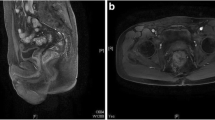
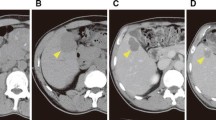
Abstract
Purpose
The objective of our systematic review was to evaluate the long-term outcomes of surgical resection as a treatment strategy for liver and pancreatic metastases from thyroid cancer (TC).
Methods
A systematic search of three electronic databases for articles published up to October 2018 was conducted. All appropriate observational studies and case reports which reported outcomes from patients with TC metastatic to the liver or pancreas were considered eligible for inclusion in the present systematic review.
Results
A total of 15 studies, which comprised of 16 patients that underwent hepatic or pancreatic resection for TC metastasis, were included in the present systematic review; among them, 5 presented with metastasis to the liver, whereas 11 had pancreatic metastatic disease. The median time interval between the initial thyroidectomy and the diagnosis of metastases (either hepatic or pancreatic) was 60 months (SE 23.8, 95% CI 13.3–106.7) for the entire cohort and the mean overall survival was 37.6 months (SE 8, 95% CI 22–53.3). Five patients with pancreatic metastases presented with recurrence whereas no recurrences were noted in patients with liver metastases.
Conclusions
Surgical resection of liver and pancreatic metastases from TC seems to be a safe and efficient treatment option for selected patients. In that setting, long-term outcomes in patients with resected TCLM are encouraging given the absence of recurrence as reported from the included studies, whereas in the case of TCPM, survival is limited due to advanced disease at diagnosis and recurrence rates.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Thyroid carcinomas (TC) are uncommon and constitute less than 1% of all human cancers, despite the high frequency of thyroid nodules [1]. The worldwide annual incidence ranges from 0.5 to 10 cases per 100,000 habitants [2], whereas distant metastases from differentiated TC have been shown to range from 4 to 23% and are described as the most frequent cause of TC-related death [3,4,5]. The most common sites for distant metastases from thyroid carcinomas include the lung, followed by the bones, the brain, and then the liver [6]. Isolated hepatic metastases are very rare with a reported frequency of less than 0.5% [7]. Moreover, the pancreas is another extremely unusual location for distant metastases from TC [8]. The incidence of pancreatic secondary tumors was 15% in autopsy cases, whereas the incidence of pancreatic metastasis from TC ranged from 0.6 to 1.0% of pancreatic secondary tumors [9]. Liver and pancreatic metastases from differentiated thyroid cancer (DTC), both follicular (FTC) and papillary (PTC), are usually closely associated with an advanced disease stage and increased aggressiveness [7].
While surgical resection of liver and pancreatic metastases from several non-colorectal non-endocrine primary lesions has been shown to benefit patients in terms of long-term outcomes, the effect of surgical resection of TC metastases in the liver and pancreas remains ill determined [10]. Moreover, several treatment strategies for the management of liver and pancreatic metastases from TC, such as surgical resection, radiofrequency ablation (RFA), radioactive iodine therapy (RAI), and chemotherapy have been reported, whatsoever the number of included patients is limited [11]. To that end, the objective of our systematic review was to evaluate the long-term outcomes of surgical resection as a treatment strategy for liver and pancreatic metastases from TC.
Materials and Methods
Study Design
All appropriate observational studies and case reports which reported outcomes from patients with TC metastatic to the liver or pancreas were considered eligible for inclusion in the present systematic review. Reviews and animal studies were excluded from analysis and tabulation. English-language trials were included. NM and APa independently performed a meticulous search of the literature, excluded overlaps, and tabulated the selected indices in structured forms.
Search Strategy and Data Collection
We systematically searched for articles published up to October 2018 using Medline (1966–2018), Scopus (2004–2018), and Google Scholar (2004–2018) databases along with the references of the articles, which were retrieved in full text. The following keywords were used for the search: “thyroid cancer,” “hepatic metastasis,” “pancreatic metastasis,” “metastatic thyroid cancer.” A minimum number of search keywords was utilized in an attempt to assess an eligible number that could be easily searched while simultaneously minimizing the potential loss of articles. Articles that fulfilled or were deemed to fulfill the inclusion criteria were retrieved; all articles published from January 2000 which described cases of patients aged > 18 years who underwent surgical management of liver or hepatic metastasis of thyroid cancer were included. The PRISMA flow diagram schematically presents the stages of article selection (Fig. 1).
Data on patient characteristics included age, gender, characteristics of the primary and metastatic tumor, and type of surgery. Concerning the main findings of the study, survival rates with regard to incidence of recurrence and cancer-related deaths as well as overall survival were appraised.
Statistical Analysis
Kaplan-Meier curves were used for the assessment of survival and the comparisons of survival among different groups were made using the log-rank test. Cox regression was used for multivariate survival analysis. All the tests were two-tailed. Results were considered significant if p value was less than 0.05. Statistical analysis was performed with the 25th edition of Statistical Package for Social Sciences (SPSS) (IBM Corporation, Armonk, NY, USA).
Results
Excluded Studies
A total of 4 studies were excluded from the present review (Fig. 1) [11,12,13,14]. Among them, two underwent only intraoperative radiofrequency ablation without parenchymal resection and were excluded [11, 12]. The study by Ibarrola et al. was excluded due to insufficient data concerning treatment strategy and long-term outcomes [13]. The study of Andreou et al. was also excluded due to inadequate specific data about patients’ surgical management and survival [14].
Included Studies
A total of 15 studies (case reports and case series), which comprised of 16 patients that underwent hepatic or pancreatic resection for TC metastasis, were included in the present systematic review. Among them, five presented with metastasis to the liver, whereas 11 had pancreatic metastatic disease [15,16,17,18,19,20,21,22,23,24,25,26,27,28,29]. The analyzed indices were structured in Table 1, which shows main demographic and tumor characteristics, along with the main outcomes after surgery according to the site of metastasis (hepatic or pancreatic).
Thyroid Cancer Liver Metastasis (TCLM)
The mean age of patients with TCLM was 54.8 ± 15.6 years whereas the male to female ratio was 0.25. One patient (20%) was diagnosed with papillary (PTC), 2 patients (40%) with follicular (FTC), 1 (20%) with both PTC and medullary (MTC), and finally one patient (20%) with differentiated primary TC. Only 1 patient (20%) presented with lymph node metastasis during the primary TC resection. The mean metastasis size was 2.7 ± 1.12 cm, whereas the liver tumors in all patients were solitary. One patient (20%) underwent laparoscopic liver resection (LLR) and four patients (80%) underwent open liver resection (OLR). Only one patient (20%) received postoperative adjuvant chemotherapy. The mean time of follow-up after hepatectomy was 22.2 ± 19.9 months. All patients were alive at last follow-up whereas no recurrence of disease was reported.
Thyroid Cancer Pancreatic Metastasis (TCPM)
The mean age of patients with TCPM was 54.8 ± 12 years whereas male to female ratio was 0.83. Seven patients (63.6%) were diagnosed with PTC, 1 (9%) with MTC, 1 (9%) with FTC, 1 (9%) had both PTC and MTC, and 1 patient (9%) was diagnosed generally with differentiated primary TC. Nine patients (81.8%) presented cervical lymph node metastasis. At the time of diagnosis, 6 patients had additional metastases at other sites including cervical lymph nodes (n = 2), bone (n = 2), brain (n = 1), and lung (n = 2). All but 1 patient underwent curative intent pancreatectomy; the patient reported by Meyer et al. underwent resection primarily due to symptom relief (massive duodenal bleeding) [16]. Six patients (54.5%) underwent Whipple procedure, 4 patients (36.3%) underwent distal pancreatectomy (DP), and 1 patient (9%) underwent enucleation of the pancreatic lesion. Three patients (27.3%) received adjuvant chemotherapy postoperatively. The mean metastasis size was 3.7 ± 2.1 cm. The mean time of follow-up after pancreatectomy was 17.7 ± 16.4 months. Five patients (45.5%) presented with recurrence of disease after pancreatectomy whereas 5 (45.5%) patients died due to disease progression. The location of recurrence in these 5 patients was the lung (n = 3), bone (n = 2), skin (n = 1), and liver (n = 2).
Survival Analysis
The median time interval between the initial thyroidectomy and the diagnosis of metastases (either hepatic or pancreatic) was 60 months (SE 23.8, 95% CI 13.3–106.7) for the entire cohort. The median time interval was 60 months (SE 26.3, 95% CI 8.5–111.5) for hepatic metastases and also 60 months (SE 45.7, 95% CI 0–149.6) for pancreatic metastases (p = 0.52). The lack of statistical significance was also confirmed in the multivariate survival analysis (p = 0.433). The only independent prognostic factor of sooner manifestation of metastasis in the multivariate survival analysis was the presence of medullary thyroid cancer (HR 18.6, 95% CI 1.3–274.2, p = 0.034).
There were 5 deaths (31%) in the entire cohort. Therefore, the median overall survival had not been reached yet. As far as metastases in liver are concerned, the median overall survival had not been reached yet and there were no deaths (0%). On the other hand, there were 5 deaths (45.45%) in cases with pancreatic metastases. Thus, the median overall survival had also not been reached yet. However, the mean overall survival was 33.2 months (SE 8.6, 95% CI 16.3–50.1). Nevertheless, this difference was not statistically significant (p = 0.093). The lack of statistical significance was also confirmed in the multivariate survival analysis (p = 0.962).
Five cases (31%) of recurrence after resection of metastases were noted in the entire cohort. Therefore, the median recurrence-free survival had not been reached yet. In regard to hepatic metastases, there were no recurrences (0%) and the median recurrence-free survival had not been reached yet. On the other hand, there were 5 recurrences (45.45%) in cases with pancreatic metastases. Thus, the median recurrence-free survival had not been reached yet. However, the mean overall survival was 37.6 months (SE 8, 95% CI 22–53.3). No significant difference between these two metastatic sites was detected (p = 0.131). The lack of statistical significance was also confirmed in the multivariate survival analysis (p = 0.702).
The results of multivariate survival analysis are listed in Table 2.
Discussion
The aim of our systematic review was to accumulate current evidence on long-term outcomes of patients with metachronous TCLM and TCPM undergoing surgical resection. According to data from the included studies, resections of metachronous TCLM and TCPM are feasible and associated with acceptable long-term outcomes particularly for the TCLM patients. The presence of primary MTC was also associated with a shorter interval of presentation of liver and pancreatic metastases. Finally, although patients with resected TCPM seem to recur more frequently than patients with resected TCLM, no difference was shown with regard to overall survival.
A plethora of biochemical markers and imaging modalities have been used for the diagnosis of metachronous metastatic lesions from TC. In our review, computed tomography (CT) was the most frequently used modality followed by magnetic resonance imaging (MRI) and 18F-fluorodeoxyglucose positron emission tomography (FDG-PET). FDG-PET and 131I imaging can be both diagnostic and prognostic, since metastatic lesions that are FDG-avid and RAI-negative indicate a high-grade tumor and thus worse prognosis. In our review, 3 out of 5 patients with TCPM that recurred had FDG-avid tumors. Additionally, the most reliable biochemical marker of recurrence of TC is elevated serum levels of thyroglobin (Tg). Ten out of 16 patients (62.5%) of our study had elevated serum values of Tg during the preoperative period and it was the indication for further chest and abdomen imaging, in order to identify the exact location of metastatic lesion [15,16,17, 19, 20, 25, 26, 28, 29]. With regard to preoperative biopsy in order to confirm the exact nature of the metastatic lesion, endoscopic ultrasound-guided fine needle aspiration (EUS-FNA) and fine needle aspirational biopsy (FNAB) have been proposed as safe and accurate modalities in the cases of TCPM and TCLM, respectively [13, 15]. Alzahrani et al. suggested performing EUS-FNA for pancreatic lesions suspicious for TC metastasis before considering resection [30].
In the present study, we reviewed only patients with TCLM and TCPM who underwent surgical resection as their main treatment strategy, whatsoever application of other invasive strategies has been reported. The results of this study demonstrate that surgical resection should be considered in patients with isolated hepatic metastasis amenable to resection. Unfortunately, a majority of hepatic metastases from TC are part of generalized metastatic disease and as a result complete resection may not be feasible. For these patients, Saito et al. reported favorable results (1-year overall survival 71.4% vs. 26.7%) after treatment with lenvatinib in patients with good physical status [31]. Wertenbroek et al. presented 3 cases of patients with TCLM that underwent open, laparoscopic, and CT-guided radiofrequency ablation, respectively [11]. Furthermore, Segkos et al. presented a 62-year-old man with a TCLM located deeply in the right lobe, which underwent laparoscopic microwave ablation [12]. Patients not deemed eligible for major resections may benefit from such minimally invasive procedures, which are accompanied by lower morbidity and mortality [11, 12], while patients with widespread disease should instead undergo systemic treatment with tyrosine-kinase inhibitors [31]. The role of pancreatic resection for TC metastasis remains to be defined, since it is generally associated with more advanced metastatic disease, considerable morbidity and almost half of included patients in this study demonstrated disease recurrence after a median period of 11 months. Careful patient selection should be applied to identify patients that may benefit from it, such as those that can tolerate pancreatic surgery and have isolated pancreatic metastasis and/or intractable symptoms. Adjuvant chemotherapy possibly has a role in patients after liver or pancreatic resections, especially in advanced disease stages but has not yet been elucidated due to the small number of patients. One out of 5 patients (20%) who underwent liver resection for TCLM received adjuvant chemotherapy [19] and 3 out of 11 patients (27.2%) who had TCPM received paclitaxel, sorafenib, and carboplatinum with adriamycin respectively [22, 25, 28].
The present study is to the best of our knowledge the first systematic review assessing outcomes from patients undergoing curative-intent surgical resection of hepatic and pancreatic metastases from TC. Whatsoever, some limitations need to be addressed before interpreting the results of the present study. Firstly, the most important being that our outcomes are derived from case reports and small case series thus precluded further definite conclusions with regard to prevalence and management of those rare metastatic sites of TC. Consequently, the small number of patients limits the strength of our statistical analysis. Finally, the fact that some parameters were omitted by some studies constitutes another limitation.
To conclude, surgical resection of TCLM and TCPM seems to be a safe and efficient treatment option for selected patients. In that setting, long-term outcomes in patients with resected TCLM are encouraging given the absence of recurrence as reported from the included studies, whereas in the case of TCPM, survival is limited due to advanced disease at diagnosis and recurrence rates. Further studies are needed in order to validate surgical resection as a beneficial treatment strategy in patients with resectable secondary hepatic and pancreatic diseases from TC, as well as to decipher the patterns of recurrence in patients undergoing curative-intent resection.
References
Jones MK. Management of papillary and follicular thyroid cancer. J R Soc Med. 2002;95:325–6.
Schlumberger MJ. Papillary and follicular thyroid carcinoma. N Engl J Med. 1998;338:297–306. https://doi.org/10.1056/NEJM199801293380506.
Haq M, Harmer C. Differentiated thyroid carcinoma with distant metastases at presentation: prognostic factors and outcome. Clin Endocrinol. 2005;63:87–93. https://doi.org/10.1111/j.1365-2265.2005.02304.x.
Hoie J, Stenwig AE, Kullmann G, Lindegaard M. Distant metastases in papillary thyroid cancer. A review of 91 patients. Cancer. 1988;61:1–6.
Shaha AR, Shah JP, Loree TR. Differentiated thyroid cancer presenting initially with distant metastasis. Am J Surg. 1997;174:474–6.
Ito Y, Miyauchi A. Prognostic factors and therapeutic strategies for differentiated carcinomas of the thyroid. Endocr J. 2009;56:177–92.
Song HJ, Xue YL, Qiu ZL, Luo QY. Uncommon metastases from differentiated thyroid carcinoma. Hell J Nucl Med. 2012;15:233–40. https://doi.org/10.1967/s002449910059.
Sperti C, Moletta L, Patane G. Metastatic tumors to the pancreas: the role of surgery. World J Gastrointest Oncol. 2014;6:381–92. https://doi.org/10.4251/wjgo.v6.i10.381.
Nakamura E, Shimizu M, Itoh T, Manabe T. Secondary tumors of the pancreas: clinicopathological study of 103 autopsy cases of Japanese patients. Pathol Int. 2001;51:686–90.
Crippa S, Angelini C, Mussi C, Bonardi C, Romano F, Sartori P, et al. Surgical treatment of metastatic tumors to the pancreas: a single center experience and review of the literature. World J Surg. 2006;30:1536–42. https://doi.org/10.1007/s00268-005-0464-4.
Wertenbroek MW, Links TP, Prins TR, Plukker JT, van der Jagt EJ, de Jong KP. Radiofrequency ablation of hepatic metastases from thyroid carcinoma. Thyroid. 2008;18:1105–10. https://doi.org/10.1089/thy.2008.0080.
Segkos K, Schmidt C, Nabhan F. Isolated liver metastasis in Hurthle cell thyroid cancer treated with microwave ablation. Case Rep Endocrinol. 2017;2017(2790741):1–4. https://doi.org/10.1155/2017/2790741.
Ibarrola de Andres C, Castellano Megias VM, Ballestin Carcavilla C, Alberti Masgrau N, Perez Barrios A, De Agustin PA. Hepatic metastases from the spindle cell variant of medullary thyroid carcinoma: report of a case with diagnosis by fine needle aspiration biopsy. Acta Cytol. 2001;45:1022–6. https://doi.org/10.1159/000328348.
Andreou A, Brouquet A, Bharathy KG, Perrier ND, Abdalla EK, Curley SA, et al. Liver resection for liver metastases from nondigestive endocrine cancer: extrahepatic disease burden defines outcome. Surgery. 2012;151:851–9. https://doi.org/10.1016/j.surg.2011.12.025.
Siddiqui AA, Olansky L, Sawh RN, Tierney WM. Pancreatic metastasis of tall cell variant of papillary thyroid carcinoma: diagnosis by endoscopic ultrasound-guided fine needle aspiration. JOP. 2006;7:417–22.
Meyer A, Behrend M. Is pancreatic resection justified for metastasis of papillary thyroid cancer? Anticancer Res. 2006;26:2269–73.
Djenic B, Duick D, Newell JO, Demeure MJ. Solitary liver metastasis from follicular variant papillary thyroid carcinoma: a case report and literature review. Int J Surg Case Rep. 2015;6C:146–9. https://doi.org/10.1016/j.ijscr.2014.11.080.
Zhou J, Luo Y, Ma BY, Ling WW, Zhu XL. Contrast-enhanced ultrasound diagnosis of hepatic metastasis of concurrent medullary-papillary thyroid carcinoma: a case report. Medicine. 2017;96:e9065. https://doi.org/10.1097/MD.0000000000009065.
Besic N, Schwarzbartl-Pevec A, Vidergar-Kralj B, Crnic T, Gazic B, Marolt Music M. Treatment and outcome of 32 patients with distant metastases of Hurthle cell thyroid carcinoma: a single-institution experience. BMC Cancer. 2016;16(162):162. https://doi.org/10.1186/s12885-016-2179-3.
Salvatori M, Perotti G, Rufini V, Maussier ML, Summaria V, Fadda G, et al. Solitary liver metastasis from Hurthle cell thyroid cancer: a case report and review of the literature. J Endocrinol Investig. 2004;27:52–6. https://doi.org/10.1007/BF03350911.
Kouso H, Ikegami T, Ezaki T, Ishida T, Aimitsu S, Fujihara M, et al. Liver metastasis from thyroid carcinoma 32 years after resection of the primary tumor: report of a case. Surg Today. 2005;35:480–2. https://doi.org/10.1007/s00595-004-2949-4.
Jobran R, Baloch ZW, Aviles V, Rosato EF, Schwartz S, LiVolsi VA. Tall cell papillary carcinoma of the thyroid: metastatic to the pancreas. Thyroid. 2000;10:185–7. https://doi.org/10.1089/thy.2000.10.185.
Angeles-Angeles A, Chable-Montero F, Martinez-Benitez B, Albores-Saavedra J. Unusual metastases of papillary thyroid carcinoma: report of 2 cases. Ann Diagn Pathol. 2009;13:189–96. https://doi.org/10.1016/j.anndiagpath.2008.02.010.
Niess H, Conrad C, Kleespies A, Haas F, Bao Q, Jauch KW, et al. Surgery for metastasis to the pancreas: is it safe and effective? J Surg Oncol. 2013;107:859–64. https://doi.org/10.1002/jso.23333.
Murakami Y, Shimura T, Okada R, Kofunato Y, Ishigame T, Yashima R, et al. Pancreatic metastasis of papillary thyroid carcinoma preoperatively diagnosed by endoscopic ultrasound-guided fine-needle aspiration biopsy: a case report with review of literatures. Clin J Gastroenterol. 2018;11:521–9. https://doi.org/10.1007/s12328-018-0875-z.
Na HJ, Kim BH, Kim JR, Oh MY, Kim SM, Song BG, et al. FDG PET/CT in the detection of pancreatic metastasis in a patient with follicular thyroid carcinoma and negative I-131 whole body scan findings. Intern Med. 2014;53:2095–9.
Li XO, Li ZP, Wang P, Li CL, Wu JH, Zhang JZ, et al. Pancreatic metastasis of papillary thyroid carcinoma: a case report with review of the literature. Int J Clin Exp Pathol. 2014;7:819–22.
Tunio MA, Alasiri M, Riaz K, Alshakweer W. Pancreas as delayed site of metastasis from papillary thyroid carcinoma. Case Rep Gastrointest Med. 2013;2013(386263):1–4. https://doi.org/10.1155/2013/386263.
Borschitz T, Eichhorn W, Fottner C, Hansen T, Schad A, Schadmand-Fischer S, et al. Diagnosis and treatment of pancreatic metastases of a papillary thyroid carcinoma. Thyroid. 2010;20:93–8. https://doi.org/10.1089/thy.2009.0026.
Alzahrani AS, AlQaraawi A, Al Sohaibani F, Almanea H, Abalkhail H. Pancreatic metastasis arising from a BRAF(V600E)-positive papillary thyroid cancer: the role of endoscopic ultrasound-guided biopsy and response to sorafenib therapy. Thyroid. 2012;22:536–41. https://doi.org/10.1089/thy.2011.0247.
Saito Y, Sugino K, Takami H, Matsuzu K, Uruno T, Ohkuwa K, et al. Clinical status and treatment of liver metastasis of differentiated thyroid cancer using tyrosine kinase inhibitors. World J Surg. 2018;42:3632–7. https://doi.org/10.1007/s00268-018-4676-9.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Paspala, A., Kostakis, I.D., Gaitanidis, A. et al. Long-Term Outcomes After Hepatic and Pancreatic Resections for Metastases from Thyroid Cancer: a Systematic Review of the Literature. J Gastrointest Canc 50, 9–15 (2019). https://doi.org/10.1007/s12029-018-00196-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-018-00196-4