Abstract
Locally advanced breast cancer (LABC) remains a clinical challenge as the majority of patients with this diagnosis develop distant metastases despite appropriate therapy. We analyzed expression of steroid and growth hormone receptor genes as well as gene associated with metabolism of chemotherapeutic drugs in locally advanced breast cancer before and after neoadjuvant chemotherapy (NACT) to study whether there is a change in gene expression induced by chemotherapy and whether such changes are associated with tumor response or non-response. Fifty patients were included with locally advanced breast cancer treated with cyclophosphamide, adriamycin, 5-fluorouracil (CAF)-based neoadjuvant chemotherapy before surgery. Total RNA was extracted from 50 match samples of pre- and post-NACT tumor tissues. RNA expression levels of epidermal growth factor receptor family genes including EGFR, ERBB2, ERBB3, androgen receptor (AR), and multidrug-resistance gene 1 (MDR1) were determined by quantitative real-time reverse transcriptase-polymerase chain reaction. Responders show significantly high levels of pre-NACT AR gene expression (P = 0.016), which reduces following NACT (P = 0.008), and hence can serve as a useful tool for the prediction of the success of neoadjuvant chemotherapy in individual cancer patients with locally advanced breast carcinoma. Moreover, a significant post-therapeutic increase in the expression levels of EGFR and MDR1 gene in responders (P = 0.026 and P < 0.001) as well as in non-responders (P = 0.055, P = 0.001) suggests that expression of these genes changes during therapy but they do not have any impact on tumor response, whereas a post-therapeutic reduction was observed in AR in responders. This indicates an independent predictive role of AR with response to NACT.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In India, a majority of the patients (50–70%) present with locally advanced breast cancer [1].
The standard mode of management for locally advanced breast cancer is neoadjuvant chemotherapy (NACT) followed by surgery in the form of modified radical mastectomy and subsequently three more cycles of adjuvant chemotherapy. NACT facilitates local as well as distant control of the disease and provides an in vivo chemosensitivity test for a particular regime. It is vital to predict response to chemotherapy in order to tailor the regime in a particular patient for an optimum response and to avoid chemotoxicity in a non-responder. Various markers like p-glycoprotein, p53, MMR, apoptotic markers, and toxicity have been studied to assess and predict response to NACT [2, 3]. In some studies, it was found that patients whose tumors lacked ER had a higher response rate to chemotherapy [4–8]. Development of resistance to chemotherapeutic agents is a major and evolving problem, and the search for an ideal predictor of response is still on [9].
Several lines of evidence suggest that type 1 growth factor receptor family (EGFR, ERBB2, ERBB3) is involved in breast cancer development and progression [10]. In primary breast cancer, increased levels of EGFR [11] and ERBB2 [12] were first reported, several thousand cases have been studied, and the clinical significance of EGFR [13, 14] and ERBB2 [15, 16] has been extensively reviewed. The expression of both genes is associated with tumor aggressiveness and is related to a lower response to treatment. Recently, therapeutic approaches based on recombinant humanized monoclonal anti-ERBB2 antibodies (herceptin; Genentech, San Francisco, CA) have been developed [17]. As demonstrated by clinical trials [18], these antibodies are well tolerated and clinically active in patients with metastatic breast cancer overexpressing ERBB2 and result in an increase in the objective clinical response rates when used in combination with chemotherapy. One pilot study described the use of pre-operative trastuzumab and paclitaxel followed by doxorubicin and cyclophosphamide in women with HER-2-positive stage II and III breast cancers [19]. ERBB2 amplification with enhanced protein expression was noted in approximately one-third of invasive human breast cancers [20–25], but until now, its association with classical prognostic factors and with clinical outcome has been poorly documented, and the results are somewhat controversial. Overexpression of ERBB3 is also frequently reported in ERBB2-altered breast cancers [26]. Human breast cancer cell lines commonly co-overexpress both ERBB2 and ERBB3, further supporting their role in breast carcinogenesis [27, 28].
The androgen receptor (AR) is detectable in the majority of tumor specimens from patients undergoing mastectomy for breast cancer [29]. AR expression in breast cancer tissue samples has been associated with an improved response to hormone therapy and longer survival in patients with breast cancer [30, 31]. Studies by Tilley’s group indicate that reduced levels of AR or impaired function of AR contributes to the failure of breast carcinoma cells to respond to progestin medroxyprogesterone acetate (MPA) [32, 33], which has been used as a second-line hormonal therapy for metastatic breast cancer. Multidrug resistance (MDR1) is a significant challenge in the treatment of breast cancer.
P-gp, the product of MDR1, was the first anticancer drug pump to be identified [34]. The MDR phenotype conferred by overexpression of MDR1 is characterized by resistance to structurally unrelated cytotoxic agents, including anthracyclines (doxorubicin and epirubicin are among the most effective anticancer drugs used in the treatment of breast cancer), epipodophyllotoxins, Vinca alkaloids, and taxanes [35]. Thus, increased expression of MDR1 is likely to contribute to clinical drug resistance in breast cancer.
Hence, this study aimed to define mRNA expression level of growth factor receptor genes (EGFR, ERBB2, ERBB3), hormone receptor gene AR, and multidrug-resistant gene MDR1 and their association with response to NACT in locally advanced breast cancer to identify possible candidate gene(s) that may predict response to treatment regimen and help in assessing the successful drug-based therapy.
Materials and methods
Tissue samples
A total of 80 patients diagnosed with locally advanced breast cancer were enrolled who underwent neoadjuvant chemotherapy between 2005 and 2009 in the Department of Surgery, Safdarjung Hospital, New Delhi. Eligibility criteria included histologically confirmed LABC with measurable locally advanced cases where paired tissue is available pre- and post-NACT. Informed consent was obtained from all participating patients, and the study was carried out with the approval of Ethical Review Committee of Safdarjung Hospital, New Delhi. From the cohort of 80 patients, in 14 cases, post-therapy tissues was not available, and 16 samples did not contain enough tumor tissue for accurate measurement. Therefore, a total of 50 patients were included in the present study.
All 50 patients received three courses of CAF (cyclophosphamide, adriamycin, and 5-fluorouracil) combination therapy. Both NACT biopsy and surgical resection material (frozen tissue) were collected for diagnosis and assessment of predictive markers. All the tissue samples were snap-frozen in liquid nitrogen till further investigation. The age of patients ranged between 26 and 65 years with a mean age of 44 years. Of total 50 cases, 38% were pre-menopausal. Patient’s characteristics are shown in Table 1.
Treatment regimen and clinical response criteria
Neoadjuvant chemotherapy (NACT) followed by breast-conserving surgery has become an acceptable option for patients with locally advanced breast cancer [36, 37]. Treatment with neoadjuvant chemotherapy consisted of the classical cyclophosphamide, adriamycin, and 5-fluorouracil (CAF) regimen (cyclophosphamide 600 mg/m2, adriamycin 50 mg/m2, 5-fluorouracil 600 mg/m2) in standard doses on the basis of body surface area. At least 3 cycles of NACT at 3-weekly intervals are administrated to the patients. Surgery is usually done after 3 weeks of the last cycles of NACT, and the patients were assessed both clinically and by USG/MRI for down-staging of the tumor in terms of tumor size and axillary lymph node status. While some patients show a partial or complete response to the above drug in the form of reduction in breast tumor size or down-staging of axillary lymph node status, others fail to do so. Thus, patients have been grouped into responders and non-responders. Clinical responders were defined as patients with a complete (CR) or partial response (PR) [CR: complete resolution of tumor, PR > 50% regression in maximum diameter of initial tumor] after 3 cycles of NACT. Non-responders are patients with a minimal response (MR ≤ 50% regression in maximum diameter of initial tumor), no change (NC), or local progression [2, 3, 38].
Real-time quantitative reverse transcriptase PCR
Total RNA was extracted from histologically confirmed breast tumors using TRIzol reagent (Invitrogen, CA, USA) in accordance with the manufacturer’s instructions. The quality and quantity of the RNA samples were determined using Agilent 2100 Bioanalyzer (Agilent Technologies, Waldbronn, Germany) and Nano-drop ND-1000 Full—spectrum UV/Vis spectrophotometer. Samples having RNA integrity number (RIN) 6 and above were selected for real-time experiment (Fig. 1).
RNA was reverse-transcribed in a final volume of 20 μl containing 2 μl 10X RT buffer, 0.8 μl 25X dNTP, 2 μl 10X random primer, 1 μl multiscribe RT, and 2 μg total RNA using high-capacity cDNA archive kit (Applied Biosystems, Foster, CA, USA). The samples were incubated at 25°C for 10 min, 37°C for 2 h, and reverse transcriptase was inactivated by heating at 95°C for 5 min and cooling to 4°C for 5 min.
Quantitative real-time PCR was performed using an ABI 7000 Sequence Detection System (Applied Biosystems, Foster, CA, USA) with cDNA as template using TaqMan probe Assay. Primers and Probe for all target genes and an internal control gene TATA box–binding protein (TBP) were designed by Applied Biosystems, (Foster city, CA, USA). A singleplex reaction mix was prepared according to the manufacturer’s protocol of Assays-on-Demand Gene Expression products and included 10 μl of TaqMan Universal PCR Master Mix, 1 μl of 20X Assays-on-Demand Gene Expression Assay Mix (all gene expression assays have a carboxyfluorescein reporter dye at the 5′-end of the TaqMan minor groove binder probe and a non-fluorescent quencher at the 3′-end of the probe), and 4 μl of cDNA(50 ng) diluted in Rnase-free water, in a total 20 μl volume. Thermal cycling conditions included an initial denaturation step at 95°C for 10 min, 40 cycles at 95°C for 15 s, and 60°C for 1 min.
The mean expression level of target gene (EGFR, ERBB2, ERBB3, AR, and MDR1) was calculated for breast tissue normalized to a housekeeping gene TBP (TATA box–binding protein). The average CT was calculated for both gene of interest and housekeeping gene (TBP). The 2−ΔΔCT method was used to calculate relative changes in gene expression determined from real-time quantitative PCR experiments. The relative gene expression level was also normalized to a calibrator consisting of a pool of normal breast tissue specimens. For this, specimen of adjacent normal breast tissue from 12 of the breast cancer patients was used as a source of normal RNA. Final results were articulated as n-fold differences in EGFR, ERBB2, ERBB3, MDR1, and AR gene expression relative to TBP gene and normal breast tissue (the calibrator).
Statistical analysis
Statistical analysis was done using non-parametric methods. Comparisons between the responders and non-responders were made using Mann–Whitney U test, and the difference in mRNA expression level of each gene in pre- and post- NACT condition was determined using Wilcoxon signed ranks test. The correlation matrix denotes the correlation between the considered biomarkers in three groups (overall, responders, and non-responders). The two-sided P < 0.05 was considered statistically significant. All of the statistical analysis was done using the SPSS version 17.0.
Results
In the present study, we analyzed the expression of type 1 growth factor receptor genes, multidrug-resistant gene, and androgen receptor gene in pre-NACT biopsies of locally advanced breast cancer and correlated their expression with response to neoadjuvant chemotherapy. Since histopathological response evaluation after neoadjuvant therapy for locally advanced breast cancer is known to be highly inconsistent, gene expressions was correlated with clinically determined tumor regression (reduction in tumor size and lymph node involvement). According to clinical criteria, 74.0% (37/50) cases were responders, and 26.0% (13/50) cases were non-responders.
The expression of the 5 genes was studied in all 50 paired (pre- and post-NACT) tissue samples. Comparison of pre- and post-NACT mRNA expression values showed decrease in AR levels in 58.0% cases and in ERBB2 level in 60% cases. On the contrary, EGFR level was found increased in 66.0% cases, ERBB3 level in 64.0% cases, and MDR1 level in 88.0% cases.
Gene expression levels in pre- and post-NACT samples
The expression of AR mRNA level was found significantly high in pre-NACT samples in responders compared with non-responders, and the outcome was statistically significant (P = 0.016, Mann–Whitney U test; Table 2). However, no significant difference in expression levels of EGFR, ERBB2, ERBB3, and MDR1 genes in pre-NACT samples was observed among responders when compared with non-responders.
On the contrary, the expression level of genes in post-NACT samples among responders and non-responders did not show any significant difference (Table 3).
Correlation of alteration in gene expression levels with drug response
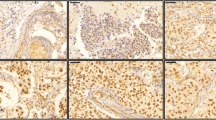
In responders, down-regulation of AR (72.9%; Fig. 2) and ERBB2 (70.2%) expression and up-regulation of EGFR (62.2%) and MDR1 (83.7%) expression were found significant (P = 0.0008, P = 0.041, P = 0.026 and P < 0.001, Wilcoxon signed ranks test; Table 4).
In non-responders, up-regulation of EGFR (76.9%), MDR1 (100%), and AR (84.6%) was statistically significant (P = 0.055, P = 0.001, and P = 0.033, Wilcoxon signed ranks test) (Table 5).
In addition, we applied correlation matrix test for all target genes in responders and non-responders, and the correlation between ERBB2 and AR among non-responders was found significant in pre-NACT tumors. In non-responders, the expression of both appears to be up-regulated after neoadjuvant chemotherapy. However, no correlation was observed in other target genes (Supplementary Table).
Discussion
There are several attempts to envisage the outcome of neoadjuvant treatment discriminating potential responders from non-responders to avoid severe side effects of an unnecessary therapy. The present study was done to understand the functional role and significance of the growth factor receptor genes, steroid receptor gene, and drug-resistance gene with the clinical response to NACT for locally advanced breast cancer cases and to find out how these molecular biomarkers play potential role to predict therapy response.
Over the past few years, cyclophosphamide, adriamycin, and 5-fluorouracil (CAF) are given as a common combination of drugs given to women with locally advanced breast cancer [39–41]. Most studies have solely used immunohistochemical (IHC) methods to determine expression of various markers in clinical samples, with considerable variation in results. Correctly defining a positive IHC result may well be one of the pitfalls in such expression studies. The study of gene expressions from tissue collected from patients before and after neoadjuvant treatment provides a lot of keys to decipher the signaling pathways and prediction of the clinical outcome of therapy. It gives more prognostic information to clinicians for better management of the disease.
In the present study, comparison of the mRNA expression level of AR gene in responders and non-responders in pre-NACT patients showed that tumors of responders had the higher AR mRNA expression levels in pre-NACT condition (P = 0.016). On the other hand, the rest of the markers (EGFR, ERBB2, ERBB3, and MDR1) did not show any differential gene expression when analyzed between responders and non-responders in patients under pre-NACT condition. The higher pre-therapeutic AR expression may have a stronger impact on drug response.
Since, chemotherapeutic agents may alter the expression levels of the genes during the course of chemotherapy and thereby may determine tumor sensitivity or resistance [42], the next analysis was done to compare expression levels of each gene before chemotherapy with expression levels after chemotherapy among responders and non-responders to detect possible therapy induced changes. The present study showed a significant increase in expression levels of EGFR and MDR1 during therapy in both responders (P = 0.026, P = <0.001) and non-responders (P = 0.055, P = 0.001). High expression of EGFR has been reported in a variety of epithelial tumors [43], whereas the overexpression of MDR1 gene is known to result in drug resistance in cancer cells. There are many proposed mechanisms, including gene amplification, which may change the expression level of a particular gene. Based on this observation, one can speculate that pre-therapeutic expression of these genes may have an impact on expression changes during therapy but not on drug response. Sequential assessment during chemotherapy of MDR1 mRNA levels in 73 breast carcinoma patients enabled prediction of clinical response to adriyamicin/doxorubicin [44]. Others, however, have found no such association or suggest that MDR1 expression is merely a measurement of advanced disease rather than an indicator of chemotherapy resistance [45, 46].
In contrast, the mRNA expression of AR in responders got reduced after neoadjuvant chemotherapy, and the difference was statistically significant (P = 0.008), whereas in non-responders, a significant up-regulation of AR expression was observed (P = 0.033).
The reason for reduction in AR mRNA expression in tumors among responders after neoadjuvant chemotherapy could be important cellular processes, e.g., DNA repair and apoptosis, which often occur within up to 48 h after chemotherapy exposure [47–50].
The down-regulation of ERBB2 expression from pre- to post-NACT in responders was also found significant (P = 0.041). In pre-NACT condition, the expression of ERBB2 gene was found higher in responders when compared with non-responders, whereas it was vice versa in case of post-NACT condition. However, ERBB2 or HER-2 oncogene is overexpressed in approximately 30% of human breast cancer specimens and is associated with a poor outcome in many studies [51, 52]. Recent data suggest that ERBB2 amplification and overexpression are associated with improved outcome after doxorubicin-based therapy (CAF) as compared with alkylator-based therapy [CMF and PF] [53, 54]. This has led to the speculation that ERBB2 confers sensitivity to doxorubicin and resistance to alkylating agents. According to a previously published report, in vitro data have shown that activation of the ERBB2, ERBB3, and ERBB4 receptors is associated with an increase in the DNA-modifying enzyme, topo IIa, which is accompanied by increased sensitivity to doxorubicin but resistance to an alkylator, cisplatin [55]. However, this finding needs validation in larger sample size.
Applying correlation matrix, a significant correlation was observed between ERBB3 and AR among non-responders in pre-NACT form (0.749) (P = <0.01). In non-responders, the expression of both appears to be up-regulated after neoadjuvant chemotherapy. Comparatively, lower expression of ERBB3 in pre-NACT condition may down-regulate the expression of AR in non-responders. ERBB3 has been reported to interact with ERBB3-binding protein 1 (Ebp1), a protein that interacts with the androgen receptor (AR) and suppresses AR-mediated gene transcription [56]. The ERBB2/ERBB3 pathway regulates AR by stabilizing AR protein levels and optimizing binding of AR to promoter/enhancer regions of androgen-regulated genes [57].
In conclusion, responders show significantly high levels of AR gene expression under pre-NACT condition which reduces following NACT, and this may be useful for the prediction of the success of neoadjuvant chemotherapy in individual cancer patients with locally advanced breast carcinoma. In pre-NACT condition, the expression of ERBB2 gene was found higher in responders compared with non-responders, suggesting its association with improved outcome after doxorubicin-based therapy. In other genes like EGFR and MDR1, the expression level increased significantly in both responders and non-responders after NACT and hence refute their predictive role for response. The major limitation of the present study is small sample size due to unavailability of paired tissue samples in few cases, but at the same time, the study might have a substantial role in finding a suitable predictive marker that can envisage the response to neoadjuvant chemotherapy for patients with locally advanced breast cancer.
References
Chopra R. The Indian scene. J Clin Oncol. 2001;19:S106–11.
Chintamani SV, Singh JP, Lyall A, Saxena S, Bansal A. Is drug-induced toxicity a good predictor of response to neo-adjuvant chemotherapy in patients with breast cancer?–a prospective clinical study. BMC Cancer 2004;4:48.
Chintamani SV, Singh JP, Mittal MK, Saxena S, Bansal A, Bhatia A, Kulshreshtha P. Role of p-glycoprotein expression in predicting response to neoadjuvantchemotherapy in breast cancer-a prospective clinical study. World J Surg Oncol. 2005;3:61.
Wallner J, Depisch D, Hopfner M, Haider K, Spona J, Ludwig H, Pirker R. MDR1 gene expression and prognostic factors in primary breast carcinomas. Eur J Cancer. 1991;27:1352–5.
Dixon AR, Bell J, Ellis IO, Elston CW, Blamey RW. P-glycoprotein expression is locally advanced breast cancer treated by neoadjuvant chemotherapy. Br J Cancer. 1992;66:537–41.
De Lena M, Zucali R, Viganotti G, Valagussa P, Bonadonna G. Combination chemotherapy, radiotherapy approach in locally advanced breast cancer. Cancer Chemother Pharmacol. 1978;1:53–9.
Sataloff DM, Mason BA, Prestipino AJ, Seinige UL, Lieber CP, Baloch Z. Pathological response to induction chemotherapy in locally advanced carcinoma of breast, a determinant of out come. J Am Coll Surg. 1995;180:297–306.
Seymour L, Bezwoda WR, Dansey RD. p-Glycoprotein immunostaining correlates with ER and with high Ki67 expression but fails to predict anthracycline resistance in patients with advanced breast cancer. Breast Cancer Res Treat. 1995;36:61–9.
Charfare H, Limongelli S, Purushotham AD. Neo-adjuvant chemotherapy in breast cancer. Br J Surg. 2005;92:14–23.
Mendelsohn J. Epidermal growth factor receptor inhibition by a monoclonal antibody as anticancer therapy. Clin Cancer Res. 1997;3:2703–7.
Sainsbury JRC, Farndon JR, Needham GK, Malcolm AJ, Harris AL. Epidermal growth factor receptor status as predictor of early recurrence and death from breast cancer. Lancet 1987;1398–1402.
Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire L. Human breast cancer. Correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science (Washington DC) 1987;235:177–182.
Klijn JGM, Berns PMJJ, Schmitz PIM, Foekens JA. The clinical significance of epidermal growth factor receptor (EGF-R) in human breast cancer: a review on 5232 patients. Endocr Rev. 1992;13:3–17.
Bhargava R, Gerald WL, Li AR, Pan Q, Lal P, Ladanyi M, et al. EGFR gene amplification in breast cancer: correlation with epidermal growth factor receptor mRNA and protein expression and HER-2 status and absence of EGFR-activating mutations. Modern Pathol. 2005;18:1027–33.
Re’villion F, Bonneterre J, Peyrat JP. ERBB2 oncogene in human breast cancer and its clinical significance. Eur J Cancer 1998;34:791–808.
Ross JS, Fletcher JA, Linette GP, Stec J, Clark E, Ayers M, et al. The Her-2/neu gene and protein in breast cancer : biomarker and target of therapy. Oncologist. 2003;8:307–25.
Tokuda Y, Okumura A, Ohta M, Kubota M, Ohnishi Y, Shmamura K, et al. A humanized Anti-c-erbB-2 monoclonal antibody for the treatment of breast cancer. Breast cancer. 1997;4:269–72.
Shak S. Overview of the trastuzumab (Herceptin) anti-HER2 monoclonal antibody clinical program in HER2-overexpressing metastatic breast cancer. Semin. Oncol. 1999;26:71–77.
Burstein HJ, Harris LN, Gelman R, et al. Preoperative therapy with trastuzumab and paclitaxel followed by sequential adjuvant doxorubicin/cyclophosphamide for HER2 over-expressing stage II or III breast cancer: a pilot study. J Clin Oncol. 2003;21:46–53.
Slamon DJ, Godolphin W, Jones LA, Holt JA, Wong SG, Keith DE, et al. Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science. 1989;244:707–12.
Paterson MC, Dietrich KD, Danyluk J, Paterson AH, Lees AW, Jamil N, et al. Correlation between c-erbB-2 amplification and risk of recurrent disease in node-negative breast cancer. Cancer Res. 1991;51:556–67.
Gullick WJ, Love SB, Wright C, Barnes DM, Gusterson B, Harris AL, et al. c-erbB-2 protein overexpression in breast cancer is a risk factor in patients with involved and uninvolved lymph nodes. Br J Cancer. 1991;63:434–8.
Andrulis IL, Bull SB, Blackstein ME, Sutherland D, Mak C, Sidlofsky S, Pritzker KP, Hartwick RW, Hanna W, Lickley L, et al. neu/erbB-2 amplification identifies a poor-prognosis group of women with node-negative breast cancer. Toronto Breast Cancer Study Group. J Clin Oncol 1998;16:1340–1349.
Thor AD, Liu S, Edgerton S, Moore D 2nd, Kasowitz KM, Benz CC, et al. Activation (tyrosine phosphorylation) of ErbB-2 (HER-2/neu): a study of incidence and correlation with outcome in breast cancer. J Clin Oncol. 2000;18:3230–9.
Penuel E, Akita RW, Sliwkowski MX. Identification of a region within the ErbB2/HER2 intracellular domain that is necessary for ligand-independent association. J Biol Chem. 2002;277:28468–73.
Gullick WJ. Prevalence of aberrant expression of epidermal growth factor receptor in human cancer cells. Br Med Bull. 1991;47:87–98.
deFazio A, Chiew YE, Sini RL, Janes PW, Sutherland RL. Expression of c-erbB receptors, heregulin and oestrogen receptor in human breast cell lines. Int J Cancer. 2000;87:487–98.
Holbro T, Beerli RR, Maurer F, Koziczak M, Barbas CF 3rd, Hynes NE. The ErbB2/ErbB3 heterodimer functions as an oncogenic unit: ErbB2 requires ErbB3 to drive breast tumor cell proliferation. Proc Natl Acad Sci USA. 2003;100(15):8933–8.
Soreide JA, Lea OA, Varhaug JE, Skarstein A, Kvinnsland S. Androgen receptors in operable breast cancer: relation to other steroid hormone receptors, correlations to prognostic factors and predictive value for effect of adjuvant tamoxifen treatment. Eur J Surg Oncol. 1992;18:112–8.
Szelei J, Jimenez J, Sotto AM, Luizzi MF, Sonnenschein C. Androgen induced inhibition of proliferation in human breast cancer MCF7 cells transfected with androgen receptor. Endocrinology. 1997;138:1406–12.
Bryan RM, Mercer RJ, Bennett RC, Rennie GC, Lie TH, Morgan FJ. Androgen receptors in breast cancer. Cancer. 1984;54:2436–40.
Buchanan G, Birrell SN, Peters AA, Bianco-Miotto T, Ramsay K, Cops EJ, et al. Decreased androgen receptor levels and receptor function in breast cancer contribute to the failure of response to medroxyprogesterone acetate. Cancer Res. 2005;65(18):8487–96.
Birrell SN, Roder DM, Horsfall DJ, Bentel JM, Tilley WD. Medroxyprogesterone acetate therapy in advanced breast cancer: the predictive value of androgen receptor expression. J Clin Oncol. 1995;13:1572–7.
Juliano RL, Ling V. A surface glycoprotein modulating drug permeability in Chinese hamster ovary cell mutants. Biochim Biophys Acta. 1976;455:152–62.
Nooter K, Herweijer H. Multidrug resistance (mdr) genes in human cancer. Br J Cancer. 1991;63:663–9.
Inaji H, Komoike Y, Motomura K, Kasugai T, Sawai Y, Koizumi M, et al. Breast-conserving treatment after neoadjuvant chemotherapy in large breast cancer. Breast cancer. 2002;9:20–5.
Tiezzi DG, Andrade JM, Marana HR, Zola FE, Peria FM. Breast conserving surgery after neoadjuvant therapy for large primary breast cancer. Eur J Surg Oncol. 2008;34:863–7.
Zhang F, Yang Y, Smith T, Kau SW, McConathy JM, Esteva FJ, et al. Correlation between HER-2 expression and response to neoadjuvant chemotherapy with 5-fluorouracil, doxorubicin, and cyclophosphamide in patients with breast carcinoma. Cancer. 2003;97:1758–65.
Favret AM, Carlson RW, Goffinet DR, Jeffrey SS, Dirbas FM, Stockdale FE. Locally advanced breast cancer. Is surgery necessary? Breast J. 2001;7:131–7.
Davidson K, Cameron DA, Dillon P, Bowman A, Stewart M, Leonard RCF. Locally advanced breast cancer: the outcome of primary polychemotherapy based on infusional 5 fluorouracil. The Breast. 1999;8:110–5.
Fukuda M, Yamaguchi S, Ohta Y, Nakayama Y, Ogata H, Shimizu K, et al. Combination therapy for advanced breast cancer: cyclophosphamide, Doxorubicin, UFT, and Tamoxifen. Oncology. 1999;7:77–81.
Schneider S, Uchida K, Brabender J, et al. Downregulation of TS, DPD, ERCC1, GST-Pi, EGFR, and HER2 gene expression after neoadjuvant three-modality treatment in patients with esophageal cancer. J Am Coll Surg. 2005;200:336–44.
Salomon DS, Brandt R, Ciardiello F, Normanno N. Epidermal growth factor-related peptides and their receptors in human malignancies. Crit Rev Oncol Hematol. 1995;19:183–232.
Chevillard S, Pouillart P, Beldjord C, Asselain B, Beuzeboc P, Magdelénat H, et al. Sequential assessment of multidrug resistance phenotype and measurement of S-phase fraction as predictive markers of breast cancer response to neoadjuvant chemotherapy. Cancer 1996;77(2):292–300.
Liu XE, Sun XD, Wu JM. Effect of NACT on MDR1 and MRP gene expression in primary breast cancer. Chin J Cancer Res. 2004;16(1):51–4.
Seymour L, Bezwoda WR, Dansey RD. P-glycoprotein immunostaining correlates with ER and with high Ki67 expression but fails to predict anthracycline resistance in patients with advanced breast cancer. Breast Cancer Res Treat. 1995;36(1):61–9.
Parton M, Krajewski S, Smith I, et al. Coordinate expression of apoptosis-associated proteins in human breast cancer before and during chemotherapy. Clin Cancer Res. 2002;8:2100–8.
Chang J, Powles TJ, Allred DC, et al. Biologic markers as predictors of clinical outcome from systemic therapy for primary operable breast cancer. J Clin Oncol. 1999;17:3058–63.
Ellis PA, Smith IE, Detre S, et al. Reduced apoptosis and proliferation and increased Bcl-2 in residual breast cancer following preoperative chemotherapy. Breast Cancer Res Treat. 1998;48:107–16.
Chang J, Ormerod M, Powles TJ, Allred DC, Ashley SE, Dowsett M. Apoptosis and proliferation as predictors of chemotherapy response in patients with breast carcinoma. Cancer (Phila). 2000;89:2145–52.
Borg A, Baldetorp B, Ferno M, Killander D, Olsson H, Sigurdsson H. ErbB2 amplification in breast cancer with a high rate of proliferation. Oncogene. 1991;6:137–43.
Marks JR, Humphrey PA, Wu K, Berry D, Bandarenko N, Kerns BJ, et al. Overexpression of p53 and HER-2/neu proteins as prognostic markers in early stage breast cancer. Ann Surg. 1994;219:332–41.
Paik S, Bryant J, Tan-Chiu E, Yothers G, Park C, Wickerham DL, et al. HER2 and choice of adjuvant chemotherapy for invasive breast cancer: NSABP B-15. J Natl Cancer Inst (Bethesda). 2000;92:1991–8.
Paik S, Bryant J, Park C, Fisher B, Tan-Chiu E, Hyams D, et al. erbB-2 and response to doxorubicin in patients with axillary lymph node-positive, hormone receptor-negative breast cancer. J Natl CancerInst (Bethesda). 1998;90:1361–70.
Harris L, Tang C, Yang C, Harris A, Lupu R. Induction of chemotherapy sensitivity in MCF-7 breast cancer cells by heregulin. Clin Cancer Res. 1998;4:1005–12.
Zhang Y, Wang XW, Jelovac D, et al. The ErbB3-binding protein Ebp1 suppresses androgen receptor-mediated gene transcription and tumorigenesis of prostate cancer cells. Proc Natl Acad Sci USA. 2005;102:9890–5.
Mellinghoff IK, Vivanco I, Kwon A, Tran C, Wongvipat J, Sawyers CL. HER2/neu kinase-dependent modulation of androgen receptor function through effects on DNA binding and stability. Cancer Cell. 2004;6:517–27.
Acknowledgments
We thank the patients and their family members without whose cooperation this study would not have been possible. Financial assistance from Indian Council of Medical Research is gratefully acknowledged.
Conflict of interest
All authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Singh, L.C., Chakraborty, A., Mishra, A.K. et al. Study on predictive role of AR and EGFR family genes with response to neoadjuvant chemotherapy in locally advanced breast cancer in Indian women. Med Oncol 29, 539–546 (2012). https://doi.org/10.1007/s12032-011-9952-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12032-011-9952-6