Abstract
Dicer, the key enzyme in the RNAi pathway, is misregulated in tumor tissues. The altered expression of Dicer is associated with clinical characteristics in patients with cancer. Liver carcinoma and adjacent non-neoplastic tissues were obtained from 36 patients with hepatocellular carcinoma (HCC) undergoing surgery. Expressions of Dicer mRNA were evaluated using the Real-time reverse transcription-PCR in 36 liver carcinoma tissues and 36 adjacent histologically non-cancerous liver tissues. Dicer mRNA levels were evaluated in relation to age, sex, tumor number, tumor size, tumor stage, and distant metastasis. Dicer mRNA level was significantly lower in malignant tissues than in the corresponding non-neoplastic tissues in 34 of the 36 patients with HCC (94.4%). The Dicer expression level was not associated with clinical characteristics, including age, sex, tumor number, tumor size, tumor stage, or distant metastasis in HCC cases. These results demonstrate that Dicer is significantly down-regulated in HCC, suggesting that reduced expression of Dicer may play an important role during the process of hepatocarcinogenesis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hepatocellular carcinoma (HCC) is one of the leading causes of cancer-related death [1, 2]. A total of 80–90% of the HCC cases in China, India, Korea, Singapore, and Vietnam are associated with Hepatitis B virus (HBV) infection [3]. Mutation, amplification, and epigenetic changes in various genes have been identified in liver cancers, suggesting that these genes are involved in hepatocarcinogenesis [4, 5]. In addition, emerging evidence suggests that components of the RNA interference (RNAi) pathway and miRNAs may play important roles in the carcinogenesis of HCC [6–16].
Dicer, the key enzyme in the RNAi pathway, is upregulated in prostate adenocarcinoma [17], ovarian serous carcinomas [18], pleomorphic adenomas of the salivary gland [19], and acute myeloid leukemia (AML) [20]. Reduced expression of Dicer is associated with poor prognosis in ovarian carcinomas and patients with lung cancer [21, 22]. However, the Dicer level in patients with AML is not associated with clinical outcomes [20]. Disruption of Dicer promotes hepatocarcinogenesis [16]. In this study, we compared the Dicer mRNA between tumor tissues and corresponding adjacent non-neoplastic tissues from patients with HCC and investigated whether the level of Dicer mRNA is associated with clinical parameters.
Materials and methods
Patients and tissues
Liver carcinoma and adjacent non-neoplastic tissues were obtained from 36 patients with HCC undergoing surgery in Southwest Hospital (Chongqing, China) between April 2008 and January 2009. None of the patients had received chemotherapy before operation. Tissue samples were immediately frozen in liquid nitrogen and stored at −80° until use. All patients gave their informed consent, and the study was approved by the ethics and scientific committees of Southwest Hospital. The tumor types and stages were determined according to the WHO classification. All HCC cases were confirmed to be HCC by clinical pathology. The clinicopathological characteristics are summarized in Table 1.
RNA isolation and quantitative real-time RT-PCR
Total RNA was prepared using TRIzol (Invitrogen) and reverse-transcribed using MML-V reverse transcriptase (Promega) according to the manufacturer’s protocol. Samples prepared without reverse transcription served as negative control templates. SYBR Green(ABI) PCR was performed in triplicate using the ABI PRISM 7300 Sequence Detection System. All samples were normalized to the signal generated from GAPDH. Primer sequences are presented in Table 2.
Western blot analysis
Total protein content was measured using the Bradford protein assay. Samples were denatured at 100°C for 5 min. Equal amounts of total protein were loaded to each well for electrophoresis in SDS–polyacrylamide gels and then transferred to polyvinylidene fluoride microporous membranes (Millipore). Membranes were then incubated with primary antibody followed by incubation with horseradish peroxidase–linked secondary antibodies. The primary antibodies included anti-Dicer (ab14601; Abcam) and anti-GAPDH (KC-5G4; Kangchen Biotech).
Statistical analysis
Statistical analysis was performed with SPSS 11.5. Statistical comparisons for significance were made with Wilcoxon signed-rank test for paired samples and with Mann–Whitney U Kruskal–Wallis test for unpaired samples. The correlation between continuous variables was examined by means of Spearman’s rank-order coefficients. A level of P < 0.05 was considered significant.
Results
Expression of Dicer mRNA in cancerous liver tissues and adjacent non-neoplastic liver tissues of HCC cases
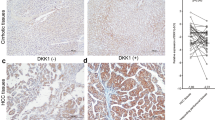
The Dicer mRNA was quantified by real-time RT-PCR in paired samples of tumor tissues and adjacent non-tumor tissues from 36 patients with HCC. The Dicer mRNA level was normalized to the level of GAPDH mRNA. In contrast to prostate adenocarcinoma [17], lung adenocarcinoma [23], ovarian serous carcinomas [18], and AML [20], where Dicer is upregulated, the Dicer mRNA level was significantly lower in malignant tissues than in the corresponding non-neoplastic tissues in 34 of the 36 patients with HCC (94.4%) (Table 1; Fig. 1). To determine whether the decreased Dicer mRNA level was mirrored by reduced protein expression, western blot was performed. As expected, our data indicated that Dicer protein was down-regulated in most of the malignant tissues. The expressions of Dicer protein from a typical case are indicated in Fig. 2.
Relationship between Dicer mRNA level and the clinical parameters in patients with HCC
Dicer expression level is associated with clinicopathological features, including clinical outcomes, in ovarian carcinoma and patients with lung cancer [12–23]. Therefore, we investigated whether the expression level of Dicer is associated with any clinicopathologic characteristics of HCC. Statistical analysis indicated that there was no significant association between Dicer expression level and clinical characteristics, including age, sex, tumor number, tumor size, tumor stage, and distant metastasis (Table 3; Figs. 3, 4).
Boxplot graphs of correlation between clinical features and relative Dicer mRNA levels in patients with HCC. The relative Dicer mRNA levels of patients with HCC have no significant correlation with sex (a), tumor numbers (b), metastasis (c), stage (d), and tumor size (cutpoint: 10 cm) (e). ★Mann–Whitney U Test, §Kruskal–Wallis Test
Discussion
Dicer is upregulated in prostate adenocarcinoma [17], ovarian serous carcinomas [18], pleomorphic adenomas of the salivary gland [19], and acute myeloid leukemia (AML) [20]. Here, we found that Dicer is down-regulated in hepatocellular carcinoma. Reduced expression of Dicer is associated with poor prognosis in ovarian carcinomas and patients with lung cancer [21, 22]. Prognosis of patients with HCC is poor [24], suggesting that reduced Dicer expression is associated with the poor outcomes of patients with HCC. However, Martin et al. [20] found that Dicer level is not associated with clinical outcomes in patients with AML, suggesting that reduced Dicer expression may not be related to the poor prognosis of patients with HCC.
Altered Dicer expression is associated with the progress of lung adenocarcinoma. In the proposed multistep carcinogenesis model, peripheral adenocarcinoma of the lung develops from a precursor lesion referred to as atypical adenomatous hyperplasia (AAH). AAH transforms into non-mucinous bronchioalveolar carcinoma (BAC), which progresses into invasive adenocarcinoma. Dicer is upregulated in AAH and BAC and down-regulated in invasive lung adenocarcinoma [23]. We found that the Dicer mRNA level is not associated with the stage of HCC. However, we cannot rule out the possibility that Dicer expression is a dynamic progress during hepatocarcinogenesis. It is possible that Dicer is upregulated in HCC at the very early stage during hepatocarcinogenesis.
Approximately 50% of all annotated human miRNAs are located in areas of the genome associated with cancer or “fragile sites” [25], and thus, miRNAs might have a crucial function in cancer progression. In fact, increasing evidence shows that expression of miRNAs is down-regulated in most of the human cancer [26, 27]. Dicer is essential for miRNA biogenesis; loss of Dicer in mice disrupts embryonic stem-cell differentiation and is lethal during early development. Low levels of Dicer mRNA also affect normal cellular development and differentiation [25–28]. Disruption of Dicer1 promotes hepatocarcinogenesis [16]. Furthermore, abnormalities in the copy number of the Dicer gene have been found in human melanoma, breast, and ovarian cancers [29]. It is therefore possible that the abnormal miRNA expression observed in HCC [6–15] is secondary to defective RNA silencing machinery and that decreased Dicer expression is a cause rather than a consequence of HCC.
Conclusions
Our data demonstrate that Dicer is significantly down-regulated in HCC, suggesting that reduced expression of Dicer may play an important role during the process of hepatocarcinogenesis.
References
Gallinger S, Langer B. Primary and secondary hepatic malignancies. Curr Opin Gen Surg. 1993;1:257–64.
Fong Y, Sun RL, Jarnagin W, et al. An analysis of 412 cases of hepatocellular carcinoma at a Western center. Ann Surg. 1999;229:790–800.
Lavanchy D. Worldwide epidemiology of HBV infection, disease burden, and vaccine prevention. J Clin Virol. 2005;34(Suppl 1):S1–3.
Herath NI, Leggett BA, MacDonald GA. Review of genetic and epigenetic alterations in hepatocarcinogenesis. J Gastroenterol Hepatol. 2006;21:15–21.
Feitelson MA, Sun B, Satiroglu Tufan NL, et al. Genetic mechanisms of hepatocarcinogenesis. Oncogene. 2002;21:2593–604.
Murakami Y, Yasuda T, Saigo K, et al. Comprehensive analysis of microRNA expression patterns in hepatocellular carcinoma and non-tumorous tissues. Oncogene. 2006;25:2537–45.
Gramantieri L, Ferracin M, Fornari F, Veronese A, et al. Cyclin G1 is a target of miR-122a, a microRNA frequently down-regulated in human hepatocellular carcinoma. Cancer Res. 2007;67:6092–9.
Meng F, Henson R, Wehbe-Janek H, et al. MicroRNA-21 regulates expression of the PTEN tumor suppressor gene in human hepatocellular cancer. Gastroenterology. 2007;133:647–58.
Varnholt H, Drebber U, Schulze F. MicroRNA gene expression profile of hepatitis C virus-associated hepatocellular carcinoma. Hepatology. 2008;47:1223–32.
Ladeiro Y, Couchy G, Balabaud C, et al. MicroRNA profiling in hepatocellular tumors is associated with clinical features and oncogene/tumor suppressor gene mutations. Hepatology. 2008;47:1955–63.
Fornari F, Gramantieri L, Ferracin M, et al. MiR-221 controls CDKN1C/p57 and CDKN1B/p27 expression in human hepatocellular carcinoma. Oncogene. 2008;27:5651–61.
Wong QW, Lung RW, Law PT, Lai PB, et al. MicroRNA-223 is commonly repressed in hepatocellular carcinoma and potentiates expression of Stathmin1. Gastroenterology. 2008;135:257–69.
Datta J, Kutay H, Nasser MW, et al. Methylation mediated silencing of MicroRNA-1 gene and its role in hepatocellular carcinogenesis. Cancer Res. 2008;68:5049–58.
Li W, Xie L, He X, et al. Diagnostic and prognostic implications of microRNAs in human hepatocellular carcinoma. Int J Cancer. 2008;123:1616–22.
Li S, Fu H, Wang Y, et al. MicroRNA-101 regulates expression of the v-fos FBJ murine osteosarcoma viral oncogene homolog (FOS) oncogene in human hepatocellular carcinoma. Hepatology. 2009;49:1194–202.
Sekine S, Ogawa R, Ito R, et al. Disruption of Dicer1 induces dysregulated fetal gene expression and promotes hepatocarcinogenesis. Gastroenterology. 2009;136:2304–15.
Chiosea S, Jelezcova E, Chandran U, et al. Up-regulation of dicer, a component of the MicroRNA machinery, in prostate adenocarcinoma. Am J Pathol. 2006;169:1812–20.
Flavin RJ, Smyth PC, Finn SP, et al. Altered eIF6 and Dicer expression is associated with clinicopathological features in ovarian serous carcinoma patients. Mod Pathol. 2008;21:676–84.
Zhang X, Cairns M, Rose B, et al. Alterations in miRNA processing and expression in pleomorphic adenomas of the salivary gland. Int J Cancer. 2009;124:2855–63.
Martin MG, Payton JE, Link DC. Dicer and outcomes in patients with acute myeloid leukemia (AML). Leuk Res. 2009;33:e127.
Merritt WM, Lin YG, Han LY, et al. Dicer, Drosha, and outcomes in patients with ovarian cancer. N Engl J Med. 2008;359(25):2641–50.
Karube Y, Tanaka H, Osada H. Reduced expression of Dicer associated with poor prognosis in lung cancer patients. Cancer Sci. 2005;96:111–5.
Chiosea S, Jelezcova E, Chandran U, et al. Overexpression of Dicer in precursor lesions of lung adenocarcinoma. Cancer Res. 2007;67:2345–50.
Schöniger-Hekele M, Müller C, Kutilek M, et al. Hepatocellular carcinoma in Central Europe: prognostic features and survival. Gut. 2001;48:103–9.
Calin GA, Sevignani C, Dumitru CD, et al. Human microRNA genes are frequently located at fragile sites and genomic regions involved in cancers. Proc Natl Acad Sci USA. 2004;101:2999–3004.
Calin GA, Croce CM. MicroRNA signatures in human cancers. Nat Rev Cancer. 2006;6:857–66.
Esquela-Kerscher A, Slack FJ. Oncomirs-microRNAs with a role in cancer. Nat Rev Cancer. 2006;6:259–69.
Cobb BS, Hertweck A, Smith J, et al. A role for Dicer in immune regulation. J Exp Med. 2006;203:2519–27.
Zhang L, Huang J, Yang N. MicroRNAs exhibit high frequency genomic alterations in human cancer. Proc Natl Acad Sci USA. 2006;103:9136–41.
Acknowledgments
This work was supported by the National Key Technologies Research and Development Program of China during the 11th 5-year Plan Period (2008ZX10002-006), National Natural Science Foundation of China (No. 30771923, 30671869, 30872250, 30801348, 30971612) and the National Basic Research Program of China (2007CB512900).
Conflict of interest statement
Authors declare that no conflicting or competing interests, of any nature, exist between the Authors of this work and their Academic activity.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wu, JF., Shen, W., Liu, NZ. et al. Down-regulation of Dicer in hepatocellular carcinoma. Med Oncol 28, 804–809 (2011). https://doi.org/10.1007/s12032-010-9520-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12032-010-9520-5