Abstract
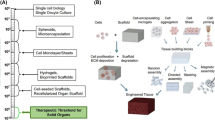
Recent advances in tissue engineering have adapted the additive manufacturing technology, also known as three-dimensional printing, which is used in several industrial applications, for the fabrication of bioscaffolds and viable tissue and/or organs to overcome the limitations of other in vitro conventional methods. 3D bioprinting technology has gained enormous attention as it enabled 3D printing of a multitude of biocompatible materials, different types of cells and other supporting growth factors into complex functional living tissues in a 3D format. A major advantage of this technology is its ability for simultaneously 3D printing various cell types in defined spatial locations, which makes this technology applicable to regenerative medicine to meet the need for suitable for transplantation suitable organs and tissues. 3D bioprinting is yet to successfully overcome the many challenges related to building 3D structures that closely resemble native organs and tissues, which are complex structures with defined microarchitecture and a variety of cell types in a confined area. An integrated approach with a combination of technologies from the fields of engineering, biomaterials science, cell biology, physics, and medicine is required to address these complexities. Meeting this challenge is being made possible by directing the 3D bioprinting to manufacture biomimetic-shaped 3D structures, using organ/tissue images, obtained from magnetic resonance imaging and computerized tomography, and employing computer-aided design and manufacturing technologies. Applications of 3D bioprinting include the generation of multilayered skin, bone, vascular grafts, heart valves, etc. The current 3D bioprinting technologies need to be improved with respect to the mechanical strength and integrity in the manufactured constructs as the presently used biomaterials are not of optimal viscosity. A better understanding of the tissue/organ microenvironment, which consists of multiple types of cells, is imperative for successful 3D bioprinting.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Tissue engineering field has seen many recent advances that led to the building of tissues and parts of organs (e.g., heart tissue, blood vessels, heart valve trachea, etc.) [1–3] that can be functional in vivo and can be used to successfully replace a defective or diseased tissue or organ. The primary approach for these advances is to seed cells, preferably stem cells, onto solid and biodegradable scaffolds, along with the needed biofactors that stimulate tissue formation. Even though the recent advances in bioscaffold engineering have shown promising results in building a number of tissues clinically [4, 5], there are many challenges that still need to be addressed in building composite tissues and organs and these include not optimal procedures for scaffold fabrication, limited biomaterial availability, and methods for growing different cell types at precise locations in a given bioscaffold to facilitate blood vessel or a neuronal generation following implantation [6]. While there are several methods for building bioscaffolds with the overall structural requirements, the precise microarchitecture—both external and internal, and the pore structure that determines the transport oxygen and nutrients for cell survival, has not yet been achieved [7].
Recent advances in tissue engineering have borrowed the additive manufacturing (AM) technology, also known as three-dimensional printing, which is used in several industrial applications, to fabricate bioscaffolds to overcome the limitations of conventional methods. This technique, developed originally to print thin layers of material sequentially followed by curing with UV light to form solid 3D structures, was named as ‘stereolithography,’ and was first described by Charles W. Hull in 1986 [8]. Further developments in this technique enabled solvent-free, aqueous-based direct printing of biological materials into 3D scaffolds that could be used for transplantation [9]. AM technology/3D bioprinting is currently being developed to manufacture biomimetic-shaped 3D structures, using organ/tissue images, from magnetic resonance imaging (MRI) and computerized tomography (CT) as templates, employing computer-aided design and manufacturing (CAD/CAM) technologies [7, 10]. Since 3D bioprinting can be combined with CAD/CAM technology to manufacture a structure with an accurate anatomical shape [11] and as this technique makes it possible to directly deposit cells in an organotypic architecture, 3D bioprinting is attracting considerable attention as a method of choice for precisely engineered tissue construction. Spatial control of the layer-by-layer precise placement of functional components such as living cells, biological materials, bioactive factors, during the fabrication of 3D biostructures, makes 3D bioprinting a unique tissue engineering technique.
Types of 3D Bioprinting Technologies
There are three major types of 3D bioprinting techniques available currently and these are inkjet bioprinting, microextrusion bioprinting, and laser-assisted bioprinting (Fig. 1). Each of these approaches has been studied widely and has many advantages and drawbacks (Table 1).
Illustration of different types of 3D bioprinting technologies. Inkjet-based bioprinters (a, b) form picoliter droplets and are mainly of two types, the thermal inkjet printers (a), which use electric heat in the print head to produce air pressure to form and eject the droplet and acoustic/piezoelectric bioprinters (b), which use pulses formed by piezoelectric or ultrasound pressure for droplet ejection. Microextrusion printers (c, d) use pressure generated with either pneumatic (c) or piston (d) to extrude continuous filaments/beads of biomaterial through a micro-nozzle. Laser-assisted printers (e) employ focused laser energy on an absorbing substrate for generating pressures in order to eject biomaterial containing cells onto a collector substrate. In all the three printers, a precise control of x–y–z axes is maintained to fabricate the desired 3D biostructure
Inkjet 3D Bioprinting
Inkjet-based bioprinting is a non-contact technique in which, picoliter droplets of biomaterials are layered onto a substratum in order to produce 2D and 3D structures [7, 12, 13]. Inkjet 3D bioprinters are also known as drop-on-demand printers and are the most commonly used. Simply stated, the ink in the cartridge is replaced with a biological material containing cells and or other biofactors, and the paper is replaced with an electronically controlled elevator stage with precisely controlled z axis position [14]. On the basis of the mechanism employed to produce the bio-ink droplet, inkjet bioprinting can be done either by thermal method, piezoelectric actuator, and by laser-induced forward transfer and pneumatic pressure [15, 16] (Fig. 1). The thermal printers cause electric heating of the print head in order to produce pressure pulses that force droplets out of the nozzle. The local heating generates a bubble in the bio-ink chamber and ejects a small droplet. Even though this localized heating in thermal printers can range from 200 to 300 °C, it lasts for a very short duration of ~2 μs and results in overall temperature increase by a maximum of 4–10 °C [17], and thus it does not exert any significant impact either on the stability of biological molecules [18], or on the viability or function of mammalian cells [19]. While thermal inkjet printers are of low cost, readily available and can give high print speeds, they suffer from the disadvantages including the lack of precise directionality and size of droplet, thermal and mechanical stress to cells and biomaterials, frequent nozzle clogging, and unreliable cell encapsulation.
The 3D bioprinters that use piezoelectric mechanism contain a piezoelectric crystal, which in response to applied voltage, induces a rapid change in shape and creates an acoustic wave inside the print head. This acoustic wave helps in breaking the liquid in the print head into many droplets and their ejection at regular intervals [20]. A combination of multiple acoustic ejectors in the format of an adjustable array can facilitate printing of different cell and biomaterial types, simultaneously [21]. One major concern in using the piezoelectric mechanism-based bioprinters is that the 15–25 kHz frequencies employed in these printers and their potential to induce cellular damage [22].
Advantages and Disadvantages of Inkjet 3D Bioprinting
Inkjet-based 3D bioprinting methods can yield high-resolution (20–100 μ) structures. Inkjet printing can alter the drop size and density and thus has the capacity to introduce concentration gradients of cells, materials, or growth factors throughout the 3D structure [23]. The drop size (from <1 to >300 pl) and drop deposition rate (1–10,000 droplets/s) can be controlled electronically [24, 25]. Patterns of single drops, each containing one or two cells, in lines ~50 μm wide, have been printed; however, because of the very low droplet volume (pl), their processing time is prolonged. Besides, high viscosity of the material in the bio-ink chamber (because of proteins, hydrogels, and living cells in high density) cannot be effectively used for delivering picoliter volumes. The low viscous materials (viscosity <10 centipoise) such as thrombin and fibrogen that have been preferably used in many cases as bio-inks for bioprinting show weak mechanical properties with low durability of the manufactured 3D structures [16, 26, 27]. Examples where the inkjet 3D bioprinting has been successfully implemented include the regeneration of functional skin and cartilage [28, 29] in situ. The inkjet bioprinting not only facilitated the deposition of primary cells and/or stem cells, with required density, throughout the area of lesion, followed by rapid crosslinking of the cell-containing material via biocompatible chemical reaction or UV light-photoinitiator and crosslinking but also maintained high cell viability and function after printing. While these examples illustrate the potential of inkjet-based 3D bioprinting for the production of functional structures, addressing the above-mentioned drawbacks can certainly make this technique an invaluable tissue engineering tool because of its versatility and low cost.
Microextrusion 3D Bioprinting
Microextrusion 3D bioprinters are the most common as they are most affordable and the printers consist of a temperature-controlled biomaterial dispensing system, a stage, that is capable of moving along the x, y, and z axes, a fiber-optic light illuminated deposition area for photoinitiator activation, a humidifier controlled by piezoelectricity, and a video camera for x–y–z command and control. Unlike the inkjet printers, which generate droplets, microextrusion procedure produces continuous beads of material that are deposited in two dimensions, as directed by the CAD-CAM software, and the deposited layer serves as a foundation for the subsequent layer while the stage or microextrusion head is moved along the z axis, finally resulting in the formation of a 3-dimensional structure. By controlling the level of pneumatic pressure or the displacement of the piston of the pump syringe pump, the amount of dispensed cell-laden hydrogel by the microextrusion printer can be adjusted [30]. Following 2D pattern printing of the hydrogels, these are solidified and stacked layer by layer, to form 3D structures.
Advantages and Disadvantages of Microextrusion 3D Bioprinting
Since high viscosity biomaterials such as hydrogels, biocompatible copolymers, and cell spheroids can be printed through the micro-nozzle [12], a wider selection of biomaterials can be used in microextrusion 3D bioprinters (Table 1) and this allows easy scale-up of 3D structure fabrication [31, 32]. While few reports indicated cell viability in the 3D structures made by microextrusion bioprinters to be >90 % [11], others reported that cell survival rates are lower than those seen with the inkjet printers, in the range of 40–86 %, with the survival rate decreasing with increasing extrusion pressure [33]. Since achieving the high physiological cell densities in tissue-engineered organs is the major goal, the capability to deposit very high cell densities is an advantage of microextrusion 3D bioprinting. Multicellular cell spheroids, which possess the mechanical and functional properties of the tissue ECM, can be deposited to self-assemble into 3D structure by the microextrusion printers [34]. This strategy of the self-assembling spheroids can potentially accelerate tissue organization and the formation of complex structures without the need for additional scaffolds.
Microextrusion bioprinters have been instrumental in the fabrication of aortic valves [35] and branched vessels [36] and more recently for the generation of adipose-derived stromal vascular fraction [37] and HepG2-cell-laden photolabile ECM-derived hydrogels [38].
Laser-Assisted 3D Bioprinting
Laser-assisted 3D bioprinting (LAB) works on the principles of laser-induced forward transfer [39]. LAB is less common than inkjet or microextrusion bioprinting, but its applications for tissue- and organ-engineering are steadily increasing. A laser-assisted 3D bioprinter consists of a pulsed laser beam with a focusing system, a ‘ribbon’ that has a donor transport support, typically made from glass covered with a laser-energy-absorbing layer (e.g., gold or titanium) and a layer of biological material containing cells and/or hydrogel, and a receiving substrate facing the ribbon. Laser-assisted 3D bioprinter focuses laser pulses on the absorbing gold-layer of the ribbon and this generates a high-pressure bubble, which in turn propels cell-containing materials toward the collector substrate. LAB can deposit cells at a density of up to 108 cells/ml with the resolution of a single cell per drop using a laser pulse, at high speed [40]. Essentially, this method makes use of laser system to generate vaporization and to produce a small droplet (Fig. 1).
Although this technique is able to produce relatively higher resolution patterns, it probably suffers from lower cell viability in the printed hydrogel in comparison to other inkjet mechanisms [11]. The problems of nozzle clogging with cells or materials, that are major drawbacks of other bioprinting technologies, are not seen with LAB, which is nozzle free. Another advantage of LAB is its compatibility with a wide range of biomaterial viscosities (1–300 mPa/s) and its ability to print mammalian cells without significant effect on cell viability and function [41]. Applications of LAB in tissue engineering include the fabrication of a cellularized skin construct [42] and deposition of nano-hydroxyapatite in a mouse calvaria 3D defect model, in vivo [43] (Table 1).
Approaches for 3D Bioprinting Using Medical Imaging
Since 3D bioprinting can be combined with CAD/CAM technology, it allows the manufacture of biomimetic-shaped 3D structures unique to the target tissue or organ, using patients’ medical images [7]. Magnetic resonance imaging, CT scans, and other radiographic images are the major source for obtaining 3D volumetric information of a defected tissue or organ. This information is stored in a digital imaging and communications in medicine (DICOM) file, which is then used to develop the CAD model by a reverse engineering process. Thus, this process transforms ‘analytical anatomy’ into ‘synthetic anatomy’ [44]. Making use of the 3D CAD model thus created from DICOM files, a fabrication code is built, which can be used to communicate with the 3D bioprinter by CAM technology.
Future Perspectives
Despite a great degree of research effort to enhance the fabrication resolution of the different kinds of 3D bioprinters, several hurdles remain in the fabrication of 3D biostructures with high resolution. Even though one cannot foresee the use of 3D bioprinting for generating fully functional organs in the near future, there is great potential and promise for the applications of this technology in the field of tissue engineering medicine. Main drawbacks of the current bioprinting technologies that need to be addressed are the lack of mechanical strength and integrity in the manufactured constructs because of the biomaterials that are being used due to viscosity issues and therefore improvements in the ability to use high viscous materials must be a priority. A better understanding of the microenvironment of the tissues and organs and their structure, which consists of multiple types of cells, with a micrometer scale resolution is imperative for successful 3D bioprinting.
References
Akhyari, P., Kamiya, H., Haverich, A., Karck, M., & Lichtenberg, A. (2008). Myocardial tissue engineering: The extracellular matrix. European Journal of Cardio-Thoracic Surgery, 34, 229–241.
Delaere, P., Vranckx, J., Verleden, G., de Leyn, P., van Raemdonck, D., & Leuven Tracheal Transplant Group. (2010). Tracheal allotransplantation after withdrawal of immunosuppressive therapy. New England Journal of Medicine, 362, 138–145.
Hamilton, N., Bullock, A. J., Macneil, S., Janes, S. M., & Birchall, M. (2014). Tissue engineering airway mucosa: A systematic review. Laryngoscope, 124, 961–968.
Macchiarini, P., Jungebluth, P., Go, T., Asnaghi, M. A., Rees, L. E., Cogan, T. A., et al. (2008). Clinical transplantation of a tissue-engineered airway. Lancet, 372, 2023–2030.
Place, E. S., Evans, N. D., & Stevens, M. M. (2009). Complexity in biomaterials for tissue engineering. Nature Materials, 8, 457–470.
Billiet, T., Vandenhaute, M., Schelfhout, J., van Vlierberghe, S., & Dubruel, P. (2012). A review of trends and limitations in hydrogel-rapid prototyping for tissue engineering. Biomaterials, 33, 6020–6041.
Seol, Y. J., Kang, T. Y., & Cho, D. W. (2012). Solid freeform fabrication technology applied to tissue engineering with various biomaterials. Soft Matter, 8, 1730–1735.
Hull, C. W. (1986). Apparatus for production of three-dimensional objects by stereolithography. GOOGLE PATENTS. US 4575330 A.
Nakamura, M., Iwanaga, S., Henmi, C., Arai, K., & Nishiyama, Y. (2010). Biomatrices and biomaterials for future developments of bioprinting and biofabrication. Biofabrication, 2, 014110.
Murphy, S. V., & Atala, A. (2014). 3d bioprinting of tissues and organs. Nature Biotechnology, 32, 773–785.
Seol, Y. J., Kang, H. W., Lee, S. J., Atala, A., & Yoo, J. J. (2014). Bioprinting technology and its applications. European Journal of Cardio-Thoracic Surgery, 46, 342–348.
Peltola, S. M., Melchels, F. P., Grijpma, D. W., & Kellomaki, M. (2008). A review of rapid prototyping techniques for tissue engineering purposes. Annals of Medicine, 40, 268–280.
Mironov, V., Kasyanov, V., & Markwald, R. R. (2011). Organ printing: From bioprinter to organ biofabrication line. Current Opinion in Biotechnology, 22, 667–673.
Xu, T., Kincaid, H., Atala, A., & Yoo, J. J. (2008). High-throughput production of single-cell microparticles using an inkjet printing technology. Journal of Manufacturing Science and Engineering, 130, 021017.
Boland, T., Xu, T., Damon, B., & Cui, X. (2006). Application of inkjet printing to tissue engineering. Biotechnology Journal, 1, 910–917.
Lee, W., Debasitis, J. C., Lee, V. K., Lee, J. H., Fischer, K., Edminster, K., et al. (2009). Multi-layered culture of human skin fibroblasts and keratinocytes through three-dimensional freeform fabrication. Biomaterials, 30, 1587–1595.
Cui, X., Dean, D., Ruggeri, Z. M., & Boland, T. (2010). Cell damage evaluation of thermal inkjet printed chinese hamster ovary cells. Biotechnology and Bioengineering, 106, 963–969.
Okamoto, T., Suzuki, T., & Yamamoto, N. (2000). Microarray fabrication with covalent attachment of DNA using bubble jet technology. Nature Biotechnology, 18, 438–441.
Xu, T., Gregory, C. A., Molnar, P., Cui, X., Jalota, S., Bhaduri, S. B., & Boland, T. (2006). Viability and electrophysiology of neural cell structures generated by the inkjet printing method. Biomaterials, 27, 3580–3588.
Tekin, E., Smith, P. J., & Schubert, U. S. (2008). Inkjet printing as a deposition and patterning tool for polymers and inorganic particles. Soft Matter, 4, 703–713.
Tasoglu, S., & Demirci, U. (2013). Bioprinting for stem cell research. Trends in Biotechnology, 31, 10–19.
Cui, X., Boland, T., D’Lima, D. D., & Lotz, M. K. (2012). Thermal inkjet printing in tissue engineering and regenerative medicine. Recent Patents on Drug Delivery and Formulation, 6, 149–155.
Phillippi, J. A., Miller, E., Weiss, L., Huard, J., Waggoner, A., & Campbell, P. (2008). Microenvironments engineered by inkjet bioprinting spatially direct adult stem cells toward muscle- and bone-like subpopulations. Stem Cells, 26, 127–134.
Sekitani, T., Noguchi, Y., Zschieschang, U., Klauk, H., & Someya, T. (2008). Organic transistors manufactured using inkjet technology with subfemtoliter accuracy. Proceedings of the National Academy of Science USA, 105, 4976–4980.
Singh, M., Haverinen, H. M., Dhagat, P., & Jabbour, G. E. (2010). Inkjet printing-process and its applications. Advanced Materials, 22, 673–685.
Cui, X., & Boland, T. (2009). Human microvasculature fabrication using thermal inkjet printing technology. Biomaterials, 30, 6221–6227.
Kim, J. D., Choi, J. S., Kim, B. S., Chan Choi, Y., & Cho, Y. W. (2010). Piezoelectric inkjet printing of polymers: Stem cell patterning on polymer substrates. Polymer, 51, 2147–2154.
Skardal, A., Mack, D., Kapetanovic, E., Atala, A., Jackson, J. D., Yoo, J., & Soker, S. (2012). Bioprinted amniotic fluid-derived stem cells accelerate healing of large skin wounds. Stem Cells Translational Medicine, 1, 792–802.
Cui, X., Breitenkamp, K., Finn, M. G., Lotz, M., & D’Lima, D. D. (2012). Direct human cartilage repair using three-dimensional bioprinting technology. Tissue Engineering Part A, 18, 1304–1312.
Khalil, S., & Sun, W. (2009). Bioprinting endothelial cells with alginate for 3d tissue constructs. Journal of Biomechanical Engineering, 131, 111002.
Fedorovich, N. E., de Wijn, J. R., Verbout, A. J., Alblas, J., & Dhert, W. J. (2008). Three-dimensional fiber deposition of cell-laden, viable, patterned constructs for bone tissue printing. Tissue Engineering Part A, 14, 127–133.
Cohen, D. L., Malone, E., Lipson, H., & Bonassar, L. J. (2006). Direct freeform fabrication of seeded hydrogels in arbitrary geometries. Tissue Engineering, 12, 1325–1335.
Chang, R., Nam, J., & Sun, W. (2008). Effects of dispensing pressure and nozzle diameter on cell survival from solid freeform fabrication-based direct cell writing. Tissue Engineering Part A, 14, 41–48.
Marga, F., Jakab, K., Khatiwala, C., Shepherd, B., Dorfman, S., Hubbard, B., et al. (2012). Toward engineering functional organ modules by additive manufacturing. Biofabrication, 4, 022001.
Duan, B., Hockaday, L. A., Kang, K. H., & Butcher, J. T. (2013). 3d bioprinting of heterogeneous aortic valve conduits with alginate/gelatin hydrogels. Journal of Biomedical Materials Research Part A, 101, 1255–1264.
Norotte, C., Marga, F. S., Niklason, L. E., & Forgacs, G. (2009). Scaffold-free vascular tissue engineering using bioprinting. Biomaterials, 30, 5910–5917.
Williams, S. K., Touroo, J. S., Church, K. H., & Hoying, J. B. (2013). Encapsulation of adipose stromal vascular fraction cells in alginate hydrogel spheroids using a direct-write three-dimensional printing system. Bioresearch Open Access, 2, 448–454.
Bertassoni, L. E., Cardoso, J. C., Manoharan, V., Cristino, A. L., Bhise, N. S., Araujo, W. A., et al. (2014). Direct-write bioprinting of cell-laden methacrylated gelatin hydrogels. Biofabrication, 6, 024105.
Barron, J. A., Ringeisen, B. R., Kim, H., Spargo, B. J., & Chrisey, D. B. (2004). Application of laser printing to mammalian cells. Thin Solid Films, 453, 383–387.
Guillotin, B., Souquet, A., Catros, S., Duocastella, M., Pippenger, B., Bellance, S., et al. (2010). Laser assisted bioprinting of engineered tissue with high cell density and microscale organization. Biomaterials, 31, 7250–7256.
Gruene, M., Pflaum, M., Hess, C., Diamantouros, S., Schlie, S., Deiwick, A., et al. (2011). Laser printing of three-dimensional multicellular arrays for studies of cell-cell and cell-environment interactions. Tissue Engineering Part C: Methods, 17, 973–982.
Michael, S., Sorg, H., Peck, C. T., Koch, L., Deiwick, A., Chichkov, B., et al. (2013). Tissue engineered skin substitutes created by laser-assisted bioprinting form skin-like structures in the dorsal skin fold chamber in mice. PLoS One, 8, e57741.
Keriquel, V., Guillemot, F., Arnault, I., Guillotin, B., Miraux, S., Amedee, J., et al. (2010). In vivo bioprinting for computer- and robotic-assisted medical intervention: Preliminary study in mice. Biofabrication, 2, 014101.
Mironov, V., Trusk, T., Kasyanov, V., Little, S., Swaja, R., & Markwald, R. (2009). Biofabrication: A 21st century manufacturing paradigm. Biofabrication, 1, 022001.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, X., Zhang, Y. Tissue Engineering Applications of Three-Dimensional Bioprinting. Cell Biochem Biophys 72, 777–782 (2015). https://doi.org/10.1007/s12013-015-0531-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-015-0531-x