Abstract
Spinal cord injury (SCI) triggers a sequel of events commonly associated with cell death and dysfunction of glias and neurons surrounding the lesion. Although astrogliosis and glial scar formation have been involved in both damage and repair processes after SCI, their role remains controversial. Our goal was to investigate the effects of the P2 receptors antagonists, PPADS and suramin, in the establishment of the reactive gliosis and the formation of the glial scar. Molecular biology, immunohistochemistry, spared tissue, and locomotor behavioral studies were used to evaluate astrogliosis, in adult female Sprague–Dawley rats treated with P2 antagonists after moderate injury with the NYU impactor device. Semi-quantitative RT-PCR confirmed the presence of P2Y1, P2Y2, P2Y4, P2Y6, P2Y12, and P2X2 receptors in the adult spinal cord. Immunohistochemistry studies confirmed a significant decrease in GFAP-labeled cells at the injury epicenter as well as a decrease in spared tissue after treatment with the antagonists. Functional open field testing revealed no significant locomotor score differences between treated and control animals. Our work is consistent with studies suggesting that astrogliosis is an important event after SCI that limits tissue damage and lesion spreading.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Spinal cord injury (SCI) is one of the major causes of disability around the world. The events following an injury are very distinctive, and plenty has been published about the progression of these molecular and cellular events (for review, Hulsebosch 2002). Tissue integrity is compromised as a result of the axotomy, edema, hemorrhage, ischemia, electrolytic shift, release of nucleotides, and excitatory amino acids that come with the injury (Ducker et al. 1971; Park et al. 2004; Franke et al. 2006; Tanhoffer et al. 2007; Sinescu et al. 2010). These events are followed by apoptosis, demyelination, and free-radical production. In addition, the upregulation of repulsive factors and inhibitory molecular signals such as proteoglycans, Slit proteins, semaphorin-3, Eph/ephrins, MAG, NOGO, and OMgp block axonal regeneration after SCI (Schnell and Schwab 1990; McKerracher et al. 1994; Fournier and Strittmatter 2001; Willson et al. 2002). Moreover, it is well known that the downregulation of growth factors and the invasion of immune cells that release cytokines and chemokines stimulate an inflammatory response (Dijkstra et al. 2001; Nakamura and Bregman 2001; Widenfalk et al. 2001; Popovich et al. 2003; Iannotti et al. 2006). These pro-inflammatory molecules that activate microglial cells and macrophages can trigger astrogliosis (Fitch et al. 1999), with the subsequent formation of the glial scar.
Astrogliosis or reactive gliosis is a term used to describe a cellular process that astrocytes undergo after pathological conditions and trauma to the central nervous system (CNS) (Kato et al. 1998; Haydon 2001; Okada et al. 2004; Williams et al. 2007; Zhang et al. 2010). Extensive hypertrophy, proliferation, and increase in the expression of the intermediate filaments, glial fibrillary acidic protein (GFAP) and vimentin, are among the factors that characterize the gliotic response (Chiu and Goldman 1985; Laping et al. 1994; Sofroniew and Vinters 2010). It is important to emphasize that the trauma to the spinal cord not only generates a gliotic response but also the upregulation of inhibitory molecules such as proteoglycans, tenascin, Eph/ephrin proteins, and semaphorin-3 by astrocytes within the glial scar (Becker et al. 2000; Pasterkamp et al. 2001; Willson et al. 2002; Cruz-Orengo et al. 2006; Figueroa et al. 2006; Cafferty et al. 2007).
Reactive astrocytes are the principal components of the glial scar, a physical and chemical barrier known to inhibit axonal regeneration (Fitch and Silver 1997). However, this cellular wall also presents some favorable functions for the injured tissue. Evidence demonstrates that glial scarring can restrict the spreading of the cyst at the lesion epicenter and isolates the injured cells from the healthy ones, preventing uncontrolled tissue damage (Faulkner et al. 2004). In addition, the glial scar may reduce the infiltration of inflammatory cells preserving motor function (Bush et al. 1999; Popovich et al. 1999).
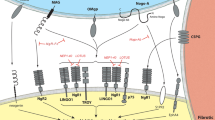
Extracellular nucleotides and nucleosides can contribute to the establishment of the reactive gliosis through the activation of metabotropic (P2Y) and ionotropic (P2X) receptors (Burnstock 1997). Studies demonstrated that purines and pyrimidines are released from dead or damaged cells following trauma, exerting a trophic effect on glial and neuronal cells (Gordon 1986; Ferrari et al. 1997; Lazarowski et al. 1997; Wang et al. 2004). Other reports confirmed that extracellular ATP lead to an increase in GFAP mRNA and protein level, as well as proliferation and stellation of cultured astrocytes (Abbracchio et al. 1994; Neary et al. 1994). In addition, in vivo studies using rat nucleus accumbens demonstrated that GFAP and astrocyte proliferation were augmented after infusion of ATP analog. This effect was blocked by P2 nucleotide receptor antagonists, indicating that these receptors mediate the trophic actions of extracellular ATP in vivo (Neary et al. 1996; Franke et al. 2001). Altogether, this evidence supports a potential role for these receptors as modulators of astrogliosis and glial scar formation after CNS injury.
P2X and P2Y receptors in astrocytes, neurons, and microglias from CNS are activated by extracellular nucleotides released after injury. These receptors are potent modulators of physiological and pathological processes in the brain and spinal cord (Erb et al. 2006; Fields and Burnstock 2006; Zimmermann 2006). Understanding the role of the extracellular nucleotides and their receptors in reactive gliosis after SCI can represent a novel therapeutic approach to decreasing the repulsive environment established by the glial scar after trauma and enhancing regeneration at the lesion epicenter. However, we could not discard the possibility that P2 receptors may play a role in controlling the spreading of the cyst at the lesion epicenter and improving the isolation of the injured cells from the healthy ones, diminishing the loss of locomotor function.
Therefore, we hypothesize that a blockade of P2 receptors after SCI should reduce the gliotic response and diminish also the formation of the glial scar. The reduced physical and chemical barrier will provide a permissive environment for axonal regeneration and locomotor recovery. On the other hand, an increase in the cavity (or reduction in spare tissue) without locomotor recovery after P2 receptors blockade support the idea that a gliotic response is necessary to contain the cyst and isolate healthy cells from damaged ones.
Materials and Methods
Spinal Cord Injury
Adult female Sprague–Dawley rats (~235 g) from Hilltop Lab (Scottdale, PA) were treated as described previously (Rodriguez-Zayas et al. 2010). Briefly, animals were anesthetized; their T10 lamina removed and their exposed spinal cord injured with the NYU impactor device from a height of 12.5 mm (Miranda et al. 1999; Cruz-Orengo et al. 2006; Figueroa et al. 2006; Santiago et al. 2009). Rats in the control group (sham) remained uncontused and were sutured after laminectomy. After surgical procedure, rats received two daily doses of the antibiotic Cefazolin (25 mg/kg, Bristol Myers Squibb, NY) subcutaneously for seven consecutive days. Two daily doses of Buprenex (buprenorphine; Reckett & Colman Pharmaceuticals, Inc. Richmond, VA; 0.05 mg/kg) was injected for 3 days and hydrated with 0.9% NaCl solution subcutaneously after surgical procedure (≈1 cm3 per 5 g weight loss). Bladders of injured rats were expressed at least three times daily until complete recovery and lodged individually on cages with absorbent bedding to convalesce. Both sham and contused groups were permitted to survive for 28 days post-injury (DPI). These protocols were approved by the University of Puerto Rico IACUC and followed NIH guidelines for the safe use and care of laboratory animals.
RNA Studies
Pentobarbital (40–50 mg/kg) was injected intraperitoneally to sham and injured rats (n = 3). After complete sedation, the rats were transcardially perfused with ice-cold 0.01 M phosphate-buffered saline (PBS), pH 7.4 (Sigma-Aldrich, St. Louis, MO) as previously described (Irizarry-Ramirez et al. 2005). The T10 lesion epicenters (5 mm) of each spinal cord were dissected and total RNA extracted using Trizol reagent (Sigma-Aldrich, Inc. St Louis, MO). To avoid genomic contamination, the extracted RNA was treated with the DNA-free kit (Ambion Inc, Austin, TX). Integrity of each RNA sample was electrophoretically confirmed by running a 1% agarose-formaldehyde gel, and quantification of total RNA was achieved using the Eppendorf BioPhotometer system (Eppendorf, AG). Reverse transcription reaction of 1 μg of RNA was performed using iScript cDNA Synthesis Kit (Bio-Rad, Hercules, CA) according to the manufacturer's instructions. Mock cDNA reaction, without RNA, was prepared and used as negative control to rule out genomic DNA contamination.
Semi-quantitative RT-PCR
cDNA amplification was performed in an Amplitron II (Barnstead/Thermolyne, Dubuque, Iowa) using a 50-μl reaction with 2 μM of primers (Table 1), 1.5 mM MgCl2, 260 μM dNTP mix, PCR buffer, and 2.5 U Taq polymerase (Promega, Madison, WI) in nuclease-free water (Ambion Inc, Austin, TX). Cycle optimization for each RT-PCR product was accomplished to establish the linear phase of gene amplification and the appropriate annealing temperature. cDNA amplification was done using the following parameters: a hot-start at 94°C for 3 min (min) and 31–38 cycles composed of 1 min at 95°C for denaturing step, 1 min annealing at 57°C or 62°C (see Table 1), and an extension at 72°C for 1 min. A post-dwell period of 5 min at 72°C was applied to complete the amplification process. Products generated were confirmed by migration to the expected position on a 1.5% agarose gel electrophoresis stained with ethidium bromide. The PCR products were purified with the QIA quick PCR purification kit (QIAGEN Inc, CA) and sequenced to confirm the identity of the products.
Intrathecal Infusion of Nucleotide Receptors Antagonists
After the T10 laminectomy and moderate contusion to the spinal cord, each rat received a second laminectomy at T12 to expose a full segment caudal to the lesion epicenter as previously reported (Cruz-Orengo et al. 2006; Figueroa et al. 2006). The dura matter was cautiously punctured with a 32-gauge needle just enough to allow the insertion of a catheter (inner diameter 0.18 mm; outer diameter 0.36 mm; Alzet, Cupertino, California, USA) that was extended rostrally to the lesion epicenter. The cannula was connected to a primed mini-osmotic pump (model 2002, Alzet, Cupertino, California, USA) that delivered a constant flow (0.5 ± 0.1 μl/h) of 10 μM pyridoxal-phosphate-6-azophenyl-2′,4′-disulfonate (PPADS) (Tocris bioscience, Missouri), 10 μM of Suramin (Tocris bioscience, Missouri), or 0.9% NaCl solution for 14 days to the lesion epicenter (n = 10–13 per group tested). These two antagonists, at the indicated concentration, have been shown to block P2 nucleotide receptors in vitro and in vivo (Charlton et al. 1996; Kharlamov et al. 2002; Tompkins and Parsons 2006). The pump was placed in a small subcutaneous pocket made over the sacral vertebrae caudal to the incision and the catheter attached to the fascia over the muscles at the incision edge. The post-operative treatment was performed as described previously.
Behavioral Assays
Animals (n = 10–13 per group) infused with antagonists and saline were evaluated weekly (7, 14, 21, and 28 DPI) by the Basso-Beattie-Bresnahan scale (BBB open field test) and horizontal grid-walking test (Basso et al. 1995; Merkler et al. 2001). The animals were previously trained for habituation and after surgery, identified by numbers and not by treatment. Two evaluators, unaware of the treatment or previous score of the animal, performed all behavioral assays as previously reported by Cruz-Orengo et al. (2006); Figueroa et al. (2006), and Santiago et al. (2009).
The BBB open field test evaluates hindlimb and forelimb movements on an open field for 4 min as described by Basso et al (1995). Sham or naïve rats had a perfect score of 21 points (stage III), but after a moderate spinal cord injury, the functional score went as low as 2–7 points (stage I) at 7 DPI and 8–13 points (stage II) at 14 DPI, remaining constant afterward (Cruz-Orengo et al. 2006; Figueroa et al. 2006).
The horizontal grid-walking test evaluates sensorimotor integration and determines the ability of the animals to place their hindlimbs over the arbitrarily located sticks of a horizontal ladder (Merkler et al. 2001). Misplacement of the paws, hindlimb footfalls, or dragging were considered as errors. The sticks placed in the horizontal ladder allow a maximum of 25 errors. Sham and naïve animals had few or no errors, but injured rats attained high number of hindlimb misplacement or errors (Cruz-Orengo et al. 2006; Figueroa et al. 2006; Santiago et al. 2009).
Immunohistochemistry
Rats completely anesthetized (n = 4–5) were perfused intracardially with ice-cold 0.01 M PBS (pH 7.4; Sigma-Aldrich, St. Louis, MO) followed by 4% paraformaldehyde (PFA) at 4°C. Spinal cord removal, post-fixation, sectioning, and immnohistochemistry protocol was performed as described by Rodriguez-Zayas et al. (2010). The primary antibodies used were mouse anti-GFAP (1:100; BD Pharmingen) for reactive astrocytes, goat anti-GAP-43 (1:500; Sigma, MO) for growth associated protein, or rabbit anti-5-HT (1:1,000; ImmunoStar Inc., Hudson, WI) for serotonergic fibers as markers of neurite outgrowth. Then, the sections were incubated with 1:250 Alexa 488 goat anti-mouse (Invitrogen), 1:250 Alexa 488 rabbit anti-goat (Molecular Probes, Oregon), or 1:250 Alexa 488 goat anti-rabbit (Molecular Probes, Oregon) as secondary antibodies for 2 h at room temperature (RT) in a covered area. Slices lacking primary antibody were used as controls. Images were visualized with a Zeiss LSM 5 PASCAL Confocal Microscope System (Carl Zeiss MicroImaging Inc., Thornwood, NY) outfitted with an argon laser emitting at 488 nm. To conclude the analysis, the ImageJ 1.43u software (NIH, MD, USA) was used to determine the mean intensity of each image taken at a magnification of 10×. Three sections from each animal spinal cord were evaluated, and the mean used as one value. The longitudinal sections represent areas in the middle and lateral sides of the epicenter.
Luxol Fast Blue/Cresyl Violet Histochemistry
The spared tissue analysis was performed as described by Santiago et al. 2009. Animals (n = 10–12 per group) were sacrificed as described before by exsanguination and perfusion with intracardial ice-cold PBS, followed by 4% PFA. Spinal cord sections (20 μm) were mounted and post-fixed in 4% PFA for 30 min at RT; then placed in 1:1 alcohol/chloroform solution overnight (ON) at RT. The sections were re-hydrated with 95% ethyl alcohol (ETOH) for 10 min and incubated for 16 h at 37°C with Luxol Fast Blue (Alfa Aesar, Shore Road, Heysham, Lancs). Excess stain was rinsed off with 95% ETOH and the sections were stained in a 0.05% lithium carbonate solution for 4 min, followed by 1 min rinse in distilled water and 4 min in 70% ETOH. When differentiation was completed, sections were washed with distilled water for an additional 1 min and counterstained with Cresyl violet solution for 40 s. Immediately, slides were rinsed with distilled water, incubated for 5 min in 95% ETOH and washed twice with 100% ETOH for 5 min each. Subsequently, the sections were rinsed in Histoclear (National Diagnostic, Atlanta, Georgia) two times for 5 min each and coverslipped using Permount (Fisher) as mounting solution. Slides were observed in a digital microscope (Fisher Scientific, Pittsburgh, PA, USA), and photomicrographs were taken using the Motic Images 2000 version 1.2 professional software. The stained sections were morphometrically analyzed (MCID, Imaging Research, Inc. Ontario, Canada) to determine the amount of white matter spared tissue (Santiago et al. 2009). Briefly, the outer border of the spinal cord, the spare white matter stained tissue, and the lesion cavity were delineated. These parameters were used to calculate the area of white matter spared tissue (density/area).
Statistical Analysis
Statistics are offered as mean ± standard error of mean. ANOVA, followed by Tukey–Kramer multiple comparisons post hoc test analysis, was applied for assessment of significant difference between control (sham) and experimental groups (at 2, 4, 7, 14, and 28 DPI). A probability <0.05 between control and experimental groups was considered to be statistically significant.
Results
P2 Receptors Expressed in Adult Spinal Cord Tissue
Expression of diverse P2 receptors mRNA in the adult spinal cord of rats was assessed by semi-quantitative RT-PCR using specific primers (Fig. 1). The amplified fragments migrated to the expected positions confirming basal levels of P2Y1, P2Y2, P2Y4, P2Y6, P2Y12, and P2X2 receptors in the adult spinal cord. The specificity of the primers and the identity of the amplified fragments were determined by sequencing of the PCR products (data not shown). β-actin was used as housekeeping gene positive control and a mock control used to discard the possibility of genomic contamination during RNA extraction.
P2 receptor mRNA expression in the adult spinal cord. Semi-quantitative RT-PCR studies were performed in mRNA samples of adult female rats to demonstrate the profile of P2 receptor expression in the spinal cord. Adult tissue exhibited a basal level of P2Y1, P2Y2, P2Y4, P2Y6, P2Y12, and P2X2 expression. Mock control was prepared to discard of genomic contamination during RNA extraction
P2 Receptors Antagonists did not Affect Functional Locomotor Recovery
Standardized behavioral assays, including BBB score and grid walking tests, revealed that blockade of P2 receptors with P2 antagonists infused intrathecally did not improve the behavioral outcomes in treated rats. As shown in Fig. 2, no changes were observed between suramin, PPADS, and control (saline) infused animals in any locomotor assay.
Absence of functional locomotor recovery after P2 antagonist treatments. BBB open field (a) and grid walking (b) behavioral tests were used to monitor the role of P2 receptor activation after SCI. No significant difference was observed between individuals treated with antagonists (PPADS and suramin) versus saline in terms of locomotive assessments (BBB and grid walking) at any of the time points studied (n = 10–13 per group). Data represent the mean ± SEM; (ANOVA followed by Tukey–Kramer multiple comparisons post-hoc test; F = 6.863; df = 11, 126 for BBB assay and F = 9.024; df = 11, 129 for grid walking test)
Statistical analysis with ANOVA demonstrated that control animals presented a BBB score of 5.125 ± 0.672 (n = 12) at 7 DPI, 8.688 ± 1.016 (n = 12) at 14 DPI, 10.604 ± 0.770 (n = 12) at 21 DPI, and 11.432 ± 0.906 (n = 11) at 28 DPI. The suramin-treated animals presented a score of 4.271 ± 0.941 (n = 12) at 7 DPI, 8.795 ± 1.267 (n = 11) at 14 DPI, 10.659 ± 1.203 (n = 11) at 21 DPI, and 12.477 ± 1.474 (n = 11) at 28 DPI; and PPADS-treated animals presented a score of 4.365 ± 1.028 (n = 13) at 7 DPI, 7.212 ± 1.105 (n = 13) at 14 DPI, 10.400 ± 1.547 (n = 10) at 21 DPI, and 11.450 ± 1.466 (n = 10) at 28 DPI. These results showed that intrathecal infusion of P2 antagonists did not improve functional locomotor recovery when compared to control samples (p > 0.05).
ANOVA analysis of grid walking behavioral assay demonstrated that control animals presented similar number of errors than P2 antagonists-treated rats. Control groups scored 24.375 ± 0.625 (n = 12) at 7 DPI, 16.625 ± 1.585 (n = 12) at 14 DPI, 15.833 ± 1.310 (n = 12) at 21 DPI, and 14.682 ± 1.702 (n = 11) errors at 28 DPI; the suramin-treated animals presented a score of 23.538 ± 0.771 (n = 13) at 7 DPI, 16.042 ± 1.558 (n = 12) at 14 DPI, 14.083 ± 1.808 (n = 12) at 21 DPI, and 12.042 ± 1.932 (n = 12) errors at 28 DPI; and PPADS-treated animals presented a score of 23.692 ± 1.227 (n = 13) at 7 DPI, 17.542 ± 1.990 (n = 12) at 14 DPI, 13.900 ± 1.560 (n = 10) at 21 DPI, and 11.550 ± 1.571 (n = 10) errors at 28 DPI, demonstrating that P2 antagonists did not reduce the number of errors when compared to control sample (p > 0.05).
Effect of P2 Antagonists in the Density of GFAP-labeled Cells at Injury Site
GFAP immunoreactivity determined the presence of reactive astrocytes in lesioned spinal cord treated with saline and the P2 antagonists: suramin and PPADS (Fig. 3a). Differences in GFAP immunoreactivity (Fig. 3b), mostly confined to the glial scar surrounding the lesion epicenter, were observed in saline treated samples (control) versus experimental samples at 28 DPI, showing a decrease in the reactive gliosis. ANOVA analysis demonstrated a significant twofold decrease in GFAP densitometric analysis after PPADS (1,530 ± 160 arbitrary units (AU), p < 0.01, n = 5) and suramin treatment (1,542 ± 390 AU, p < 0.05, n = 4) when compared with saline samples (2,906 ± 272 AU, n = 5). In addition, blockade of P2 receptors did not affect significantly GAP-43 and serotonin (5-HT) immunostaining (data not shown) at the lesion epicenter and regions rostral or caudal to it.
P2 antagonists decrease gliotic response at the injury site. Immunohistochemistry, confocal microscopy (a), and densitometric analyses were used to determine the amount of GFAP-positive cells at the lesion epicenter in 28 DPI spinal cord samples treated with saline (n = 5), PPADS (n = 5), or suramin (n = 4). Inset: High magnification picture at the lesion epicenter, representative of the samples analyzed. Densitometric analysis (b) of GFAP intensity per area confirmed a down-regulation of this intermediate filament after treatment with P2 antagonist. Data represent the mean ± SEM; **p < 0.01, *p < 0.05 significant difference versus saline group (ANOVA followed by Tukey–Kramer multiple comparisons post hoc test; F = 8.682; df = 2, 11)
Spared Tissue Level Decreased with P2 Antagonist Treatment
Spinal cord sections from the lesion epicenter were stained with luxol fast blue-cresyl violet and morphometrically analyzed to determine the extent of white matter spared after treatment with saline, suramin, and PPADS. Suramin and PPADS treatments exacerbated the loss of tissue after SCI and provoked a significant decrease in the amount of spared white matter when compared to the saline group (Fig. 4). ANOVA analysis demonstrated a significant decrease after suramin treatment (46 ± 4 arbitrary units (AU), p < 0.001, n = 11) and after PPADS treatment (54 ± 3 AU, p < 0.05, n = 10) when compared with saline sample (67 ± 4 AU, n = 12).
Spared tissue decreased with P2 antagonist treatment. a Photo micrographs were captured with a Sony Progressive 3CDD camera and morphometrically analyzed (MCID, Imaging Research) to determine the extent of spared white matter after treatment. The spared tissue was reduced and the cavity was augmented with P2 antagonist treatment. b Densitometric analysis of tissue's density per area confirmed a decrease in the amount of spare tissue after treatment with P2 antagonist, PPADS (n = 10), and suramin (n = 11) relative to control-saline treated animals (n = 12). Data represent the mean ± SEM; ***p < 0.001, *p < 0.05 significant difference versus saline group (ANOVA followed by Tukey–Kramer multiple comparisons post-hoc test; F = 9.064; df = 2, 30)
Discussion
Astrocytes are essential in maintaining homeostasis, tissue integrity, and functional outcome in the CNS. Among its divergent roles are supporting neuronal and synaptic function, regulation of extracellular ions, neurotransmitters, and fluid balance (Fields and Stevens-Graham 2002; Hansson and Ronnback 2003; Laird et al. 2008). However, the role of reactive astrocytes after SCI is still a subject of debate. Extensive studies associate scar-forming astrocytes with inhibition of axonal regeneration and elongation after trauma. The upregulation of repellent proteins expressed by astrocytes after being activated, added to the downregulation of growth factors and the infiltration of inflammatory cells, may be recognized by regenerating axons, thus blocking axonal elongation at the injury site. Some researchers have proposed that astrogliosis generated after trauma may produce a physical barrier for regenerative axons (McGraw et al. 2001). Despite this, other studies confirm that astrocytes are involved in maintaining an optimal environment for CNS function (Sidoryk-Wegrzynowicz et al. 2010). Reactive astrocytes perform a leading role in maintaining neural tissue and restricting infiltration of inflammatory cells during crucial times after trauma to the CNS, thus preserving motor function (Myer et al. 2006).
Injury to the CNS causes release of nucleotides into extracellular milieu that can reach up to millimolar concentrations (Wang et al. 2004; Franke and Illes 2006). The increased levels of these nucleotides stimulate astrocytes proliferation and contribute to the process of astrogliosis (Abbracchio et al. 1994; Franke et al. 2001). Studies by Wang et al. (2004) reported a high ATP release through extensive peritraumatic zones for hours after SCI, probably due to a continuous production of ATP and failure to regulate efflux pathways. Although the molecular mechanism behind the nucleotide response of reactive astrocytes after trauma is uncertain, it has been associated with the activation of the ATP-dependent astrocytic calcium signaling and the P2 nucleotide receptors (Fam et al. 2000; Scemes et al. 2000). Therefore, these receptors represent a new target to deal with reactive gliosis in SCI.
As a first approach, the expression of the mRNA for various P2 receptors in the spinal cord was evaluated. The presence of P2Y1, P2Y2, P2Y4, P2Y6, P2Y12, and P2X2 receptors in the adult spinal cord was evident. Also, the existence of other P2 receptors, such as P2X4, P2X6, P2X7, and P2Y14, have been reported in the spinal cord (Collo et al. 1996; Burnstock 2000; Kobayashi et al. 2006). In general, P2Y nucleotide receptors have been related to survival responses in nervous tissues and to the support of cellular growth, proliferation, and differentiation (Rathbone et al. 1999; Weisman et al. 2005; Franke and Illes 2006). In contrast, P2X receptor activation has been associated with inflammation and apoptotic signaling (Cavaliere et al. 2003; Skaper et al. 2006). Altogether, this family of receptors is activated via extracellular nucleotides and is considered strong modulator of normal and pathological processes in the CNS.
Knowing that several P2 receptors are expressed in the adult spinal cord and the abundant release of extracellular nucleotides after injury, we decided to use two non-specific P2 antagonists, PPADS and suramin, to block the activation of diverse P2 receptors. In the past, suramin and PPADS have been extensively used as antagonist of P2Y1, P2Y2, P2Y4, P2Y6, P2Y12, and P2X2 receptors and for other receptors such as P2X7 (Lambrecht et al. 2002; Abbracchio et al. 2006; Jacobson et al. 2009). The intrathecal administration of antagonists and the blockade of the P2 receptors activation do not improve locomotor behavior in our injured rats. These results suggest that the role of these receptors after SCI is not associated directly with regenerative events associated with locomotor behavior because GAP-43 and serotonergic fibers were not significantly affected. However, we cannot exclude the possibility that the decrease in reactive gliosis and the increases in the lesion cavity, caused by the infusion of antagonists, can also be associated with the lack of locomotor improvement.
In vivo studies using Brilliant blue G (BBG), a selective antagonist of P2X7 receptor, reported reduction in local activation of astrocytes and microglia, reduction in the inflammatory response, as well as reduction in lesion size. They also reported an improvement in locomotor activity after systemic administration of the antagonist (Peng et al. 2009). But also, they attribute these overall protections to the fact that the intravenous route of delivery increases the number of cell types potentially affected by the receptor blockade, launching the possibility of off-target effects. In addition, because BBG is a selective antagonist for the P2X7 receptor, any interference with P2Y family receptors or other P2X receptors was not expected. These data indicate that there exists a subtle line between blockade and activation of different P2Y and P2X family receptors when combined as well as the route of antagonist/agonist delivery in order to obtain the expected effect.
GFAP-labeled cells analysis revealed a significant decrease after PPADS and suramin treatment when compared with the saline control group, which leads to an increase of the cavity size and a reduction in spare tissue. Results confirmed by the morphometrical analysis of samples stained with luxol fast blue-cresyl violet revealed that suramin and PPADS treatments aggravate the size of the cavity and decreased significantly the amount of spared tissue when compared with saline control samples. Previous studies in rat nucleus accumbens showed a reduction in the injury-induced astroglial proliferation via P2Y1 activation after traumatic injury and after microinfusion of agonist (ADP beta S) when pretreated with PPADS (Franke et al. 2009). Other studies with primary cultures of rat cortical astrocytes demonstrated that treatment with suramin diminished the phosphorylation of STAT3 at Ser-727 through P2 receptor, which resulted in reduced astrocytes proliferation and reactive astrogliosis (Washburn and Neary 2006). In addition, studies demonstrated that suramin interrupts the gliotic response via nucleotide receptors activation after injury suggesting a role of these receptors in the reactive gliosis initiated by trauma to the CNS (Di Prospero et al. 1998).
In summary, our results suggest that blockade of P2 receptors with the antagonists, suramin and PPDS, disrupted the mitogenic and morphogenic changes that typify the reactive gliosis, causing an increase in the cavity size and tissue loss. A diminished and less effective encapsulation of the cyst can lead to infiltration of inflammatory cells and uncontrolled spreading of tissue damage. In addition, neuronal metabolic and trophic support provided by astrocytes is going to be lost and glial–glial or neuronal–glial interactions could be interrupted, promoting neurodegeneration at the injury site.
References
Abbracchio MP, Saffrey MJ, Hopker V, Burnstock G (1994) Modulation of astroglial cell proliferation by analogues of adenosine and ATP in primary cultures of rat striatum. Neuroscience 59:67–76
Abbracchio MP, Burnstock G, Boeynaems JM et al (2006) International Union of Pharmacology LVIII: update on the P2Y G protein-coupled nucleotide receptors: from molecular mechanisms and pathophysiology to therapy. Pharmacol Rev 58:281–341
Basso DM, Beattie MS, Bresnahan JC (1995) A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma 12:1–21
Becker T, Anliker B, Becker CG et al (2000) Tenascin-R inhibits regrowth of optic fibers in vitro and persists in the optic nerve of mice after injury. Glia 29:330–346
Burnstock G (1997) The past, present and future of purine nucleotides as signalling molecules. Neuropharmacology 36:1127–1139
Burnstock G (2000) P2X receptors in sensory neurones. Br J Anaesth 84:476–488
Bush TG, Puvanachandra N, Horner CH et al (1999) Leukocyte infiltration, neuronal degeneration, and neurite outgrowth after ablation of scar-forming, reactive astrocytes in adult transgenic mice. Neuron 23:297–308
Cafferty WB, Yang SH, Duffy PJ, Li S, Strittmatter SM (2007) Functional axonal regeneration through astrocytic scar genetically modified to digest chondroitin sulfate proteoglycans. J Neurosci 27:2176–2185
Cavaliere F, Florenzano F, Amadio S et al (2003) Up-regulation of P2X2, P2X4 receptor and ischemic cell death: prevention by P2 antagonists. Neuroscience 120:85–98
Charlton SJ, Brown CA, Weisman GA, Turner JT, Erb L, Boarder MR (1996) PPADS and suramin as antagonists at cloned P2Y- and P2U-purinoceptors. Br J Pharmacol 118:704–710
Chiu FC, Goldman JE (1985) Regulation of glial fibrillary acidic protein (GFAP) expression in CNS development and in pathological states. J Neuroimmunol 8:283–292
Collo G, North RA, Kawashima E et al (1996) Cloning OF P2X5 and P2X6 receptors and the distribution and properties of an extended family of ATP-gated ion channels. J Neurosci 16:2495–2507
Cruz-Orengo L, Figueroa JD, Velazquez I et al (2006) Blocking EphA4 upregulation after spinal cord injury results in enhanced chronic pain. Exp Neurol 202:421–433
Di Prospero NA, Zhou XR, Meiners S, McAuliffe WG, Ho SY, Geller HM (1998) Suramin disrupts the gliotic response following a stab wound injury to the adult rat brain. J Neurocytol 27:491–506
Dijkstra S, Geisert EE Jr, Dijkstra CD, Bar PR, Joosten EA (2001) CD81 and microglial activation in vitro: proliferation, phagocytosis and nitric oxide production. J Neuroimmunol 114:151–159
Ducker TB, Kindt GW, Kempf LG (1971) Pathological findings in acute experimental spinal cord trauma. J Neurosurg 35:700–708
Erb L, Liao Z, Seye CI, Weisman GA (2006) P2 receptors: intracellular signaling. Pflugers Arch 452:552–562
Fam SR, Gallagher CJ, Salter MW (2000) P2Y(1) purinoceptor-mediated Ca(2+) signaling and Ca(2+) wave propagation in dorsal spinal cord astrocytes. J Neurosci 20:2800–2808
Faulkner JR, Herrmann JE, Woo MJ, Tansey KE, Doan NB, Sofroniew MV (2004) Reactive astrocytes protect tissue and preserve function after spinal cord injury. J Neurosci 24:2143–2155
Ferrari D, Chiozzi P, Falzoni S et al (1997) ATP-mediated cytotoxicity in microglial cells. Neuropharmacology 36:1295–1301
Fields RD, Burnstock G (2006) Purinergic signalling in neuron–glia interactions. Nat Rev Neurosci 7:423–436
Fields RD, Stevens-Graham B (2002) New insights into neuron–glia communication. Science 298:556–562
Figueroa JD, Benton RL, Velazquez I et al (2006) Inhibition of EphA7 up-regulation after spinal cord injury reduces apoptosis and promotes locomotor recovery. J Neurosci Res 84:1438–1451
Fitch MT, Silver J (1997) Glial cell extracellular matrix: boundaries for axon growth in development and regeneration. Cell Tissue Res 290:379–384
Fitch MT, Doller C, Combs CK, Landreth GE, Silver J (1999) Cellular and molecular mechanisms of glial scarring and progressive cavitation: in vivo and in vitro analysis of inflammation-induced secondary injury after CNS trauma. J Neurosci 19:8182–8198
Fournier AE, Strittmatter SM (2001) Repulsive factors and axon regeneration in the CNS. Curr Opin Neurobiol 11:89–94
Franke H, Illes P (2006) Involvement of P2 receptors in the growth and survival of neurons in the CNS. Pharmacol Ther 109:297–324
Franke H, Krugel U, Schmidt R, Grosche J, Reichenbach A, Illes P (2001) P2 receptor-types involved in astrogliosis in vivo. Br J Pharmacol 134:1180–1189
Franke H, Krugel U, Illes P (2006) P2 receptors and neuronal injury. Pflugers Arch 452:622–644
Franke H, Sauer C, Rudolph C, Krügel U, Hengstler JG, Illes P (2009) P2 receptor-mediated stimulation of the PI3-K/Akt-pathway in vivo. Glia 57:1031–1045
Gordon JL (1986) Extracellular ATP: effects, sources and fate. Biochem J 233:309–319
Hansson E, Ronnback L (2003) Glial neuronal signaling in the central nervous system. FASEB J 17:341–348
Haydon PG (2001) GLIA: listening and talking to the synapse. Nat Rev Neurosci 2:185–193
Hulsebosch CE (2002) Recent advances in pathophysiology and treatment of spinal cord injury. Adv Physiol Educ 26:238–255
Iannotti C, Zhang YP, Shields LB et al (2006) Dural repair reduces connective tissue scar invasion and cystic cavity formation after acute spinal cord laceration injury in adult rats. J Neurotrauma 23:853–865
Irizarry-Ramirez M, Willson CA, Cruz-Orengo L et al (2005) Upregulation of EphA3 receptor after spinal cord injury. J Neurotrauma 22:929–935
Jacobson KA, Ivanov AA, de Castro S, Harden TK, Ko H (2009) Development of selective agonists and antagonists of P2Y receptors. Purinergic Signal 5:75–89
Kato S, Gondo T, Hoshii Y, Takahashi M, Yamada M, Ishihara T (1998) Confocal observation of senile plaques in Alzheimer's disease: senile plaque morphology and relationship between senile plaques and astrocytes. Pathol Int 48:332–340
Kharlamov A, Jones SC, Kim DK (2002) Suramin reduces infarct volume in a model of focal brain ischemia in rats. Exp Brain Res 147:353–359
Kobayashi K, Fukuoka T, Yamanaka H et al (2006) Neurons and glial cells differentially express P2Y receptor mRNAs in the rat dorsal root ganglion and spinal cord. J Comp Neurol 498:443–454
Laird MD, Vender JR, Dhandapani KM (2008) Opposing roles for reactive astrocytes following traumatic brain injury. Neurosignals 16:154–164
Lambrecht G, Braun K, Damer M et al (2002) Structure–activity relationships of suramin and pyridoxal-5′-phosphate derivatives as P2 receptor antagonists. Curr Pharm Des 8:2371–2399
Laping NJ, Teter B, Nichols NR, Rozovsky I, Finch CE (1994) Glial fibrillary acidic protein: regulation by hormones, cytokines, and growth factors. Brain Pathol 4:259–275
Lazarowski ER, Homolya L, Boucher RC, Harden TK (1997) Direct demonstration of mechanically induced release of cellular UTP and its implication for uridine nucleotide receptor activation. J Biol Chem 272:24348–24354
McGraw J, Hiebert GW, Steeves JD (2001) Modulating astrogliosis after neurotrauma. J Neurosci Res 63:109–115
McKerracher L, David S, Jackson DL, Kottis V, Dunn RJ, Braun PE (1994) Identification of myelin-associated glycoprotein as a major myelin-derived inhibitor of neurite growth. Neuron 13:805–811
Merkler D, Metz GA, Raineteau O, Dietz V, Schwab ME, Fouad K (2001) Locomotor recovery in spinal cord-injured rats treated with an antibody neutralizing the myelin-associated neurite growth inhibitor Nogo-A. J Neurosci 21:3665–3673
Miranda JD, White LA, Marcillo AE, Willson CA, Jagid J, Whittemore SR (1999) Induction of Eph B3 after spinal cord injury. Exp Neurol 156:218–222
Myer DJ, Gurkoff GG, Lee SM, Hovda DA, Sofroniew MV (2006) Essential protective roles of reactive astrocytes in traumatic brain injury. Brain 129:2761–2772
Nakamura M, Bregman BS (2001) Differences in neurotrophic factor gene expression profiles between neonate and adult rat spinal cord after injury. Exp Neurol 169:407–415
Neary JT, Baker L, Jorgensen SL, Norenberg MD (1994) Extracellular ATP induces stellation and increases glial fibrillary acidic protein content and DNA synthesis in primary astrocyte cultures. Acta Neuropathol 87:8–13
Neary JT, Rathbone MP, Cattabeni F, Abbracchio MP, Burnstock G (1996) Trophic actions of extracellular nucleotides and nucleosides on glial and neuronal cells. Trends Neurosci 19:13–18
Okada S, Nakamura M, Mikami Y et al (2004) Blockade of interleukin-6 receptor suppresses reactive astrogliosis and ameliorates functional recovery in experimental spinal cord injury. J Neurosci Res 76:265–276
Park E, Velumian AA, Fehlings MG (2004) The role of excitotoxicity in secondary mechanisms of spinal cord injury: a review with an emphasis on the implications for white matter degeneration. J Neurotrauma 21:754–774
Pasterkamp RJ, Anderson PN, Verhaagen J (2001) Peripheral nerve injury fails to induce growth of lesioned ascending dorsal column axons into spinal cord scar tissue expressing the axon repellent Semaphorin3A. Eur J Neurosci 13:457–471
Peng W, Cotrina ML, Han X et al (2009) Systemic administration of an antagonist of the ATP-sensitive receptor P2X7 improves recovery after spinal cord injury. Proc Natl Acad Sci USA 106:12489–12493
Popovich PG, Guan Z, Wei P, Huitinga I, van Rooijen N, Stokes BT (1999) Depletion of hematogenous macrophages promotes partial hindlimb recovery and neuroanatomical repair after experimental spinal cord injury. Exp Neurol 158:351–365
Popovich PG, van Rooijen N, Hickey WF, Preidis G, McGaughy V (2003) Hematogenous macrophages express CD8 and distribute to regions of lesion cavitation after spinal cord injury. Exp Neurol 182:275–287
Rathbone MP, Middlemiss PJ, Gysbers JW et al (1999) Trophic effects of purines in neurons and glial cells. Prog Neurobiol 59:663–690
Rodriguez-Zayas AE, Torrado AI, Miranda JD (2010) P2Y2 receptor expression is altered in rats after spinal cord injury. Int J Dev Neurosci 28:413–421
Santiago JM, Rosas O, Torrado AI, Gonzalez MM, Kalyan-Masih PO, Miranda JD (2009) Molecular, anatomical, physiological, and behavioral studies of rats treated with buprenorphine after spinal cord injury. J Neurotrauma 26:1783–1793
Scemes E, Suadicani SO, Spray DC (2000) Intercellular communication in spinal cord astrocytes: fine tuning between gap junctions and P2 nucleotide receptors in calcium wave propagation. J Neurosci 20:1435–1445
Schnell L, Schwab ME (1990) Axonal regeneration in the rat spinal cord produced by an antibody against myelin-associated neurite growth inhibitors. Nature 343:269–272
Sidoryk-Wegrzynowicz M, Wegrzynowicz M, Lee E, Bowman A, Aschner M (2010) Role of astrocytes in brain function and disease. Toxicol Pathol 39(1):115–123
Sinescu C, Popa F, Grigorean VT et al (2010) Molecular basis of vascular events following spinal cord injury. J Med Life 3:254–261
Skaper SD, Facci L, Culbert AA et al (2006) P2X(7) receptors on microglial cells mediate injury to cortical neurons in vitro. Glia 54:234–242
Sofroniew MV, Vinters HV (2010) Astrocytes: biology and pathology. Acta Neuropathol 119:7–35
Tanhoffer RA, Yamazaki RK, Nunes EA et al (2007) Glutamine concentration and immune response of spinal cord-injured rats. J Spinal Cord Med 30:140–146
Tompkins JD, Parsons RL (2006) Exocytotic release of ATP and activation of P2X receptors in dissociated guinea pig stellate neurons. Am J Physiol Cell Physiol 291:C1062–C1071
Wang X, Arcuino G, Takano T et al (2004) P2X7 receptor inhibition improves recovery after spinal cord injury. Nat Med 10:821–827
Washburn KB, Neary JT (2006) P2 purinergic receptors signal to STAT3 in astrocytes: difference in STAT3 responses to P2Y and P2X receptor activation. Neuroscience 142:411–423
Weisman GA, Wang M, Kong Q et al (2005) Molecular determinants of P2Y2 nucleotide receptor function: implications for proliferative and inflammatory pathways in astrocytes. Mol Neurobiol 31:169–183
Widenfalk J, Lundstromer K, Jubran M, Brene S, Olson L (2001) Neurotrophic factors and receptors in the immature and adult spinal cord after mechanical injury or kainic acid. J Neurosci 21:3457–3475
Williams A, Piaton G, Lubetzki C (2007) Astrocytes—friends or foes in multiple sclerosis? Glia 55:1300–1312
Willson CA, Irizarry-Ramirez M, Gaskins HE et al (2002) Upregulation of EphA receptor expression in the injured adult rat spinal cord. Cell Transplant 11:229–239
Zhang D, Hu X, Qian L, O’Callaghan JP, Hong JS (2010) Astrogliosis in CNS pathologies: is there a role for microglia? Mol Neurobiol 41:232–241
Zimmermann H (2006) Nucleotide signaling in nervous system development. Pflugers Arch 452:573–588
Acknowledgements
The authors thank Luz C. Arocho and Laurivette Mosquera for the excellent technical assistance during surgeries and post-operatory procedures. Special thanks also to the personnel of the Animal Resources Center (University of Puerto Rico, Medical Science Campus) and the Experimental Surgery facilities. Our gratitude to Jose O. Garcia, Ph.D. for his critiques in the manuscript. This work was in partial fulfillment of Ana E. Rodríguez-Zayas doctoral dissertation and was supported by NIH-MRISP (2 R24 MH 48190–14), NIH-SNRP (NS39405), MBRS- SCORE (S06-GM008224), MBRS-RISE (GM-68138) and the Associated Deanship of Biomedical Sciences and Graduate Studies of the UPR School of Medicine. Editorial support was provided by Dr. Mary Helen Mays, Puerto Rico Clinical and Translational Research Consortium, funded by the National Center for Research Resources (NCRR) (1U54RR026139-01A1), a component of the National Institutes of Health (NIH). Its contents are solely the responsibility of the authors and do not necessarily represent the official view of NCRR or NIH.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rodríguez-Zayas, A.E., Torrado, A.I., Rosas, O.R. et al. Blockade of P2 Nucleotide Receptors After Spinal Cord Injury Reduced the Gliotic Response and Spared Tissue. J Mol Neurosci 46, 167–176 (2012). https://doi.org/10.1007/s12031-011-9567-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-011-9567-6