Abstract
It is ironic that the liver, which serves a critical function in immune tolerance, itself becomes the victim of an autoimmune attack. Indeed, liver autoimmunity and the autoimmune diseases associated with both innate and adaptive responses to hepatocytes and/or cholangiocytes are models of human autoimmunity. For example, in primary biliary cirrhosis, there exists a well-defined and characteristic autoantibody and considerable homogeneity between patients. In autoimmune hepatitis, there are clinical characteristics that allow a rigorous subset definition and well-defined inflammatory infiltrates. In both cases, there are defects in a variety of immune pathways and including regulatory cells. In primary sclerosing cholangitis, with its characteristic overlap with inflammatory bowel disease, there are unique defects in innate immunity and particular important contribution of lymphoid homing to disease pathogenesis. In these diseases, as with other human autoimmune processes, there is the critical understanding that pathogenesis requires a genetic background, but is determined by environmental features, and indeed the concordance of these diseases in identical twins highlights the stochastic nature of immunopathology. Unfortunately, despite major advances in basic immunology and in immunopathology in these diseases, there remains a major void in therapy. The newer biologics that are so widely used in rheumatology, neurology, and gastroenterology have not yet seen success in autoimmune liver disease. Future efforts will depend on more rigorous molecular biology and systems analysis in order for successful application to be made to patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
In 1963, the first textbook on autoimmune diseases was published and written by Ian R. Mackay and F. Macfarlane Burnet. Mackay is perhaps the best known figure in liver autoimmunity and was a pioneer in a variety of contributions, including epidemiology, serology, and clinical diagnosis. At the time, Mackay worked at the Walter and Eliza Hall Institute (WEHI) for medical research. Macfarlane Burnet was the Director of WEHI and had received the Nobel Prize for his thesis on the forbidden clone.
This textbook is noteworthy for many features, not the least of which is that it was published in the Living Chemistry series. Because of the myriad and diversity of manuscripts within this special volume, we think it is appropriate to quote from the first three paragraphs of the preface [1]. “One of the greatest developments in Medicine during recent years has been the growing recognition of the importance of processes in which the immune mechanisms of the body are, as it were, turned against the body’s own components. This topic is in many ways in a pre-scientific phase. Many clinical scientists are skeptical of the reality of autoimmune processes and would prefer to look for antigens derived from the environment to account for the serological results observed. They are inclined to look on autoimmune disease as a currently fashionable diagnostic label, which can be attached to many diseases of still undefined etiology. We disagree strongly with this view, and in this short monograph, we have tried to provide an up to date and reasonably self-consistent account of those diseases for which an autoimmune etiology has been established either as a primary or an associated factor, or in which there is some likelihood that autoimmune processes are concerned. Allergic diseases, which are associated with extrinsic antigens, were not considered to be within the scope of this monograph. During the last 5 years, work in this Institute has centered on theoretical, experimental, and clinical aspects of immunity and an important part in the development of elective theories of immunity by Burnet came from association with the problems arising in clinical work. This has been the main stimulus for the production of this monograph. We believe that the central concept expressed in the term ‘forbidden clone’ is a valuable help to the understanding of autoimmune disease, and this concept is used as the basis of our present approach. The book is written more for physicians than for immunologists, and we have thought it would be helpful to include a glossary of immunological terms. Some of these terms have been used in a slightly different sense from the usual, but only when this was necessary to clarify the point of view we have adopted.”
Indeed, this pioneering textbook was only 323 pages long and included a glossary in which so-called “frequently used immunological terms” were included. Interestingly, among those terms was antigenic determinant, which we now know as epitopes, and autoclasia, a term which we no longer use, but which implies “a vicious cycle of immunologically induced damage, liberation of more antigen, fresh antigenic stimulation, further damage and so on”; perhaps an early term for apoptosis. But among the glossary is the key phrase “immunological tolerance.” Indeed, it is ironic that the liver, which itself is the basis of immune tolerance, becomes the victim in autoimmune liver disease.
In this respect, autoimmune/immune-mediated hepatobiliary diseases are attracting increasing attention based in no small part on the enormous contributions in basic biologic science that have allowed us to define more appropriate diagnostic and prognostic markers and an increasing recognition of the clinical significance of disease. It is also particularly important in understanding the geoepidemiology of autoimmunity.
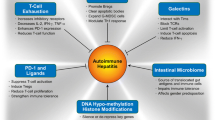
Autoimmunity is clearly a result of a genetic predisposition superimposed on environmental factors. These are best illustrated in studies of concordance in identical twins as well as the data that suggests that the only thing non-identical about identical twins is the immune response. On the other hand, the genetic predisposition, whose solution was once thought to be the defining moment in our understanding of autoimmunity, has become a disappointing quagmire despite enormous efforts in multiple autoimmune diseases [2–15]. For example, with rare exceptions, the major genetic contribution to the risk of autoimmunity remains the major histocompatibility complex. There have been no smoking guns in autoimmune liver disease and, in fact, in both primary biliary cirrhosis and autoimmune hepatitis, as well as numerous other human autoimmune diseases, the concepts of epigenetics are gaining increasing recognition [16–25]. Furthermore, despite a significant body of data on the microenvironment of the liver, the role of effector pathways, the development of animal models, and the use of biologics in other human autoimmune diseases, therapeutic advances in autoimmune liver disease, have been few and not of the rigor as in, for example, rheumatoid arthritis [2, 26–52].
Primary biliary cirrhosis (PBC), as the most frequent of the autoimmune/immune-mediated hepatobiliary diseases, reaches a prevalence of about 25–40 per 100,000 in North America and Northern Europe [53–55]. Genetic [56, 57], environmental [58], and endogenous factors [59–61] potentially contributing to the pathogenesis of PBC have been unraveled. Appropriate animal models are still in need [47, 62]. The association of PBC with numerous other autoimmune conditions is striking [63, 64] as is the female preponderance [65, 66]. The rates of extrahepatic malignancies associated with PBC [63] are much less intimidating than in primary sclerosing cholangitis (PSC), but deserve attention. Ursodeoxycholic acid (UDCA) remains the mainstay of therapy and enables a normal life expectancy for two of three patients when adequately responding to UDCA therapy [67–70]. New therapies, mostly in combination with UDCA, are under evaluation in phase 2 and 3 studies for treatment of those patients who do not adequately respond to UDCA alone [71–73]. The most frequent extrahepatic manifestations of PBC, fatigue [74] and pruritus [75], remain by far the major burden for the patients.
PSC has an about three times lower prevalence than PBC and affects most patients at younger age than PBC [54]. Two of three patients are male, and two of three suffer from inflammatory bowel disease (IBD). Genome-wide association studies have disclosed various risk genes in PSC [76–78], the majority of which do affect immune responses, but some may notably also affect cholangiocyte and intestinal barrier function [76, 79, 80] and the stability of the “biliary bicarbonate umbrella” [59, 81]. The search for adequate animal models of PSC is ongoing [62, 82], and the immunopathogenesis of PSC is increasingly unraveled [61, 83]. In clinical practice, a differentiation of PSC from various causes of secondary sclerosing cholangitis remains challenging, particularly when patients do not suffer from IBD, a characteristic feature of PSC, but not secondary forms of sclerosing cholangitis [54]. For PSC patients, pruritus is a major and sometimes barely tolerable complaint. An enormous burden for PSC patients is the high risk of hepatobiliary and extrahepatic malignancies, among them cholangiocarcinoma, gallbladder carcinoma, and colon carcinoma, which require annual (or biennial in selected cases) screening efforts [54, 84]. No medical treatment of proven benefit for long-term prognosis has been documented so far. UDCA improves serum liver tests and prognostic surrogate markers of PSC, but no survival benefit has been shown, whereby limited numbers of patients included and/or limited periods of follow-up may have contributed to the so far negative results [54]. The ongoing demand and unchanged rates of liver transplantation in PSC underline the urgent need for novel treatment options in PSC. Of note, the so far largest population-based analysis revealed a median survival of 21 years for a large cohort of 590 PSC patients from the Netherlands, 90 % of them under treatment with standard dose UDCA, a markedly longer median survival than that reported in previous selected cohorts mainly from tertiary centers.
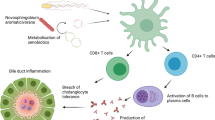
IgG4-related disease (IgG4-RD) with its biliary and pancreatic manifestations, IgG4-associated cholangitis (IAC) and autoimmune pancreatitis (AIP), has most recently received attention as an important differential diagnosis of PSC, but also of cholangiocarcinoma and pancreatic carcinoma [85]. The recent finding of a few dominant IgG4+ B cell clones in blood and affected tissue of patients with IgG4-RD [86], meanwhile confirmed extramurally [87], allows a new look on the so far elusive pathogenesis of this rare multi-organ disorder and makes the potential risk of long-term exposure to environmental toxins such as solvents, oil products, and paints, for developing IgG4-RD [88] more plausible. Still, the exact role of IgG4 and B and T cells in IgG4-RD remains obscure. The striking effect of immunosuppressive treatment in IAC and rapid disappearance of IgG4+ B cell clones [85, 86] reminds clinical hepatologists of autoimmune hepatitis (AIH) as does the frequent relapse after tapering down corticosteroids [89].
Finally, physicians are increasingly aware of the extrahepatic manifestations of autoimmune liver disease, including the roles of inflammation, the development of hepatocarcinoma, and, interestingly, the co-existence in many patients of Sjogren’s syndrome and scleroderma [64, 90–92]. There are several papers in this issue which are devoted to this theme, and, importantly, they illustrate the contributions of geoepidemiology in our understanding of immune tolerance. Of greatest importance is the absence of a major therapeutic advance. Certainly, the use of liver transplantation has been life-saving for many patients but, on the other hand, there are numerous new molecules and pathways being described in the immune response and hepatobiliary pathophysiology and it will be only through the molecular dissection of these pathways that will allow for adequate medical treatment. This is what we owe our patients and this is what we all strive for.
References
Mackay IR, Burnet F (1963) Autoimmune Diseases. Charles C. Thomas, Springfield
Berrih-Aknin S (2014) Myasthenia Gravis: paradox versus paradigm in autoimmunity. J Autoimmun 52:1–28
Collin R, Dugas V, Chabot-Roy G, et al (2015) Autoimmunity and antibody affinity maturation are modulated by genetic variants on mouse chromosome 12. J Autoimmun
Cui Y, Sheng Y, Zhang X (2013) Genetic susceptibility to SLE: recent progress from GWAS. J Autoimmun 41:25–33
de Boer YS, van Gerven NM, Zwiers A et al (2014) Genome-wide association study identifies variants associated with autoimmune hepatitis type 1. Gastroenterology 147:443–452, e445
Gatselis NK, Zachou K, Koukoulis GK, Dalekos GN (2015) Autoimmune hepatitis, one disease with many faces: etiopathogenetic, clinico-laboratory and histological characteristics. World J Gastroenterol 21:60–83
Hirschfield GM, Karlsen TH (2014) Genetic risks link autoimmune hepatitis to other autoimmune liver disease. Gastroenterology 147:270–273
Juran BD, Lazaridis KN (2014) Environmental factors in primary biliary cirrhosis. Semin Liver Dis 34:265–272
Kopec AK, Sullivan BP, Kassel KM, Joshi N, Luyendyk JP (2014) Toxicogenomic analysis reveals profibrogenic effects of trichloroethylene in autoimmune-mediated cholangitis in mice. Toxicol Sci 141:515–523
Lu Q (2013) The critical importance of epigenetics in autoimmunity. J Autoimmun 41:1–5
Lugli EB, Correia R, Fischer R et al (2015) Expression of citrulline and homocitrulline residues in the lungs of non-smokers and smokers: implications for autoimmunity in rheumatoid arthritis. Arthritis Res Ther 17:9
Wang L, Wang FS, Chang C, Gershwin ME (2014) Breach of tolerance: primary biliary cirrhosis. Semin Liver Dis 34:297–317
Yang W, Yao Y, Yang YQ et al (2014) Differential modulation by IL-17A of Cholangitis versus Colitis in IL-2Ralpha deleted mice. PLoS One 9:e105351
Zhang P, Zhao M, Liang G et al (2013) Whole-genome DNA methylation in skin lesions from patients with psoriasis vulgaris. J Autoimmun 41:17–24
Hirschfield GM, Gershwin ME (2013) The immunobiology and pathophysiology of primary biliary cirrhosis. Annu Rev Pathol 8:303–330
Agmon-Levin N, Arango MT, Kivity S et al (2014) Immunization with hepatitis B vaccine accelerates SLE-like disease in a murine model. J Autoimmun 54:21–32
Castiella A, Zapata E, Lucena MI, Andrade RJ (2014) Drug-induced autoimmune liver disease: a diagnostic dilemma of an increasingly reported disease. World J Hepatol 6:160–168
Chen RC, Naiyanetr P, Shu SA et al (2013) Antimitochondrial antibody heterogeneity and the xenobiotic etiology of primary biliary cirrhosis. Hepatology 57:1498–1508
Coit P, Yalavarthi S, Ognenovski M, et al (2015) Epigenome profiling reveals significant DNA demethylation of interferon signature genes in lupus neutrophils. J Autoimmun
deLemos AS, Foureau DM, Jacobs C, Ahrens W, Russo MW, Bonkovsky HL (2014) Drug-induced liver injury with autoimmune features. Semin Liver Dis 34:194–204
Fan Y, Gualtierotti G, Tajima A et al (2014) Compromised central tolerance of ICA69 induces multiple organ autoimmunity. J Autoimmun 53:10–25
Katoh H, Zheng P, Liu Y (2013) FOXP3: genetic and epigenetic implications for autoimmunity. J Autoimmun 41:72–78
Leung PS, Wang J, Naiyanetr P et al (2013) Environment and primary biliary cirrhosis: electrophilic drugs and the induction of AMA. J Autoimmun 41:79–86
Wang Q, Selmi C, Zhou X et al (2013) Epigenetic considerations and the clinical reevaluation of the overlap syndrome between primary biliary cirrhosis and autoimmune hepatitis. J Autoimmun 41:140–145
Zhao M, Liu S, Luo S et al (2014) DNA methylation and mRNA and microRNA expression of SLE CD4+ T cells correlate with disease phenotype. J Autoimmun 54:127–136
Adutler-Lieber S, Zaretsky I, Platzman I et al (2014) Engineering of synthetic cellular microenvironments: implications for immunity. J Autoimmun 54:100–111
Ando Y, Yang GX, Kenny TP et al (2013) Overexpression of microRNA-21 is associated with elevated pro-inflammatory cytokines in dominant-negative TGF-beta receptor type II mouse. J Autoimmun 41:111–119
Bowlus CL, Gershwin ME (2014) The diagnosis of primary biliary cirrhosis. Autoimmun Rev 13:441–444
Chang CH, Chen YC, Yu YH et al (2014) Innate immunity drives xenobiotic-induced murine autoimmune cholangitis. Clin Exp Immunol 177:373–380
Fuchs S, Aricha R, Reuveni D, Souroujon MC (2014) Experimental autoimmune myasthenia gravis (EAMG): from immunochemical characterization to therapeutic approaches. J Autoimmun 54:51–59
Gershwin ME, Krawitt EL (2014) Autoimmune hepatitis: 50 years of (slow) progress. Hepatology 59:754–756
Huang W, Kachapati K, Adams D et al (2014) Murine autoimmune cholangitis requires two hits: cytotoxic KLRG1(+) CD8 effector cells and defective T regulatory cells. J Autoimmun 50:123–134
Hudspeth K, Pontarini E, Tentorio P et al (2013) The role of natural killer cells in autoimmune liver disease: a comprehensive review. J Autoimmun 46:55–65
Kar SP, Seldin MF, Chen W et al (2013) Pathway-based analysis of primary biliary cirrhosis genome-wide association studies. Genes Immun 14:179–186
Kawata K, Tsuda M, Yang GX et al (2013) Identification of potential cytokine pathways for therapeutic intervention in murine primary biliary cirrhosis. PLoS One 8:e74225
Kawata K, Yang GX, Ando Y et al (2013) Clonality, activated antigen-specific CD8(+) T cells, and development of autoimmune cholangitis in dnTGFbetaRII mice. Hepatology 58:1094–1104
Kurth MJ, Yokoi T, Gershwin ME (2014) Halothane-induced hepatitis: paradigm or paradox for drug-induced liver injury. Hepatology 60:1473–1475
Lleo A, Oertelt-Prigione S, Bianchi I et al (2013) Y chromosome loss in male patients with primary biliary cirrhosis. J Autoimmun 41:87–91
Lleo A, Zhang W, McDonald WH et al (2014) Shotgun proteomics: identification of unique protein profiles of apoptotic bodies from biliary epithelial cells. Hepatology 60:1314–1323
Mayer CT, Ghorbani P, Kuhl AA et al (2014) Few Foxp3(+) regulatory T cells are sufficient to protect adult mice from lethal autoimmunity. Eur J Immunol 44:2990–3002
Ridgway WM, Gershwin ME (2014) Prometheus unbound: NKT cells inhibit hepatic regeneration. Hepatology 60:1133–1135
Selmi C, Cavaciocchi F, Lleo A et al (2014) Genome-wide analysis of DNA methylation, copy number variation, and gene expression in monozygotic twins discordant for primary biliary cirrhosis. Front Immunol 5:128
Sthoeger Z, Sharabi A, Mozes E (2014) Novel approaches to the development of targeted therapeutic agents for systemic lupus erythematosus. J Autoimmun 54:60–71
Tanaka H, Yang GX, Iwakoshi N et al (2013) Anti-CD40 ligand monoclonal antibody delays the progression of murine autoimmune cholangitis. Clin Exp Immunol 174:364–371
Tanaka H, Zhang W, Yang GX et al (2014) Successful immunotherapy of autoimmune cholangitis by adoptive transfer of forkhead box protein 3(+) regulatory T cells. Clin Exp Immunol 178:253–261
Wang J, Budamagunta MS, Voss JC et al (2013) Antimitochondrial antibody recognition and structural integrity of the inner lipoyl domain of the E2 subunit of pyruvate dehydrogenase complex. J Immunol 191:2126–2133
Wang J, Yang GX, Tsuneyama K, Gershwin ME, Ridgway WM, Leung PS (2014) Animal models of primary biliary cirrhosis. Semin Liver Dis 34:285–296
Wang L, Sun Y, Zhang Z et al (2015) CXCR5(+) CD4(+) T follicular helper cells participate in the pathogenesis of primary biliary cirrhosis. Hepatology 61:627–638
Xu X, Zhang S, Jin R et al (2015) Retention and tolerance of autoreactive CD4(+) recent thymic emigrants in the liver. J Autoimmun 56:87–97
Yang CY, Ma X, Tsuneyama K et al (2014) IL-12/Th1 and IL-23/Th17 biliary microenvironment in primary biliary cirrhosis: implications for therapy. Hepatology 59:1944–1953
Yao Y, Yang W, Yang YQ et al (2014) Distinct from its canonical effects, deletion of IL-12p40 induces cholangitis and fibrosis in interleukin-2Ralpha(−/−) mice. J Autoimmun 51:99–108
Zhang J, Zhang W, Leung PS et al (2014) Ongoing activation of autoantigen-specific B cells in primary biliary cirrhosis. Hepatology 60:1708–1716
Lindor KD, Gershwin ME, Poupon R, Kaplan M, Bergasa NV, Heathcote EJ (2009) Primary biliary cirrhosis. Hepatology 50:291–308
European Association for the Study of the Liver (2009) EASL clinical practice guidelines: management of cholestatic liver diseases. J Hepatol 51:237–267
Boonstra K, Beuers U, Ponsioen CY (2012) Epidemiology of primary sclerosing cholangitis and primary biliary cirrhosis: a systematic review. J Hepatol 56:1181–1188
Hirschfield G M, Siminovitch K A, (2014) Genetics in PBC: what do the “risk genes” teach us? Clin Rev Allergy Immunol
Dong M, Li J, Tang R, et al (2015) Multiple genetic variants associated with primary biliary Cirrhosis in a Han Chinese Population. Clin Rev Allergy Immunol
Gershwin ME, Mackay IR (2008) The causes of primary biliary cirrhosis: convenient and inconvenient truths. Hepatology 47:737–745
Beuers U, Hohenester S, de Buy Wenniger LJ, Kremer AE, Jansen PL, Elferink RP (2010) The biliary HCO(3)(−) umbrella: a unifying hypothesis on pathogenetic and therapeutic aspects of fibrosing cholangiopathies. Hepatology 52:1489–1496
Medina JF, Martinez A, Vazquez JJ, Prieto J (1997) Decreased anion exchanger 2 immunoreactivity in the liver of patients with primary biliary cirrhosis. Hepatology 25:12–17
Liaskou E, Hirschfield GM, Gershwin ME (2014) Mechanisms of tissue injury in autoimmune liver diseases. Semin Immunopathol 36:553–568
Pollheimer M J, Fickert P, (2014) Animal models in primary biliary cirrhosis and primary sclerosing cholangitis. Clin Rev Allergy Immunol
Floreani A, Spinazze A, Caballeria L, et al (2014) Extrahepatic malignancies in primary biliary cirrhosis: a comparative study at two European centers. Clin Rev Allergy Immunol
Sun Y, Zhang W, Li B, Zou Z, Selmi C, Gershwin M E, (2015) The coexistence of sjogren’s syndrome and primary biliary cirrhosis: a comprehensive review. Clin Rev Allergy Immunol
Sun Y, Haapanen K, Li B, Zhang W, Van de Water J, Gershwin, M E (2014) Women and primary biliary cirrhosis. Clin Rev Allergy Immunol
Floreani A, Infantolino C, Franceschet I, et al (2014) Pregnancy and primary biliary cirrhosis: a case–control study. Clin Rev Allergy Immunol
Corpechot C, Abenavoli L, Rabahi N et al (2008) Biochemical response to ursodeoxycholic acid and long-term prognosis in primary biliary cirrhosis. Hepatology 48:871–877
Corpechot C, Chazouilleres O, Poupon R (2011) Early primary biliary cirrhosis: biochemical response to treatment and prediction of long-term outcome. J Hepatol 55:1361–1367
Lammers WJ, van Buuren HR, Hirschfield GM et al (2014) Levels of alkaline phosphatase and bilirubin are surrogate end points of outcomes of patients with primary biliary cirrhosis: an international follow-up study. Gastroenterology 147:1338–1349.e5, quiz e1315
Pares A, Caballeria L, Rodes J (2006) Excellent long-term survival in patients with primary biliary cirrhosis and biochemical response to ursodeoxycholic Acid. Gastroenterology 130:715–720
Floreani A, Franceschet I, Perini L, Cazzagon N, Gershwin M E, Bowlus C L, (2014) New therapies for primary biliary cirrhosis. Clin Rev Allergy Immunol
Dyson J K, Hirschfield G M, Adams D H, et al (2015) Novel therapeutic targets in primary biliary cirrhosis. Nat Rev Gastroenterol Hepatol
Beuers U, Trauner M, Jansen P, Poupon R (2015) New paradigms in the treatment of hepatic cholestasis: from UDCA to FXR, PXR and beyond. J Hepatology 62, in press
Griffiths L, Jones DE (2014) Pathogenesis of primary biliary cirrhosis and its fatigue. Dig Dis 32:615–625
Beuers U, Kremer AE, Bolier R, Elferink RP (2014) Pruritus in cholestasis: facts and fiction. Hepatology 60:399–407
Folseraas T, Liaskou E, Anderson C A, Karlsen T H (2014) Genetics in PSC: What do the “risk genes” teach us? Clin Rev Allergy Immunol
Karlsen TH, Franke A, Melum E et al (2010) Genome-wide association analysis in primary sclerosing cholangitis. Gastroenterology 138:1102–1111
Folseraas T, Melum E, Rausch P et al (2012) Extended analysis of a genome-wide association study in primary sclerosing cholangitis detects multiple novel risk loci. J Hepatol 57:366–375
Maroni L, van de Graaf S F, Hohenester S D, Oude Elferink R P, Beuers U (2014) Fucosyltransferase 2: a genetic risk factor for primary sclerosing cholangitis and crohn’s disease-a comprehensive review. Clin Rev Allergy Immunol
Keitel V, Reich M, Haussinger D (2014) TGR5: pathogenetic role and/or therapeutic target in fibrosing cholangitis? Clin Rev Allergy Immunol
Hohenester S, Wenniger LM, Paulusma CC et al (2012) A biliary HCO3- umbrella constitutes a protective mechanism against bile acid-induced injury in human cholangiocytes. Hepatology 55:173–183
Fickert P, Pollheimer MJ, Beuers U et al (2014) Characterization of animal models for primary sclerosing cholangitis (PSC). J Hepatol 60:1290–1303
Liaskou E, Jeffery LE, Trivedi PJ et al (2014) Loss of CD28 expression by liver-infiltrating T cells contributes to pathogenesis of primary sclerosing cholangitis. Gastroenterology 147(221–232):e227
Chapman R, Fevery J, Kalloo A et al (2010) Diagnosis and management of primary sclerosing cholangitis. Hepatology 51:660–678
Hubers L M, Maillette de Buy Wenniger L J, Doorenspleet M E, et al (2014) IgG4-associated cholangitis: a comprehensive review. Clin Rev Allergy Immunol
de Buy M, Wenniger LJ, Doorenspleet ME, Klarenbeek PL et al (2013) Immunoglobulin G4+ clones identified by next-generation sequencing dominate the B cell receptor repertoire in immunoglobulin G4 associated cholangitis. Hepatology 57:2390–2398
Wallace ZS, Mattoo H, Carruthers M et al (2015) Plasmablasts as a biomarker for IgG4-related disease, independent of serum IgG4 concentrations. Ann Rheum Dis 74:190–195
de Buy Wenniger LJ, Culver EL, Beuers U (2014) Exposure to occupational antigens might predispose to IgG4-related disease. Hepatology 60:1453–1454
Ghazale A, Chari ST, Zhang L et al (2008) Immunoglobulin G4-associated cholangitis: clinical profile and response to therapy. Gastroenterology 134:706–715
Gong YZ, Nititham J, Taylor K et al (2014) Differentiation of follicular helper T cells by salivary gland epithelial cells in primary Sjogren’s syndrome. J Autoimmun 51:57–66
Invernizzi P, Gershwin ME (2014) New therapeutics in primary biliary cirrhosis: will there ever be light? Liver Int 34:167–170
Seror R, Theander E, Bootsma H et al (2014) Outcome measures for primary Sjogren’s syndrome: a comprehensive review. J Autoimmun 51:51–56
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Beuers, U., Gershwin, M.E. Unmet Challenges in Immune-Mediated Hepatobiliary Diseases. Clinic Rev Allerg Immunol 48, 127–131 (2015). https://doi.org/10.1007/s12016-015-8484-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12016-015-8484-9