Abstract
Purpose
To assess the safety and efficacy of microwave ablation (MWA) for primary papillary thyroid microcarcinoma (PTMC) with a large sample of 185 patients.
Methods
A total of 185 patients underwent MWA for 206 primary PTMC nodules. They received ultrasound follow-up at 1, 3, 6, and 12 months after MWA and every 6 months thereafter. Nodule volumes were calculated at each follow-up and compared with those before MWA. Additionally, the volume reduction rate (VRR) of the nodules was calculated. Patients’ thyroid functions were tested before and 1 month after MWA.
Results
The mean follow-up time of the 185 patients was 20.7 ± 8.8 months (range 12–36 months). During the follow-up period, the mean volume of the 206 nodules was 100.1 ± 92.9 mm3 (range 3.6–423.9) before MWA, which decreased to 2.2 ± 5.6 mm3 (range 0–20.3 mm3) after MWA (P = 0.000). The mean VRR of the nodules was 98.65 ± 3.60% after MWA (range 83.85–100%). One hundred and seventy four of 206 nodules (84.5%) were fully absorbed. Compared with the preoperative results, no significant variation in thyroid function was observed 1 month after MWA. Thirty-eight patients (20.5%) had different types of complications, ranging from minor to major. Five patients (2.7%) had hoarseness, 11 patients (5.9%) had bleeding, 21 patients (11.4%) had earache or toothache, and one patient had another lesion 1 month after MWA.
Conclusions
This preliminary study suggests that MWA is safe and effective in the treatment of primary PTMC and offers a new alternative for clinical treatment.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Papillary thyroid microcarcinoma (PTMC) is a main cause of the high incidence of thyroid carcinoma in recent years [1,2,3]. Due to the trauma of surgery and possible complications, e.g., permanent damage to thyroid function, a growing number of researchers are opposed to surgery and support observation for the treatment of PTMC [4,5,6,7]. However, many patients diagnosed with PTMC remain uneasy because observation does not effectively treat the disease. Therefore, a method that is minimally invasive and effective in protecting thyroid function is urgently needed to treat these patients.
As a minimally invasive method, microwave ablation (MWA) has not only been successfully applied in other lesions, e.g., liver, kidney, lung, and breast [8,9,10,11], but has also achieved excellent results in treating benign thyroid tumors [12,13,14,15]. Recently, some scholars have employed MWA to treat PTMC [16,17,18]. However, these studies have small sample sizes, and to the best of our knowledge, there has been no large-scale study of MWA in the treatment of PTMC.
The aim of this study was to assess the safety and efficacy of a large sample of patients with PTMC treated by MWA.
Materials and methods
Study oversight
This retrospective study was approved by the Ethics Committee. Before MWA, all patients signed informed consent. Additionally, they understood that MWA was not the first-line treatment in current evidence and that recurrence or cervical metastasis could not be thoroughly avoided. All nodules were diagnosed as PTMC by fine-needle aspiration (FNA) or core needle biopsy (CNB).
Patients
The inclusion criteria were as follows: (1) patients diagnosed with papillary thyroid carcinoma by FNA or CNB; (2) maximum diameter of the lesion was less than 10 mm; (3) no cervical lymph node metastasis was found; and (4) patients were reluctant to undergo surgery or could not bear surgery.
The exclusion criteria were as follows: (1) cervical lymph node metastasis or distant metastasis; (2) pregnant women; (3) severe coagulation dysfunction; (4) severe cardiopulmonary abnormalities; or (5) other malignant diseases.
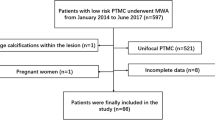
From January 2015 to June 2017, 4789 patients were diagnosed with PTMC in our hospital. Among them, 189 patients with 210 nodules were treated with MWA, four of these patients with four lesions were lost to follow-up, and the remaining 185 patients with 206 lesions were included here. The mean follow-up time of the 185 patients was 20.7 ± 8.8 months (range 12–36 months). Among the recruited patients, there were 41 lesions in 40 male patients and 165 lesions in 145 female patients. The average age of the recruited patients was 42.2 ± 11.7 years (range 17–72 years).
Equipment
The Siemens Acuson S2000 (Siemens Mountainview, USA), Esaote MyLab Twice (Esaote, Italy) and Mindray Resona 7 (MINDRAY, Shenzhen, China) ultrasound diagnostic instruments were employed for ultrasonic image acquisition and MWA guidance. Line array probes for detecting superficial organs were also applied here. An Ego ECO-100A1 machine (YIGAO Microwave System Engineering Co Ltd., Nanjing, Jiangsu Province, China) and ECO-100AI3 superficial organ ablation needle were applied here, which is consistent with our previous study [17].
Procedure before MWA
Image acquisition consisted of observing and recording the size, location, neighboring structures, composition, echo, edge, aspect ratio, and calcification. All images were collected by a senior doctor (with over 20 years of experience in thyroid ultrasound examination). The level of thyroid function was tested. The volume of the nodules was calculated using the following formula: V = πabc/6 (V: volume, a: the largest diameter, b and c: the other two perpendicular diameters).
MWA procedure
The patient was placed in a supine position to fully expose the neck. Lidocaine 1% was applied for local anesthesia. After using “Teng’s needle puncture method” [19] for puncturing the skin, the ablation electrode was implanted into the thyroid lesion, and the ablation was initiated at an output power of 20 W. For lesions in a dangerous location, normal saline served as the spacer fluid to separate the normal tissues from the lesions. The spacer fluid should be fully capable of preventing damage to the surrounding important organs or tissues. During the ablation process, if the spacer fluid was absorbed, it was replenished. Fixed ablation and moving-shot techniques [20] were employed during ablation. The ablation range was 5 mm beyond the lesions if it is possible. After ablation, contrast-enhanced ultrasonography (CEUS) was performed immediately. If the ablation range was not satisfactory, ablation was continued; if the ablation range was satisfactory, ablation was complete. A thyroid function test was required before MWA. During MWA, ECG, blood pressure, and blood oxygen were monitored. The presence of bleeding, hoarseness, and skin burns were noted during and after MWA.
Follow-up
Precise ultrasound examination was performed at 1, 3, 6, and 12 months after MWA and every 6 months thereafter. The size and volume reductions of the ablation area and cervical lymph nodes were observed and recorded. The volume reduction rate (VRR) of the lesion was calculated by VRR (%) = ([initial volume–final volume] × 100%)/initial volume. The thyroid function was retested at 1 month after MWA, and levothyroxine sodium was administered in accordance with the TSH level to maintain the TSH level under 0.1 mIU/L.
Statistical analysis
SPSS 22.0 was applied for statistical analysis. The size of the lesions and age of the patients are described by the mean ± SD. To compare the changes in nodule volume and thyroid function before and after the MWA, paired t-tests were performed. The difference was considered statistically significant at P < 0.05.
Results
Clinical characteristics
A total of 206 lesions in 185 patients underwent MWA. The outcomes of MWA treatment are listed in Table 1. These 185 patients included 40 males and 145 females. The average age was 42.2 ± 11.7 years (range 17–72 years). Among them, 167 patients had one lesion, 15 had two lesions, and three had three lesions, and all the lesions were confirmed as papillary thyroid carcinoma by pathology. There were 110 lesions on the right side and 96 lesions on the left side. The mean maximum diameter of the lesions was 5.3 ± 1.91 mm (range 2.1–9.9 mm), and the mean volume of the lesions was 100.1 ± 92.9 mm3 (range 3.6–423.9 mm3).
MWA procedure
During the operation, 182 of 185 patients (98.4%) underwent local anesthesia with good tolerance, and no other analgesic drugs were employed after surgery. The other three patients (1.6%) were converted to general anesthesia; two could not bear surgical pain, and the other one had a vagus nerve reflex.
Follow-up
The mean follow-up time of the 185 patients was 20.7 ± 8.8 months (range 12–36 months). Twenty-six patients with 28 lesions received a 36-month follow-up. Fifty patients with 58 lesions were followed up for 30 months. A total of 78 patients with 91 lesions received a 24-month follow-up. A total of 113 patients with 129 lesions were followed up for 18 months. A total of 185 patients with 206 lesions received a 12-month follow-up. The mean volume of the lesions before MWA was 100.1 ± 92.9 mm3 (range 3.6–423.9 mm3). The mean volumes of the nodules at 1, 3, 6, 12, 18, 24, 30, and 36 months after MWA were 1014.4 ± 756.3 mm3 (range 25.2–3350.2 mm3), 369.9 ± 375.1 mm3 (range 0–1992.2 mm3), 119.9 ± 173.2 mm3 (range 0–1000.9 mm3), 36.6 ± 75.1 mm3 (range 0–476.6 mm3), 13.8 ± 39.7 mm3 (range 0–230.7 mm3), 6.6 ± 18.2 mm3 (range 0–112.0 mm3), 3.2 ± 9.0 mm3 (range 0–57.4 mm3) and 2.2 ± 5.6 mm3 (range 0–20.3 mm3), respectively. Due to the expanded lesions during the ablation, the volume of the nodules increased at 1 month after MWA compared with that before MWA. As the follow-up time was prolonged, the volume of the nodules decreased gradually. Compared with the volume of the nodules before the procedure, the volume at 6 months later was similar, and the volume of the nodules was significantly smaller at 12 months later than before the procedure (P = 0.000) (Table 1). The changes in volume before and after MWA are shown in Fig. 1.
The variation of volumes and the VRR after microwave ablation (MWA), a the volumes of the 206 nodules at 12 months after MWA; b the volumes of the 129 nodules at 18 months; c the volumes of the 91 nodules at 24 months; d the volumes of the 58 nodules at 30 months; e the volumes of the 28 nodules at 36 months; f mean VRR at the 12th, 18th, 24th, 30th, and 36th months follow-up
The mean VRR of the nodules at 1, 3, 6, 12, 18, 24, 30, and 36 months after MWA were −1770.85 ± 2161.54% (range −47.07 to −17042.68%), −534.58 ± 758.92% (range −4853.74 to 100%), −92.99 ± 321.79% (range −1963.91 to 100%), 51.09 ± 96.11% (range −645.15 to 100%), 84.72 ± 45.67% (range −186.81 to 100%), 93.43 ± 18.16% (range −5.45 to 100%), 98.12 ± 6.90% (range 56.04–100%) and 98.65 ± 3.60% (range 83.85–100%), respectively. The mean VRR of the nodules after MWA is shown in Fig. 1f.
Of all the nodules, the numbers of completely absorbed nodules were 17, 68, 124, 165, 172, 173, and 174 at 3, 6, 12, 18, 24, 30, and 36 months after MWA, and the complete absorption rates were 8.3%, 33.0%, 60.2%, 80.1%, 83.5%, 84.0% and 84.5%, respectively (Fig. 2).
A 39-years-old woman had a nodule diagnosed as papillary thyroid microcarcinoma (PTMC) in the right thyroid lobe. The images of the nodule in preoperative, intraoperative, and postoperative follow-up are given, a before MWA, a nodule with 5.8 mm × 6.0 mm × 4.2 mm in size and 76.5 mm3 in volume was found in the right lobe; b the ablation electrode was implanted into the nodule, and the ablation mode was started at a output power of 20 w; c 1 month after MWA, the ablation area was 13.4 mm × 7.2 mm × 11.3 mm in size and 570.6 mm3 in volume; d 3 month after MWA, the ablation area was 7.4 mm × 4.0 mm × 5.3 mm and 82.1 mm3; e 6 months after MWA, the ablation are was fully absorbed (arrow)
All 185 patients underwent thyroid function examination before and 1 month after MWA. Before MWA, the average values of T3, T4, FT3, FT4, and TSH were 2.28 ± 0.53 nmol/L (range 1.36–3.12 nmol/L), 112.1 ± 25.8 nmol/L (range 70.0–155.8 nmol/L), 4.95 ± 1.12 pmol/L (range 3.12–6.80 pmol/L), 17.2 ± 2.7 pmol/L (range 12.0–22.0 pmol/L) and 2.68 ± 1.29 mU/L (range 0.42 to 4.94 mU/L), respectively. One month after MWA, the mean values of T3, T4, FT3, FT4, and TSH were 2.24 ± 0.50 nmol/L (range 1.37–3.14 nmol/ L), 113.4 ± 23.7 nmol/L (range 70.1–155.6 nmol/L), 4.88 ± 1.05 pmol/L (range 3.10–6.77 pmol/L), 16.8 ± 3.0 pmol/L (range 12.0–21.9 pmol/L), and 2.58 ± 1.28 mU/L (range 0.38–4.94 mU/L), respectively. The P values for the differences between the preoperative values and those 1 month after MWA were 0.397, 0.619, 0.559, 0.109, and 0.465, respectively. No significant variation in thyroid function was shown 1 month after MWA (Table 2).
Complications and recurrence
In this study, 38 patients had complications during or after MWA (Table 3). Five patients (2.7%) had hoarseness after MWA, and their lesions were in high-risk areas, e.g., near the trachea and adjacent to the dorsal thyroid gland. Among these five patients, four recovered over 3 months, and one recovered 2 days after MWA.
Eleven patients (5.9%) had bleeding during the operation, and nine of them (4.9%) stopped bleeding after compression. In addition, the compression time was <15 min. The other two patients (1.1%) failed to stop bleeding after compression. Accordingly, MWA was performed on the two patients for hemostasis with 2 min of ablation at a power of 40 W. Subsequently, hemostasis was confirmed, with over 30 min of observation by ultrasound and CEUS.
During the operation, 21 patients (11.4%) had earache or toothache on the same side as the nodules, and the pain was relieved within 2 h after the treatment.
In one patient (0.5%) with one nodule ablated, another small nodule of 2.1 × 2.4 × 2.1 mm in size was found at the 1-month follow-up. The FNA pathological diagnosis of the nodule showed papillary thyroid cancer, requiring secondary MWA. After secondary ablation, the follow-up of the two lesions continued for more than 18 months with a VRR of 100%.
No lymph node metastasis or distant metastases was found in any of the 185 patients reexamined by CT or US every 6 months after MWA.
Discussion
In this study, 206 nodules of primary PTMC in 185 patients were treated with MWA, and these patients received follow-up for an average of more than 20 months. According to the preliminary results, MWA was safe and effective for the treatment of primary PTMC.
Over the years, thermal ablation has shown good performance in treating thyroid disorders. The results of benign thyroid nodules were outstanding [12,13,14,15]. Treatment in metastatic lymph nodes was encouraging [19, 21, 22]. In recent years, some studies with the largest sample of 64 nodules have introduced laser ablation for PTMC treatment and have achieved excellent effectiveness [23,24,25,26]. Some scholars also employed radiofrequency ablation to treat PTMC [27, 28] and achieved favorable effectiveness, among which the largest sample size was 89 patients with 92 nodules. For the past few years, some scholars have also applied MWA to treat PTMC. For instance, Yue et al. [16] applied 40 W high-power microwave to treat 21 nodules and achieved good effectiveness. In addition, our previous study [17] employed 20 W low-power microwave to treat 21 PTMC nodules in 15 patients. However, these previous MWA studies were all small samples, and 206 lesions in 185 patients were included here, which has been the largest cohort thus far.
In the present study, MWA treatment in PTMC achieved excellent effectiveness after 12–36 months of follow-up, and the nodules were all significantly shrunk. Among 206 nodules, 174 (84.5%) were fully absorbed. The results here were similar to those of our previous studies [17] but with a higher success rate than that of other previous studies [18]. This outcome is probably because we used a lower output power of 20 W and performed a longer follow-up, which had been discussed in our previous study [17]. Among the 32 cases of incomplete absorption, 27 were followed up for less than 36 months, which probably led to incomplete absorption. The other five cases of incompletely absorbed lesions were followed up for 36 months, and a few residual lesions remained. Macrocalcification was found in all of the remaining five lesions at preoperative ultrasonography. Thus, it is believed that macrocalcification can affect the absorption after MWA.
No lymph node metastasis or distant metastasis were found during follow-up in our study. The reason for the excellent results is considered as follows: on the one hand, all lesions were expanded to a safe margin of 5 mm around the lesion at the farthest extent. On the other hand, only the nodules with clearly diagnosed PTMC were recruited, and the nodules that FNA or CNB could not identify were excluded. However, we should note that two nodules with capsular invasion in two patients were included in this study. Both were young women, and they refused to undergo surgery as a cosmetic concern. Although the two patients had no lymph node metastasis or distant metastasis during follow-up, we did not recommend this treatment to patients with capsular invasion as conventional.
This study showed that a low-power MWA of 20 W for PTMC was safe. In Yue’s study [16], four (19%) had major complications with a power of 40 W, while two patients (4.3%) in Li’s study [18] had major complications with a power of 30 W. In our previous study [17], 15 patients were treated, and no serious complications were found, which may be associated with the small sample size and lower power.
In the current study, five patients (2.7%) with hoarseness were found out of 185 patients with 206 nodules. Among them, one patient (0.5%) recovered 1 day after MWA, and the cause was considered the edema compression after MWA. Four patients (2.2%) recovered after 3–6 months and were considered to have recurrent laryngeal nerve heat injury. Among these patients, the lesions were close to the trachea and in the posterior thyroid region adjacent to the capsule, approaching the region where the recurrent laryngeal nerve traverses. This outcome suggested that when treating thyroid nodules in this area, it is necessary to use hydrodissection to protect the nerve. If the ablation time was too long and the isolation liquid was absorbed, it was important to replenish in time to avoid recurrent laryngeal nerve injury. Moreover, to observe the patient’s pronunciation, communication with patients during the operation is necessary. If hoarseness occurs, the operation should be stopped immediately. To reduce the incidence or severity of nerve injury, injecting cold saline as an isolation solution around the thyroid gland could be helpful. Finally, choosing the needle path from the isthmus during ablation also contributed to reducing the damage of the recurrent laryngeal nerve [20].
Eleven patients (5.9%) had bleeding during MWA; nine (4.9%) stopped bleeding, and two (1.1%) failed to stop bleeding with compression. The two patients were successfully ablated by MWA for hemostasis. In this study, the nine patients with compression hemostasis did not form hematomas in the surrounding tissue due to a short bleeding time and low volume of bleeding. The other two patients who received MWA for hemostasis formed hematomas around the thyroid gland, and the hematomas absorbed within one week after ablation. The incidence of hematoma here was 1.1% (2/185), which was close to the incidence observed with radiofrequency ablation in a previous study [29]. On one hand, the cause of bleeding may be associated with the needle passing through small blood vessels during the operation, which can be avoided by careful observation with color Doppler ultrasound during the puncture. On the other hand, the bleeding may have been caused by swallowing or coughing during the MWA procedure. During the operation, if a patient needed to swallow or to cough, the patient gently patted the operating table to remind the surgeon that the ablation electrode could be pulled out to a safe position in time to reduce the occurrence of bleeding in this situation. Accordingly, it is necessary to fully communicate with patients before MWA.
Twenty-one patients (11.4%) developed ear pain and toothache during the operation, and all were relieved within 2 h after ablation. The pain was thought to result from thyroid parenchymal edema, capsule heat injury or anterior cervical muscle involvement pain. The degree of pain was tolerable in most patients. When the lesions were close to the anterior and lateral thyroid capsule, injection of isolation fluid around the capsule was helpful to reduce the occurrence of this symptom.
A special case (0.5%) was found in the current study. A patient with one nodule was treated by MWA 1 month prior. During the follow-up at 1 month after the ablation, another small lesion with a size of 2.1 × 2.4 × 2.1 mm was found on the same side as the previous nodule. A second MWA procedure was performed on the patient. After 18 months of follow-up, both lesions were fully absorbed, and no new lesions were found. By reviewing the imaging data of the first ablation procedure, the lesion was not found. Because the location of the new nodule was far from the previous lesion during the first ablation and the new nodule was found shortly thereafter, it was considered that an omission occurred in the first MWA procedure. Because of the multicenter tendency of primary PTMC, careful ultrasound examination of the thyroid is very important before MWA. The gland should be carefully observed, and FNA should be performed in all suspicious areas to avoid missing nodules.
This study revealed that local anesthesia was an effective method for MWA in primary PTMC. Among the 185 patients, 182 were successfully treated with local anesthesia, and it was well tolerated. Only three patients (1.6%) underwent general anesthesia. Local anesthesia should be the preferred method of anesthesia in thyroid MWA. Because patients with local anesthesia can communicate with the surgeon during the operation, the surgeon can observe ear pain, toothache, voice changes in time and reduce the incidence of complications.
The thyroid function of 185 patients was reviewed 1 month after MWA, and there was no significant change compared with that before MWA. One month after MWA, levothyroxine sodium tablets were taken to suppress TSH levels under 0.1 mIU/L to reduce the risk of tumor recurrence [16, 30]. Due to the use of levothyroxine sodium tablets, the follow-up study on thyroid function was not conducted after 1 month post-MWA.
This study still has some limitations. First, primary PTMC is a slow-growing disease. Further and longer observation for local recurrence, lymph node metastasis and distant metastasis are still required. Although our results with a short-term follow-up are encouraging, a longer follow-up of at least 10 years is necessary in further research. Second, this is only a single center study, and statistical analysis of more central samples is still required. Finally, the postoperative thyroid function changes were not followed up for a long period here. The effects of MWA on thyroid function still require further study.
Conclusion
This preliminary study suggests that MWA is safe and effective in the treatment of primary PTMC and offers a new alternative for clinical therapy.
References
L.G. Morris, R.M. Tuttle, L. Davies, Changing Trends in the Incidence of Thyroid Cancer in the United States. JAMA Otolaryngol.-- Head Neck Surg. 142(7), 709–711 (2016). https://doi.org/10.1001/jamaoto.2016.0230
B. Aschebrook-Kilfoy, R.B. Schechter, Y.C. Shih, E.L. Kaplan, B.C. Chiu, P. Angelos, R.H. Grogan, The clinical and economic burden of a sustained increase in thyroid cancer incidence. Cancer Epidemiol. Biomark. Prev. 22(7), 1252–1259 (2013). https://doi.org/10.1158/1055-9965.epi-13-0242
S. Vaccarella, S. Franceschi, F. Bray, C.P. Wild, M. Plummer, L. Dal Maso, Worldwide thyroid-cancer epidemic? The increasing impact of overdiagnosis. N. Engl. J. Med. 375(7), 614–617 (2016). https://doi.org/10.1056/NEJMp1604412
Y. Ito, A. Miyauchi, H. Oda, Low-risk papillary microcarcinoma of the thyroid: A review of active surveillance trials. Eur. J. Surg. Oncol. (2017). https://doi.org/10.1016/j.ejso.2017.03.004
A. Griffin, J.P. Brito, M. Bahl, J.K. Hoang, Applying criteria of active surveillance to low-risk papillary thyroid cancer over a decade: how many surgeries and complications can be avoided? Thyroid 27(4), 518–523 (2017). https://doi.org/10.1089/thy.2016.0568
G.C. Haser, R.M. Tuttle, H.K. Su, E.E. Alon, D. Bergman, V. Bernet, E. Brett, R. Cobin, E.H. Dewey, G. Doherty, L.L. Dos Reis, J. Harris, J. Klopper, S.L. Lee, R.A. Levine, S.J. Lepore, I. Likhterov, M.A. Lupo, J. Machac, J.I. Mechanick, S. Mehra, M. Milas, L.A. Orloff, G. Randolph, T.A. Revenson, K.J. Roberts, D.S. Ross, M.E. Rowe, R.C. Smallridge, D. Terris, R.P. Tufano, M.L. Urken, Active surveillance for papillary thyroid microcarcinoma: new challenges and opportunities for the health care system. Endocr. Pract. 22(5), 602–611 (2016). https://doi.org/10.4158/ep151065.ra
H.G. Welch, G.M. Doherty, Saving thyroids - overtreatment of small papillary cancers. N. Engl. J. Med. 379(4), 310–312 (2018). https://doi.org/10.1056/NEJMp1804426
P. Liang, Y. Wang, X. Yu, B. Dong, Malignant liver tumors: treatment with percutaneous microwave ablation--complications among cohort of 1136 patients. Radiology 251(3), 933–940 (2009). https://doi.org/10.1148/radiol.2513081740
J. Yu, P. Liang, X.L. Yu, Z.G. Cheng, Z.Y. Han, X. Zhang, J. Dong, M.J. Mu, X. Li, X.H. Wang, US-guided percutaneous microwave ablation versus open radical nephrectomy for small renal cell carcinoma: intermediate-term results. Radiology 270(3), 880–887 (2014). https://doi.org/10.1148/radiol.13130275
L. Sidoff, D.E. Dupuy, Clinical experiences with microwave thermal ablation of lung malignancies. Int. J. Hyperth. 33(1), 25–33 (2017). https://doi.org/10.1080/02656736.2016.1204630
J. Xu, H. Wu, Z. Han, J. Zhang, Q. Li, J. Dou, C. An, E. Qi, J. Yu, P. Liang, Microwave ablation of benign breast tumors: a prospective study with minimum 12 months follow-up. Int. J. Hyperth. 1–9 (2018). https://doi.org/10.1080/02656736.2018.1494340
X. Zhi, N. Zhao, Y. Liu, J.B. Liu, C. Teng, L. Qian, Microwave ablation compared to thyroidectomy to treat benign thyroid nodules. Int. J. Hyperth. 34(5), 644–652 (2018). https://doi.org/10.1080/02656736.2018.1456677
B.W. Zheng, J.F. Wang, J.X. Ju, T. Wu, G. Tong, J. Ren, Efficacy and safety of cooled and uncooled microwave ablation for the treatment of benign thyroid nodules: a systematic review and meta-analysis. Endocrine 62(2), 307–317 (2018). https://doi.org/10.1007/s12020-018-1693-2
Z. Cheng, Y. Che, S. Yu, S. Wang, D. Teng, H. Xu, J. Li, D. Sun, Z. Han, P. Liang, US-Guided Percutaneous Radiofrequency versus Microwave Ablation for Benign Thyroid Nodules: A Prospective Multicenter Study. Sci. Rep. 7(1), 9554 (2017). https://doi.org/10.1038/s41598-017-09930-7
A.P. Mainini, C. Monaco, L.C. Pescatori, C. De Angelis, F. Sardanelli, L.M. Sconfienza, G. Mauri, Image-guided thermal ablation of benign thyroid nodules. J. Ultrasound 20(1), 11–22 (2017). https://doi.org/10.1007/s40477-016-0221-6
W. Yue, S. Wang, S. Yu, B. Wang, Ultrasound-guided percutaneous microwave ablation of solitary T1N0M0 papillary thyroid microcarcinoma: initial experience. Int. J. Hyperth. 30(2), 150–157 (2014). https://doi.org/10.3109/02656736.2014.885590
D. Teng, G. Sui, C. Liu, Y. Wang, Y. Xia, H. Wang, Long-term efficacy of ultrasound-guided low power microwave ablation for the treatment of primary papillary thyroid microcarcinoma: a 3-year follow-up study. J. Cancer Res. Clin. Oncol. 144(4), 771–779 (2018). https://doi.org/10.1007/s00432-018-2607-7
J. Li, Y. Liu, J. Liu, L. Qian, Ultrasound-guided percutaneous microwave ablation versus surgery for papillary thyroid microcarcinoma. Int. J. Hyperth. 34(5), 653–659 (2018). https://doi.org/10.1080/02656736.2018.1453092
D. Teng, L. Ding, Y. Wang, C. Liu, Y. Xia, H. Wang, Safety and efficiency of ultrasound-guided low power microwave ablation in the treatment of cervical metastatic lymph node from papillary thyroid carcinoma: a mean of 32 months follow-up study. Endocrine 62(3), 648–654 (2018). https://doi.org/10.1007/s12020-018-1711-4
J.H. Kim, J.H. Baek, H.K. Lim, H.S. Ahn, S.M. Baek, Y.J. Choi, Y.J. Choi, S.R. Chung, E.J. Ha, S.Y. Hahn, S.L. Jung, D.S. Kim, S.J. Kim, Y.K. Kim, C.Y. Lee, J.H. Lee, K.H. Lee, Y.H. Lee, J.S. Park, H. Park, J.H. Shin, C.H. Suh, J.Y. Sung, J.S. Sim, I. Youn, M. Choi, D.G. Na, 2017 Thyroid Radiofrequency Ablation Guideline: Korean Society of Thyroid Radiology. Korean J. Radiol. 19(4), 632–655 (2018). https://doi.org/10.3348/kjr.2018.19.4.632
Y. Guang, Y. Luo, Y. Zhang, M. Zhang, N. Li, Y. Zhang, J. Tang, Efficacy and safety of percutaneous ultrasound guided radiofrequency ablation for treating cervical metastatic lymph nodes from papillary thyroid carcinoma. J. Cancer Res. Clin. Oncol. 143(8), 1555–1562 (2017). https://doi.org/10.1007/s00432-017-2386-6
G. Mauri, L. Cova, T. Ierace, A. Baroli, E. Di Mauro, C.M. Pacella, S.N. Goldberg, L. Solbiati, Treatment of metastatic lymph nodes in the neck from papillary thyroid carcinoma with percutaneous laser ablation. Cardiovasc. Interv. Radiol. 39(7), 1023–1030 (2016). https://doi.org/10.1007/s00270-016-1313-6
W. Zhou, S. Jiang, W. Zhan, J. Zhou, S. Xu, L. Zhang, Ultrasound-guided percutaneous laser ablation of unifocal T1N0M0 papillary thyroid microcarcinoma: preliminary results. Eur. Radiol. 27(7), 2934–2940 (2017). https://doi.org/10.1007/s00330-016-4610-1
L. Zhang, W. Zhou, W. Zhan, Y. Peng, S. Jiang, S. Xu, Percutaneous laser ablation of unifocal papillary thyroid microcarcinoma: utility of conventional ultrasound and contrast-enhanced ultrasound in assessing local therapeutic response. World J. Surg. 42(8), 2476–2484 (2018). https://doi.org/10.1007/s00268-018-4500-6
R. Valcavi, S. Piana, G.S. Bortolan, R. Lai, V. Barbieri, R. Negro, Ultrasound-guided percutaneous laser ablation of papillary thyroid microcarcinoma: a feasibility study on three cases with pathological and immunohistochemical evaluation. Thyroid 23(12), 1578–1582 (2013). https://doi.org/10.1089/thy.2013.0279
E. Papini, R. Guglielmi, H. Gharib, I. Misischi, F. Graziano, M. Chianelli, A. Crescenzi, A. Bianchini, D. Valle, G. Bizzarri, Ultrasound-guided laser ablation of incidental papillary thyroid microcarcinoma: a potential therapeutic approach in patients at surgical risk. Thyroid 21(8), 917–920 (2011). https://doi.org/10.1089/thy.2010.0447
J.H. Kim, J.H. Baek, J.Y. Sung, H.S. Min, K.W. Kim, J.H. Hah, D.J. Park, K.H. Kim, B.Y. Cho, D.G. Na, Radiofrequency ablation of low-risk small papillary thyroidcarcinoma: preliminary results for patients ineligible for surgery. Int. J. Hyperth., 1–11 (2016). https://doi.org/10.1080/02656736.2016.1230893
M. Zhang, Y. Luo, Y. Zhang, J. Tang, Efficacy and safety of ultrasound-guided radiofrequency ablation for treating low-risk papillary thyroid microcarcinoma: a prospective study. Thyroid 26(11), 1581–1587 (2016). https://doi.org/10.1089/thy.2015.0471
D.G. Na, J.H. Lee, S.L. Jung, J.H. Kim, J.Y. Sung, J.H. Shin, E.K. Kim, J.H. Lee, D.W. Kim, J.S. Park, K.S. Kim, S.M. Baek, Y. Lee, S. Chong, J.S. Sim, J.Y. Huh, J.I. Bae, K.T. Kim, S.Y. Han, M.Y. Bae, Y.S. Kim, J.H. Baek, Radiofrequency ablation of benign Thyroid nodules and recurrent Thyroid cancers: Consensus statement and recommendations. Kor. J. Radiol. 13(2), 117–125 (2012). https://doi.org/10.3348/kjr.2012.13.2.117
B.R. Haugen, E.K. Alexander, K.C. Bible, G.M. Doherty, S.J. Mandel, Y.E. Nikiforov, F. Pacini, G.W. Randolph, A.M. Sawka, M. Schlumberger, K.G. Schuff, S.I. Sherman, J.A. Sosa, D.L. Steward, R.M. Tuttle, L. Wartofsky, 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 26(1), 1–133 (2016). https://doi.org/10.1089/thy.2015.0020
Acknowledgements
This study was funded by the Finance Department of Jilin Province (No. SCZSY201701), the Jilin Provincial Health and Family Planning Commission (NO. 2016ZC032) and Jilin Province Science and Technology Department (NO. 20170414042GH).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The studies have been approved by the appropriate institutional research ethics committee and have been performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. This manuscript does not contain any studies with animals by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Teng, DK., Li, HQ., Sui, GQ. et al. Preliminary report of microwave ablation for the primary papillary thyroid microcarcinoma: a large-cohort of 185 patients feasibility study. Endocrine 64, 109–117 (2019). https://doi.org/10.1007/s12020-019-01868-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-019-01868-2