Abstract
The major pelvic ganglia (MPG) contain both parasympathetic and sympathetic postganglionic neurons and provide much of the autonomic innervation to urogenital organs and components of the lower bowel. Whereas many parasympathetic neurons were found to express vasoactive intestinal polypeptide (VIP), no MPG neurons exhibited immunoreactivity for pituitary adenylate cyclase-activating polypeptide (PACAP). However, in 3-day cultured MPGs, numerous PACAP-IR cells and nerve fibers were present, and transcript levels for PACAP increased significantly. In 3-day cultured MPGs, PACAP immunoreactivity was seen in cells that were also immunoreactive for VIP or neuronal nitric oxide synthase, but not tyrosine hydroxylase, indicating that PACAP expression occurred preferentially in MPG parasympathetic postganglionic neurons. Transcript levels for the VPAC2, but not VPAC1 or PAC1 receptor, also increased significantly following 3 days in culture. Transcript levels of activating transcription factor 3 (ATF-3), a marker of cellular injury, were increased 64-fold in 3-day explants, and ATF-3-IR nuclei were evident in both TH-IR and nNOS-IR neurons as well as in non-neuronal cells. In sum, these results demonstrate that, although only the parasympathetic neurons in explant cultured MPGs increase expression of PACAP, both sympathetic and parasympathetic postganglionic neurons in the cultured MPG whole-mount increase expression of ATF-3.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The major pelvic ganglia (MPG) provide the majority of the autonomic nerve supply to urogenital organs and components of the lower bowel (Keast 2006). This ganglion is a mixed ganglion containing both catecholaminergic sympathetic postganglionic and cholinergic parasympathetic postganglionic neurons. In many species, subpopulations of the parasympathetic cholinergic neurons in the MPG also express vasoactive intestinal polypeptide (VIP) or neuronal nitric oxide synthase (nNOS). In the peripheral nervous system, pituitary adenylate cyclase-activating polypeptide (PACAP) often is co-localized with VIP (Fahrenkrug and Hannibal 2004). However, no prior studies have determined whether PACAP is present within the mouse MPG. Therefore, experiments were done to test whether neurons and/or nerve fibers immunoreactive for PACAP are present in the male mouse MPG.
It is well-established that autonomic postganglionic neurons increase expression of a number of neuropeptides, including VIP and PACAP in response to injury (Hyatt-Sachs et al. 1993; Mohney et al. 1994; Moller et al. 1997; Suarez et al. 2006). The increased expression of these neuropeptides is postulated to support cell survival and regeneration (Hyatt-Sachs et al. 1993; Mohney et al. 1994; Moller et al. 1997; Girard et al. 2007). However, no studies have analyzed injury-induced changes in neuropeptide expression in MPG neurons. Consequently, we have completed studies using an explant cultured mouse MPG whole-mount preparation to identify injury-induced changes in the neuronal expression of VIP, PACAP, and the receptors activated by these closely related peptides. We also tested, during explant culture, whether the MPG neurons increase expression of activating transcription factor 3 (ATF-3), a well-established injury marker (Tsujino et al. 2000), which is up-regulated in sympathetic neurons in vivo following axotomy (Hyatt-Sachs et al. 2007) and in vitro during explant culture of guinea pig cardiac ganglia (Young et al. 2008).
Our results demonstrated that PACAP, but not VIP, transcript levels increased during culture. In addition, PACAP was preferentially expressed in cultured MPG parasympathetic postganglionic neurons, whereas ATF-3 expression increased in all MPG neurons. Our results also indicated that transcript levels for the VIP/PACAP receptor 2 (VPAC2), but not the VIP/PACAP receptor 1 (VPAC1) or the PACAP selective receptor (PAC1), increased in explant cultured ganglia.
Experimental Procedures
Preparation
Experiments were performed in vitro on whole-mount preparations containing the MPG from 26 male C57BL6 mice (4–5 weeks). Protocols for use of mice were approved by the University of Vermont IACUC and followed NIH guidelines. When cultured, the MPGs were removed under sterile conditions and maintained at 37°C in culture media consisting of DMEM–F12 (1:1) containing 10% horse serum, gentamicin (10 g/ml), amphotericin B (3.75 g/ml), penicillin (100 U/ml), and streptomycin (100 g/ml; Sigma, St. Louis, MO). The preparations were pinned on a sylgard-coated petri dish, which was placed on a wave platform shaker in a 5% CO2–95% air incubator (37°C) and kept for 4, 8, 24, 48, or 72 h, with the culture media replaced every 24 h.
Immunohistochemistry
MPG whole-mount preparations were fixed in 2% paraformaldehyde containing 0.2% picric acid for 2 h at 4°C. When mouse primary antibodies were used, the petri dish containing the pinned MPG whole-mount was placed on the wave platform shaker and the preparations bathed for 4 h in the culture media prior to fixation. The tissues were then fixed and rinsed in phosphate-buffered saline, permeabilized with 0.5% Triton X-100, and incubated at 4°C overnight with combinations of the primary antiserum. The primary antisera were then removed, and the whole-mounts were incubated for 2 h at room temperature with fluorescein isothiocyanate (FITC)-conjugated or indocarbocyanine (Cy3)-conjugated secondary antiserum (Jackson Immunoresearch Laboratories, West Grove, PA), washed again, and mounted with Citifluor (UKA Chemical Laboratory, Canterbury, England). The whole-mount preparations were viewed with an Olympus AX70 fluorescence microscope equipped with HBO 100-W UV light source and filters for FITC and Cy3. Digital images were obtained with a CCD camera (MagnaFire SP; Optronics; Optical Analysis Corp., Nashua, NH) and imported into Adobe Photoshop CS3 (Adobe Corporation, Mountain View, CA) to assemble figures, which were minimally adjusted for contrast and brightness.
Antibodies
Primary antisera used in this study included: a rabbit anti-ATF-3, 1:1,000 (Santa Cruz Biotechnology, Santa Cruz, CA; lot no. C0707); a mouse monoclonal anti-PACAP 1:10, from Dr. Jan Fahrenkrug, Copenhagen, Denmark; a rabbit anti-VIP 1:2,000, from Dr. John Walsh, UCLA School of Medicine, USA; a mouse anti-brain nitric oxide synthase 1:500, (Sigma, St Louis, MO; lot no. 037K4765); a rabbit anti-nNOS, 1:500 (Sigma, St Louis, MO; lot no. D30859); a mouse anti-microtubule associated protein 2 (MAP-2), 1:200 (Millipore, Billerica, MA; lot no.1284959); and a mouse anti-S-100 (beta subunit) 1/1,000 (Sigma, St Louis, MO; clone SH-D4).
All antisera used are well-characterized and have been employed in our prior studies (Mawe et al. 1996; Calupca et al. 2000a, b; Young et al. 2008). In addition, neuronal staining was not observed when whole-mounts were treated only with primary or secondary antiserum. No VIP immunolabeling was seen in MPG neurons from VIP knockout mice (Studeny et al. 2008).
Real-Time Quantitative Reverse Transcription-Polymerase Chain Reaction
MPG preparations were dissected under RNase-free conditions, and total RNA was extracted from individual preparations using Tri reagent (Sigma). The total RNA quantity for each whole-mount preparation was determined with a Nanodrop ND-1000 spectrophotometer (Nanodrop Technologies, Wilmington, DE). One microgram of RNA per sample was used to synthesize complementary DNA with the Omniscript reverse transcription (Qiagen, Valencia, CA) and a mix of oligo-dT and random hexamer primers. Amplified MPG DNA product from specific primers was ligated into pCR2.1 TOPO using TOPO TA cloning kit (Invitrogen, Carlsbad, CA) to generate plasmid standards. The nucleotide sequences of the inserts were verified by automated fluorescent dideoxy dye terminator sequencing (Vermont Cancer Center DNA Analysis Facility), and tenfold serial dilutions of stock plasmid were prepared to generate assay standard curves. Amplification of the mouse cDNA templates and plasmid standards was performed using HotStart IT® SYBR® Green qPCR Master Mix (USB). Real-time quantitative PCR was performed on an ABI 7500 Fast Real-Time PCR System (Applied Biosystems, Norwalk, CT). All quantitative data were normalized to 18S relative expression. Data are represented as a fold change compared with the average 0 day time point level. All of the primers have been used in previous studies (Braas and May 1999; Girard et al. 2002, 2006; Young et al. 2008).
Statistical Analyses
Student's t test was used to evaluate differences among groups. Differences were considered statistically significant at P ≤ 0.05.
Results
Many VIP-IR, but No PACAP-IR, Neurons Are Present in the MPG
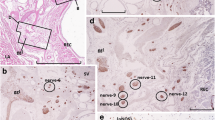
Many mouse MPG neurons exhibit immunoreactivity to VIP (Wanigasekara et al. 2003; Tompkins et al. 2010). These neurons are thought to be parasympathetic postganglionic neurons (Wanigasekara et al. 2003). A significant percentage of the MPG neurons were also labeled by an antibody directed against nNOS and in double-labeled preparations; many of the neuronal nitric oxide synthase-immunoreactive (nNOS-IR) neurons exhibited immunoreactivity for VIP, indicating the nNOS-IR neurons are parasympathetic postganglionic neurons (Fig. 1a, b). However, not all vasoactive intestinal polypeptide-immunoreactive (VIP-IR) cells were nNOS immunoreactive, and not all nNOS-IR neurons were immunoreactive for VIP, an observation indicating that VIP and nNOS can label different subgroups of parasympathetic postganglionic neurons. Although many VIP-IR and nNOS-IR neurons were present in freshly dissected and in 4-h cultured MPGs, there was no evidence of any pituitary adenylate cyclase-activating polypeptide-immunoreactive (PACAP-IR) neurons in the 4-h MPG cultures (data not shown).
Many MPG neurons are immunoreactive to both VIP and nNOS. a, b a 4-h rinsed MPG double-labeled with an antiserum directed against VIP (a) and an antiserum directed against nNOS (b). Many of the MPG neurons, examples indicated by arrows in a and b, are immunostained by both antisera. c, d A 4-h rinsed MPG double-labeled with an antiserum directed against VIP (c) and an antiserum directed against TH (d). None of the MPG neurons are immunostained by both antisera. e, f A 4-h rinsed MPG double-labeled with an antiserum directed against nNOS (e) and an antiserum directed against TH (f). None of the MPG neurons are immunostained by both antisera. Calibration equals 50 µm
In additional experiments, we determined that neither VIP nor nNOS was co-localized in tyrosine hydroxylase-immunoreactive (TH-IR) neurons (Fig. 1c–f), an observation providing further support that VIP and nNOS are present only in the parasympathetic postganglionic neurons as previously described (Wanigasekara et al. 2003).
PACAP is Expressed in 3-Day Explant Cultured MPG Neurons
Because PACAP expression increased in cultured guinea pig cardiac ganglia (Girard et al. 2007), we tested using QPCR whether transcript levels for PACAP increased in cultured MPGs. Transcript levels for PACAP were low in extracts from acutely isolated MPGs, but increased 16-fold (P ≤ 0.004) after 3 days in culture (Fig. 2a). In contrast, there was no significant difference in VIP transcript levels in extracts from acutely isolated MPGs and 3-day cultured MPGs (Fig. 2b).
Comparison of mRNA levels for PACAP and VIP, and PAC1, VPAC receptors, and ATF-3 in extracts of acutely isolated and 3-day explant cultured MPG whole-mount preparations. The transcript levels were normalized to the transcript level for 18S. a PACAP transcript levels were significantly increased in 3-day cultured MPGs (16-fold, P = 0.004) whereas mRNA levels for VIP (b) did not increase significantly. Data represent mean + SEM from six acutely isolated ganglia (0d) and six 3-day cultured ganglia (3d). c ATF-3 mRNA levels increase 84-fold after 3-days in explant culture. Data represent mean ± SEM from six acutely isolated ganglia (0d) and six 3-day cultured (3d) ganglia. The change in transcript level is significant (P = 0.004). Normalized PAC1 (d) and VPAC1 (e) are not different in extracts from acutely isolated or 3-day cultured MPGs whereas normalized VPAC2 (f) transcript expression is increased sixfold in extracts from 3-day cultured MPGs (P = 0.04; n = 4 for both 0 day and 3-day cultures MPGs)
In 3-day cultured MPG whole-mounts, many neurons exhibited immunoreactivity to PACAP (Fig. 3a, b). Also, many PACAP-IR fibers were evident in these 3-day cultured MPG explants. The neuropeptide-positive fibers were observed within the MPG and in nerve connectives with the intensity of fluorescent staining for PACAP being most intense at the ends of the fibers (Fig. 3c).
Increased PACAP Expression in the 3-Day Cultured MPG Occurs Primarily in Parasympathetic Postganglionic Neurons
As the MPG contains both sympathetic and parasympathetic postganglionic neurons, we determined whether the increased expression of PACAP occurred in both neuron types or preferentially in one type. Three-day cultured MPG whole-mounts were double-labeled using an antiserum directed against PACAP and antiserum directed against tyrosine hydroxylase (TH), nNOS, or VIP. No PACAP-IR neurons were identified that were also TH immmunoreactive (Fig. 4a1, 2). In contrast, many PACAP-IR neurons were also immunoreactive for nNOS (Fig. 4b1, 2) or VIP (Fig. 4c1, 2). We also noted that there were PACAP-IR cells that were not nNOS immunoreactive and vice versa. In addition, there were PACAP-IR neurons that were not VIP immunoreactive and vice versa.
PACAP-IR neurons in 3-day cultured MPGs, exhibit immunoreactivity for nNOS and VIP, but not TH. a Example micrographs from 3-day cultured MPGs double-labeled with antiserum directed against PACAP and antiserum directed against TH. a 1 Examples of PACAP-IR neurons. a 2 These neurons are not TH-IR although many TH-IR neurons are evident. b Example micrographs from 3-day cultured MPGs double-labeled with antiserum directed against PACAP and antiserum directed against nNOS. b 1 PACAP-R neurons and fibers. b 2 nNOS-IR neurons and fibers in the same MPG. Note that the PACAP-IR neuron indicated by the arrow in b 1 does not exhibit nNOS immunoreactivity (indicated by arrow in b 2), whereas the neuron indicated by an arrow head in b 1 and b 2 exhibits immunoreactivity for both PACAP and nNOS. Other nNOS-IR neurons in b 2 are not PACAP-immunoreactive. c Example micrographs from 3-day cultured MPGs double-labeled with antiserum directed against PACAP and antiserum directed against VIP. c 1 A group of PACAP-IR neurons. c 2 Some, but not all, of the PACAP-IR neurons in c 1 (indicated by arrows) also exhibit immunoreactivity for VIP (indicated by arrows in c 2). Calibration equals 50 µm
VPAC2, but Not VPAC1 or PAC1, Transcript Levels Increase in 3-Day Explant Cultured MPGs
Tompkins et al. (2010) determined that transcripts for VPAC1, VPAC2, and PAC1 receptors were present in the mouse MPG. Furthermore, VIP, PACAP, and maxidilan, the PAC1-selective agonist, all altered the electrical properties of MPG neurons, an observation suggesting all three of the VIP/PACAP receptors are expressed by the MPG neurons (Tompkins et al. 2010). Consequently, we tested whether transcript levels of these receptors changed during explant culture. QPCR results presented in Fig. 2f indicate that only the transcript level for the VPAC2 receptor was increased in extracts from 3-day cultured MPGs.
ATF-3 Expression Increases in Explant Cultured MPG Whole-Mounts
Because the expression of PACAP occurred preferentially in parasympathetic neurons in the 3-day cultured MPGs, we questioned whether the sympathetic neurons might have become unresponsive during explant culture. To test this, ATF-3 transcript levels were compared in extracts from freshly isolated and 3-day cultured MPG whole-mounts. In the control preparations, ATF-3 transcript levels were very low, but the transcript level increased 84-fold (P ≤ 0.004) after 3 days in culture (Fig. 2c).
Next, using immunostaining for ATF-3, we determined the time-dependence of the increase in ATF-3 expression. In freshly isolated or 4-h cultured MPG preparations, no neuronal or non-neuronal cell nuclei were ATF-3 immunoreactive (Fig. 5a). However, over time in explant culture, more nuclei of both the neurons and non-neuronal cells progressively exhibited ATF-3 immunoreactivity. By 8 h, most neuronal and non-neuronal cell nuclei exhibited ATF-3 immunoreactivity (Figs. 5b; 6a, b), and after 24 h or longer in culture, all neurons had ATF- 3 immunoreactive nuclei (Fig. 5c, d).
ATF-3 expression increases over time in explant cultured MPG neurons. a–d Micrographs of four different mouse MPGs cultured for 4, 8, 24, or 48 h, respectively, and immunolabeled with an antiserum directed against ATF-3. Note that, at 4 h, no neuronal nuclei are ATF-3-IR, whereas after 8 h in explant culture many of the neuronal nuclei are ATF-3-IR. Calibration bar equals 200 µm
Explant cultured (1 and 2 day) MPG were immunostained with a mouse monoclonal antibody against S-100, a glial cell marker, and with the rabbit anti-ATF-3 antiserum. The pattern of S-100 immunostaining was similar to that reported by Hyatt-Sachs et al. (2007) in mouse and rat superior cervical ganglion (SCG; data not shown). Based on the staining pattern, we suggest that small, elongated ATF-3 nuclei within nerve bundles were Schwann cell nuclei, and small, round activating transcription factor 3-immunoreactive (ATF-3-IR) nuclei which closely surrounded the ganglion cells were satellite cell nuclei.
ATF-3-IR nuclei were evident in neurons exhibiting TH immunoreactivity (Fig. 6c) and in nNOS-IR neurons (Fig. 6d). Thus, in cultured MPG, both sympathetic and parasympathetic postganglionic MPG neurons exhibited ATF-3 immunoreactivity.
Discussion
PACAP expression increased in MPG neurons maintained in explant culture. This was determined from both a change in transcript level as well as an increase in the number of immunolabeled cells. In contrast, in 3-day cultured MPGs, VIP transcript levels did not change. The MPG contains both sympathetic and parasympathetic postganglionic neurons. However, the increased expression of PACAP occurred preferentially in the parasympathetic postganglionic neurons. This finding was supported by the demonstration of PACAP in VIP or nNOS-IR neurons, but not in TH-IR neurons in 3-day cultured MPG.
PACAP expression increases following axotomy in rat sympathetic neurons and in cultured guinea pig parasympathetic cardiac neurons (Moller et al. 1997; Girard et al. 2007). We were surprised that, in the cultured MPG, the increased PACAP expression was limited to the parasympathetic postganglionic neurons. In contrast, both the parasympathetic and sympathetic postganglionic MPG neurons exhibited ATF-3-IR nuclei. Increased ATF-3 expression occurs consistently in autonomic and sensory neurons in response to axotomy (Tsujino et al. 2000; Boeshore et al. 2004; Hyatt-Sachs et al. 2007; Young et al. 2008). Thus, the sympathetic postganglionic neurons exhibit the injury marker, but do not respond to injury by increasing PACAP.
In explant cultured MPG, all neuronal and glial nuclei exhibited ATF-3 immunoreactivity. Similarly, in the rat SCG following axotomy or in the explant cultured guinea pig cardiac ganglia, both neuronal and non-neuronal nuclei exhibit ATF-3 immunoreactivity (Hyatt-Sachs et al. 2007; Young et al. 2008). The expression of ATF-3 occurred sooner in glial nuclei than in neuronal nuclei in the cultured mouse MPG as was also reported for explant cultured cardiac ganglia (Young et al. 2008). Recent results on dorsal root ganglion cells suggest that the up-regulation of ATF-3 increases the intrinsic growth state, thereby promoting axonal regeneration (Nakagomi et al. 2003; Seijffers et al. 2006, 2007). An injury-induced increased ATF-3 expression in the MPG very likely could be a key step in setting up the neuronal regenerative process.
Up-regulation of PACAP occurs in vivo and in vitro following axotomy of autonomic and sensory neurons (Mohney et al. 1994; Moller et al. 1997; Girard et al. 2007). The increased expression of PACAP has been suggested to promote cell survival and regeneration (Moller et al. 1997; Suarez et al. 2006) although the mechanism(s) through which this increased expression might support cell survival and regeneration is not established. During explant culture, transcript levels of the VPAC2, but not VPAC1 or PAC1 receptor, also increased. The function of the increased VPAC2 expression remains to be determined. In future experiments, we will test how an increased expression of VPAC2 receptors along with the increased PACAP expression in MPG might support neuropeptide-induced regenerative mechanisms.
ATF-3 immunoreactivity is evident in non-neuronal cell nuclei and nuclei of both TH-IR and nNOS-IR neurons. a, b 8-h cultured MPG whole-mounts were double-labeled with an antiserum directed against MAP-2, a marker for neurons, and an antiserum directed against ATF-3. a Most, but not all, neuronal nuclei are ATF-3 positive. Arrows indicate MAP-2 positive neurons with nuclei that are ATF-3 negative. b Non-neuronal cell nuclei also express ATF-3. Note that the non-neuronal cells, presumed to be Schwann cells, have small oblong ATF-3-IR nuclei. c A 24-h cultured MPG whole-mount double-labeled with antiserum against ATF-3 (Cy3 secondary) and antiserum against TH (FITC secondary). Note that nuclei of TH-IR neurons also are ATF-3 immunoreactive. d Another 24-h cultured MPG whole-mount double-labeled with antiserum against ATF-3 (Cy3 secondary) and antiserum against nNOS (FITC secondary). Note that nuclei of nNOS-IR neurons also are ATF-3-immunoreactive. Scale bars represent 50 µm
References
Boeshore KL, Schreiber RC, Vaccariello SA et al (2004) Novel changes in gene expression following axotomy of a sympathetic ganglion: a microarray analysis. Eur J NeuroSci 59:216–235
Braas KM, May V (1999) Pituitary adenylate cyclase-activating polypeptides directly stimulate sympathetic neuron neuropeptide Y release through PAC(1) receptor isoform activation of specific intracellular signaling pathways. J Biol Chem 274(39):27702–27710
Calupca MA, Vizzard MA, Parsons RL (2000a) Origin of pituitary adenylate cyclase-activating polypeptide (PACAP)-immunoreactive fibers innervating guinea pig parasympathetic cardiac ganglia. J Comp Neurol 423:26–39
Calupca MA, Vizzard MA, Parsons RL (2000b) Origin of neuronal nitric oxide synthase (nNOS)-immunoreactive fibers in guinea pig parasympathetic cardiac ganglia. J Comp Neurol 426:493–504
Fahrenkrug J, Hannibal J (2004) Neurotransmitters co-existing with VIP or PACAP. Peptides 25:393–401
Girard BM, May V, Bora SH, Fina F, Braas KM (2002) Regulation of neurotrophic peptide expression in sympathetic neurons: quantitative analysis using radioimmunoassay and real-time quantitative polymerase chain reaction. Regul Pept 109(1–3):89–101
Girard BM, Lelievre V, Braas KM et al (2006) Noncompensation in peptide/receptor gene expression and distinct behavioral phenotypes in VIP- and PACAP-deficient mice. J Neurochem 99(2):499–513
Girard BM, Young B, Buttolph T, White S, Parsons RL (2007) Regulation of pituitary adenylate cyclase-activating polypeptide (PACAP) expression during culture of guinea-pig cardiac ganglia. Neuroscience 146:584–593
Hyatt-Sachs H, Schreiber RC, Bennett TA, Zigmond RE (1993) Phenotypic plasticity in adult sympathetic ganglia in vivo: effects of deafferentation and axotomy on the expression of vasoactive intestinal peptide. J Neurosci 13(4):1642–1653
Hyatt-Sachs H, Schreiber RC, Shoemaker SE, Sabe A, Reed E, Zigmond RE (2007) Activating transcription factor 3 induction in sympathetic neurons after axotomy: response to decreased neurotrophin availability. Neuroscience 15(4):887–897
Keast JR (2006) Plasticity of pelvic autonomic ganglia and urogenital innervation. Int Rev Cytol 248:141–208
Mawe GM, Talmage EK, Lee KP, Parsons RL (1996) Expression of choline acetyltransferase immunoreactivity in guinea pig cardiac ganglia. Cell Tissue Res 285:281–286
Mohney RP, Siegel RE, Zigmond RE (1994) Galanin and vasoactive intestinal peptide messenger RNAs increase following axotomy of adult sympathetic neurons. J Neurobiol 25(2):108–118
Moller K, Reimer M, Ekblad E et al (1997) The effects of axotomy and preganglionic denervation on the expression of pituitary adenylate cyclase activating peptide (PACAP), galanin and PACAP type 1 receptors in the rat superior cervical ganglion. Brain Res 775:166–182
Nakagomi S, Suzuki Y, Namikawa K, Kiryu-Seo S, Kiyama H (2003) Expression of the activating transcription factor 3 prevents c-Jun N-terminal kinase-induced neuronal death by promoting heat shock protein 27 expression and Akt activation. J Neurosci 23:5187–5196
Seijffers R, Allchorne AJ, Woolf CJ (2006) The transcription factor ATF-3 promotes neurite outgrowth. Mol Cell Neurosci 32:143–154
Seijffers R, Mills CD, Woolf CJ (2007) ATF3 increases the intrinsic growth state of DRG neurons to enhance peripheral nerve regeneration. J Neurosci 27(30):7911–7920
Studeny S, Cheppudira BP, Meyers S et al (2008) Urinary bladder function and somatic sensitivity in vasoactive intestinal polypeptide (VIP)-/- mice. J Mol Neurosci 36(1–3):175–187
Suarez V, Guntinas-Lichius O, Streppel M et al (2006) The axotomy-induced neuropeptides galanin and pituitary adenylate cyclase-activating peptide promote axonal sprouting of primary afferent and cranial motor neurones. Eur J NeuroSci 24:1555–1564
Tompkins JD, Girard BM, Vizzard MA, Parsons RL (2010) VIP and PACAP effects on mouse major pelvic ganglia neurons. J Mol Neurosci (in press)
Tsujino H, Kondo E, Fukuoka T et al (2000) Activating transcription factor 3 (ATF3) induction by axotomy in sensory and motoneurons: a novel neuronal marker of nerve injury. Mol Cell Neurosci 15(2):170–182
Wanigasekara Y, Kepper ME, Keast JR (2003) Immunohistochemical characterisation of pelvic autonomic ganglia in male mice. Cell Tissue Res 311(2):175–185
Young BA, Girard B, Parsons RL (2008) Neurturin suppresses injury-induced neuronal activating transcription factor 3 (ATF-3) expression in cultured guinea pig cardiac ganglia. J Comp Neurol 508:795–805
Acknowledgements
The study was supported in part by NCRR Grant P20 RR16435 (RLP) and R01 DK060481 and R01 DK051369 (MAV). We thank Dr. Jan Fahrenkrug for kindly providing the PACAP antiserum.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Girard, B.M., Galli, J.R., Young, B.A. et al. PACAP Expression in Explant Cultured Mouse Major Pelvic Ganglia. J Mol Neurosci 42, 370–377 (2010). https://doi.org/10.1007/s12031-010-9359-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-010-9359-4