Abstract
Purpose
Polycystic ovarian syndrome (PCOS) is most commonly presented with insulin resistance (IR). Simple anthropometric indices may serve as surrogate markers of these conditions with population-based cut-off values. The present study suggests the cut-off values of waist-to-height ratio (WHtR) and body mass index (BMI) in early prediction of PCOS and IR in PCOS women based in Kolkata, a major metropolitan city in India.
Methods
This cross-sectional study included 66 women (aged 16–30 years) from Kolkata, India, with confirmed PCOS, using Rotterdam criteria. IR was defined following the homeostasis model assessment (HOMA). Anthropometric and biochemical data were obtained using standard protocol and compared among the PCOS subjects grouped as per IR prevalence, BMI, and WHtR values. The receiver operating characteristics (ROC) curve was applied to evaluate and compare the cut-off values of WHtR and BMI in the prediction of PCOS and IR in women with PCOS.
Results
As per ROC analysis, WHtR showed significantly higher AUC in the detection of PCOS and IR in PCOS subjects respectively, than that of BMI. The cut-off values of WHtR and BMI for PCOS were 0.560 and 28.47 respectively, and for IR in PCOS patients, were 0.620 and 29.14 respectively.
Conclusions
The present study suggests a cut-off value of WHtR to be used as an inexpensive and noninvasive screening tool for early prediction of PCOS and IR among PCOS afflicted women based in Kolkata, India and for this prediction, the study also claims WHtR as a better index than BMI.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Polycystic ovary syndrome (PCOS) is the most prevalent gynecological and heterogeneous endocrine disorder, which is manifested by recurrent anovulation and cutaneous symptoms of the excess androgens [1]. Its prevalence varies between 2 and 26% of women of reproductive age of different populations [1, 2], and in India, it affects 3.7–22.5% of women, including a substantial percentage from the metropolitan city of Kolkata [3]. It is also considered as a metabolic syndrome (MetS) [4], complementing 50–70% cases of insulin resistance (IR) [5]. In Kolkata, 37.9% PCOS women manifest MetS [6]. IR can evolve in several metabolic disorders such as diabetes mellitus (DM), dyslipidemia, MetS, and sleep apneas [7]. Diagnosis of IR should be regarded as one of the ways to detect the growing metabolic dysfunctions in the long-term treatment of women with PCOS. MetS is highly prevalent in women with PCOS compared to healthy women of same age range [8]. Moreover, for obese women, MetS is more severe than in non-obese PCOS females [9, 10].
It is also supposed that obesity intensifies IR and may cause glucose intolerance in PCOS patients [11] assuming that increase in prevalence of obesity leads to higher PCOS incidence followed by its consequences [12]. In India, obesity has reportedly affected more than 135 million people [13]. In the host state of the Metropolitan city of Kolkata, West Bengal, 19.9% women are reported to be obese compared to 14.2% in men [14]. In Kolkata, likewise the global prevalence, this is a growing public health concern of recent times owing to its impact on health and the economy [15, 16].
Thus, the above evidences suggest that obesity and PCOS coexistence is associated with incidence of IR and MetS. These also correlate positively with other comorbidies such as cardiovascular diseases, whose early management may be aided by early detection of IR and MetS [17]. Thus, a simple rapid yet accurate test for early detection of IR or MetS will be beneficial for clinical practice as well as for research purposes. For identification and classification of obesity, body mass index (BMI) is a widely accepted anthropometric index [18]. However, using BMI as predictive index for obesity has certain limitations, one of which is a lack of fat distribution assessment [19]. Thus, a more accurate measure of central adiposity may be the waist circumference (WC) and Waist-to-hip ratio (WHR) that may replace BMI for prediction of obesity and related health issues [20]. In this aspect, the waist-to-height ratio (WHtR), which is defined as the ratio of WC to height of an individual, is a well-known clinical measure for body fat distribution as it is correlated with abdominal obesity [21]. WHtR has been shown to be a stronger indicator of metabolic disorders (including hyperlipidemia, hypertension, hyperuricemia, and hyperglycemia) and risk factors for cardiovascular disease than BMI, WC, and WHR [22,23,24]. In predicting MetS in women with PCOS, WHtR has been reported to be better predictor than WHR [25]. Definite values of WHtR for predicting IR in women with PCOS in Kolkata, however, are unavailable. This cross-sectional study has, therefore, been conducted to analyze the accuracy of WHtR as possible modified alternative to BMI in predicting IR and to find the optimal cut-off points for PCOS patients in the metropolitan city of Kolkata.
Methods
Ethical considerations and study population
The Institutional Human Ethical Committee of Burdwan University (IECH/OCH/02/CC) has approved the study proposal. Ethics Committee of S. K. Pharma has also approved the study and all the participants agreed for the investigation protocol. The subjects were obtained from the outdoor clinic of Dr. Lipika Das (Mukhopadhyay) situated in S. K. Pharma, Santoshpur Avenue, Survey Park, Kolkata, India within the duration of November 1, 2018–April 30, 2019. This population-based cross-sectional study was conducted with 66 unmarried female aged between 16 and 30 years with clinically confirmed polycystic ovary following the diagnosis with transabdominal ultrasonography, also clinically matched signs and symptoms of PCOS as per the Rotterdam 2003 criteria [26], and age-matched 66 control subjects (comprised of 34 adolescents and 32 adults) were selected who were having some other gynecological problems except the PCOS. In India, vaginal ultrasonography is not allowed in unmarried girls for social reasons [27]. The exclusion criteria for this study were (a) abnormal renal or hepatic functions, (b) individuals with hyperprolactinemia/hypothyroidism, (c) individuals with secondary causes of androgen excess (d) individuals who had any hormone treatment in the preceding 3 months, (e) individuals with gynecological age <3 years, (f) use of any medications, including hormonal contraceptives, anti-androgenic drugs, and treatment for IR within the previous 3 months, (g) women suffering from genetic disorders, such as primary hypopituitarism, Turner’s syndrome, premature ovarian failure and (h) primary IR. Girls under the age of 16 years and pregnant women were also excluded from the study.
Anthropometric and biochemical measurements
Height, body weight, WC, and hip circumference were measured for every participant in bare feet on a plane surface and with minimal garments. Portable electronic scales were used for measuring body weight (measured to the nearest 0.1 kg) by resetting to zero before each measurement to get the accuracy of the measurement and the subjects were instructed to look forward during both the measurements. For height measurement, portable stadiometers were employed (measured to the nearest 1 cm), with feet of the participants placed together, with heels, hips and shoulder blades touched against the stick and positioning of the head on the horizontal plane. For measurement of waist and hip circumferences, inextensible anthropometric tape was used while the participants were instructed to stand erect with arms at their sides and feet close together, as recommended by Legro et al. [28].
The BMI (or Quetelet Index) was measured by using the following formula: BMI = weight (kg)/(Height in m)2 [29]. The patients were advised for the assessment of following biochemical examinations from an authorized concern diagnostic center by maintaining the standard protocol. The biochemical parameters include fasting blood sugar, fasting insulin, and lipid profile. Other glycemic parameters were derived by using the standard formula as mentioned by Gutch et al. [30] and all the derived lipid parameters were calculated as explained by Olamoyegun et al. [31].
Terms definition
PCOS was diagnosed as per the 2003 Rotterdam criteria [26]. The MetS was defined according to the joint interim statement (JIS) [32], as “the presence of any three or more of following five risk factors: (i) fasting TG ≥ 150 mg/dL or specific treatment, (ii) fasting HDL ≤ 50 mg/dL or specific treatment, (iii) raised systolic blood pressure ≥130 mmHg or raised diastolic blood pressure ≥85 mmHg or specific treatment, (iv) fasting plasma glucose ≥100 mg/dL or treatment and (v) IR was also estimated by the homeostasis model assessment (HOMA-IR) as a surrogate reference standard for measurement of IR according to the following formula: (fasting glucose [mg/dL]) × [fasting insulin] ÷ 405)” [33].
Statistical analysis
Statistical analysis was done assuming a confidence level of 95%, with a sample size, N = 66 (PCOS patients) and obtained data were arranged in Microsoft Office Excel spreadsheet 2007, analyzed by SPSS (v. 22.0, IBM, Chicago, IL, USA) and MedCalc (v. 19.05, Ostend, Belgium). Means ± SD were provided for the continuous variables. Descriptive statistics, one-way ANOVA, and Students’ t test were applied to analyze the obtained data. The statistically significant level was fixed at P < 0.05.
Receiver operator characteristic (ROC) analysis was performed using WHtR and BMI as continuous variables and PCOS and IR as the categorical variables to obtain and compare the area under the curves (AUCs), sensitivities, specificities, Youden’s indices and cut-off values. For all analyses, P < 0.05 was considered statistically significant.
Results
BMI and WHtR in adolescent and adult PCOS subjects with and without IR
Among 66 subjects undergoing gynecological treatment, age of 22 subjects were ≤19.0 (33.3%) years, 44 subjects were ≥19.1 years (66.7%) [34, 35]. Subjects when categorized into four groups based on heights where 51.5% had height of 152.5–162.4 cms, 25.8% of 162.5–172.5 cms, 18.2% of 142.5–152.4 cms and 4.5% of 132.5–142.4 cms. Half of the of the study subjects had body weight of 67–86 kgs, while 27.3% of 87–105 kgs and 22.7% of 46–66 kgs.
As per the WHO guidelines, a BMI over 25 kg/m2 is internationally considered as cut-off to define overweight [29]. Since Indians exhibit high tendency toward abdominal obesity due to genetic constitution and lifestyle factors, consensus statement for optimum cut-off for BMI for Asian Indians have been proposed by Mishra et al. (2009) [36], based on which the subjects were categorized into three groups, with highest BMI (≥25) found among 57.6% of the study subjects followed by moderate BMI (23–24.99) in 31.8% of the subjects and normal BMI (18.5–22.99) only in the rest 10.6% subjects. The descriptive information about BMI among the five different study groups and control subjects has been expressed in Table 1. Incidence of PCOS is higher in the young adults than in the adolescents. In the adolescents, higher number of PCOS subjects are overweight (BMI 23–24.99), rather than obese (BMI ≥ 25), while in young adults, higher number of PCOS subjects are obese (BMI ≥ 25), than overweight (BMI 23–24.99) or normal body weight (BMI 18–22.99). IR is prevalent in obese (BMI ≥ 25) PCOS women.
Table 1 also shows distribution of subjects as per the cut-off value of WHtR. Out of 66 PCOS subjects 62 had WHtR more than the cut-off value of 0.5. WHtR value showed similar pattern in sub-groups, with 21 out of 22 adolescent and 41 out of 44 young adults with PCOS among had WHtR more than the cut-off value. In agreement to these observations, all the PCOS subjects with IR and 31 out of 35 non-IR PCOS subjects showed higher WHtR value than the cut-off.
Anthropometric and biochemical parameters
In healthy subjects and PCOS patients
Mean comparison of anthropometric and biochemical parameters between control (N = 66) and PCOS (N = 66) groups have been depicted in Table 2. Independent t-test was performed, and the level of significance was considered at minimum value of P < 0.05. Parameters except height, BMI, fasting sugar levels, and LDL levels were significantly different in the PCOS group as compared to the control. Again, among these parameters, particularly, G:I ratio and HDL were significantly lower, while other parameters were significantly higher in the PCOS group than the control group.
In BMI-categorized PCOS patients
Comparison of anthropometric and biochemical parameters among three groups of subjects with PCOS categorized based on BMI values (group I, Group II, and Group III) are shown in Table 3. One-way ANOVA was applied for the test at 95% confidence interval, level of significance was considered when P < 0.05. In adolescent PCOS subjects, there are statistically significant differences between the means of all parameters, except the G:I ratio, of the three groups. In young adults with PCOS, means of the parameters, except insulin, HOMA, TC, and LDL showed significant differences among the three groups as per the BMI values.
In WHtR-categorized PCOS patients
Comparison of anthropometric and biochemical parameters two groups of PCOS patients categorized based on WHtR values (WHtR ≤ 0.5 and WHtR > 0.5) are shown in Table 4. Independent t-test was performed, and the level of significance was considered at minimum value of P < 0.05. The means of all parameters except age, height, HC, AIP index, and AC differed significantly between the two groups. Parameters other than GI ratio and HDL were significantly higher in the PCOS subjects belonging in the group with WHtR > 0.5 as compared to the other group.
Prevalence of dyslipidemia among the subjects with PCOS
Types and occurrence of dyslipidemia among the study groups of subjects with PCOS are shown in Table 5, considering the following physiological values as normal, TC < 200 mg/dl, LDL < 130 mg/dl, HDL > 50 mg/dl, and TG < 150 mg/dl. The table depicts that 28.78% of the subjects with PCOS had TC higher than physiological levels and all of them were young adults and consisted of 25.80% of PCOS subjects with IR and 28.57% of non-IR PCOS subjects. Higher LDL and TG than physiological values were more prevalent in the young adults, while majority of PCOS subjects of all the study groups showed HDL lower than the considered physiological value.
Anthropometric and biochemical parameters in IR and Non-IR PCOS subjects
Comparison of anthropometric and biochemical parameters between IR and non-IR group of PCOS subjects are shows in Table 6. Independent t-test was performed, and the level of significance was considered at minimum value of P < 0.05. The mean of parameters, body weight, BMI, WC, WHR, WHtR ratio, fasting insulin, and HOMA were significantly higher, while the G:I ratio was found significantly lower in IR group as compared to non-IR group. Other anthropometric and lipid profile parameters did not show any statistically significant differences the two groups.
WHtR and BMI as predictors of PCOS
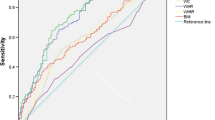
The comparison of ROC curves of WHtR and BMI for prediction of PCOS in women are presented in Fig. 1A. The ROC curve analysis for WHtR in predicting PCOS among all the study subjects (n = 132) showed AUCs (CI 95%) of 0.907 (0.844–0.950) with the cut-off value of 0.560, sensitivity of 74.2 and specificity of 97.0. The AUCs (CI 95%) for BMI were 0.782 (0.702–0.849) with the cut-off value of 28.47, sensitivity of 74.2 and specificity of 71.21. It indicated low predictive power of BMI for PCOS in women. The WHtR had the significantly higher AUC with difference of 0.125 (0.0314–0.218) and Youden index compared to BMI in prediction of PCOS in women, which was significant at P = 0.0088 (Table 7).
WHtR and BMI as predictors of IR in women with PCOS
The ROC curves of WHtR and BMI for prediction of IR in PCOS patients are presented in Fig. 1B. The ROC curve analysis for WHtR in predicting IR in PCOS patients showed AUCs (CI 95%) of 0.982 (0.915–0.999), cut-off value of 0.620, with 96.8% sensitivity and 91.4% specificity. ROC curve to analyze the predictive value of BMI for IR in PCOS patients, showed AUCs (CI 95%) of 0.585 (0.457–0.705) with the optimal cut-off value of 29.14 with 74.2% sensitivity and 48.6% specificity. WHtR had significantly higher AUC value with difference of 0.392 (0.244–0.539) and Youden index compared to BMI in prediction of IR in PCOS patients, at P < 0.0001 (Table 7).
Discussion
PCOS is among the most common and concerning global health issue affecting one in ten women of reproductive age [37]. Insulin resistance (IR) renders females with PCOS in reproductive ages at higher risk for cardiometabolic consequences as compared to healthy women as evidenced through different population-based studies [38]. The preliminary observation of the present study is concordant with previous studies depicting that majority of PCOS patients also have IR. Therefore, the early diagnosis of IR in PCOS patients is of great clinical importance. Euglycemic clamp technique is the gold standard IR detection method [39]. Since the method is expensive and time-consuming, the surrogate methods are used to detect IR and among them, the HOMA, is extensively used for its linear correlation with Euglycemic clamp technique [40]. There have been several studies aiming to come up with less invasive anthropometric markers for IR, such as BMI, WC, WHR and WHtR in different populations [41,42,43,44,45]. The present cross-sectional study is the first study on PCOS patients based in Kolkata (third-most populous metropolitan area in India) [46] that compares the diagnostic value of WHtR and BMI along with recommendation of their cut-off values in identifying risk of IR in PCOS patients. This study finds high relevance in the current scenario where obesity has been strongly associated with incidence of PCOS, while there lacks consensus on the accurate markers predicting risk of disorders as consequences of co-occurrence of obesity and PCOS. Use of simple anthropometric indices as screening tool is always a great advantage given its reliability of prediction of metabolic disorders, simple protocol as well as cost-effectiveness.
The study reveals that incidence of PCOS is higher in the young adults than in adolescents. As a common finding of increased BMI, in our study also, most of the individuals are either obese or overweight according to the BMI score which indicates the appearance of obesity among the PCOS patients. In this study BMI was showing significant association with two other anthropometric parameters body weight and height among the patients as it is a best-known derived parameter for obesity and depends on same two parameters. BMI has been demonstrated to reflect overall obesity rather than abdominal obesity, while WHtR included both WC and height. WHtR has been described as a parameter for central obesity and which was considered as screening tool for obesity assessment during clinical practice [47].
Both the anthropometric indices WHtR and BMI were significantly positively correlated with IR which are showcased in other studies as well [48, 49]. In the present study, higher number of PCOS cases are observed in the overweight adolescents (BMI 23–24.99) than in the obese group (BMI ≥ 25), while in young adults PCOS incidence is higher in the obese group. The distribution of subjects as per the WHtR cut-off (0.5) [50, 51] showed better homogeneity in both adolescent and adult groups (Table 1) with majority of subjects with higher WHtR being PCOS patients with IR. Moreover, several types of dyslipidemia were projected among the study groups. Lower serum HDL was most frequently observed type of dyslipidemia among all the patient groups followed by high plasma levels of TG, LDL, and TC accordingly which are also common resultants of obesity related diseases. In consensus with other studies, our study also showed that BMI and WHtR are associated with dyslipidemia in PCOS patients with and without IR [52].
In the present study, the comparative ROC curve using WHtR and BMI as continuous variables and PCOS and IR as the categorical variables, demonstrated WHtR as a better predictor of incidence of PCOS among all the study subjects (n = 132) as well as of occurrence of IR among the PCOS patients (n = 66) (Fig. 1). WHtR showed significantly higher AUC as well as higher Youden’s index, specificity and sensitivity than BMI, in prediction of PCOS as well as IR in PCOS women (Table 7) and the findings of the present study are consistent with previous reports in other populations where WHtR showed advantages over the use of other anthropometric indices in the assessment of IR in PCOS patients [25, 49, 53, 54]. WHtR also had a relatively better sensitivity and specificity than BMI. The results showed cut-off values of 28.47 and 29.14 for BMI in predicting PCOS and IR in PCOS respectively, which are close enough to suggest that BMI cannot discriminate between PCOS patients with IR and those without IR. In contrary, the ROC curve analysis provided suggestive cut-off value of 0.560 for WHtR in prediction of PCOS in women and 0.620 in prediction of IR in PCOS patients, for Kolkata population, while the cut-off values ranged between 0.5 and 0.52 as per other population-based studies that used WHtR as marker for abdominal obesity, metabolic risk factors, and cardiovascular disease [51, 55, 56]. The most possible reasons for the variations in WHtR cut-off values in different study populations may be the differences in race, food habits and other lifestyle factors, genetic compositions, geographical factors among other factors.
Conclusions
Our study shows that the WHtR is a potential marker and a better anthropometric indicator than BMI, for early detection of PCOS and IR in PCOS patients. Thus, based on our findings, we hereby recommend a WHtR cut-off value of 0.560 for prediction of PCOS and 0.620 in predicting IR in PCOS patients, in Kolkata, India.
References
S.M. Sirmans, K.A. Pate, Epidemiology, diagnosis, and management of polycystic ovary syndrome. Clin. Epidemiol. 6, 1 (2014)
N.M. Clark, A.J. Podolski, E.D. Brooks, D.R. Chizen, R.A. Pierson, D.C. Lehotay, M.E. Lujan, Prevalence of polycystic ovary syndrome phenotypes using updated criteria for polycystic ovarian morphology: an assessment of over 100 consecutive women self-reporting features of polycystic ovary syndrome. Reprod. Sci. 21(8), 1034–1043 (2014)
M.A. Ganie, V. Vasudevan, I.A. Wani, M.S. Baba, T. Arif, A. Rashid, Epidemiology, pathogenesis, genetics & management of polycystic ovary syndrome in India. Indian J. Med. Res. 150(4), 333 (2019)
S. Chhabra, N. Gangane, Coexistence of Endometrial Cancer, Polycystic Ovarian Syndrome and Metabolic Syndrome. EC Endocrinol. Metab. Res. 4, 91–97 (2019)
G. Hartmann, B. McEwen, Insulin resistance and Polycystic ovary syndrome (PCOS): part 2. Diet and Nutritional Medicine. J. Austr. Trad. Med. Soc. 25(1), 18 (2019)
S.M. Bhattacharya, Prevalence of metabolic syndrome in women with polycystic ovary syndrome, using two proposed definitions. Gynecol. Endocrinol. 26(7), 516–520 (2010)
S.M. Grundy, Metabolic syndrome update. Trends Cardiovas. Med. 26(4), 364–373 (2016)
J.A. Boyle, J. Cunningham, R.J. Norman, T. Dunbar, K. O’Dea, Polycystic ovary syndrome and metabolic syndrome in Indigenous Australian women. Int. Med. J. 45(12), 1247–1254 (2015)
M. Zaki, W. Basha, H.T. El-Bassyouni, S. El-Toukhy, T. Hussein, Evaluation of DNA damage profile in obese women and its association to risk of metabolic syndrome, polycystic ovary syndrome and recurrent preeclampsia. Gen. Dis. 5(4), 367–373 (2018)
F. Sigit, D. Tahapary, E. Sartono, S. Trompet, M. Yazdanbakhsh, F. Rosendaal, R. de Mutsert, The Prevalence Of Metabolic Syndrome And Its Association With Body Fat Distribution In A Dutch And Indonesian Population. Atherosclerosis 287, e135–e136 (2019)
J. Rojas, M. Chávez, L. Olivar, M. Rojas, J. Morillo, J. Mejías, M. Calvo, V. Bermúdez, Polycystic ovary syndrome, insulin resistance, and obesity: navigating the pathophysiologic labyrinth. Int. J. Reprod. Med. 2014, 1–17 (2014). https://doi.org/10.1155/2014/719050
R.S. Legro, Obesity and PCOS: implications for diagnosis and treatment. Semin Reprod. Med. 30(6), 496 (2012)
R. Ahirwar, P.R. Mondal, Prevalence of obesity in India: a systematic review. Clin. Res. Rev. 13(1), 318–321 (2019)
NFHS. Key findings: 2015-16 India: National Family Health Survey, India; 2012 [http://rchiips.org/NFHS/factsheet_NFHS-4.shtml.] [Last accessed on 19 Jul 2020]
M. Bhadra, A. Mukhopadhyay, K. Bose, Overweight and obesity among adult Bengalee Hindu women of Kolkata, India. Hum. Ecol. 13, 77–83 (2005)
P. Sengupta, P. Chaudhuri, K. Bhattacharya, Screening obesity by direct and derived anthropometric indices with evaluation of physical efficiency among female college students of Kolkata. Ann. Med. Health Sci. Res. 3(4), 517–522 (2013)
R.S. Legro, V.D. Castracane, R.P. Kauffman, Detecting insulin resistance in polycystic ovary syndrome: purposes and pitfalls. Obstet. Gynecol. Surv. 59(2), 141–154 (2004)
L.J. Aronne, Classification of obesity and assessment of obesity‐related health risks. Obes. Res. 10(S12), 105S–115S (2002)
F. Haghighatdoost, N. Sarrafzadegan, N. Mohammadifard, S. Asgary, M. Boshtam, L. Azadbakht, Assessing body shape index as a risk predictor for cardiovascular diseases and metabolic syndrome among Iranian adults. Nutrition 30(6), 636–644 (2014)
J.S. Mokha, S.R. Srinivasan, P. DasMahapatra, C. Fernandez, W. Chen, J. Xu, G.S. Berenson, Utility of waist-to-height ratio in assessing the status of central obesity and related cardiometabolic risk profile among normal weight and overweight/obese children: the Bogalusa Heart Study. BMC Pedia. 10(1), 73 (2010)
G. Belarmino, R.S. Torrinhas, P. Sala, L.M. Horie, L. Damiani, N.C. Lopes, S.B. Heymsfield, D.L. Waitzberg, A new anthropometric index for body fat estimation in patients with severe obesity. BMC Obes. 5(1), 25 (2018)
B. Dong, Z. Wang, L.W. Arnold, Y. Song, H.J. Wang, J. Ma, Simplifying the screening of abdominal adiposity in Chinese children with waist‐to‐height ratio. Am. J. Hum. Biol. 28(6), 945–949 (2016)
Z.P. Huang, B.X. Huang, H. Zhang, M.F. Zhu, H.L. Zhu, Waist-to-Height Ratio Is a Better Predictor of Hyperuricemia than Body Mass Index and Waist Circumference in Chinese. Ann. Nutr. Metab. 75(3), 187–194 (2019)
H. Yang, Z. Xin, J.P. Feng, J.K. Yang, Waist-to-height ratio is better than body mass index and waist circumference as a screening criterion for metabolic syndrome in Han Chinese adults. Medicine. 96(39), e8192 (2017)
E.C. Costa, J.C. Ferezini de Sá, E.M. Mafaldo Soares, T.M. Araújo Moura Lemos, T.M. de Oliveira Maranhão, G. Dantas, Azevedo, Anthropometric indices of central obesity how discriminators of metabolic syndrome in Brazilian women with polycystic ovary syndrome. Gynecol. Endocrinol. 28(1), 12–15 (2012)
The Rotterdam ESHRE/ASRM‐sponsored PCOS consensus workshop group, Revised 2003 consensus on diagnostic criteria and long‐term health risks related to polycystic ovary syndrome (PCOS). Hum. Reprod. 19(1), 41–47 (2004)
S. Bhattacharya, M. Ghosh, Insulin resistance and adolescent girls with polycystic ovary syndrome. J. Pedia. Adoles. Gynecol. 23(3), 158–161 (2010)
R.S. Legro, A.R. Kunselman, W.C. Dodson, A. Dunaif, Prevalence and predictors of risk for type 2 diabetes mellitus and impaired glucose tolerance in polycystic ovary syndrome: a prospective, controlled study in 254 affected women. J. Clin. Endocrinol. Metab. 84(1), 165–169 (1999)
World Health Organization, BMI classification. Global Database on body mass index. Switzerland (World Health Organization, 2006)
M. Gutch, S. Kumar, S.M. Razi, K.K. Gupta, A. Gupta, Assessment of insulin sensitivity/resistance. Indian J. Endocrinol. Metab. 19(1), 160 (2015)
M.A. Olamoyegun, R. Oluyombo, S.O. Asaolu, Evaluation of dyslipidemia, lipid ratios, and atherogenic index as cardiovascular risk factors among semi-urban dwellers in Nigeria. Ann. Afr. Med. 15(4), 194 (2016)
K. Alberti, R.H. Eckel, S.M. Grundy, P.Z. Zimmet, J.I. Cleeman, K.A. Donato, J.-C. Fruchart, W.P.T. James, C.M. Loria, S.C. Smith Jr, Harmonizing the metabolic syndrome: a joint interim statement of the international diabetes federation task force on epidemiology and prevention; national heart, lung, and blood institute; American heart association; world heart federation; international atherosclerosis society; and international association for the study of obesity. Circulation 120(16), 1640–1645 (2009)
D. Matthews, J. Hosker, A. Rudenski, B. Naylor, D. Treacher, R. Turner, Homeostasis model assessment: insulin resistance and β-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28(7), 412–419 (1985)
A. Ramachandran, C. Snehalatha, R. Vinitha, M. Thayyil, C.S. Kumar, L. Sheeba, S. Joseph, V. Vijay, Prevalence of overweight in urban Indian adolescent school children. Diab. Res. Clin. Prac. 57(3), 185–190 (2002)
D. Sacks, C.P. Society, A.H. Committee, Age limits and adolescents. Paediat. Child Heal. 8(9), 577–577 (2003)
A. Misra, P. Chowbey, B. Makkar, N. Vikram, J. Wasir, D. Chadha, S.R. Joshi, Consensus statement for diagnosis of obesity, abdominal obesity and the metabolic syndrome for Asian Indians and recommendations for physical activity, medical and surgical management. J. Assoc. Phys. India 57(2), 163–170 (2009)
US Department of Health and Human Services, Polycystic ovary syndrome. 2016 [cited 2020 31.05.2020]; https://www.womenshealth.gov/a-z-topics/polycystic-ovary-syndrome [last accessed on 19 July, 2020]
H.J. Teede, S. Hutchison, S. Zoungas, C. Meyer, Insulin resistance, the metabolic syndrome, diabetes, and cardiovascular disease risk in women with PCOS. Endocrine 30(1), 45–53 (2006)
E. Bonora, G. Targher, M. Alberiche, R.C. Bonadonna, F. Saggiani, M.B. Zenere, T. Monauni, M. Muggeo, Homeostasis model assessment closely mirrors the glucose clamp technique in the assessment of insulin sensitivity: studies in subjects with various degrees of glucose tolerance and insulin sensitivity. Diab. Care 23(1), 57–63 (2000)
B. Singh, A. Saxena, Surrogate markers of insulin resistance: a review. World J. Diab. 1(2), 36 (2010)
A. Vasques, L. Rosado, G. Rosado, R. Ribeiro, S. Franceschini, B. Geloneze, Anthropometric Indicators Of Insulin Resistance [indicadores Antropométricos De Resistência à Insulina]. Arq. Brasil. Cardiol. 95(1), e14-e23 (2010)
K. Kondaki, E. Grammatikaki, D.J. Pavón, Y. Manios, M. González-Gross, M. Sjöstrom, F. Gottrand, D. Molnar, L.A. Moreno, A. Kafatos, Comparison of several anthropometric indices with insulin resistance proxy measures among European adolescents: The Helena Study. Eur. J. Pedia. 170(6), 731–739 (2011)
M.I.B. Silva, C.C. da Silva Lemos, M.R.S.G. Torres, R. Bregman, Waist-to-height ratio: an accurate anthropometric index of abdominal adiposity and a predictor of high HOMA-IR values in nondialyzed chronic kidney disease patients. Nutrition 30(3), 279–285 (2014)
A. Nadeem, A.K. Naveed, M.M. Hussain, S.I. Raza, Cut-off values of anthropometric indices to determine insulin resistance in Pakistani adults. Age 51(12.1), 51.16–10.62 (2013)
C.M.Y. Lee, R.R. Huxley, R.P. Wildman, M. Woodward, Indices of abdominal obesity are better discriminators of cardiovascular risk factors than BMI: a meta-analysis. J. Clin. Epidemiol. 61(7), 646–653 (2008)
Q. A. Acton, Issues in global environment-Climate and Climate Change. Climate Research, ed. Q. A. Acton (Atlanta, Georgia, ScholarlyEditions, 2013)
E.G. Yoo, Waist-to-height ratio as a screening tool for obesity and cardiometabolic risk. Kor. J. Pedia. 59(11), 425 (2016)
J. Cresswell, R.B. Fraser, C. Bruce, P. Egger, D. Phillips, D.J. Barker, Relationship between polycystic ovaries, body mass index and insulin resistance. Acta Obstet. Gynecol. Scand. 82(1), 61–64 (2003)
S. Behboudi-Gandevani, F.R. Tehrani, L. Cheraghi, F. Azizi, Could “a body shape index” and “waist to height ratio” predict insulin resistance and metabolic syndrome in polycystic ovary syndrome? Eur. J. Obstet. Gynecol. Reprod. Biol. 205, 110–114 (2016)
M. Ashwell, S. Gibson, Waist-to-height ratio as an indicator of ‘early health risk’: simpler and more predictive than using a ‘matrix’based on BMI and waist circumference. BMJ Open 6(3), e010159 (2016)
L.M. Browning, S.D. Hsieh, M. Ashwell, A systematic review of waist-to-height ratio as a screening tool for the prediction of cardiovascular disease and diabetes: 0· 5 could be a suitable global boundary value. Nutr. Res. Rev. 23(2), 247–269 (2010)
M. Van Hemelrijck, H. Ulmer, G. Nagel, R.S. Peter, J. Fritz, R. Myte, B. Van Guelpen, B. Föger, H. Concin, C. Häggström, Longitudinal study of body mass index, dyslipidemia, hyperglycemia, and hypertension in 60,000 men and women in Sweden and Austria. PloS ONE. 13(6), e0197830 (2018)
F.R. Tehrani, S. Minooee, F. Azizi, Comparison of various adiposity indexes in women with polycystic ovary syndrome and normo-ovulatory non-hirsute women: a population-based study. Eur. J. Endocrinol. 171, 199–207 (2014)
T. Liu, Q. Wang, W. Huang, J. Tan, D. Liu, T. Pei, X. Li, G. Zhou, Anthropometric indices to predict insulin resistance in women with polycystic ovary syndrome in China. Reprod. Biomed. Online 38(1), 101–107 (2019)
F. Bacopoulou, V. Efthymiou, G. Landis, A. Rentoumis, G.P. Chrousos, Waist circumference, waist-to-hip ratio and waist-to-height ratio reference percentiles for abdominal obesity among Greek adolescents. BMC Pedia. 15(1), 50 (2015)
X. Guan, G. Sun, L. Zheng, W. Hu, W. Li, Y. Sun, Associations between metabolic risk factors and body mass index, waist circumference, waist‐to‐height ratio and waist‐to‐hip ratio in a Chinese rural population. J. Diab. Investig. 7(4), 601–606 (2016)
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The Institutional Human Ethical Committee of Burdwan University (IECH/OCH/02/CC) has approved the present study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bhattacharya, K., Sengupta, P., Dutta, S. et al. Waist-to-height ratio and BMI as predictive markers for insulin resistance in women with PCOS in Kolkata, India. Endocrine 72, 86–95 (2021). https://doi.org/10.1007/s12020-020-02555-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-020-02555-3