spatial learning and memory impairment, which occurred in parallel with activation of GSK-3β and an increase of tau phosphorylation, in 5xFAD AD model mice. Notably, the effect of DCP-LA was obtained by oral administration for only 7 days. This suggests that DCP-LA is capable of exhibiting very prompt and beneficial effect on AD dementia. In contrast, the acetylcholine esterase inhibitor galanthamine, which is clinically used for treatment of mild to moderate AD, exhibited little/less beneficial effect (Supplementary Fig. S4c, d). This suggests that acetylcholine esterase inhibitors have no potential to improve dementia accompanied by tauopathies.
Tau-targeted drugs for AD therapy under development include Hsp90 inhibitors to degrade hyperphosphorylated tau [37], tau aggregation inhibitors such as methylthioninium chloride and methylthioninium [38], O-GlcNAcase inhibitors to prevent tau phosphorylation and aggregation [39], inhibitors of tau fibrillization [40], and microtubule stabilizing agents [41]. In a series of our studies, DCP-LA induces a long-lasting facilitation of hippocampal synaptic transmission by stimulating vesicular transport of α7 ACh receptor, involving release of neurotransmitters including glutamate, and AMPA receptor, involving excitatory synaptic transmission, toward presynaptic terminals and postsynaptic cells, respectively [42–46]. The facilitatory action of DCP-LA may account for improvement of Aβ- or scopolamine-induced spatial learning and memory impairment or age-related cognitive deterioration [25–27]. The results of the present study show that DCP-LA could ameliorate AD dementia by the mechanism distinct from that previously proposed; DCP-LA suppresses Aβ-induced GSK-3β activation and Tau phosphorylation due to PKCε activation relevant to Akt-independent direct and Akt-dependent indirect inactivation of GSK-3β and to PTP1B inhibition relevant to enhancement of an IRS-1/PI3K/PDK1/Akt pathway. Consequently, combination of PKCε activation and PTP1B inhibition must be an innovative strategy for AD therapy targeting Aβ and tau.
References
Alonso A, Zaidi T, Novak M, Grundke-Iqbal I, Iqbal K (2001) Hyperphosphorylation induces self-assembly of tau into tangles of paired helical filaments/straight filaments. Proc Natl Acad Sci U S A 98(12):6923–6928
Hirokawa N, Takemura R (2005) Molecular motors and mechanisms of directional transport in neurons. Nat Rev Neurosci 6(3):201–214
Mandelkow E, von Bergen M, Biernat J, Mandelkow EM (2007) Structural principles of tau and the paired helical filaments of Alzheimer's disease. Brain Pathol 17(1):83–90
Trinczek B, Ebneth A, Mandelkow EM, Mandelkow E (1999) Tau regulates the attachment/detachment but not the speed of motors in microtubule-dependent transport of single vesicles and organelles. J Cell Sci 112(Pt14):2355–2367
Garcia ML, Cleveland DW (2001) Going new places using an old MAP: tau, microtubules and human neurodegenerative disease. Curr Opin Cell Biol 13(1):41–48
Binder LI, Guillozet-Bongaarts AL, Garcia-Sierra F, Berry RW (2005) Tau, tangles, and Alzheimer's disease. Biochim Biophys Acta 1739(2–3):216–223
Cuchillo-Ibanez I, Seereeram A, Byers HL, Leung KY, Ward MA, Anderton BH, Hanger DP (2008) Phosphorylation of tau regulates its axonal transport by controlling its binding to kinesin. FASEB J 22(9):3186–3195
Drubin DG, Kirschner MW (1986) Tau protein function in living cells. J Cell Biol 103(6 Pt2):2739–2746
Braak H, Braak E (1991) Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol 82(4):239–259
Selkoe DJ (1986) Altered structural proteins in plaques and tangles: what do they tell us about the biology of Alzheimer’s disease? Neurobiol Aging 7(6):425–432
Ballatore C, Lee VM, Trojanowski JQ (2007) Tau-mediated neurodegeneration in Alzheimer’s disease and related disorders. Nat Rev Neurosci 8(9):663–672
Ma QL, Yang F, Rosario ER, Ubeda OJ, Beech W, Gant DJ, Chen PP, Hudspeth B et al (2009) β-amyloid oligomers induce phosphorylation of tau and inactivation of insulin receptor substrate via c-Jun N-terminal kinase signaling: suppression by omega-3 fatty acids and curcumin. J Neurosci 29(28):9078–9089
Hurtado DE, Molina-Porcel L, Iba M, Aboagye AK, Paul SM, Trojanowski JQ, Lee VM (2010) Aβ accelerates the spatiotemporal progression of tau pathology and augments tau amyloidosis in an Alzheimer mouse model. Am J Pathol 177(4):1977–1988
Héraud C, Goufak D, Ando K, Leroy K, Suain V, Yilmaz Z, De Decker R, Authelet M et al (2014) Increased misfolding and truncation of tau in APP/PS1/tau transgenic mice compared to mutant tau mice. Neurobiol Dis 62:100–112
Stancu IC, Ris L, Vasconcelos B, Marinangeli C, Goeminne L, Laporte V, Haylani LE, Couturier J et al (2014) Tauopathy contributes to synaptic and cognitive deficits in a murine model for Alzheimer's disease. FASEB J 28(6):2620–2631
Patrick GN, Zukerberg L, Nikolic M, de la Monte S, Dikkes P, Tsai LH (1999) Conversion of p35 to p25 deregulates Cdk5 activity and promotes neurodegeneration. Nature 402(6762):615–622
Jeganathan S, Hascher A, Chinnathambi S, Biernat J, Mandelkow EM, Mandelkow E (2008) Proline-directed pseudo-phosphorylation at AT8 and PHF1 epitopes induces a compaction of the paperclip folding of Tau and generates a pathological (MC-1) conformation. J Biol Chem 283(46):32066–32076
Pei JJ, Björkdahl C, Zhang H, Zhou X, Winblad B (2008) p70 S6 kinase and tau in Alzheimer's disease. J Alzheimers Dis 14(4):385–392
Takashima A (2006) GSK-3 is essential in the pathogenesis of Alzheimer's disease. J Alzheimers Dis 9(3 Suppl):309–317
Tsuchiya A, Kanno T, Nagaya H, Shimizu T, Tanaka A, Nishizaki T (2014) PTP1B inhibition causes Rac1 activation by enhancing receptor tyrosine kinase signaling. Cell Physiol Biochem 33(4):1097–1105
Kanno T, Yamamoto H, Yaguchi T, Hi R, Mukasa T, Fujikawa H, Nagata T, Yamamoto S et al (2006) The linoleic acid derivative DCP-LA selectively activates PKC-ε, possibly binding to the phosphatidylserine binding site. J Lipid Res 47(6):1146–1156
Shimizu T, Kanno T, Tanaka A, Nishizaki T (2011) α, β-DCP-LA selectively activates PKC-ε and stimulates neurotransmitter release with the highest potency among 4 diastereomers. Cell Physiol Biochem 27(2):149–158
Wada A (2009) Lithium and neuropsychiatric therapeutics: neuroplasticity via glycogen synthase kinase-3β, β-catenin, and neurotrophin cascades. J Pharmacol Sci 110(1):14–28
Tsuchiya A, Kanno T, Nishizaki T (2013) Diacylglycerol promotes GLUT4 translocation to the cell surface in a PKCε-dependent and PKCλ/ι and -ζ-independent manner. Life Sci 93(5–6):240–246
Nagata T, Tomiyama T, Mori H, Yaguchi T, Nishizaki T (2010) DCP-LA neutralizes mutant amyloid β peptide-induced impairment of long-term potentiation and spatial learning. Behav Brain Res 206(1):151–154
Nagata T, Yamamoto S, Yaguchi T, Iso H, Tanaka A, Nishizaki T (2005) The newly synthesized linoleic acid derivative DCP-LA ameliorates memory deficits in animal models treated with amyloid-β peptide and scopolamine. Psychogeriatrics 5(4):122–126
Yaguchi T, Nagata T, Mukasa T, Fujikawa H, Yamamoto H, Yamamoto S, Iso H, Tanaka A et al (2006) Linoleic acid derivative DCP-LA improves learning impairment in SAMP8. Neuroreport 17(1):105–108
Oakley H, Cole SL, Logan S, Maus E, Shao P, Craft J, Guillozet-Bongaarts A, Ohno M et al (2006) Intraneuronal β-amyloid aggregates, neurodegeneration, and neuron loss in transgenic mice with five familial Alzheimer's disease mutations: potential factors in amyloid plaque formation. J Neurosci 26(40):10129–10140
Rizzo G, Martinelli P, Manners D, Scaglione C, Tonon C, Cortelli P, Malucelli E, Capellari S et al (2008) Diffusion-weighted brain imaging study of patients with clinical diagnosis of corticobasal degeneration, progressive supranuclear palsy and Parkinson's disease. Brain 131(Pt10):2690–2700
Nussbaum JM, Seward ME, Bloom GS (2013) Alzheimer disease: a tale of two prions. Prion 7(1):14–19
Duyckaerts C (2011) Tau pathology in children and young adults: can you still be unconditionally baptist. Acta Neuropathol 121(2):145–147
Mukaetova-Ladinska EB, Garcia-Siera F, Hurt J, Gertz HJ, Xuereb JH, Hills R, Brayne C, Huppert FA et al (2000) Staging of cytoskeletal and β-amyloid changes in human isocortex reveals biphasic synaptic protein response during progression of Alzheimer's disease. Am J Pathol 157(2):623–636
Kanno T, Tsuchiya A, Shimizu T, Nakao S, Tanaka A, Nishizaki T (2013) Effects of newly synthesized DCP-LA-phospholipids on protein kinase C and protein phosphatases. Cell Physiol Biochem 31(4–5):555–564
Bayascas JR, Alessi DR (2005) Regulation of Akt/PKB Ser473 phosphorylation. Mol Cell 18(2):143–145
Gao T, Furnari F, Newton AC (2005) PHLPP: a phosphatase that directly dephosphorylates Akt, promotes apoptosis, and suppresses tumor growth. Mol Cell 18(1):13–24
Razmara M, Heldin CH, Lennartsson J (2013) Platelet-derived growth factor-induced Akt phosphorylation requires mTOR/Rictor and phospholipase C-γ1, whereas S6 phosphorylation depends on mTOR/Raptor and phospholipase D. Cell Commun Signal 11(1):3
Salminen A, Ojala J, Kaarniranta K, Hiltunen M, Soininen H (2011) Hsp90 regulates tau pathology through co-chaperone complexes in Alzheimer's disease. Prog Neurobiol 93(1):99–110
Wischik CM, Harrington CR, Storey JM (2014) Tau-aggregation inhibitor therapy for Alzheimer's disease. Biochem Pharmacol 88(4):529–539
Graham DL, Gray AJ, Joyce JA, Yu D, O'Moore J, Carlson GA, Shearman MS, Dellovade TL et al (2014) Increased O-GlcNAcylation reduces pathological tau without affecting its normal phosphorylation in a mouse model of tauopathy. Neuropharmacology 79:307–313
Ballatore C, Brunden KR, Piscitelli F, James MJ, Crowe A, Yao Y, Hyde E, Trojanowski JQ et al (2010) Discovery of brain-penetrant, orally bioavailable aminothienopyridazine inhibitors of tau aggregation. J Med Chem 53(9):3739–3747
Brunden KR, Ballatore C, Lee VM, Smith AB 3rd, Trojanowski JQ (2012) Brain-penetrant microtubule-stabilizing compounds as potential therapeutic agents for tauopathies. Biochem Soc Trans 40(4):661–666
Tanaka A, Nishizaki T (2003) The newly synthesized linoleic acid derivative FR236924 induces a long-lasting facilitation of hippocampal neurotransmission by targeting nicotinic acetylcholine receptors. Bioorg Med Chem Lett 13(6):1037–1040
Yamamoto S, Kanno T, Nagata T, Yaguchi T, Tanaka A, Nishizaki T (2005) The linoleic acid derivative FR236924 facilitates hippocampal synaptic transmission by enhancing activity of presynaptic α7 acetylcholine receptors on the glutamatergic terminals. Neuroscience 130(1):207–213
Kanno T, Tanaka A, Nishizaki T (2012) Linoleic acid derivative DCP-LA stimulates vesicular transport of α7 ACh receptors towards surface membrane. Cell Physiol Biochem 30(1):75–82
Kanno T, Tsuchiya A, Tanaka A, Nishizaki T (2013) The linoleic acid derivative DCP-LA increases membrane surface localization of the α7 ACh receptor in a protein 4.1N-dependent manner. Biochem J 450(2):303–309
Kanno T, Yaguchi T, Nagata T, Tanaka A, Nishizaki T (2009) DCP-LA stimulates AMPA receptor exocytosis through CaMKII activation due to PP-1 inhibition. J Cell Physiol 221(1):183–188
Conflict of interest
The authors state that there are no actual or potential conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
(PDF 198 kb)
Supplementary Fig. 2
(PDF 64 kb)
Supplementary Fig. 3
(PDF 68 kb)
Supplementary Fig. 4
(PDF 285 kb)
Supplementary Fig. 5
(PDF 135 kb)
Supplementary Fig. 6
(PDF 220 kb)
Supplementary Fig. 7
(PDF 81 kb)
Rights and permissions
About this article
Cite this article
Kanno, T., Tsuchiya, A., Tanaka, A. et al. Combination of PKCε Activation and PTP1B Inhibition Effectively Suppresses Aβ-Induced GSK-3β Activation and Tau Phosphorylation. Mol Neurobiol 53, 4787–4797 (2016). https://doi.org/10.1007/s12035-015-9405-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-015-9405-x