Abstract
The global prevalence of obesity has significantly increased in most industrialized countries. Anti-obesity drugs are scarce, and indications to change their life style are impractical. Therefore, to identify diets able to produce significantly and maintained weight loss is mandatory. The present work evaluated the efficacy of a very low-calorie-ketogenic (VLCK) diet in obesity. A group of obese patients were randomized into two groups: the VLCK diet group and a standard low-calorie diet (LC group). The follow-up period was 12 months. Both groups received external support, counseling, to perform physical activity and adhered to the diet. The VLCK diet induced a 30–45 days of mild ketosis and significant effects on body weight within 15 days. At 2 months, the weight reductions in the VLCK diet and LC diet groups were 13.6 ± 3.9 and 4.8 ± 2.7 kg, respectively (p < 0.0001). At the end of the study, at 12 months, the weight reductions were 19.9 ± 12.3 and 7.0 ± 5.6 kg, respectively (p < 0.0001), and more than 88 % of patients in the VLCK diet group lost more of 10 % of their initial weight. Lean mass was practically unaffected. The VLCK diet was well tolerated and the side effects were moderate and transitory. In a group of obese patients, the VLCK diet was significantly more effective than a standard LC diet. At one year follow-up in the group with VLCK diet, most of the patients loss more than 10 % of their initial weight and lean mass was well preserved.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obesity has been defined as a global epidemic, and its prevalence is now maintained or even accelerated in most industrialized countries [1–3]. Furthermore, it is forecasted that those countries that exhibit a lower prevalence than the above-mentioned countries will reach the same levels once social inequities are settled [4]. The genetic background of the human species will not change over the next decades, and the environmental situation will not return to more active times with expensive food; the logical forecast is that prevalence levels of obesity will be maintained or increased, thereby increasing its associated diseases and complications, such as type 2 diabetes mellitus (DMT2), cardiovascular mortality, cancer, and so on [5–7]. Thus, prevention and treatment of obesity are mandatory to reduce the burden of the disease in both the individuals and the society.
The promotion of lifestyle changes has a solid rationale, but this policy is doomed to failure and is unsuitable in the large-scale [8] and bariatric surgery had been shown to be unequivocally effective long term and able to reduce cardiovascular morbidity and mortality [9–11], but such techniques are unsuitable as population–wide treatments [12]. Hence, effective medical treatments are needed. However, most of the drugs that have been used to treat obesity have been withdrawn from the market due to incorrect use or side effects [13–15], and the new anti-obesity drugs approved or under evaluation by the FDA or the EMA have not yet been tested in the large scale [16]. Thus, health stakeholders are facing a situation without precedent by fighting an epidemic without tools.
An increase in physical activity is one way to prevent or fight obesity, but the efficacy of this intervention is scarce without a simultaneous reduction in the food intake [17]. As a result, many dietary regimens which operate through various mechanisms have been proposed to reduce appetite or for weight control [18]. The leading non-pharmacological approach is the use of diets, particularly low-calorie and very low-calorie diets [19–21]. In the last few years, the low-carbohydrate diet has gained progressive recognition over other dietary treatments [22], and the low-carbohydrate ketogenic diet in particular has been closely examined [23, 24].
In the present work, a commercially available very low-calorie-ketogenic (VLCK) diet was tested against the standard low-calorie (LC) diet that is commonly practiced in many hospitals. The two main objectives of this study were to compare the efficacy of both treatments in the induction of weight loss, and to evaluate their efficacy after a one year follow-up period. The secondary targets were to evaluate the rate of dropouts and side effects, and to determine the effects of these diets on fat mass (preservation of lean mass) and cardiovascular risk factors.
Materials and methods
This study was a nutritional intervention clinical trial, open, randomized, controlled, prospective for one year, and performed in a single center. The patients attending the Obesity Unit at the Hospital Gregorio Marañon of Madrid to receive treatment for obesity were consecutively enrolled in this study. Apart from obesity and prediabetes, participants were generally healthy individuals. The inclusion criteria were, age 18–65 years, body mass index (BMI) ≥ 30, stable body weight in the previous 3 months, desire to lose weight, and history of failed dietary efforts. None of the participants had serious medical condition.
The main exclusion criteria were, type 1 diabetes mellitus or insulin therapy, obesity induced by other endocrine disorders or by drugs, and use of any weight loss diet or pills in the previous 6 months. Secondary exclusion criteria were, severe depression or any other psychiatric disease, abuse of narcotics or alcohol, severe hepatic insufficiency, any type of renal insufficiency or gout episodes, neoplasia (except basal cell skin cancer), previous events of cardiovascular or cerebrovascular disease, kidney ltiasis, uncontrolled hypertension, and hydroelectrolytic alterations. Females with child-bearing potential, who were pregnant, breast-feeding, intending to become pregnant, or not using adequate contraceptive methods were excluded. All participants provided written informed consent and the Institutional Review Board (Comite Etico de Investigacion Clinica, Hospital Gregorio Marañon, Madrid) approved the study (C.I 143/09, protocol PRO–PRO 2009-02, Foundation code 143/09 EO). Participants received no monetary incentive. A total of 79 subjects were participated in the study (Table 1).
Interventions
Using a controlled open design, the patients were randomized and allocated to receive either a low-calorie diet (thereafter “LC diet”) or a very low-calorie-ketogenic diet (thereafter “VLCK diet”) as part of a commercial weight loss program (Pronokal Method), which included lifestyle and behavioral modification support. The intervention for both groups included an evaluation by the specialist physician conducting the study, an assessment by an expert dietician, group meetings, and exercise recommendations. The group meetings and evaluations took place in a hospital setting at 0.5, 2, 4, 6, 8, 10, and 12 months. At 6 and 12 months, the participant′s satisfaction with the diet was assessed. In these meetings, the patients received diet instructions, individual supportive counsel, and encouragement to exercise on a regular basis using a formal exercise program. In addition, a program of telephone reinforcement calls was instituted, and a phone number to address any doubts was provided to all participants.
Low-calorie diet
The standard LC diet of the Obesity Unit was provided to this group. This equilibrated diet had a caloric value 10 % below the total metabolic expenditure of each individual. The total metabolic expenditure was calculated from the basal metabolic expenditure (based on the formula FAO/WHO/UN) [25] multiplied by the coefficient of activity, which was calculated according to the physical activity of each participant. The calories provided to this group ranged between 1,400 and 1,800 kcal/day. The ration of macronutrients provided was 45–55 % carbohydrates, 15–25 % proteins, and 25–35 % fat [26], in addition to a recommended intake of 20–40 g/day of fiber in the form of vegetables and fruits. A ratio exchange model was followed.
Very low-calorie-ketogenic diet
The VLCK diet group, followed a very low-calorie-ketogenic diet according to a commercial weight loss program (PronoKal method) based on a high-biological-value protein preparations diet and natural foods. Each protein preparation contained 15 g protein, 4 g carbohydrates, and 3 g fat, and provided 90–100 kcla [27] “Fig 1”. This method has three stages: active, re-education, and maintenance.
The active stage consists of a very low-calorie diet (600–800 kcal/day), low in carbohydrates (<50 g daily from vegetables) and lipids (only 10 g of olive oil per day). The amount of high-biological-value proteins ranged between 0.8 and 1.2 g per each Kg of ideal body weight, to ensure meeting the minimal body requirements and to prevent the loss of lean mass. This method produces three ketogenic phases. In phase 1, the patients eat high-biological-value protein preparations five times a day, and vegetables with low glycemic index. In phase 2, one of the protein servings is substituted by a natural protein (e.g., meat and fish) either at lunch or at dinner. In the phase 3, a second serve of the natural protein low in fat substituted the second serve of biological protein preparation. Throughout these ketogenic phases, supplements of vitamins and minerals, such as K, Na, Mg, Ca, and omega-3 fatty acids, were provided in accordance to international recommendations [28]. This active stage is maintained until the patient loses most of weight loss target, ideally 80 %. Hence, the ketogenic phases were variable in time depending on the individual and the weight loss target, but they lasted between 30 and 45 days in total.
In the re-education stage, the ketogenic phases were ended by the physician in charge of the patient based on the amount of weight lost, and started a low-calorie diet. At this point, the patients underwent a progressive incorporation of different food groups and participated in a program of alimentary re-education to guarantee the long-term maintenance of the weight lost (see “Fig. 1”). The maintenance stage, consist of an eating plan balanced in carbohydrates, protein, and fat, that lasted one year. Depending on the individual the calories consumed ranged between 1,500 and 2,000 kcal/day and the target was to maintain the lost weight and promote healthy life styles.
Primary outcome measures
The body weight, waist circumference (WC), and BMI were the primary outcome measures. At each visit, patients were weighed on the same calibrated scale (Seca 220 scale, Medical Resources, EPI Inc OH, USA) wearing light clothing and no shoes. BMI was calculated as body weight in Kg, divided by height in meters squared. WC was recorded with a standard flexible non-elastic metric tape over the midpoint between the last rib and the iliac crest, with the patient standing and exhaling [26].
Secondary outcome measures
Cardiovascular risk factors
Blood pressure and pulse rate were measured in the non-dominant arm after the participants sat by at least 15 min. Two measurements were collected on each visit and averaged. Serum lipids were measured before starting the intervention and at regular intervals thereafter.
Adherence
Adherence to the diet and exercise recommendations in VLCK diet group was determined through self-reports, food records, and in the ketogenic phases by urinary ketone qualitative assessment. Interacetona dipstick (Carulla Vekar Madrid) was used for ketone analysis and the patients recorded these values that were analyzed at each visit to the hospital. Adherence in the LC diet group was assessed according to the patients self-reports.
Body composition
Body composition was measured by Dual-Energy-X-ray Absorptiometry (DXA Scan). Total body imaging was acquired using the GE Healthcare Lunar (iDXA Madison, WI, USA), and analyzed using enCORE software version 13.2. Subjects were scanned using standard imaging and positioning protocols. The GE Lunar Body Composition Software option (body composition) used on GE Lunar iDXA bone densitometer measures the regional and whole body bone mineral density (BMD), lean, and fat tissue mass, and calculates derivative values of bone mineral content (BMC), area, soft tissues mass, regional soft tissue mass, total soft tissue mass, fat free mass, regional/total soft tissue mass ratio, % fat, region % fat, total body % fat, Android % fat, and Gynoid % fat. Android/Gynoid ratio (A/G ratio) is automatically generated. During the procedure, patients wore only light clothes.
Other metabolic parameters
Serum tests for glucose, HbA1c, cholesterol-HDL and cholesterol-LDL, potassium, chloride, sodium, urea, nitrogen, creatinine, calcium, phosphorus, total proteins, albumin, liver transaminases, bilirubin, uric acid, thyroid hormones, alkaline phosphatase, iron, hemoglobin, red cell and white cells counts, plasma lipids, and glomerular filtration rate were performed using automatic standard procedures (Cobas c711, Roche-Spain, Madrid, Spain) and a Coulter LH 750 Hematology Analyzer, (Beckman Coulter, Inc.; Brea CA, USA) at basal and at regular intervals during and after the intervention programs.
Adverse effects, dropouts, and satisfaction with the treatments
During all visits, the patients completed a questionnaire of side effects and dropouts, and the reasons provided were recorded. A questionnaire of patient satisfaction with the diets, with a single question “How satisfied are you with the diet that you are following”, using 5-point Likert scale format (1 = very dissatisfied, 2 = dissatisfied, 3 = indifferent, 4 = satisfied, 5 = very satisfied) was provided to the patients. This questionnaire was undertaken at visits 6–12 months.
Statistical analysis
This study was a nutritional intervention clinical trial, open, randomized, controlled, prospective for one year, and performed in a single center. A total of 79 obese subjects were studied, 39 patients were in the VLCK diet group and 40 patients were in the LC diet group, because it was calculated that the groups should have at least 30 patients on each arm to detect differences in the mean weight reduction of 3 kg, with a potency of 90 %. The significance level was established at p < 0.05. Weight loss was primarily analysed in the patients that complete the study (completers), and results were also evaluated with an intention-to-treat (ITT) analysis using the baseline observation carried forward (BOCF). In sensitivity analyses, used the last observation carried forward (LOCF) and multiple imputations (MI) as age, sex, and baseline values were used to predict any missing values at the 1 year time point and determine whether the findings were affected by the choice of data-imputation method.
The data are presented as mean ± SD. The differences between two groups were determined using Fisher′s exact test for categorical variables and Student′s t test. Alternatively, ANOVA was used for comparison of continuous variables between three or more groups. Variables that were not normally distributed were analyzed by non-parametric tests (Mann–Whitney U test or Kruskal–Wallis test). For completers-only analysis, there were no formal imputations, therefore, all the estimations were obtained using all the available data (available data only-ADO). Analyses were performed using the SAS software version 9.1.3 (SAS Institute Inc). The level of significance was set to 5 %.
Funding source
The funding for the study as well as the high-biological-value protein preparations was provided by Protein Supplies, S.L., (Barcelona, Spain) free of charge to the patients. The funding source had no involvement in the study design, recruitment of patients, study interventions, data collection, or interpretation of the results.
Results
Of the 79 participants that were initially enrolled in the study, a total of 12 in the VLCK diet group and 14 in the standard LC diet group dropped out of the study within 12 months. The 53 patients who completed the study, (completers), exhibited the following characteristics at enrollment: mean age 45.3 ± 8.9 year (range 24–65), BMI 35.1 ± 4.9 (range 29.5–47.3), and waist circumference 109.8 ± 12.4 cm (range 90.0–151.0). In addition, 88.6 % were women, and all the patients were obese (Table 1).
The completers-only analysis revealed that starting at day 15, the reduction in weight was twofold higher in the VLCK diet than in the LC diet group. This difference was most evident at 2 months with a reduction of 13.6 ± 3.9 and 4.8 ± 2.7 kg in the VLCK and LC groups, respectively (p < 0.0001); “Fig. 2”. The maximum weight loss was observed after 8 months of treatment, 22.8 ± 11.4 kg in the VLCK diet group and 7.1 ± 5.3 kg in the LC group. At 12 months, the difference between the groups was still evident, with a reduction of 19.9 ± 12.3 kg for the VLCK, whereas it was of 7.0 ± 5.6 kg in the LC group (p < 0.0001), even though the weight regain in VLCK diet group was 12.4 %, while in the LC diet group was only 1.4 %.
Evolution of the weight loss after the initiation of treatment and during the 12-months follow-up. Data from the completers groups are presented and the data obtained through the intention-to-treat (ITT) analysis are also shown. last observation carried (LOC) forward; basal observation carried (BOC) forward; and multiple imputations (MI). *p < 0.001 compared with the value at baseline; ++p < 0.001 when compared between groups
When the weight loss at one year was analysed as intention-to-treat with multiple imputations (ITT-MI), the values for the VLCK versus LC diets were 19.9 ± 12.6 versus 7.1 ± 4.7 kg (p < 0.0001). Similarly, the differences between both groups were 17.3 ± 11.9 and 5.7 ± 5.6 kg (p < 0.0001) in the ITT-last observation carried forward (ITT-LOCF); and 13.7 ± 13.8 versus 4.5 ± 5.6 kg (p < 0.0002), in the ITT-baseline intervention carried forward (ITT-BOCF), “Fig. 1”. All of the analyses showed that the VLCK diet induced a higher weight reduction of more than 10 kg compared with the LC diet.
As shown in “Fig. 3”, the study of the categories of weight loss percentages demonstrated that the VLCK diet induced a reduction of more than 10 % of the original weight at 2 months in 96 % of the patients, compared with 3 % of the patients in the LC group (p < 0.0001). This striking difference in the efficacy of the diets was maintained at 4 months, and at the end of the study (12 months), 88.9 % of the patients in the VLCK group exhibited a weight loss of at least 10 % of the original weight, compared with 34.6 % of the patients in the LC group (p < 0.0001).
Although both groups exhibited a fairly similar BMI (35.1) at the beginning of the study, the BMI absolute values within 4 months were 28.0 ± 3.2 in the VLCK group and 32.5 ± 5.6 in the LC group, (p < 0.0008), and persisted throughout the study (Fig 4a). This observation resulted in final reductions at 12 months of 7.0 ± 3.9 and 2.6 ± 2.2 BMI units for the VLCK and the LC groups, respectively (p < 0.00001) “Fig. 4b”. Similar results were observed when the data were analysed as absolute values of body weight in Kg, or as excess of body weight in percentage “Fig 4c, d”. Similar observations were obtained with the intention-to-treat analysis, ITT-MI, ITT-LOCF, and ITT-BOCF.
Evaluation of the weight loss in the ketogenic and hypocaloric diet groups as completers and as intention-to-treat (ITT) analysis is presented. a absolute values of body mass index (BMI), b Changes in BMI, c Absolute values of body weight in Kg, d Absolute values of excess of weight loss. LOC forward; BOC forward and MI
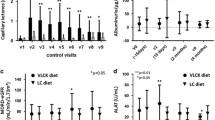
To evaluate the toll imposed by both diets on the different body compartments, i.e., fat mass or lean mass, the waist circumference was measured as an indirect measurement of the fat mass or more precisely the central or visceral fat mass. As shown in “Fig. 5a”, the VLCK diet induced a reduction in waist circumference from 111.3 ± 13.4 to 92.8 ± 8.0 cm (p < 0.0001) at 12 months, whereas the LC diet induced a reduction from 108.2 ± 11.4 to 101.2 ± 13.3 cm, (p < 0.0473), at 12 months. Using absolute changes in waist circumference “Fig. 5b”, net reductions of 18.4 ± 10.4 and 7.0 ± 6.3 cm were obtained for the VLCK and the LC group, respectively (p < 0.0001). A more sophisticated analysis was performed using the DEXA scan evaluation. As shown in “Figs. 5c–d”, the VLCK diet induced a reduction in fat mass from 44.3 to 28.3 kg, (whereas, the LC diet induced a reduction from 42.3 to 36.7 kg). None of the diets affected the lean mass because no significant changes were found throughout the 12-months study.
Evaluation of the body composition of the patients in the VLCK diet and the LC diet groups at the end of the study. a Absolute values of the waist circumference, b Changes the waist circumference (in cm), c Percentage of the loss of fat-mass evaluated by DXA scan, and d Absolute values of lean body mass (LBM) and fat body mass (FBM) determined by DXA scan
Biochemical and metabolic parameters measured during the treatment and follow-up periods are depicted in (Table 2). As shown, the VLCK diet induced a significant change in some of the biological parameters that were clinically meaningful, such as HbA1c, total cholesterol and micro-albuminuria, and some of these changes were still evident at 1 year follow-up.
Side effects and dropout rates at the 1 year follow-up
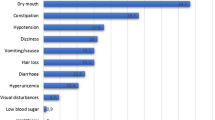
Table 3 shows the most frequent side effects that were observed in both groups. The VLCK diet induced more frequent manifestations of asthenia, fatigue, headache, muscle weakness, constipation, hyper-uricemia, and nausea that that of the LC diet. However, these manifestations were of mild intensity and transient (most disappear after the first few weeks). In fact, the number of patients who dropped out of the study was negligible at the beginning of the study, which was when more frequent side effects were observed. In addition, none of the patients who dropped out attributed that to these collateral effects. In total 12, (33.3 %) in the VLCK diet group, and 14, (35 %) in the LC diet group dropped out throughout the course of the study.
Sensitivity analysis
To minimize the effect of the method used to handle missing data in the final results obtained for the group of patients who completed the study, all the analyses were performed again through sensitivity analysis using the last observation carried forward, basal observation carried forward, and multiple imputations, in the intention-to-treat analysis. None of the alternative data imputation methods altered the results of the main analysis.
Discussion
The main finding of this work is that, compared with a standard low-calorie diet (LC diet), a VLCK diet was significantly more effective in inducing loss of body weight, and that the weight loss was mainly related to the loss of fat mass. As ancillary findings, the reduction in weight was rapid, the side effects were transitory and well tolerated. Interesting enough, when evaluated at one year follow-up, the VLCK diet group showed than more than 88 % of the patients presented a reduction of initial body weight higher than 10 % and that lean mass was well preserved.
In the present work, the VLCK diet induced a rapid, intense, and persistent weight reduction compared with the LC diet. In fact, it is remarkable that this diet group exhibited a twofold higher weight reduction within 15 days of starting the program. Although, in absolute terms, the maximum loss (−22.8 kg) was observed at 8 months, the effect was rapid and maintained because the loss at 4 months (−22.0 kg) and the ensuing values of (−22.0) and (−20.7 kg) obtained at 6 and 10 months were all not significantly different than those at the 8 months. This result is remarkable considering that the ketogenic phase ended at 30–45 days after starting the method, therefore, the above results were obtained in the re-education stage and out of ketosis. It has been previously found that a greater initial weight loss improves the long-term maintenance of the weight loss [29, 30]. This hypothesis was confirmed in the present work: patients eagerly adhered to the treatment and evaluations mostly because they found the rapid weight loss highly encouraging and increasingly motivating. The effect was persistent long term because the final weight loss of (−19.9 kg) was obtained at 12 months, i.e., time after the ketosis combined with very low-caloric treatment had ended and patients were on a maintenance diet. Because weight regain in successfully treated obese patients is the most common drawback of any treatment [31, 32], the long-term maintenance of weight here observed is another positive effect of the ketogenic approach, it was interesting to find that patients on the conventional treatment also exhibited a significant weight reduction, which indicate that the multidisciplinary approach used in this study for both groups, which include diet counseling, physical activity, and psychological support was effective.
The VLCK diet method was found to be superior to the standard LC diet, and the average weight loss of 20 kg that was measured in this study was higher than the weight loss that has been observed with most of the anti-obesity drugs that have been studied [33–38]. The analysis of the percentage of the body weight loss revealed that more than 96 % of the patients in the VLCK diet group lost more than 10 % of the initial body weight within 2 months compared with 3.8 % of the patients in the LC-diet group. In addition, these differences were maintained at 4 and 12 months. Although initially the VLCK diet was lower in calories than the LC diet which implies a greater efficacy, the main point of this work is that patients were able to tolerate and gladly adhere to the calorie restriction. In the true that during the first phase of the study there was a difference in the total caloric value of the diets in the VLCK study group and the LCD control group, but after that short period, and up to a maximum of a 4 months after the beginning of the study, calorie intake was increased until it reached that of the control group (LC diet), as a similar calorie intake was followed by both groups until 12 months the differences between them faded away. The initial effectiveness for the VLCK group was mainly due to the calorie limitations specified in the dietary guidelines. The proportion of macronutrients in the diet provides a percentage of fats which covers essential requirements, a not too severe restriction of carbohydrates (50 g) and the intake of high biological value proteins (75 g per day). This proportion of macronutrients, which may induce ketosis, has been associated with a greater feeling of fullness and a greater tolerability by the patient (23). Interesting enough, the ketogenic diet mainly reduced body fat and did not affect lean mass. In fact, both the indirect data provided by waist circumference and the direct information provided by DXA scan clearly showed that the VLCK diet induces a strong reduction in fat mass (measured either as a percent or in absolute values, i.e., total Kg), whereas, the lean mass was no significantly affected by either treatments. No clear explanation is apparent for the observed preservation of lean mass in the presence of the rapid weight reduction, but is likely that the combination of protein administration and the exercise program explain these results [39, 40].
There are some concerns regarding the use of very low-calorie diets with or without ketosis. Such restricted calorie diets, as well as bariatric surgery, have been associated with adverse events, such as gallstones formation and sudden deaths [40, 41]. In addition, the regain of weight is a concern of very low-calorie diets [42]. It is possible that their risks may be reduced by informing the patients of these risks and the substantial efforts required to maintain the weight loss. The dropout rate in this study was similar to that expected in any program with obese individuals, but the dropout rate due to side effects was negligible. Many of the side effects were well tolerated and transient, i.e., they disappeared after the first few weeks of treatment.
This study exhibits a number of strengths and limitations. It involves a unicenter analysis which may have introduced some selection bias and baselines differences. However, the results were consistent when analyzed as total weight loss (in Kg) or as percentage of weight loss. Moreover, the results obtained from the BMI-stratified analysis were also consistent. The data were collected in consecutively enrolled participants in a real life setting. In addition, although the main analysis was performed with the completers, the results obtained when the data were analyzed using an intention-to-treat (ITT) approach [43, 44], the results were quite similar. The analysis of ITT-last observation carried forward and the ITT-basal observation carried forward showed weight reductions of −17.3 and −13.7 kg, respectively, and the ITT with multiple imputations also yielded a similar result of −19.9 kg. The results were higher encouraging toward the use of VLCK diets in obese patients although in the LC diet group the weight reduction was positive suggesting that the supporting method was very effective. However, a more extended follow-up of this type of diets is required to further evaluate its efficacy.
Conclusion
In summary, the present work demonstrated that a VLCK diet was significantly superior in the induction of weight loss in otherwise healthy obese patients compared with a standard LC diet. The induced weight loss was rapid, intense, and persistent, i.e., the same weight loss was obtained in the follow-up evaluation. The treatment was well tolerated and exhibited small dropout rate, and none of the patients who dropped out of the program cited collateral effects as the reason. Furthermore, the results at one year after starting the treatment showing that most of the patients still maintain a weight reduction higher than 10 % of the initial weight and that lean mass was preserved in the VLCK diet group, address an important concern in the current diet treatment methods of obesity [45–47].
References
K.M. Flegal, M.D. Carroll, B.K. Kit, C.L. Ogden, Prevalence of obesity and trends in the distribution of body mass index among US adults, 1999–2010. JAMA 307, 491–497 (2012)
F.F. Casanueva, B. Moreno, R. Rodríguez-Azeredo, C. Massien, P. Conthe, X. Formiguera et al., Relationship of abdominal obesity with cardiovascular disease, diabetes and hyperlipidaemia in Spain. Clin. Endocrinol. 73, 35–40 (2010)
J.L. Gutierrez-Fisac, P. Guallar-Castillon, L.M. Leon-Muñoz, A. Graciani, J.R. Banegas, F. Rodriguez-Artalejo, Prevalence of general and abdominal obesity in the adult population of Spain, 2008–2010: the ENRICA study. Obes. Rev. 13, 388–392 (2012)
J.C. Seidell, Obesity, insulin resistance and diabetes–a worldwide epidemic. Br. J. Nutr. 83(Suppl. 1), S5–S8 (2000)
X. Pi-Sunyer, The medical risks of obesity. Postgrad. Med. 121, 21–33 (2009)
L. Velazquez-Lopez, E. Gonzalez-Figueroa, P. Medina-Bravo, I. Pineda-Del Aguila, L. Avila-Jimenez, R. Ramos-Hernandez et al., Low calorie and carbohydrate diet: to improve the cardiovascular risk indicators in overweight or obese adults with prediabetes. Endocrine 43, 593–602 (2013)
M. Bergman, Inadequacies of current approaches to prediabetes and diabetes prevention. Endocrine 44, 623–633 (2013)
E.W. Gregg, H. Chen, L.E. Wagenknecht, J.M. Clark, L.M. Delahanty, J. Bantle et al., Association of an intensive lifestyle intervention with remission of type 2 diabetes. JAMA 308(23), 2489–2496 (2012)
B. Laferrère, Do we really know why diabetes remits after gastric bypass surgery? Endocrine 40, 162–167 (2011)
A.P. Shukla, S.M. Ahn, R.T. Patel, M.W. Rosenbaum, F. Rubino, Surgical treatment of type 2 diabetes: the surgeon perspective. Endocrine 40, 151–161 (2011)
L. Sjöström, M. Peltonen, P. Jacobson, C.D. Sjöström, K. Karason, H. Wedel et al., Bariatric surgery and long-term cardiovascular events. JAMA 307, 56–65 (2012)
G.B. Dodell, J.B. Albu, L. Attia, J. McGinty, F.X. Pi-Sunyer, B. Laferrère, The bariatric surgery patient: lost to follow-up; from morbid obesity to severe malnutrition. Endocr. Pract. 18, 21–25 (2012)
J.C. Halford, E.J. Boyland, J.E. Blundell, T.C. Kirkham, J.A. Harrold, Pharmacological management of appetite expression in obesity. Nat. Rev. Endocrinol 6, 255–269 (2010)
G. Di Dalmazi, V. Vicennati, R. Pasquali, U. Pagotto, The unrelenting fall of the pharmacological treatment of obesity. Endocrine 44, 598–609 (2013)
W.P. James, I.D. Caterson, W. Coutinho, N. Finer, L.F. Van Gaal, SCOUT Investigators. Effect of sibutramine on cardiovascular outcomes in overweight and obese subjects. N. Engl. J. Med. 363, 905–917 (2010)
R.V. Dvorak, A.M. Sharma, A. Astrup, Anti-obesity drugs: to be or not to be? Obes. Rev. 11, 833–834 (2010)
M. Hopkins, N.A. King, J.E. Blundell, Acute and long-term effects of exercise on appetite control: is there any benefit for weight control? Curr. Opin. Clin. Nutr. Metab. Care 13, 635–640 (2010)
D. Paddon-Jones, E. Westman, R.D. Mattes, R.R. Wolfe, A. Astrup, M. Westerterp-Plantenga, Protein, weight management, and satiety. Am. J. Clin. Nutr. 87, 1558S–1561S (2008)
A. Astrup, T. Meinert Larsen, A. Harper, Atkins and other low carbohydrate diets: hoax or an effective tool for weight loss? Lancet 364, 897–899 (2004)
A.B. Crujeiras, E. Goyenechea, I. Abete, M. Lage, M. Carreira, J.A. Martinez, F.F. Casanueva, Weight regain after a diet-induced loss is predicted by higher baseline leptin and lower ghrelin plasma levels. J. Clin. Endocrin. Metab. 95, 5037–5044 (2010)
E. Hemmingsson, K. Johansson, J. Eriksson, J. Sundstrom, M. Neovius, C. Marcus, Weight loss and dropout during a commercial weight-loss program including a very-low-calorie diet, a low-calorie diet or restricted normal food: observational cohort study. Am. J. Clin. Nutr. 96, 953–961 (2012)
E.C. Westman, W.S. Yancy, J.S. Edman, K.F. Tomlin, C.E. Perkins, Effect of low-calorie versus low-carbohydrate ketogenic diet in type 2 diabetes. Am. J. Med. 113, 30–36 (2002)
A.M. Johnstone, G.W. Horgan, D.M. Murison, D.M. Bremner, G.E. Lobley, Effects of a high-protein ketogenic diet on hunger, appetite, and weight loss in obese men feeding ad libitum. Am. J. Clin. Nutr. 87, 44–55 (2008)
T.A. Hussain, T.C. Mathew, A.A. Dashti, S. Asfar, N. Al-Zaid, H.M. Dashti, Effect of low-calorie versus low-carbohydrate ketogenic diet in type 2 diabetes. Nutrition 28, 1016–1021 (2012)
FAO/WHO/UNU Energy and protein requirements. Technical Report Series No 724. WorldHealthOrganization, Geneva, 1985
Recomendaciones nutricionales basadas en la evidencia para la prevencion y el tratamiento del sobrepeso y la obesidad en adultos, Rev. Esp. Obes. 10, suppl. 1 (2011)
SCOOP-VLCD task 7.3. Reports on tasks for scientific cooperation. Collection of data on products intended for use in very-low-calorie-diets. Report. Brussels. European Commission, September 2002
A. Astrup, S. Rossner, Lessons from obesity management programmes: greater initial weight loss improves long-term maintenance. Obes. Rev. 1, 17–19 (2000)
T.A. Wadden, R.H. Neiberg, R.R. Wing, J.M. Clark, L.M. Delahanty, J.O. Hill et al., Four-year weight losses in the look AHEAD study: factors associated with long-term success. Obesity (Silver Spring) 19, 1987–1998 (2011)
R.R. Wing, S. Phelan, Long-term weight loss maintenance. Am. J. Clin. Nutr. 82, 222S–225S (2005)
R.R. Wing, D.F. Tate, A.A. Gorin, H.A. Raynor, J.L. Fava, A self-regulation program for maintenance of weight loss. N. Engl. J. Med. 355, 1563–1571 (2006)
L. Sjöström, A. Rissanen, T. Andersen, M. Boldrin, A. Golay, H.P. Koppeschaar, M. Krempf, Randomised placebo-controlled trial of orlistat for weight loss and prevention of weight regain in obese patients. European multicentre orlistat study group. Lancet 352, 167–172 (1998)
J.P. Despres, A. Golay, L. Sjostrom, Effects of rimonabant on metabolic risk factors in overweight patients with dyslipidemia. N. Engl. J. Med. 353, 2121–2134 (2005)
L.F. Van Gaal, Rissanen AM, Scheen AJ, Ziegler O, Rossner S, for the RIO-Europe Study Group. Effects of the cannabinoid-1 receptor blocker rimonabant on weight reduction and cardiovascular risk factors in overweight patients: 1 year experience from the RIO-Europe study. Lancet 365, 1389–1397 (2005)
S. Smith, N.J. Weissman, C.M. Anderson, M. Sanchez, E. Chuang, S. Subbe et al., Multicenter, placebo-controlled trial of lorcaserin for weight management. N. Engl. J. Med. 363, 245–256 (2010)
M. Glandt, I. Raz, Present and future: pharmacologic treatment of Obesity. J. Obesity (2011). doi:10.1155/2011/636181
G.A. Bray, D.H. Ryan, D. Gordon, S. Heidingsfelder, F. Cerise, K. Wilson, A double-blind randomized placebo-controlled trial of sibutramine. Obes. Res. 4, 263–270 (1996)
W.H. Saris, Very low calorie diets and sustained weight loss. Obes. Res. 9(suppl. 4), 295S–301S (2001)
A.G. Tsai, T.A. Wadden, The evolution of very low calorie diets: an update and meta-analysis. Obesity 14, 1283–1293 (2006)
J.E. Everhart, Contributions of obesity and weight loss to gallstone disease. Ann. Intern. Med. 119, 1029–1035 (1993)
M.M. Finucane, G.A. Stevens, M.J. Cowan, G. Danaei, J.K. Lin, C.J. Paciorek et al., National, regional, and global trends in body-mass index since 1980: systematic analysis of health examination surveys and epidemiological studies with 960 country-years and 9.1 million participants. Lancet 377, 557–567 (2011)
D. Moher, S. Hopewell, K.F. Schulz, V. Montori, P.C. Gøtzsche, P.J. Devereaux, D. Elbourne, M. Egger, D.G. Altman, CONSORT 2010 explanation and elaboration: updated guidelines for reporting parallel group randomised trials. BMJ 340, c869 (2010)
K.F. Schulz, D.G. Altman, D. Moher, CONSORT 2010 statement: updated guidelines for reporting parallel group randomised trials. PLoSMed 7, e1000251 (2010)
N.B. Bueno, I.S. Vieira, S. Lima, T. Rocha, Very-low-carbohydrate ketogenic diet v. low-fat diet for long term weight loss: a meta-analysis of randomised control trials. Brit. J. Nutr. 110(7), 1178–1187 (2013)
C.B. Ebbeling, J.F. Swain, H.A. Feldman, W.W. Wong, D.L. Hachey, E. Garcia-Lago, D.S. Ludwig, Effects of dietary composition on energy expenditure during weight-loss maintenance. JAMA 307, 2627–2634 (2012)
S.G.J.A. Camps, S.P.M. Verhoef, K.R. Westerterp, Weight loss, weight maintenance, and adaptive thermogenesis. Am J Nutr 97, 990–994 (2013)
Acknowledgments
We acknowledge the Pronokal Division of Protein Supplies SL Spain for providing free of charge the diet of the ketosis group.
Disclosure
BM, DB, AG, and FFC received advisory board fees and or research grants from Pronokal Protein Supplies Spain
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moreno, B., Bellido, D., Sajoux, I. et al. Comparison of a very low-calorie-ketogenic diet with a standard low-calorie diet in the treatment of obesity. Endocrine 47, 793–805 (2014). https://doi.org/10.1007/s12020-014-0192-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-014-0192-3