Abstract
The objective of this study is to assess the levels of heavy metals (cadmium, lead, antimony, mercury, and arsenic), which are also called endocrine-disrupting chemicals, and trace elements (chromium-III, chromium-VI, zinc, copper, and selenium) vs. monocyte to HDL ratio among pregnant women with gestational diabetes mellitus (GDM). A total of 112 pregnant women (60 with GDM and 52 healthy women) were included in this case-control study. Analysis of heavy metals and trace elements were performed in inductively coupled plasma mass spectrometer. Heavy metals (cadmium, lead, antimony, mercury, and arsenic), trace elements (chromium-III, chromium-VI, zinc, copper, and selenium), and metabolic parameters were assessed in both groups. It was determined that the levels of cadmium, lead, antimony, and copper were higher (p < 0.05) and levels of chromium-III, zinc, and selenium were lower (p < 0.05) among the GDM group compared to the control group, whereas there was a statistically insignificant difference between the two groups, regarding the levels of copper, mercury, and arsenic (p > 0.05). Moreover, the monocyte to HDL ratio was higher in the GDM group (p < 0.05), and the insulin resistance was significantly higher as well (p < 0.05). The results of our study demonstrated that environmental factors could be effective in the etiology of GDM. Toxic heavy metals, through inducing Cu, OS, and chronic inflammation, and other trace elements, either directly by impacting insulin secretion or through weakening the body’s antioxidant defense system, could play a role in the occurrence of GDM.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Gestational diabetes mellitus (GDM) is one of the most prevalent complications of pregnancy, which is diagnosed for the first time during pregnancy and progresses with different degrees of glucose intolerance [1]. It is a public health issue with an increasing prevalence, in line with the increasing prevalence of obesity and diabetes mellitus across the world. Even though its prevalence is higher in Far East Asian countries, the International Diabetes Federation estimates this rate as 14% worldwide [2, 3]. GDM prevalence in studies conducted in Turkey is between 4.8 and 6.2% [4, 5]. GDM, with its short- and long-term complications, threatens maternal and fetal health. Risk factors of GDM include advanced maternal age, obesity, race, medical history of GDM in previous pregnancies, and history of type 2 diabetes mellitus (T2DM) in parents; however, information related to the impact of environmental factors on GDM is limited [6].
Heavy metals are used in industrial products. Heavy metal exposure occurs through food intake, drinking water, and inhaling polluted air [7]. It has now been determined that environmental chemical exposure could lead to an increase in the incidence of GDM [8, 9]. The fact that it can show toxic effects even in small amounts indicates the significance of heavy metal exposure. Some heavy metals such as cadmium (Cd), arsenic (As), mercury (Hg), antimony (Sb), and lead (Pb) have been termed as endocrine disruptors. It has been revealed that Cd damages β cells through increasing oxidative stress in the pancreas [10]. It has been determined in animal trials that Pb can induce hyperglycemia and glucose intolerance through hepatic gluconeogenesis [11]. Although it is not clearly understood how As affects the formation of GDM, experimental studies have shown that As can affect β cell functions [12]. Moreover, the negative effects of Sb exposure on glucose metabolism during pregnancy have been demonstrated [13]. It has been determined that Hg may play a role in the development of diabetes mellitus through inflammation and oxidative stress [14].
Trace elements, which consist of elements such as copper (Cu), selenium (Se), zinc (Zn), and molybdenum (Mo), are mostly taken with foods. The level of trace elements decreases with oxidative stress, which increases in diabetes mellitus [15]. Se is necessary for the synthesis of selenoproteins, which are known with their antioxidant and cytoprotective properties. Besides, Se has effects such as facilitating the entry of glucose into the cell and regulating the metabolic process, namely, insulin-mimetic [16]. Chromium (Cr) manifests its impact generally through the insulin receptor [17]. It has been determined that the level of Cr in the blood decreases with oxidative stress, which occurs secondary to hyperglycemia [18]. Zn plays an active role in a wide range of different processes, from cytokine production to apoptosis. There are also studies in the literature in which the increase of trace elements compared to the control group is associated with GDM [19].
Oxidative stress (OS) draws the attention as a remarkable component in the pathophysiology of many illnesses [20, 21]. It has been shown that OS increases in GDM, albeit the relationship between GDM and OS has not been explained precisely [21]. Monocyte to high-density lipoprotein cholesterol (HDL-C) ratio (MHR) was considered to be a marker for oxidative stress and inflammation, since monocytes play a vital role in the secretion of circulating pro-inflammatory and oxidant cytokines and due to the fact that the anti-inflammatory and antioxidant properties of HDL-C have been found out [22,23,24]. To the best of our knowledge, hitherto MHR has not been studied in GDM.
Although there are studies in the literature related to the relationship of heavy metal exposure and levels of trace elements with GDM, their results are confounding. We aimed in this study to assess the levels of heavy metals (Cd, Pb, Sb, Hg, and As), which are also called endocrine disruptors, and trace elements (Cr-III, Cr-VI, Zn, Cu and Se) vs. MHR among pregnant women with GDM. Besides, we intended to reveal the correlation between these elements and the metabolic parameters of MHR and GDM.
Materials and Method
Our study is a case-control trial and was performed at the Obstetrics and Gynecology Department of Yozgat Bozok University Faculty of Medicine, upon obtaining the approval of the local Ethics Committee (2017-KAEK-189_2018.02.27-09). Written informed consent was obtained from the participants, and the ethical principles of the Declaration of Helsinki were complied with in all phases of the study.
Population of the Study
Our study consists of 2 groups, namely, pregnant women with GDM and healthy (control) pregnant women. Pregestational body mass index (BMI), blood pressure, smoking status, iron, and vitamin supplements were questioned during antenatal follow-ups of pregnant women. Moreover, pregnant women with type I-II diabetes mellitus, hypertension, chronic renal failure, hypo-hyperthyroidism, and heart diseases and those who smoke were excluded from the study.
Diagnosis of GDM
The diagnosis of GDM was made in two phases in our study. Both tests are conducted during fasting. Fifty-gram oral glucose tolerance test (OGTT) was performed to all pregnant women who were between the gestational weeks of 24th and 28th, during routine antenatal follow-ups in our clinic. As a result of this test, 100-g OGTT is applied to pregnant women whose 1st hour serum glucose value (SG) is ≥ 140 mg/dL. There is at least 3 days of interval between the two tests, and pregnant women who will undergo 100-g OGTT are asked to consume at least 150 mg of carbohydrate per day throughout this period. The pregnant women are diagnosed with GDM in the presence of 2 of the following criteria in the 100-g OGTT test [25]: fasting SG > 95 mg/dL, 1st hour SG > 180 mg/dL, 2nd hour SG > 155 mg/dL, and 3rd hour > 140 mg/dL.
Measurement of Biochemical Parameters
Venous blood samples were taken from all participants following 10 to 12 h of fasting, and the samples were centrifuged within 30 to 60 min and kept frozen at − 80 °C. Furthermore, fasting serum glucose (FSG), total cholesterol (TC), triglyceride (TG), HDL cholesterol (HDL-C), and 2nd hour SG were analyzed using assay kits (Abbott®, IL, USA) with an autoanalyzer (Aeroset®, Abbott®, IL, USA). Fasting serum insulin (FSI) was measured by the electrochemiluminescence immunoassay (ECLIA) using commercial kits (Abbott Laboratories) suitable for the autoanalyzer (Architect i2000; Abbott Laboratories, Abbott Park, IL, USA). HbA1c levels were measured using high-performance liquid chromatography (Variant II Turbo, Bio-Rad, Hercules, CA). The cutoff value of SG, HbA1c, TC, TG, and HDL-C are listed below: 109 mg/dL, 6.5%, 200 mg/dL, 200 mg/dL, and 45 mg/dL. The coefficient of variation of SG, FSI, HbA1c, TC, TG, and HDL-C are respectively as follows: 0.7%, 2.0%, 1.6%, 1.1%, 1.1%, and 1.8%. An automated hematology analyzer (BC-6800, Mindray, Shenzhen, China) was used for complete blood count results. Homeostasis model assessment of insulin resistance (HOMA-IR) was calculated with this formula: FSG (mg/dL) × FSI (mU/mL)/405. IR was considered HOMA-IR > 2.5.
Measurement of Heavy Metals and Trace Elements
Serum samples were collected for heavy metal and trace element levels measurements and stored at − 80 °C until being transferred. Subsequently, samples were transferred to Science and Technology Application and Research Center (STARC) Yozgat Bozok University for analysis in a cold chain setting. The samples were pretreated before analysis at this center. In order to perform this phase, 1 mL of each sample was taken and placed in Teflon tubes of microwave digestion unit, which are available in the laboratory (Milestone Ethos, USA). Subsequently, 5 mL of suprapur nitric acid (HNO 3) and 5 mL of ultrapure water were added to these samples, respectively. Samples were dissolved at 180 °C for 40 min (Table 1). Afterward, these analyzed samples were transferred into 50-mL polypropylene tubes and completed with 20 mL of ultrapure water, and the samples were stored at + 4 o C until analysis.
Analysis of heavy metals and trace elements was performed in inductively coupled plasma mass spectrometer (ICP, Thermo Scientific ICAPQc, USA). An 11-point calibration curve was generated for each element, and internationally validated certified standard samples (CRM, Seronorm, Whole Blood L-2) were used for the validation of these techniques. Plasma power was set to 1500 W and nebulizer pressure to 3.01 bar, and the spray chamber temperature was measured to be 2.9 oC. High purity argon gas was used as gas; plasma gas was set to 0.7 L/min, and nebulizer gas was set to 0.9 L/min. The measurement was performed in 0.01 s. Measurements of samples and standard samples were repeated a total of 3 times. The results were obtained as the mean of 3 readings, and it was determined that the relative standard deviation (RSD) did not surpass 5%. Measurement of the blank was performed using 3% nitric acid solution. The r 2 values of the calibration curves were determined to be at least 0.9988.
Statistical Analysis
The statistical analysis was conducted using the software of SPSS 20 (IBM Corp. released 2011. IBM SPSS Statistics for Windows, version 20.0, Armonk, NY: IBM Corp.). Data are presented as mean ± SD, median (interquartile range), and n (%). The normality of the data was tested with the Kolmogorov-Smirnov/Shapiro-Wilks tests. Student t test or the Mann-Whitney U test was performed to compare the continuous variables of the groups, depending on whether they are normally distributed or not. Univariate correlations were analyzed through Spearman’s rho test. Categorical data were compared with the chi-square test. The diagnostic performance of the parameters was evaluated by the analysis of the receiver operating characteristic (ROC) curve. The results were considered statistically significant at p < 0.05.
Results
Of the 112 pregnant women who participated in our study, 60 were in the GDM, and 52 were in the control group. As presented in Table 2, values of age, pregestational BMI, gravida, and parity were significantly different between the two groups. It was higher among the GDM group, compared to the control group (p < 0.05).
FSG, FSI, PPSG, HOMA-IR, HbA1c, triglyceride, HDL-C, and MHR were significantly different between groups. It was determined that FSG, FSI, PPSG, HOMA-IR, HbA1c, triglyceride, and MHR were higher among the GDM group, while HDL-C was higher among the control group (Table 2) (p < 0.05). The IR rate was significantly higher in the GDM group (Table 2) (p < 0.05).
The levels of trace elements and heavy metals are presented in Table 3. As shown in Table 3, the levels of trace elements including Cr-III, Zn, and Se were lower among GDM group, whereas the level of Cu level was higher among the GDM group (p < 0.05). Moreover, the difference between the two groups, regarding the Cr-VI level, was statistically insignificant (p > 0.05). Of the heavy metals, Cd, Pb, and Sb levels were higher in the GDM group (p < 0.05), whereas the difference between the two groups, regarding the levels of Hg and As, was statistically insignificant (p > 0.05).
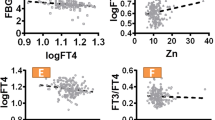
The correlations between metabolic parameters and trace elements and heavy metals are presented in Table 4. There is a negative correlation between FSG and Cr-III (r = − 0.274, p = 0.003), whereas, there is a positive correlation between Cu (r = 0.294, p = 0.002), Pb (r = 0.205, p = 0.030), and Sb (r = 0.198, p = 0.037) (Table 4, Fig. 1). A negative correlation was determined between PPSG and Cr-III and Zn (r = −0.294, p = 0.002) (r = − 0.214, p = 0.023), respectively. Of the heavy metals, a positive correlation was found out to be between Pb, Sb, and PPSG (r = 0.278, p = 0.003) (r = 0.265, p = 0.005), respectively. A negative correlation was detected between HbA1c and Cr-III and Zn, whereas a positive correlation was detected between HbA1c and Cu, (r = − 0.373, p < 0.001) (r = − 0.259, p = 0.006) (r = 0.255, p = 0.007), respectively. It has been determined that HOMA-IR was negatively correlated with Cr-III, whereas it was positively correlated with Cu and Sb (r = − 0.255, p = 0.007) (r = 0.242, p = 0.010) (r = 0.224, p = 0.017), respectively.
Furthermore, it was found out that MHR, which is an oxidative stress marker, was negatively correlated with Cr-III and Se (r = − 0.419, p < 0.001) (r = − 0.337, p < 0.001), respectively, whereas it was positively correlated with Cu, Cd, Pb, and Sb (r = 0.229, p = 0.015) (r = 0.562, p < 0.001) (r = 0.396, p < 0.001) (r = 0.236, p = 0.012), respectively (Table 4). In addition to that, a strong relationship was determined between IR and Cr-III, Cu, Sb, and MHR (Fig. 2).
It was found out as a result of the ROC curve analysis of Cr-III and MHR that the cutoff value for Cr-III was 0.213 μg/L with a sensitivity of 82.7% and a specificity of 83.3% (AUC: 0.917) (Fig. 3), while the cutoff value for MHR was 8.97 with a sensitivity of 83.3% sensitivity and a specificity of 69.2% (AUC: 0.840) (Fig. 3).
Discussion
The primary objective of our study is to investigate the correlation of GDM with heavy metal exposure during pregnancy and the levels of trace elements. To the best of our knowledge, our research is the first study in the literature to concurrently assess the heavy metal exposure and trace element levels (especially both forms of chromium) in GDM. It was found out in this study that the serum levels of trace elements such as Cr-III, Se, and Zn were lower among the GDM group, whereas the serum levels of Cu, which is a trace element, and Cd, Pb, and Sb were higher among the GDM group. Moreover, a correlation was determined between trace elements and heavy metals and glucose metabolism parameters.
Trace elements, which are found in the body as a component of metalloproteins, play an active role in many structural and enzymatic processes. Furthermore, they are a significant part of the antioxidant defense system. The requirement for trace elements increases typically during pregnancy. Cr, which is known for its favorable effects on glucose metabolism, is better absorbed as Cr-III. Cr, which acts through chromodulin that enhances insulin signal [26], also increases insulin receptor count and β cell sensitivity [27]. Król et al. [28] found out that serum glucose level enhanced and Cu to Zn ratio decreased in rats thanks to the Cr-III supplementation. Cr-III supplementation has a favorable impact on individuals with impaired lipid and glucose metabolism rather than healthy individuals [29]. Zhou et al. [30] determined that the serum level of Cr was decreased in T2DM and its complications, such as diabetic nephropathy. The increased levels of Cu are toxic to the cell, albeit it is a trace element. Se is included in the structure of glutathione peroxidase, while Cu is included in the structure of superoxide dismutase and cytochrome-c oxidase [31]. Cu might lead to oxidative stress directly through the Fenton-like reaction or by reducing the glutathione level [32, 33]. It is well-known that Zn plays a key role in cell division and differentiation. Moreover, it impacts the functions of various hormones, including insulin. The requirement for trace elements increases typically during pregnancy. The potential increase in Cu levels paradoxically decreases Zn absorption [34]. Wilson et al. [35] investigated the correlation between pregnancy complications and serum trace element levels during early pregnancy. They found out as a result of this study that the increased levels of Cu and Zn were associated with pregnancy complications. Another study, which associated Se level with birth weight, also revealed a decreased Se level in preterm births [36]. The levels of Cr-III, Zn, and Se were determined to be lower and Cu levels to be higher in our study, upon comparing women with GDM and healthy pregnant women (p < 0.05). On the other hand, the difference between the two groups, regarding Cr-VI, was statistically insignificant. It has been underscored in a meta-analysis, which assessed the correlation between Se and GDM, that the decreased levels of Se are associated with GDM [37]. Another study found out that the increased level of Cu heightens the risk of glucose dysregulation [38]. Mishu et al. [39] identified similar results to ours in patients with GDM.
Cd exposure occurs predominantly through food and smoking cigarettes [40]. It has been well-known for nearly 50 years that Cd has diabetogenic effects [41]. The association of Cd with T2DM and impaired fasting glucose was revealed for the first time in the study of NHANES III [42]. Ting et al. [43] compared 122 newly diagnosed T2DM patients with 429 healthy controls and determined that certain metal levels in plasma, including Cd, were significantly higher among the T2DM group. It has been revealed that Cd induces hyperglycemia and is negatively correlated with the level of insulin [44]. Liu et al. [8] determined in their study, in which they investigated the relationship between Cd level and GDM, that maternal urinary Cd level could be associated with GDM. Soomro et al. [45] found out analogous results in another study. In contrast, Oguri et al. [46] put forward that there was no correlation between Cd exposure and GDM. Cd was determined to be significantly higher among the GDM group in this study. Besides, Cd has the strongest correlation with MHR along with Sb.
Sb exposure, as with the case of other heavy metal exposures, is associated with industrial development. The majority of the population is exposed to chronic low dose Sb. Iyengar et al. [47] has demonstrated that American men aged between 25 to 30 are exposed to a mean of 5 μg/day Sb. In another study, Sb was detected to be above the range of baseline values for American adults in blood samples of neonates in China [48]. The results of studies, which investigate the correlation between T2DM and Sb, are contradictory. Menke et al. [49] found out a correlation between T2DM and Sb in their study, whereas in another study, a negative correlation was determined between T2DM and plasma Sb level [50]. Zhang et al. [51] assessed urinary Sb levels in a prospective cohort study in which 2093 pregnant women were included. Ultimately, they highlighted that an increased Sb exposure could be associated with GDM. It has been revealed in another study that the risk of GDM increases even with lower levels of Sb exposure, and it could lead to deterioration of glucose metabolism in pregnant women. Our study also supports these results. In addition to these similar findings, deterioration was also detected in metabolic parameters such as FSG, PPSG, and HOMA-IR, proportional with the Sb level.
The effects of Pb exposure occur in a dose dependent manner. Pb inhibits glutathione reductase and delta-aminolevulinic acid dehydrogenase, which are members of the antioxidant defense system [52]. High dose Pb exposure has an increased risk for GDM, when high dose exposure and low dose exposure are compared [45]. Excluding occupational exposure, the community is subjected to Pb exposure through environmental pollution and smoking [53, 54]. Pb levels in maternal blood samples were compared with obstetric results. Jelliffe-Pawlowski et al. [55] found out that a Pb level of 10 mg/dL in maternal blood increased the risk of preterm delivery and low birth weight. Whereas some studies have found out a correlation between maternal Pb level and GDM [45, 56], other studies have determined that the maternal Pb level is not correlated with the GDM [46, 57]. Maternal Pb levels were significantly higher among the GDM group in our study. Moreover, it has been found out that it correlates positively with FSG and PPSG.
Hg and As are among the toxic heavy metals. It is well-documented that the risk of diabetes increases in the environments, which are remarkably contaminated with As [58]. MIREC study has indicated that the As exposure could be a risk factor for GDM [57]. The same research did not determine a correlation between Hg and GDM [57]. In another study, no correlation was identified between inorganic As and GDM among 230 pregnant women whose urine samples were taken [59]. Ettinger et al. [60] underscored in their study, in which they assessed the As level in maternal hair samples, that As exposure might be associated with the increased risk of GDM. A review, which has examined the metabolic effects of Hg exposure, suggested that the risk of diabetes mellitus and metabolic syndrome may rise with the increased Hg exposure, yet there is not any compelling evidence to explain this cause-effect relationship [61]. No significant difference was determined between the two groups, regarding Hg and As levels, in our study.
OS might occur secondary to hyperglycemia, as it might also lead to GDM. Heavy metals could also cause GDM through increasing OS. For instance, it is well-documented that Cd damages β cells via OS [10]. MHR is a method that demonstrates oxidative stress and chronic inflammation [62,63,64]. To the best of our knowledge, MHR has not been previously assessed in patients with GDM. We determined that MHR was significantly higher among the GDM group. In addition to this, MHR is negatively correlated with Se and Cr-III and positively correlated with Cu, Cd, Pb, and Sb. These findings suggest that OS might play a role in the pathophysiology of GDM, regardless of its cause.
Heavy metal exposure and trace elements may play a key role in GDM, albeit the mechanism is not fully understood. Oxidative stress, which is caused by heavy metal exposure, inflammation, inhibition of peroxisome proliferator-activated receptor-gamma, and modification of diabetes-related genes are considered to be the mechanisms that may be effective in the occurrence of GDM [45, 65]. Trace elements can affect this process positively in a direct way through insulin secretion and receptors, and they can also be effective by boosting tissues in their fight against the oxidative stress. Moreover, even though Cu is a trace element, increased levels of it could lead to the occurrence of GDM through increasing oxidative stress [26, 27, 31,32,33].
Our study has some limitations: Firstly, it has a small sample size. Secondly, demonstrating OS with other OS markers apart from MHR could increase the strength of the study. Another limitation was that the measurement of heavy metal and trace element levels was performed with the material, which was taken from a single sample. Hair, urine, umbilical cord blood, and meconium specimens could be used as well. Furthermore, the robustness of the study was tried to be enhanced by excluding the circumstances that could create a situation of risk for GDM, change the levels of heavy metals, and lead to chronic inflammation, when generating the trial and control groups. In addition to that, various heavy metals and trace elements were assessed in the same participants.
The results of our study demonstrated that environmental factors could be effective in the etiology of GDM. Toxic heavy metals, through inducing Cu, OS, and chronic inflammation, and other trace elements, either directly by impacting insulin secretion or through weakening the body’s antioxidant defense system, could play a role in the occurrence of GDM. From this point of view, our results will shed light on further studies; nevertheless, studies with larger sample groups are required to validate our results.
Data Availability
The data set has been uploaded to the system.
References
Care D (2018) Medical care in diabetes (2018). Diabetes Care 41(1):S105–S118
Zhu WW, Yang HX, Wei YM, Yan J, Wang ZL, Li XL, Wu HR, Li N, Zhang MH, Liu XH, Zhang H, Wang YH, Niu JM, Gan YJ, Zhong LR, Wang YF, Kapur A (2013) Evaluation of the value of fasting plasma glucose in the first prenatal visit to diagnose gestational diabetes mellitus in China. Diabetes Care 36(3):586–590. https://doi.org/10.2337/dc12-1157
Edition I International Diabetes Federation (2017) IDF Diabetes Atlas, 8th edn. International Diabetes Federation, Brussels
Akgol S, Obut M, Baglı İ, Kahveci B, Budak MS (2019) An evaluation of the effect of a one or two-step gestational diabetes mellitus screening program on obstetric and neonatal outcomes in pregnancies. Gynecology Obstetrics & Reproductive Medicine 25(2):62–66
Akgol S, Budak MS (2019) Obstetric and neonatal outcomes of pregnancies with mild gestational hyperglycemia diagnosed at gestational diabetes mellitus screening. Gynecology Obstetrics & Reproductive Medicine 25(3):138–141
McIntyre HD, Catalano P, Zhang C, Desoye G, Mathiesen ER, Damm P (2019) Gestational diabetes mellitus. Nat Rev Dis Primers 5(1):47. https://doi.org/10.1038/s41572-019-0098-8
Rehman K, Fatima F, Waheed I, Akash MSH (2018) Prevalence of exposure of heavy metals and their impact on health consequences. J Cell Biochem 119(1):157–184. https://doi.org/10.1002/jcb.26234
Liu W, Zhang B, Huang Z, Pan X, Chen X, Hu C, Liu H, Jiang Y, Sun X, Peng Y, Xia W, Xu S, Li Y (2018) Cadmium body burden and gestational diabetes mellitus: a prospective study. Environ Health Perspect 126(2):027006. https://doi.org/10.1289/ehp2716
Farzan SF, Gossai A, Chen Y, Chasan-Taber L, Baker E, Karagas M (2016) Maternal arsenic exposure and gestational diabetes and glucose intolerance in the New Hampshire birth cohort study. Environ Health 15(1):106. https://doi.org/10.1186/s12940-016-0194-0
Chang KC, Hsu CC, Liu SH, Su CC, Yen CC, Lee MJ, Chen KL, Ho TJ, Hung DZ, Wu CC, Lu TH, Su YC, Chen YW, Huang CF (2013) Cadmium induces apoptosis in pancreatic β-cells through a mitochondria-dependent pathway: the role of oxidative stress-mediated c-Jun N-terminal kinase activation. PLoS One 8(2):e54374. https://doi.org/10.1371/journal.pone.0054374
Tyrrell JB, Hafida S, Stemmer P, Adhami A, Leff T (2017) Lead (Pb) exposure promotes diabetes in obese rodents. J Trace Elem Med Biol 39:221–226. https://doi.org/10.1016/j.jtemb.2016.10.007
Maull EA, Ahsan H, Edwards J, Longnecker MP, Navas-Acien A, Pi J, Silbergeld EK, Styblo M, Tseng CH, Thayer KA, Loomis D (2012) Evaluation of the association between arsenic and diabetes: a National Toxicology Program workshop review. Environ Health Perspect 120(12):1658–1670. https://doi.org/10.1289/ehp.1104579
Zhang Q, Li X, Liu X, Dong M, Xiao J, Wang J, Zhou M, Wang Y, Ning D, Ma W, Zhu W, Liu T, Zhang B (2020) Association between maternal antimony exposure and risk of gestational diabetes mellitus: a birth cohort study. Chemosphere 246:125732. https://doi.org/10.1016/j.chemosphere.2019.125732
Tinkov AA, Ajsuvakova OP, Skalnaya MG, Popova EV, Sinitskii AI, Nemereshina ON, Gatiatulina ER, Nikonorov AA, Skalny AV (2015) Mercury and metabolic syndrome: a review of experimental and clinical observations. Biometals 28(2):231–254. https://doi.org/10.1007/s10534-015-9823-2
Dubey P, Thakur V, Chattopadhyay M (2020) Role of minerals and trace elements in diabetes and insulin resistance. Nutrients 12(6). https://doi.org/10.3390/nu12061864
Stapleton SR (2000) Selenium: an insulin-mimetic. Cell Mol Life Sci 57(13–14):1874–1879. https://doi.org/10.1007/pl00000669
Anderson RA (1997) Nutritional factors influencing the glucose/insulin system: chromium. J Am Coll Nutr 16(5):404–410. https://doi.org/10.1080/07315724.1997.10718705
Rajendran K, Manikandan S, Nair LD, Karuthodiyil R, Vijayarajan N, Gnanasekar R, Kapil VV, Mohamed AS (2015) Serum chromium levels in type 2 diabetic patients and its association with Glycaemic control. J Clin Diagn Res 9(11):Oc05–Oc08. https://doi.org/10.7860/jcdr/2015/16062.6753
Zheng Y, Zhang C, Weisskopf MG, Williams PL, Claus Henn B, Parsons PJ, Palmer CD, Buck Louis GM, James-Todd T (2020) Evaluating associations between early pregnancy trace elements mixture and 2nd trimester gestational glucose levels: a comparison of three statistical approaches. Int J Hyg Environ Health 224:113446. https://doi.org/10.1016/j.ijheh.2019.113446
Elliot MG (2016) Oxidative stress and the evolutionary origins of preeclampsia. J Reprod Immunol 114:75–80. https://doi.org/10.1016/j.jri.2016.02.003
Rueangdetnarong H, Sekararithi R, Jaiwongkam T, Kumfu S, Chattipakorn N, Tongsong T, Jatavan P (2018) Comparisons of the oxidative stress biomarkers levels in gestational diabetes mellitus (GDM) and non-GDM among Thai population: cohort study. Endocr Connect 7(5):681–687. https://doi.org/10.1530/ec-18-0093
Ancuta P, Wang J, Gabuzda D (2006) CD16+ monocytes produce IL-6, CCL2, and matrix metalloproteinase-9 upon interaction with CX3CL1-expressing endothelial cells. J Leukoc Biol 80(5):1156–1164. https://doi.org/10.1189/jlb.0206125
Parthasarathy S, Barnett J, Fong LG (1990) High-density lipoprotein inhibits the oxidative modification of low-density lipoprotein. Biochim Biophys Acta 1044(2):275–283. https://doi.org/10.1016/0005-2760(90)90314-n
Canpolat U, Çetin EH, Cetin S, Aydin S, Akboga MK, Yayla C, Turak O, Aras D, Aydogdu S (2016) Association of monocyte-to-HDL cholesterol ratio with slow coronary flow is linked to systemic inflammation. Clin Appl Thromb Hemost 22(5):476–482. https://doi.org/10.1177/1076029615594002
Coustan DR, Carpenter MW (1998) The diagnosis of gestational diabetes. Diabetes Care 21(Suppl 2):B5–B8
Abebe W, Liu JY, Wimborne H, Mozaffari MS (2010) Effects of chromium picolinate on vascular reactivity and cardiac ischemia-reperfusion injury in spontaneously hypertensive rats. Pharmacol Rep 62(4):674–682
Wang ZQ, Zhang XH, Russell JC, Hulver M, Cefalu WT (2006) Chromium picolinate enhances skeletal muscle cellular insulin signaling in vivo in obese, insulin-resistant JCR:LA-cp rats. J Nutr 136(2):415–420. https://doi.org/10.1093/jn/136.2.415
Król E, Krejpcio Z, Okulicz M, Śmigielska H (2020) Chromium(III) glycinate complex supplementation improves the blood glucose level and attenuates the tissular copper to zinc ratio in rats with mild hyperglycaemia. Biol Trace Elem Res 193(1):185–194. https://doi.org/10.1007/s12011-019-01686-7
Lewicki S, Zdanowski R, Krzyżowska M, Lewicka A, Dębski B, Niemcewicz M, Goniewicz M (2014) The role of chromium III in the organism and its possible use in diabetes and obesity treatment. Ann Agric Environ Med 21(2):331–335. https://doi.org/10.5604/1232-1966.1108599
Zhou Q, Guo W, Jia Y, Xu J (2019) Comparison of chromium and Iron distribution in serum and urine among healthy people and Prediabetes and diabetes patients. Biomed Res Int 2019:3801639–3801638. https://doi.org/10.1155/2019/3801639
Kilinc M, Coskun A, Bilge F, Imrek SS, Atli Y (2010) Serum reference levels of selenium, zinc and copper in healthy pregnant women at a prenatal screening program in southeastern Mediterranean region of Turkey. J Trace Elem Med Biol 24(3):152–156. https://doi.org/10.1016/j.jtemb.2010.01.004
Prousek J (2007) Fenton chemistry in biology and medicine. Pure Appl Chem 79(12):2325–2338
Speisky H, Gómez M, Burgos-Bravo F, López-Alarcón C, Jullian C, Olea-Azar C, Aliaga ME (2009) Generation of superoxide radicals by copper-glutathione complexes: redox-consequences associated with their interaction with reduced glutathione. Bioorg Med Chem 17(5):1803–1810. https://doi.org/10.1016/j.bmc.2009.01.069
Gibson RS (2007) The role of diet- and host-related factors in nutrient bioavailability and thus in nutrient-based dietary requirement estimates. Food Nutr Bull 28(1 Suppl International):S77–S100. https://doi.org/10.1177/15648265070281s108
Wilson RL, Bianco-Miotto T, Leemaqz SY, Grzeskowiak LE, Dekker GA, Roberts CT (2018) Early pregnancy maternal trace mineral status and the association with adverse pregnancy outcome in a cohort of Australian women. J Trace Elem Med Biol 46:103–109. https://doi.org/10.1016/j.jtemb.2017.11.016
Tsuzuki S, Morimoto N, Hosokawa S, Matsushita T (2013) Associations of maternal and neonatal serum trace element concentrations with neonatal birth weight. PLoS One 8(9):e75627. https://doi.org/10.1371/journal.pone.0075627
Askari G, Iraj B, Salehi-Abargouei A, Fallah AA, Jafari T (2015) The association between serum selenium and gestational diabetes mellitus: a systematic review and meta-analysis. J Trace Elem Med Biol 29:195–201. https://doi.org/10.1016/j.jtemb.2014.09.006
Zheng Y, Zhang C, Weisskopf M, Williams PL, Parsons PJ, Palmer CD, Buck Louis GM, James-Todd T (2019) A prospective study of early pregnancy essential metal(loid)s and glucose levels late in the second trimester. J Clin Endocrinol Metab 104(10):4295–4303. https://doi.org/10.1210/jc.2019-00109
Mishu FA, Boral N, Ferdous N, Nahar S, Sultana GS, Yesmin MS, Khan NZ (2019) Estimation of serum zinc, copper and magnesium levels in Bangladeshi women with gestational diabetes mellitus attending in a tertiary care hospital. Mymensingh Med J 28(1):157–162
Faroon O, Ashizawa A, Wright S, Tucker P, Jenkins K, Ingerman L, Rudisill C (2012) Toxicological profile for cadmium. Atlanta (GA): Agency for Toxic Substances and Disease Registry (US); 2012 Sep.
Ithakissios DS, Ghafghazi T, Mennear JH, Kessler WV (1975) Effect of multiple doses of cadmium on glucose metabolism and insulin secretion in the rat. Toxicol Appl Pharmacol 31(1):143–149
Schwartz GG, Il’yasova D, Ivanova A (2003) Urinary cadmium, impaired fasting glucose, and diabetes in the NHANES III. Diabetes Care 26(2):468–470
Li XT, Yu PF, Gao Y, Guo WH, Wang J, Liu X, Gu AH, Ji GX, Dong Q, Wang BS, Cao Y, Zhu BL, Xiao H (2017) Association between plasma metal levels and diabetes risk: a case-control study in China. Biomed Environ Sci 30(7):482–491
Edwards JR, Prozialeck WC (2009) Cadmium, diabetes and chronic kidney disease. Toxicol Appl Pharmacol 238(3):289–293. https://doi.org/10.1016/j.taap.2009.03.007
Soomro MH, Baiz N, Huel G, Yazbeck C, Botton J, Heude B, Bornehag CG, Annesi-Maesano I (2019) Exposure to heavy metals during pregnancy related to gestational diabetes mellitus in diabetes-free mothers. Sci Total Environ 656:870–876. https://doi.org/10.1016/j.scitotenv.2018.11.422
Oguri T, Ebara T, Nakayama SF, Sugiura-Ogasawara M, Kamijima M (2019) Association between maternal blood cadmium and lead concentrations and gestational diabetes mellitus in the Japan environment and children's study. Int Arch Occup Environ Health 92(2):209–217. https://doi.org/10.1007/s00420-018-1367-7
Iyengar G, Tanner J, Wolf W, Zeisler R (1987) Preparation of a mixed human diet material for the determination of nutrient elements, selected toxic elements and organic nutrients: a preliminary report. Sci Total Environ 61:235–252
Filella M, Belzile N, Chen Y-W (2013) Human exposure to antimony. IV Contents in human blood. Critical Reviews in Environmental Science and Technology 43(19):2071–2105
Menke A, Guallar E, Cowie CC (2016) Metals in urine and diabetes in U.S. adults. Diabetes 65(1):164–171. https://doi.org/10.2337/db15-0316
Yuan Y, Xiao Y, Yu Y, Liu Y, Feng W, Qiu G, Wang H, Liu B, Wang J, Zhou L, Liu K, Xu X, Yang H, Li X, Qi L, Zhang X, He M, Hu FB, Pan A, Wu T (2018) Associations of multiple plasma metals with incident type 2 diabetes in Chinese adults: the Dongfeng-Tongji cohort. Environ Pollut 237:917–925. https://doi.org/10.1016/j.envpol.2018.01.046
Zhang G, Wang X, Zhang X, Li Q, Xu S, Huang L, Zhang Y, Lin L, Gao D, Wu M, Sun G, Song Y, Zhong C, Yang X, Hao L, Yang H, Yang L, Yang N (2019) Antimony in urine during early pregnancy correlates with increased risk of gestational diabetes mellitus: a prospective cohort study. Environ Int 123:164–170. https://doi.org/10.1016/j.envint.2018.11.072
Hoffman DJ, Heinz GH, Sileo L, Audet DJ, Campbell JK, Obrecht HH III (2000) Developmental toxicity of lead-contaminated sediment in Canada geese (Branta canadensis). J Toxicol Environ Health A 59(4):235–252
Pinto E, Cruz M, Ramos P, Santos A, Almeida A (2017) Metals transfer from tobacco to cigarette smoke: evidences in smokers' lung tissue. J Hazard Mater 325:31–35. https://doi.org/10.1016/j.jhazmat.2016.11.069
Augustsson AL, Uddh-Söderberg TE, Hogmalm KJ, Filipsson ME (2015) Metal uptake by homegrown vegetables - the relative importance in human health risk assessments at contaminated sites. Environ Res 138:181–190. https://doi.org/10.1016/j.envres.2015.01.020
Jelliffe-Pawlowski LL, Miles SQ, Courtney JG, Materna B, Charlton V (2006) Effect of magnitude and timing of maternal pregnancy blood lead (Pb) levels on birth outcomes. J Perinatol 26(3):154–162. https://doi.org/10.1038/sj.jp.7211453
Wang Y, Zhang P, Chen X, Wu W, Feng Y, Yang H, Li M, Xie B, Guo P, Warren JL, Shi X, Wang S, Zhang Y (2019) Multiple metal concentrations and gestational diabetes mellitus in Taiyuan, China. Chemosphere 237:124412. https://doi.org/10.1016/j.chemosphere.2019.124412
Shapiro GD, Dodds L, Arbuckle TE, Ashley-Martin J, Fraser W, Fisher M, Taback S, Keely E, Bouchard MF, Monnier P, Dallaire R, Morisset A, Ettinger AS (2015) Exposure to phthalates, bisphenol a and metals in pregnancy and the association with impaired glucose tolerance and gestational diabetes mellitus: the MIREC study. Environ Int 83:63–71. https://doi.org/10.1016/j.envint.2015.05.016
Tseng CH, Tseng CP, Chiou HY, Hsueh YM, Chong CK, Chen CJ (2002) Epidemiologic evidence of diabetogenic effect of arsenic. Toxicol Lett 133(1):69–76. https://doi.org/10.1016/s0378-4274(02)00085-1
Muñoz MP, Valdés M, Muñoz-Quezada MT, Lucero B, Rubilar P, Pino P, Iglesias V (2018) Urinary inorganic arsenic concentration and gestational diabetes mellitus in pregnant women from Arica, Chile. Int J Environ Res Public Health 15(7). https://doi.org/10.3390/ijerph15071418
Ettinger AS, Zota AR, Amarasiriwardena CJ, Hopkins MR, Schwartz J, Hu H, Wright RO (2009) Maternal arsenic exposure and impaired glucose tolerance during pregnancy. Environ Health Perspect 117(7):1059–1064. https://doi.org/10.1289/ehp0800533
Roy C, Tremblay PY, Ayotte P (2017) Is mercury exposure causing diabetes, metabolic syndrome and insulin resistance? A systematic review of the literature. Environ Res 156:747–760. https://doi.org/10.1016/j.envres.2017.04.038
Yılmaz M, Kayançiçek H (2018) A new inflammatory marker: elevated monocyte to HDL cholesterol ratio associated with smoking. J Clin Med 7(4). https://doi.org/10.3390/jcm7040076
Chen JW, Li C, Liu ZH, Shen Y, Ding FH, Shu XY, Zhang RY, Shen WF, Lu L, Wang XQ (2019) The role of monocyte to high-density lipoprotein cholesterol ratio in prediction of carotid intima-media thickness in patients with type 2 diabetes. Diabetes. Front Endocrinol (Lausanne) 10:191. https://doi.org/10.3389/fendo.2019.00191
Ganjali S, Gotto AM Jr, Ruscica M, Atkin SL, Butler AE, Banach M, Sahebkar A (2018) Monocyte-to-HDL-cholesterol ratio as a prognostic marker in cardiovascular diseases. J Cell Physiol 233(12):9237–9246. https://doi.org/10.1002/jcp.27028
Senturk S, Kagitci M, Guven ESG (2018) Thrombocyte alterations in pregnant women with gestational diabetes mellitus. Gynecology Obstetrics & Reproductive Medicine 24(1):12–16. https://doi.org/10.21613/GORM.2017.711
Funding
This study was supported by Yozgat Bozok University Project Coordination Application and Research Center (6602a-TF/18-201).
Author information
Authors and Affiliations
Contributions
Concept: M.K., E.S.Y., V.A.T.
Design: M.D.C., T.O.
Data collection or processing: T.O., E.B., D.A.K., V.A.T.
Analysis or interpretation: T.O., D.A.K., E.S.Y.
Writing: T.O., M.D.C.
Corresponding author
Ethics declarations
The study was approved by the Yozgat Bozok University Clinical Research Ethics Committee (2017-KAEK-189_2018.02.27-09), and an informed consent was obtained from all participants.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Onat, T., Demir Caltekin, M., Turksoy, V.A. et al. The Relationship Between Heavy Metal Exposure, Trace Element Level, and Monocyte to HDL Cholesterol Ratio with Gestational Diabetes Mellitus. Biol Trace Elem Res 199, 1306–1315 (2021). https://doi.org/10.1007/s12011-020-02499-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-020-02499-9