Abstract
The contamination of seafood like narrow-barred Spanish mackerel (Scomberomorus commerson) fillets by potentially toxic elements (PTEs) has converted to worldwide health concerns. In this regard, the related citations regarding the concentration of PTEs in fillets of narrow-barred Spanish mackerel were collected through some of the international databases such as Scopus, Cochrane, PubMed, and Scientific Information Database (SID) up to 10 March 2020. The concentration of PTEs in fillets of narrow-barred Spanish mackerel fish was meta-analyzed and the health risk (non-carcinogenic risk) was estimated by the total target hazard quotient (TTHQ). The meta-analysis of data indicated that the rank order of PTEs in fillet of narrow-barred Spanish mackerel was Fe (10,853.29 μg/kg-ww) > Zn (4007.00 μg/kg-ww) > Cu (1005.66 μg/kg-ww) > total Cr (544.14 μg/kg-ww) > Mn (515.93 μg/kg-ww) > Ni (409.90 μg/kg-ww) > Pb (180.99 μg/kg-ww) > As (93.11 μg/kg-ww) > methyl Hg (66.60 μg/kg-ww) > Cd (66.03 μg/kg-ww). The rank order of health risk assessment based on the country by the aid of TTHQ for adult consumers was Malaysia (0.22251) > Philippines (0.21912) > Egypt (0.08684) > Taiwan (0.07430) > Bahrain (0.04893) > Iran (0.03528) > China (0.00620) > Pakistan (0.00316) > Yemen (0.00157) > India (0.00073). In addition, the rank order of health risk assessment based on the country by the aid of TTHQ for child consumers was Malaysia (1.03838) > Philippines (1.02257) > Egypt (0.40523) > Taiwan (0.34674) > Bahrain (0.22832) > Iran (0.16466) > China (0.02892) > Pakistan (0.01474) > Yemen (0.00731) > India (0.00340). Therefore, the children in Malaysia and the Philippines were at considerable non-carcinogenic risk. Hence, approaching the recommended control plans in order to decrease the non-carcinogenic risk associated with the ingestion of PTEs via the consumption of narrow-barred Spanish mackerel fish fillets is crucial.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The health of humans is associated with feeding habits [1]. A healthy diet should not only contain vitamins and essential trace elements but also should be free of chemical contaminants (heavy metals and persistent organic pollutants) and pathogenic microorganisms (bacteria and virus) [2,3,4]. While some of the food products, such as fish, attracted considerable attention due to their rich content of protein, their contamination by pollutants is a matter of concern. Fish is in the marine feeding chain’s highest loops and is one of the most important food sources for humans [5]. Fish fillet has high protein with low-calorie content, constitutes 6% and 17% of protein requirement and animal protein, respectively, as compared to other food sources [5]. At the same time, the fish fillet consumption (twice a week) is recommended due to its high omega-3 fatty acids content like eicosapentaenoic acid [6], which can significantly reduce the risk of cardiovascular disease [7, 8] besides the proposed role in preventing thrombosis and arrhythmia and following decreasing triglyceride levels [9]. Despite the several benefits, fish fillet consumption plays a considerable role in transmitting environmental contaminants to humans [10, 11]. Natural sources like crust corrosion can contaminate aquatic environments, or mainly anthropogenic sources consist of agricultural, industrial, and municipal wastewater and solid waste disposal, industrial processes, smelting procedures, mining, and application of herbicides [12,13,14].
Potentially harmful elements (PTEs) are elements are categorized into two groups of essential elements like zinc(Zn), iron (Fe), copper (Cu), calcium (Ca), and selenium (Se) and heavy metals like as mercury (Hg), cadmium (Cd), arsenic (As), lead(Pb), nickel(Ni), and copper (Cu) [15, 16]. However, the essential elements in low concentrations are posing some benefits for humans’ health; in high concentrations, they can be harmful to health; however, non-essential elements (heavy metals) can endanger health, while in low concentrations [15]. The non-essential elements are non-biodegradable, demonstrating the stable properties among the food chain [17]. The exposure to PTEs leads to adverse health effects on humans’ nervous system, kidney, liver, cardiovascular, and bone [18,19,20]. Besides, they pose teratogenesis, mutagenesis, and carcinogenesis properties [21,22,23].
Narrow-barred Spanish mackerel (Scomberomorus commerson) is a large predatory fish that live on the coast of southeast Asia, the east coast of Africa, the Persian Gulf, northern coastal Indian ocean, and southwest Pacific ocean [24]. Narrow-barred Spanish mackerel is a popular and high-consumption marine food resource in the world due to its delicious meat [25,26,27]. Moreover, other similar seafood consumptions, such as narrow-barred Spanish mackerel fish, have increased in recent years [24,25,26,27].
However, many investigations have been conducted to measure the PTEs in narrow-barred Spanish mackerel worldwide [28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44]; no systematic review study was performed. Hence, the current study’s aim was a meta-analysis of the concentration of PTEs, including inorganic As, Methyl Hg, Total Cr, Cd, Pb, Cu, Zn, Fe, Mn, and Ni in narrow-barred Spanish mackerel fillet on a global scale. Moreover, the related health risk assessment for exposed population-based countries was estimated.
Material and Method
Search Strategy
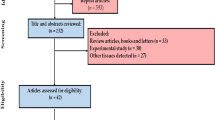
Cochrane protocol was used to conduct systematic review [45], and the following selection process of studies was done according to PRISMA (Fig. 1) [46]. Scopus, Cochrane, and PubMed, scientific information database (SID) databases, were used to search articles on the concentration of PTEs in the fillet of narrow-barred Spanish mackerel up to 10 March 2020. Keywords consist of “heavy metals” OR “trace metals” OR “toxic metal” OR metals OR metal (oid) s OR element AND “marine food” OR fish OR seafood’s OR “Narrow-barred Spanish mackerel” OR “Scomberomorus commerson were used. The reference articles were screened to retrieve more citations.
Selection Criteria and Data Extraction
The inclusion criteria consisted of analysis concentration of PTEs in the fillet of narrow-barred Spanish mackerel; descriptive study; present of average or range concentration of PTEs; present of sample size; and access English language full text. Books, review articles, letters, and conferences were excluded based on protocol. Moreover, experimental studies, like studies on the bioaccumulation of PTEs indifferent fish tissues of narrow-barred Spanish mackerel, were excluded. Country, year of study, location of sampling, sample size, the average concentration PTEs, standard deviation (SD) of PTEs, and range of concentration of PTEs and method of detection were extracted.
Meta-analysis of Data
The concentration of PTEs in fillets narrow-barred Spanish mackerel was meta-analyzed using the standard error (SE) equation (Table 1). The I2 index and chi-square were used to analysis of heterogeneity. If I2 > 50%, heterogeneity is considerable [53,54,55]; hence, a random effect model (REM) was used for meta-analysis. A meta-analysis of data was conducted using Stata, version 14 (Stata Corporation, College Station, TX).
Health Risk Assessment
Health risk assessment can provide more interpretive results regarding human exposure to contaminated food products [56,57,58]. All equations used in health risk assessment are presented in Table 1 and per capita consumption of fish in Table 2, followed by the included studies’ main characteristics presented in Table 1S.
Results and Discussion
The Concentration of PTEs in the Fillet of Narrow-Barred Spanish Mackerel in Different Countries
The rank order of PTEs in fillet of narrow-barred Spanish mackerel was Fe (10,853.29 μg/kg-ww) > Zn (4007.00 μg/kg-ww) > Cu (1005.66 μg/kg-ww) > Cr (544.14 μg/kg-ww) > Mn (515.93 μg/kg-ww) > Ni (409.90 μg/kg-ww) > Pb (180.99 μg/kg-ww) > As (93.11 μg/kg-ww) > Hg (66.60 μg/kg-ww) > Cd (66.03 μg/kg-ww). The concentration of toxic metals (As, Hg, and Cd) is much lower than the essential elements (Fe, Zn, and Cu), which justifies the latter’s higher affinity to enter the fish body [59, 60]. In the present study, Fe showed the highest concentration among investigated PTEs because this element is the most abundant metal present on the earth’s crust, which is essential for most biological functions in the fish and other animals’ bodies [61]. In this regard, Biswas et al. (2012) stated that terrestrial erosion is the main mechanism enhancing Fe concentration in the seawater [43]. It was also reported that high amounts of Zn in the fillet of narrow-barred Spanish mackerel were derived from food supplies compared to the water [29].
PTEs can be stored in the fish through the water, food, and sediments. Therefore, fish is considered as an indicator of PTEs contamination in the coastal environment [43]. PTEs observed in the fish body are resulted from two main sources: (1) the natural presence of PTEs in the earth’s crust and are introduced to the water medium through weathering mechanism, and (2) the introduced PTEs to the aquatic medium through anthropogenic activities (i.e., through urban run-off, agricultural wastes, industrial effluents, and emissions or unintentional spill of oil from boats and ships) mainly in the coastal areas [34, 35, 38, 40, 62].
The essential elements need to be taken up from the water, food, or sediment for fish’s normal metabolism. However, non-essential elements are also taken up by fish simultaneously and accumulate in their tissues [29, 35, 38]. It is worth mentioning that the affinity of different PTEs is not similar, influencing their bioaccumulation degree within the fish body. In this regard, Heshmati et al. (2019) noted that toxic metals’ affinity was significantly lower than that of essential metals [63]. For instance, essential elements (like Fe and Zn) had the highest affinity rate since they are essential elements, and fish take up them as much as needed from the surrounded milieu [61]. Therefore, the level of toxic metals in the fillet of narrow-barred Spanish mackerel is much lower than that of the essential metals [15].
It should bear in mind that different PTEs considerably depend on their concentrations and their bioavailability in the water media and exposure time [64]. Furthermore, factors like ingestion quantity, storage level, and excretion percentages of PTEs in the fish body effectively affect their bioaccumulation after absorption [65, 66].
Numerous factors are influencing PTE concentrations in fish tissues, including biological factors related to fish (like species, growth-dependent factors like age, length, sex and body weight, tissue, habitat, trophic level, feeding behavior, and migration) and ecological factors (water chemistry like salinity, hardness, pH as well as geographical conditions and season) [35,36,37, 40, 41, 43, 67].
Narrow-barred Spanish mackerel (Scomberomorus commerson)is grouped as pelagic and carnivores on macrofauna [32, 41]. They exist and feed by small fishes like anchovies, clupeids, carangids, and squids and shrimps in the sea [36]. Sary & Velayatzadeh (2014) studied Pb and Zn levels in some fish species, including the Scomberomorus commerson from the Persian Gulf in Iran. Their results showed that both metals’ concentration is higher in the gills and liver of Scomberomorus commerson than in the muscle. Furthemore, Bibak et al. (2020) also showed that PTE concentrations in the liver of 2 fish species were much more than muscle [68]. The PTE levels also in 2001 were higher than those in 2011 [67]. It should bear in mind that a higher concentration of most PTEs in the liver results from the fact that this organ has the primary physiological role in fish metabolism and shows higher metabolic activity in detoxification of these contaminants [29, 37, 69].
Generally, PTE levels increase with the size and length of fish [36]. The fish size impacts the value of intake, distribution, translocation, and elimination of PTEs in the fish body. Older fish show higher PTE concentrations than younger because of extensive exposure time [36]. For example, Abdallah (2008) observed that the PTE concentrations reduced considerably with fish’s body weight. However, a positive association with body weight was detected for Zn, Cu, and Pb concentrations. For example, significant negative correlations with body weight were found for Cd and Pb; however, a positive correlation was found for Cr in Scomberomorus commerson [41]. Furthermore, Anual (2018) reported that Hg concentrations in fish with body length below 20 cm were 1.5 times lower than fish with more than 20 cm [36]. The accumulation of PTEs in different parts of the fish body is also related to the type of metal. In this regard, Khaled (2004) noted that Fe, Zn, and Cu had the highest concentrations in the liver while Cd, Cr, and Pb had their highest levels in the kidneys, gills, and bones [37].
It is worthy to note that the type of PTEs in each area is different. In this regard, Abdallah (2008) observed that the concentrations of Cu and Cr were lower in fishes from El-Mex Bay than Eastern Harbor, while the reverse trend was reported for Cd, Pb, and Zn, representing the close sources of toxic elements present in El-Mex shoreline region (Egypt) which is exposed to significant inputs of anthropogenic and agricultural pollution during the past years. There are different industrial plants in this area including chloralkali, iron, steel factories, tanneries, and petroleum refining [41]. Furthermore, Bibak et al. (2020) investigated the concentration of PTEs in fishes caught from 4 different Persian Gulf areas. Their results showed that fish samples of Emam Hassan Port were more contaminated than other ports due to pollutant discharge from oil refinery [68]. It was stated that season is an important factor affecting PTE concentration in the fish tissues due to alterations in water temperature and salinity as well as growing and reproductive phases [29]. For instance, Saei-Dehkordi et al. (2010 and 2011) reported that most of all fish samples had more PTEs (Hg and As) concentration in winter than in summer. In winter, the higher PTE level could be due to high rainfall, which rinsed the wastes [32, 40]. In contrast, Khaled (2009) observed opposite results [29].
Nevertheless, muscle tissues cannot store high amounts of most PTEs, but in contaminated zones, such toxic metals in muscles may increase [69]. Thus, the PTE concentrations in the fish living in the water medium close to industrialized areas are much higher than in other areas [38]. Shwafi (2002) reported that the differences in the concentration of PTEs in the fish muscle were mostly attributed to rocks’ mechanical and chemical eroding rather than anthropogenic activities [70]. Pilehvarian et al. (2015) also revealed that industries close to the Persian Gulf could introduce many PTEs to the water environment [35]. Furthermore, Ahmed et al. (2015) observed that the PTE concentrations in 2010–2011 were considerably higher than those in 2006–2007 [38]. Prudente (1997) also reported that Manila Bay in China accepts major discharges of urban and industrial disposals [71]. Yasmeen et al. (2016) also indicated that in the Port located near the Karachi, disposal of industrial and municipal waste is the main reason for the high concentration of PTEs in the fish tissues [44].
Concentration of Pb
Pb does not have positive or nutritional influences on organisms as it is toxic [41]. After its entrance to the body through gills, and is accumulated in gills, liver, and muscles [67]. Pb can negatively affect cognitive growth and intellectual functioning in children [72]. It could also increase blood pressure and heart-related diseases in adults [70], anemia, renal failure, and liver injury in humans [34].
The results of the current study showed that the ranking of countries based on Pb concentration in fillet of narrow-barred Spanish mackerel fish was Egypt (851.81 μg/kg-ww) > Iran (458.97 μg/kg-ww) > Pakistan (167.25 μg/kg-ww) > Yemen (52.03 μg/kg-ww) > Philippines (27.30 μg/kg-ww) > Bahrain (10.00 μg/kg-ww) > China (5.91 μg/kg-ww) > India (0.63 μg/kg-ww) (Table 3).
Pb′s mean concentration in all investigated countries was lower than the permissible limit (FAO: 500 μg/kg-ww) [81], except in Egypt. In Egypt, fish are caught and collected either from wild sources or aquacultures distributed in different localities worldwide, and many industries discharge their polluted effluent into water sources such as the Nile River [84,85,86,87,88]. Lead is used in different industrial activities and is a major residue from gasoline-burning, which diffuses the atmosphere in a large amount [41]. Pb enters into water throughout industrial waste (like painting, battery manufacturing, dyeing, and oil refineries) [72]. Furthermore, Pb could enter the sea from natural supplies and the atmosphere [67]. Thus, Pb concentration increases due to industrial activities as well as using pesticides and fertilizers [72]. For example, using gasoline additives and lubricants high in the lead in the mining and industrial parts could increase Pb concentration in the food chain [35, 44].
Ni Concentration
Ni poisoning complications include headache, insomnia, nausea, dizziness, skin inflammation, and lung cancer [89]. The maximum limit of Ni in fish tissue recommended by USFDA is 80,000 μg/kg ww [83]. The rank of Ni concentration in narrow-barred Spanish mackerel fish in different countries was as Iran (1358.34 μg/kg) > Egypt (587.85 μg/kg) > Pakistan (630.00 μg/kg) > China (32.27 μg/kg) > Bahrain (32.27 μg/kg). Therefore, according to Table 3, Ni concentration in all investigated countries was considerably lower than the permissible level.
Ni is widely distributed in the environment through fossil fuels, mining and refineries, waste incineration, and land erosion by rainfall into water resources [90].
Fe Concentration
Fish is a significant iron source, an important essential element for the body’s metabolism, and has a leading role in transporting oxygen and cellular respiration [16]. Iron deficiency leads to anemia [34, 43]. Fe has an effective function in forming hemoglobin in combination with protein and Cu [44]. However, too much concentration of Fe is toxic and dangerous to humans health [91].
The rank of Fe concentration in narrow-barred Spanish mackerel fish in different countries was as Egypt (18,055.87 μg/kg) > Pakistan (15,885.94 μg/kg) > China (5470.00 μg/kg) > Bahrain (5000 μg/kg) > India (3990.00 μg/kg) > Iran (550.20 μg/kg). The observed Fe concentrations in narrow-barred Spanish mackerel fish from all studied countries (Table 3) were much lower than acceptable standards set by WHO (100,000 μg/kg ww) [92]. Fe is the main constituent of the earth’s outer and inner core and the fourth most common element in the crust [93].
Cd Concentration
Cd is a prevalent toxic metal that is not essential in the fish body, and it is hard to excrete after ingested [41]. After Cd′s ingestion through food consumption, it may trigger kidney disorders, skeletal impairment, and reproductive shortages [34]. It boosts the generation of renal stones and secretion of calcium through urine [44].
The rank of Cd concentration in fillet of narrow-barred Spanish mackerel fish in different countries was Pakistan (199.94 μg/kg) > Egypt (181.00 μg/kg) > Yemen (121.79 μg/kg) > Iran (45.87 μg/kg) > Bahrain (10 μg/kg) > Philippines (2 μg/kg) > China (1.57 μg/kg) (Table 3).
The Food and Agriculture Organization (FAO) allows maximum concentrations of 500 μg/kg Cd [81]. Our results showed that the Cd concentration in all investigated countries was lower than the FAO’s acceptable level. This pollutant is naturally present in surface and groundwater [94]. However, most of it enters the aquatic ecosystems through soil and bedrock erosion, polluted sediments derived from industrial plants, wastewater from contaminated areas, and sludge and fertilizer in agriculture [95, 96]. Tepanosyan et al. (2017) attributed high Cd concentrations in the environment to vehicle tires and the burning of fossil fuels [97]. At the same time, Najm et al. (2014) emphasized the widespread use of phosphate fertilizers in agriculture as the main reason for the increase of Cd in water, sediments, and finally, the aquatic life of Babolsar Coastal Waters [98].
Total Cr Concentration
Cr is effective in macronutrient metabolism and considers as an essential element in humans [43]. Cr is the seventh most abundant element in the earth’s crust and has various oxidation states, of which forms Cr (III), and Cr (VI) can be frequent and more stable in the environment. Cr (III) is an important component in the balance of human and animal diets, and its deficiency leads to disruption of glucose and fat metabolism [99]. However, Cr (VI) is highly toxic and carcinogenic, causing death in humans, animals, plants, shortness of breath, and inflammation of the mouth, nose, and lungs, and inflammation of the skin, problems with digestion, and damage to the kidneys and liver [98].
The ranking of countries based on total Cr concentration in fillet of narrow-barred Spanish mackerel fish in different countries was China (820.00 μg/kg-ww) > Egypt (746.00 μg/kg-ww) > Pakistan (330.00 μg/kg-ww) > India (81.90 μg/kg-ww) (Table 3).
The FAO and WHO suggested 1000 and 50,000 μg/kg as maximum limits for Cr in fish, respectively [81, 100]. As shown in Table 3, all investigated countries in terms of Cr concentration in the fillet of narrow-barred Spanish fish have reasonable conditions compared with a regulated limit. Type of species, age and developmental stage, temperature, Cr concentration, pH, alkalinity, salinity, and hardness of water can influence Cr concentration in fish tissues [101]. Cr element naturally exists in soil, rocks, animals, and plants [102]. However, anthropologic activities like manufacturing processes of iron and steel industries and cooling towers discharge Cr into the environment [103]. Furthermore, Cr is used in the inks, leather, and steel industries [41].
Zn Concentration
Zn is probably the least toxic of all the PTEs [37]. It is a vital metal in humans and other animals, which acts as an enzyme activator [72]. It can become toxic in excess concentration [43]. It is contributed to several metabolic routes in the human body, and its deficiency can lead to appetite decrement, growth inhibition, skin disorders, and immunological malfunctions [34].
Results showed that the order of different countries based on Zn concentration in fillet of narrow-barred Spanish mackerel fish was Philippines (14,595.00 μg/kg-ww) > Pakistan (6740.00 μg/kg-ww) > Egypt (5429.09 μg/kg-ww) > India (4515.00 μg/kg-ww) > China (4240.00 μg/kg-ww) > Iran (3421.20 μg/kg-ww) > Yemen (454.34 μg/kg-ww). The Zn concentration in all the investigated countries is lower than the standard value (30,000 μg/kg-ww) regulated by FAO/WHO in fish (Table 3) [104]. The high concentration of Zn in the fillet of narrow-barred Spanish mackerel is perhaps due to the incidence of an enormous number of fishing containers that apply galvanized element coatings to avoid rusting, which finally enters into the environment due to leaching [43]. Zn enters into aquatic ecosystems through metal smelting industries, the effect of acid rain on building materials containing zinc, drilling activity, metal condensation, sewage sludge, compost, chemical fertilizers, and insecticides [105]. Leung et al. (2014) carried out a study in India and they reported that the main source of Zn (140 mg/kg) detected in the Estuary and the surrounding coastal area sediments was the Pearl River delta, where is an important area for agricultural, commercial, and industrial development [90]. Additionally, Agah et al. (2008) noted that the high amounts of heavy metal, including Zn, in fishes captured from the Persian Gulf (Iran) could be attributed to growing industrial pressures on the Persian Gulf environment [106].
Mn Concentration
Mn, an essential element in biological systems, plays a coenzyme role in the human body bound to some enzymes such as pyruvate carboxylase and superoxide dismutase to act as an activator in accelerating reactions [107]. Mn is a low toxicity metal. Its increased levels cause severe brain damage with physiological and neurological disorders such as poor reproductive performance, Parkinson (muscle stiffness), and abnormal bone and cartilage [108, 109]. In this study, the rank of Mn concentration in fillet of narrow-barred Spanish mackerel fish in different countries was Philippines (2793.00 μg/kg-ww) > China (1720 μg/kg-ww) > Egypt (736.48.00 μg/kg-ww) > India (105.00 μg/kg-ww) > Iran (84.00 μg/kg-ww) > Bahrain (40.00 μg/kg-ww). The observed Mn concentrations in narrow-barred Spanish mackerel fish from all studied countries (Table 3) were much lower than the legal standards set by FAO (300,000 μg/kg ww) [81]. Therefore, there is no concern in the case of Mn in the fillet of narrow-barred Spanish mackerel fish. Mn could find its way to enter into the environment from industries of iron, alloys, and steel products [37]. Furthermore, fertilizers, fungicides, and dry-cell batteries are considered other Mn sources [43].
Cu Concentration
Copper is an essential metal for animals because it contributes to enzyme function and takes part in respiratory routes [41]. It plays a vital role in the activation of numerous enzymes [43]. It is necessary for the growth and development of bones and human health [44]. However, its high intake can cause adverse health problems such as liver and kidney damage [34].
The rank of Cu concentration in fillet of narrow-barred Spanish mackerel fish in different countries was Bahrain (4000 μg/kg-ww) > Pakistan (2807.41 μg/kg-ww) > Egypt (922.44 μg/kg-ww) > Philippines (577.00 μg/kg-ww) > India (525 μg/kg-ww) > China (370.00 μg/kg-ww) > Iran (201.01 μg/kg-ww) > Yemen (64.85 μg/kg-ww) (Table 3).
The FAO/WHO suggested 30,000 μg/kg-ww as maximum limits for Cu in fish [81]. In the case of Cu, its concentration in fish tissue was significantly lower than the standard limit, so there is no concern. Generally, Cu concentration observed in studied countries originates from natural components (e.g., forest fire, volcanoes, and plant decomposition), as well as human activities (e.g., fertilizers, pesticides, mining, municipal and industrial wastewater industries) [110]. Yi et al. (2011) revealed that the sources of Hg, Cd, Pb, Cr, Cu, and Zn are anthropogenic components and may originate from similar pollution sources, mainly from metal processing, electroplating industries, industrial wastewater, and domestic sewage [111]. Bervoets et al. (2001) concluded that Cu concentration in fish mussels was positively correlated with sediment and water levels [112].
Inorganic As Concentration
Arsenic is a ubiquitous toxic metal that causes adverse biological effects like several types of cancer (liver, skin, and bladder) [40]. Researches on the concentration of As level in the body of narrow-barred Spanish mackerel fish are very limited. Among the countries, the concentration of As in narrow-barred Spanish mackerel fish has been studied only in three studies in Iran (Table 3), in which the average amount of inorganic As mentioned was 93.11 μg/kg ww. There is no information about maximum As levels in fish samples in US standards, while the acceptable limit of As in fish tissue in Singapore and Malaysia is 1 ppm [113]. The maximum arsenic level permitted for fishes is 1000 μg/kg, according to Australia standard [113, 114]. According to these, the inorganic As the level in the investigated country (Table 3) is lower than the allowable limit, and there is no considerable concern. Shah et al. (2009) reported the concentration range of 1.01–15.2 μg/g in freshwater fish species [115], while Tuzen (2009) expressed As in the range of 0.0–1.72 mg/kgN ww in canned fishes [113]. As contamination may derive from natural environmental sources percolating into the water, mining activities, and other manufacturing processes [35].
Industrial wastewater from tanneries, ore mining, and dyeing are also significant sources of surface water resource contamination by As [116, 117].
Methylmercury
Over 90% of metallic mercury can be transformed into MeHg by some microorganisms. MeHg, a lipophilic organic substance, is a recognized neurotoxicant related to children’s developmental interruptions [118]. Methyl mercury, an extremely poisonous and absorbable kind of mercury, mostly affects the nervous system [31, 35, 40].
According to the Joint FAO/WHO Expert Committee on Food Additives/JECFA, the maximum allowable limit has been reported 500 μg/kg for Hg in fish [82]. The rank of methyl mercury concentration in fillet of narrow-barred Spanish mackerel fish in different countries was Philippines (153.30 μg/kg) > Malaysia (108 μg/kg) > Bahrain (100.00 μg/kg-ww) > Taiwan (60.00 μg/kg-ww) > Egypt (50.40 μg/kg-ww) > Iran (21.90 μg/kg-ww) (Table 3). Considering to Hg concentration in the fillet of narrow-barred Spanish mackerel fish of different countries, it was found that all investigated countries meet the regulated standards.
Natural sources of methylmercury entering the aquatic environment include weathering rocks, erosion of the earth’s crust, and volcanoes [119]. Critical anthropologic sources of methylmercury result from rapid urbanization and industrialization, including coal combustion, paper and cellulose industries, the mining industry and by-products, agricultural fertilizers, and fungicides, and solid waste incineration [120]. Driscoll et al. (2013) noted that mercury’s most important anthropogenic sources in the environment are coal combustion [121].
Health Risk Assessment
A total target hazard quotient (TTHQ) equal or less than 1, the non-carcinogenic risk is a safe range, while for TTHQ higher than 1, adverse health effects are considerable [122]. The rank of countries based on the TTHQ in adults was the rank order of health risk assessment by country based on the TTHQ for adult consumers was Malesia(0.22251) > Philippines (0.21912) > Egypt (0.08684) > Taiwan(0.07430) > Bahrain(0.04893) > Iran (0.03528) > China(0.00620) > Pakistan (0.00316) > Yemen(0.00157) > India(0.00073), while the corresponding values for child consumers was Malaysia (1.03838) > Philippines (1.02257) > Egypt (0.40523) > Taiwan (0.34674) > Bahrain (0.22832) > Iran (0.16466) > China (0.02892) > Pakistan (0.01474) > Yemen (0.00731) > India (0.00340) (Table 4). Children in Malaysia and the Philippines were at considerable non-carcinogenic risk [122,123,124,125] due to the ingestion of PTEs via the consumption of narrow-barred Spanish mackerel fish (Table 2). The high TTHQ in Malaysia and the Philippines can be correlated with the high concentration of PTEs in the targeted food products (Table 3) and high ingestion rate (Table 2).
Similarly, some studies were conducted regarding PTEs in foods such as seafood, meat products, cereal-based foods, and vegetables and associated health risks in adults and children in Iran [123, 126,127,128,129,130]; TTHQ in the child consumers was ~ 4.68 times higher than adults. The higher TTHQ in children is related to their lower BW, and hence, children are at a higher health risk than adults [125, 131,132,133,134].
Differences in concentrations of PTEs in narrow-barred Spanish mackerel fish, ingestion rate, exposure time, exposure frequency, exposures duration, average life-time, body weight, and toxicity of PTEs play significant roles in the health risk of consumers. Hence, we observed various TTHQ values for PTEs in the countries investigated [125, 131, 133,134,135].
Conclusions
This study was designed to retrieve all the studies on the concentration of PTEs, including inorganic As, methyl Hg, Total Cr, Cd, Pb, Cu, Zn, Fe, Mn, and Ni in fillets of narrow-barred Spanish mackerel fish. Concentrations of PTEs were meta-analyzed according to the country; finally, the exposed population’s health risk was estimated. The lowest and highest concentrations in fillets of narrow-barred Spanish mackerel fish were related to Cd and Fe, respectively. Children in Malaysia and the Philippines were at considerable non-carcinogenic risk; hence, control plans for reducing the concentration of PTEs in fillets of narrow-barred Spanish mackerel fish are necessary.
References
Korkmaz C, Ay Ö, Ersoysal Y, Köroğlu MA, Erdem C (2019) Heavy metal levels in muscle tissues of some fish species caught from north-east Mediterranean: evaluation of their effects on human health. J Food Compos Anal 81:1–9
Fair PA, White ND, Wolf B, Arnott SA, Kannan K, Karthikraj R, Vena JE (2018) Persistent organic pollutants in fish from Charleston Harbor and tributaries, South Carolina, United States: a risk assessment. Environ Res 167:598–613
Borzoei M, Zanjanchi MA, Sadeghi-aliabadi H, Saghaie L (2018) Optimization of a methodology for determination of iron concentration in aqueous samples using a newly synthesized chelating agent in dispersive liquid-liquid microextraction. Food Chem 264:9–15
Mo WY, Man YB, Zhang F, Wong MH (2019) Fermented food waste for culturing jade perch and Nile tilapia: growth performance and health risk assessment based on metal/loids. J Environ Manag 236:236–244
Copat C, Arena G, Fiore M, Ledda C, Fallico R, Sciacca S, Ferrante M (2013) Heavy metals concentrations in fish and shellfish from eastern Mediterranean Sea: consumption advisories. Food Chem Toxicol 53:33–37
Fuentes-Gandara F, Herrera-Herrera C, Pinedo-Hernández J, Marrugo-Negrete J, Díez S (2018) Assessment of human health risk associated with methylmercury in the imported fish marketed in the Caribbean. Environ Res 165:324–329
Go AS, Mozaffarian D, Roger VL, Benjamin EJ, Berry JD, Blaha MJ, Dai S, Ford ES, Fox CS, Franco S (2014) Executive summary: heart disease and stroke statistics—2014 update: a report from the American Heart Association. Circulation 129(3):399–410
Hu FB, Manson JE, Willett WC (2001) Types of dietary fat and risk of coronary heart disease: a critical review. J Am Coll Nutr 20(1):5–19
Korkmaz C, Ay Ö, Çolakfakioğlu C, Cicik B, Erdem C (2017) Heavy metal levels in muscle tissues of Solea solea, Mullus barbatus, and Sardina pilchardus marketed for consumption in Mersin, Turkey. Water Air Soil Pollut 228(8):315
Ferrante M, Zanghì G, Cristaldi A, Copat C, Grasso A, Fiore M, Signorelli SS, Zuccarello P, Conti GO (2018) PAHs in seafood from the Mediterranean Sea: an exposure risk assessment. Food Chem Toxicol 115:385–390
Zhao YG, Wan HT, Law AY, Wei X, Huang YQ, Giesy JP, Wong MH, Wong CK (2011) Risk assessment for human consumption of perfluorinated compound-contaminated freshwater and marine fish from Hong Kong and Xiamen. Chemosphere 85(2):277–283
Cobelo-Garcı́a A, Prego R, Labandeira A (2004) Land inputs of trace metals, major elements, particulate organic carbon and suspended solids to an industrial coastal bay of the NE Atlantic. Water Res 38 (7):1753–1764. https://doi.org/10.1016/j.watres.2003.12.038
Korn MGA, dos Santos GL, Rosa SM, Teixeira LSG, de Oliveira PV (2010) Determination of cadmium and lead in cetacean Dolphinidae tissue from the coast of Bahia state in Brazil by GFAAS. Microchem J 96(1):12–16. https://doi.org/10.1016/j.microc.2010.01.001
Tran TAM, Leermakers M, Hoang TL, Elskens M (2018) Metals and arsenic in sediment and fish from Cau Hai lagoon in Vietnam: ecological and human health risks. Chemosphere 210:175–182
McEneff G, Quinn B, Bennion M, Dolan S, O'Rourke K, Morrison L (2017) Bioaccumulation of metals in juvenile rainbow trout (oncorhynchus mykiss) via dietary exposure to blue mussels. Chemosphere 188:548–556. https://doi.org/10.1016/j.chemosphere.2017.08.141
Borzoei M, Zanjanchi MA, Sadeghi-aliabadi H, Saghaie L (2019) Trace determination of Iron in real waters and fruit juice samples using rapid method: optimized dispersive liquid-liquid microextraction with synthesized nontoxic chelating agent. Biol Trace Elem Res 192(2):319–329
Saha N, Mollah MZI, Alam MF, Safiur Rahman M (2016) Seasonal investigation of heavy metals in marine fishes captured from the Bay of Bengal and the implications for human health risk assessment. Food Control 70:110–118. https://doi.org/10.1016/j.foodcont.2016.05.040
Ghaneian MT, Bhatnagar A, Ehrampoush MH, Amrollahi M, Jamshidi B, Dehvari M, Taghavi M (2017) Biosorption of hexavalent chromium from aqueous solution onto pomegranate seeds: kinetic modeling studies. Int J Environ Sci Technol 14(2):331–340. https://doi.org/10.1007/s13762-016-1216-8
Djahed B, Taghavi M, Farzadkia M, Norzaee S, Miri M (2018) Stochastic exposure and health risk assessment of rice contamination to the heavy metals in the market of Iranshahr, Iran. Food and Chemical Toxicology
Moradi A, Mirzaei R, Mr A, Bay A, Ghaderpoori M, Asadi A, Fakhri Y, Sorooshian A, Mousavi Khaneghah A (2020) The concentration, characteristics, and probabilistic health risk assessment of potentially toxic elements (PTEs) in street dust: a case study of Kashan, Iran. Toxin Rev 5(2):1–20
Fakhri Y, Mousavi Khaneghah A, Hadiani MR, Keramati H, Hosseini Pouya R, Moradi B, da Silva BS (2017) Non-carcinogenic risk assessment induced by heavy metals content of the bottled water in Iran. Toxin Rev 36(4):313–321
Khan A, Khan S, Khan MA, Qamar Z, Waqas M (2015) The uptake and bioaccumulation of heavy metals by food plants, their effects on plants nutrients, and associated health risk: a review. Environ Sci Pollut Res 22(18):13772–13799
Fathabad AE, Shariatifar N, Moazzen M, Nazmara S, Fakhri Y, Alimohammadi M, Azari A, Khaneghah AM (2018) Determination of heavy metal content of processed fruit products from Tehran's market using ICP-OES: a risk assessment study. Food Chem Toxicol 115:436–446
Rainer F, Daniel P (2018) Scomberomorus commerson in fish base
Roa-Ureta RH, Lin Y-J, Rabaoui L, Al-Abdulkader K, Qurban MA (2019) Life history traits of the narrow-barred Spanish mackerel (Scomberemorus commerson) across jurisdictions of the southeast Arabian peninsula: implications for regional management policies. Reg Stud Mar Sci 31:100797
Grandcourt EM A review of the fisheries, biology, status and management of the narrow-barred Spanish mackerel (Scomberomorus commerson) in the Gulf Cooperation Council countries (Bahrain, Kuwait, Oman, Qatar, Saudi Arabia and the United Arab Emirates)
Motlagh SAT, Shojaei MG (2009) Population dynamics of narrow–barred Spanish mackerel (Scomberomorus commerson) in the Persian Gulf, Bushehr Province, Iran. Indian J Fish 56(1):7–11
Khoshnoud MJ, Mobini K, Javidnia K, Hosseinkhezri P, Aeen Jamshid KJIJoPS (2011) Heavy metals (Zn, Cu, Pb, Cd and Hg) contents and fatty acids ratios in two fish species (Scomberomorus commerson and Otolithes ruber) of the Persian Gulf 7 (3):191–196
Khaled A (2009) Trace metals in fish of economic interest from the west of Alexandria. Egypt. 25(4):229–246
Musaiger AO, D’Souza RJPJBS (2008) Chemical composition of raw fish consumed in Bahrain 11:55–61
Chien L-C, Yeh C-Y, Jiang C-B, Hsu C-S, Han B-CJC (2007) Estimation of acceptable mercury intake from fish in. Taiwan. 67(1):29–35
Saei-Dehkordi SS, Fallah AAJMJ (2011) Determination of copper, lead, cadmium and zinc content in commercially valuable fish species from the Persian Gulf using derivative potentiometric stripping analysis 98 (1):156–162
Sobhanardakani S, Tayebi L, Farmany A, Cheraghi MJEm, assessment (2012) Analysis of trace elements (Cu, Cd, and Zn) in the muscle, gill, and liver tissues of some fish species using anodic stripping voltammetry 184 (11):6607–6611
Gu Y-G, Lin Q, Wang X-H, Du F-Y, Yu Z-L, Huang H-HJMpb (2015) Heavy metal concentrations in wild fishes captured from the South China Sea and associated health risks 96 (1–2):508–512
Pilehvarian AA, Malekirad AA, Bolandnazar N-S, Rezaei MJTr (2015) Heavy metal bioaccumulation in different fish species in the coast of the Persian Gulf, Iran. 34 (4):215–219
Anual ZF, Maher W, Krikowa F, Hakim L, Ahmad NI, Foster SJMJ (2018) Mercury and risk assessment from consumption of crustaceans, cephalopods and fish from West Peninsular Malaysia. 140:214-221
Khaled A (2004) Heavy metals concentrations in certain tissues of five commercially important fishes from El-Mex Bay. Alexandria, Egypt
Ahmed Q, Bat L, Yousuf F, Ali QM, Nazim KJOBSJ (2015) Accumulation of heavy metals (Fe, Mn, Cu, Zn, Ni, Pb, Cd and Cr) in tissues of narrow-barred Spanish mackerel (family-Scombridae) fish marketed by Karachi Fish Harbor. 1 (1)
Sobhanardakani S, Tayebi L, Farmany AJWASJ (2011) Toxic metal (Pb, Hg and As) contamination of muscle, gill and liver tissues of Otolithes rubber, Pampus argenteus, Parastromateus niger, Scomberomorus commerson and Onchorynchus mykiss 14 (10):1453–1456
Saei-Dehkordi SS, Fallah AA, Nematollahi AJF, Toxicology C (2010) Arsenic and mercury in commercially valuable fish species from the Persian Gulf: influence of season and habitat 48 (10):2945–2950
Abdallah MAMJJoMS (2008) Trace element levels in some commercially valuable fish species from coastal waters of Mediterranean Sea, Egypt. 73 (1–2):114–122
Sary AA, Velayatzadeh MJAiEB (2012) Lead and zinc levels in Scomberomorus guttatus, Scomberomorus commerson and Otolithes ruber from Hendijan, Iran.843-849
Biswas S, Prabhu RK, Hussain KJ, Selvanayagam M, Satpathy KKJEm, assessment (2012) Heavy metals concentration in edible fishes from coastal region of Kalpakkam, southeastern part of India 184 (8):5097–5104
Yasmeen K, Mirza MA, Khan NA, Kausar N, Rehman A-u, Hanif MJS (2016) Trace metals health risk appraisal in fish species of Arabian Sea. 5 (1):859
Higgins JP, Green S (2011) Cochrane handbook for systematic reviews of interventions, vol 4. John Wiley & Sons,
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J, Moher D (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med 6(7):15–25
Hedges LV, Gurevitch J, Curtis PS (1999) The meta-analysis of response ratios in experimental ecology 80 (4):1150–1156. https://doi.org/10.1890/0012-9658(1999)080[1150:Tmaorr]2.0.Co;2
FAO (2018) Fish and seafood consumption per capita. https://ourworldindata.org/grapher/fish-and-seafood-consumption-per-capita
Njuguna SM, Makokha VA, Yan X, Gituru RW, Wang Q, Wang J (2019) Health risk assessment by consumption of vegetables irrigated with reclaimed waste water: a case study in Thika (Kenya). J Environ Manag 231:576–581. https://doi.org/10.1016/j.jenvman.2018.10.088
Barnes DG, Dourson M, Preuss P, Bellin J, Derosa C, Engler R, Erdreich L, Farber T, Fenner-Crisp P, Francis E (1988) Reference dose (RfD): description and use in health risk assessments. Regul Toxicol Pharmacol 8(4):471–486
EPA (2000) Risk-based concentration table. J Philadelphia PA: United States Environmental Protection Agency, Washington DC
EPA (2011) Exposure factors handbook: 2011 edition. EPA/600/R-09,
Quan H, Zhang J (2003) Estimate of standard deviation for a log-transformed variable using arithmetic means and standard deviations. Stat Med 22(17):2723–2736
Higgins J, White IR, Anzures-Cabrera J (2008) Meta-analysis of skewed data: combining results reported on log-transformed or raw scales. Stat Med 27(29):6072–6092
Higgins., Thompson S (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21 (11):1539–1558
Moradi Q, Mirzaei R, Alipour M, Bay A, Ghaderpoori M, Asadi A, Fakhri Y, Sorooshian A, Mousavi Khaneghah A (2020) The concentration, characteristics, and probabilistic health risk assessment of potentially toxic elements (PTEs) in street dust: a case study of Kashan, Iran. Toxin Rev:1–10
Mostafaii GR, Moravveji A, Hajirostamloo B, Hesami Arani M, Dehghani M, Heidarinejad Z, Fakhri Y, Khaneghah AM (2020) The concentration and risk assessment of potentially toxic elements (PTEs) in unrefined salt: a case study of Aran and Bidgol Lake, Iran. Int J Environ Anal Chem :1–13
Samiee S, Fakhri Y, Sadighara P, Arabameri M, Rezaei M, Nabizadeh R, Shariatifar N, Mousavi Khaneghah A (2020) The concentration of polycyclic aromatic hydrocarbons (PAHs) in the processed meat samples collected from Iran’s market: a probabilistic health risk assessment study. Environ Sci Pollut Res :1–14
Fakhri Y, Nematollahi A, Abdi-Moghadam Z, Daraei H, Ghasemi SM (2020) Concentration of potentially harmful elements (PHEs) in trout fillet (rainbow and brown) fish: a global systematic review and meta-analysis and health risk assessment. Biol Trace Elem Res:1–13
Pirsaheb M, Irandost M, Asadi F, Fakhri Y, Asadi A (2020) Evaluation of polycyclic aromatic hydrocarbons (PAHs) in fish: a review and meta-analysis. Toxin Rev 39(3):205–213
Varol M, Kaya GK, Alp SA, Sünbül MR (2018) Trace metal levels in rainbow trout (Oncorhynchus mykiss) cultured in net cages in a reservoir and evaluation of human health risks from consumption. Biol Trace Elem Res 184(1):268–278
Cilingir-Yeltekin A (2018) Comparison of toxic metal, trace element and macro element levels in trout cultivated in Latvia and Turkey. FEB-FRESENIUS environmental bulletin:7039
Heshmati A, Sadati R, Ghavami M, Khaneghah AM (2019) The concentration of potentially toxic elements (PTEs) in muscle tissue of farmed Iranian rainbow trout (Oncorhynchus mykiss), feed, and water samples collected from the west of Iran: a risk assessment study. Environ Sci Pollut Res 26(33):34584–34593
Barrientos C, Tapia J, Bertrán C, Peña-Cortés F, Hauenstein E, Fierro P, Vargas-Chacoff L (2019) Is eating wild rainbow trout safe? The effects of different land-uses on heavy metals content in Chile. Environ Pollut 254:112995
Bat L, Oztekin A, Yardim O (2018) Metal levels in large sea trout from Sinop fish market. Fresenius Environ Bull 27(12):8505–8508
Jiang H, Qin D, Mou Z, Zhao J, Tang S, Wu S, Gao L (2016) Trace elements in farmed fish (Cyprinus carpio, Ctenopharyngodon idella and Oncorhynchus mykiss) from Beijing: implication from feed. Food Addit Contam Part B 9(2):132–141
Sary AA, Velayatzadeh M (2014) Determination of lead and zinc in king mackerel (Scomberomorus guttatus Bloch & Schneider, 1801), Spanish mackerel (Scomberomorus commerson Lacepède, 1800) and tiger-toothed croaker (Otolithes ruber Bloch and Schneider, 1801) from Persian Gulf, Iran in 2001 and 2011. J Biodivers Environ Sci 5(1):322–329
Bibak M, Sattari M, Tahmasebi S, Kafaei R, Sorial GA, Ramavandi B (2020) Trace and major elements concentration in fish and associated sediment–seawater, northern shores of the Persian Gulf. Biol Trace Elem Res :1–13
Hosseini M, Naderi M, Gholami S, Hadipour M (2018) Toxic metals in the muscle and liver of five main commercially-important fishes from the Persian Gulf, Southern Iran. Caspian J Environ Sci 16(2):191–198
Shwafi A (2002) Heavy metals concentration levels in some fish species in the red sea and gulf of Aden-Yemen
Prudente M, Kim E-Y, Tanabe S, Tatsukawa R (1997) Metal levels in some commercial fish species from Manila Bay, the Philippines. Mar Pollut Bull 34(8):671–674
Gu Y-G, Lin Q, Wang X-H, Du F-Y, Yu Z-L, Huang H-H (2015) Heavy metal concentrations in wild fishes captured from the South China Sea and associated health risks. Mar Pollut Bull 96(1–2):508–512
Sadeghi E, Mohammadi M, Sharafi K, Bohlouli SJBCC (2015) Determination and assessment of three heavy metal content (Cd, Pb and Zn) in Scomberomorous commerson fish caught from the Persian Gulf. 47:220–223
Hosseini M, Naderi M, Gholami S, Hadipour MJCJES (2018) Toxic metals in the muscle and liver of five main commercially-important fishes from the Persian Gulf. Southern Iran 16(2):191–198
Pazira A, fard OK (2016) Comparison of biological accumulation of nickel and cadmium heavy metals in muscle tissue of two species of milk fish Scomberomorus guttatus Scomberomorus commerson in Bushehr Mar Biol Res 6:79–89
Sari AE, Ghasempouri SM, Yaghoupzadeh Y (2001) Measurement Comparison of heavy elements (cadmium, nickel, lead and zinc) in some commercial fish and Penaeus semisulcatus green tiger shrimp in Bushehr region. Ecology 27:15–23
Sary AA, Velayatzadeh MJJoB, Sciences E (2014) Determination of lead and zinc in king mackerel (Scomberomorus guttatus Bloch & Schneider, 1801), Spanish mackerel (Scomberomorus commerson Lacepède, 1800) and Tiger-toothed Croaker (Otolithes ruber Bloch and Schneider, 1801) from Persian Gulf, Iran in 2001 and 2011 5 (1):322–329
Khaled AJC, Ecology (2009) Trace metals in fish of economic interest from the west of Alexandria, Egypt. 25 (4):229–246
Al-Atoom AA, Farah H, El-Badrawy E Some heavy metals levels in tissues of some fish species consumed in Taif region
Prudente M, Kim E-Y, Tanabe S, Tatsukawa RJMPB (1997) Metal levels in some commercial fish species from Manila Bay, the Philippines 34 (8):671–674
Nauen CE (1983) Compilation of legal limits for hazardous substances in fish and fishery products. FAO Fisheries Circular (FAO) no 764
FAO (2017) World Health Organization. WHO Expert Committee on Food Additives. Evaluation of certain food additives: eighty-fourth report of the Joint FAO. World Health Organization,
Tabinda A, Hussain M, Ahmed I, Yasar AJPJZ (2010) Accumulation of toxic and essential trace metals in fish and prawns from Keti Bunder Thatta District. Sindh. 42(5)
Masoud MS, El-Samra MI, El-Sadawy MMJC, Ecology (2007) Heavy-metal distribution and risk assessment of sediment and fish from El-Mex Bay, Alexandria, Egypt. 23 (3):201–216
Nabawi A, Heinzow B, Kruse HJBECT (1987) As, Cd, Cu, Pb, Hg, and Zn in fish from the Alexandria region. Egypt. 39(5)
El-Moselhy KM, Othman A, Abd El-Azem H, El-Metwally MJEjob, sciences a (2014) Bioaccumulation of heavy metals in some tissues of fish in the Red Sea, Egypt 1 (2):97–105
Fakhri Y, Djahed B, Toolabi A, Raoofi A, Gholizadeh A, Eslami H, Taghavi M, Mr A, Mousavi Khaneghah A (2020) Potentially toxic elements (PTEs) in fillet tissue of common carp (Cyprinus carpio): a systematic review, meta-analysis and risk assessment study. Toxin Rev:1–13
Okyere H, Voegborlo RB, Agorku SE (2015) Human exposure to mercury, lead and cadmium through consumption of canned mackerel, tuna, pilchard and sardine. Food Chem 179:331–335. https://doi.org/10.1016/j.foodchem.2015.01.038
Lavilla I, Vilas P, Bendicho C (2008) Fast determination of arsenic, selenium, nickel and vanadium in fish and shellfish by electrothermal atomic absorption spectrometry following ultrasound-assisted extraction. Food Chem 106(1):403–409. https://doi.org/10.1016/j.foodchem.2007.05.072
Leung HM, Leung AOW, Wang HS, Ma KK, Liang Y, Ho KC, Cheung KC, Tohidi F, Yung KKL (2014) Assessment of heavy metals/metalloid (As, Pb, Cd, Ni, Zn, Cr, Cu, Mn) concentrations in edible fish species tissue in the Pearl River Delta (PRD), China. Mar Pollut Bull 78(1):235–245. https://doi.org/10.1016/j.marpolbul.2013.10.028
Parida S, Barik SK, Mohanty B, Muduli PR, Mohanty SK, Samanta S, Pattanaik AK (2017) Trace metal concentrations in euryhaline fish species from Chilika lagoon: human health risk assessment. Int J Environ Sci Technol 14(12):2649–2660. https://doi.org/10.1007/s13762-017-1334-y
WHO (1989) World Health Organization. Lead: environmental aspects-environmental health criteria 85
Tiimub BM, Afua MADJAijob (2013) Determination of selected heavy metals and iron concentration in two common fish species in Densu River at Weija District in Grater Accra region of Ghana 1 (1):45–55
Biswas S, Prabhu RK, Hussain KJ, Selvanayagam M, Satpathy KK (2012) Heavy metals concentration in edible fishes from coastal region of Kalpakkam, southeastern part of India. Environ Monit Assess 184(8):5097–5104
Pereira L, Ribas J, Vicari T, Silva S, Stival J, Baldan A, Domingos FV, Grassi M, Cestari M, de Assis HSJE, safety e (2016) Effects of ecologically relevant concentrations of cadmium in a freshwater fish 130:29–36
Chen J, Xu Y, Han Q, Yao Y, Xing H, Teng X (2019) Immunosuppression, oxidative stress, and glycometabolism disorder caused by cadmium in common carp (Cyprinus carpio L.): application of transcriptome analysis in risk assessment of environmental contaminant cadmium. J Hazard Mater 366:386–394. https://doi.org/10.1016/j.jhazmat.2018.12.014
Tepanosyan G, Maghakyan N, Sahakyan L, Saghatelyan A (2017) Heavy metals pollution levels and children health risk assessment of Yerevan kindergartens soils. Ecotoxicol Environ Saf 142:257–265. https://doi.org/10.1016/j.ecoenv.2017.04.013
Najm M, Shokrzadeh M, Fakhar M, Sharif M, Hosseini SM, RahimiEsboei B, Habibi FJJoMUoMS (2014) Concentration of heavy metals (Cd, Cr and Pb) in the tissues of Clupeonella cultriventris and Gasterosteus aculeatus from Babolsar coastal waters of Mazandaran Province, Caspian Sea 24 (113):185–192
Gholizadeh A, Salmani MH, Ebrahimi AA, Hosseini SS, Ehrampoush MH, Miri M, Nikoonahad A, Pasalari H (2018) Improved power density and Cr/Pb removal using ozone in a microbial desalination cell. Environ Chem Lett 16(4):1477–1485. https://doi.org/10.1007/s10311-018-0760-5
Organization WH (1989) Heavy metals environmental aspects. Environ Health Criteria 85
Velma V, Vutukuru SS, Tchounwou PB (2009) Ecotoxicology of hexavalent chromium in freshwater fish: a critical review. Rev Environ Health 24(2):129–145. https://doi.org/10.1515/reveh.2009.24.2.129
Shah N, Khisroon M, Shah SSA (2020) Assessment of copper, chromium, and lead toxicity in fish (Ctenopharyngodon idella Valenciennes, 1844) through hematological biomarkers. Environ Sci Pollut Res 27:33259–33269. https://doi.org/10.1007/s11356-020-09598-z
Lescord GL, Johnston TA, Heerschap MJ, Keller W, Southee FM, O’Connor CM, Dyer RD, Branfireun BA, Gunn JM (2020) Arsenic, chromium, and other elements of concern in fish from remote boreal lakes and rivers: drivers of variation and implications for subsistence consumption. Environ Pollut 259:113878. https://doi.org/10.1016/j.envpol.2019.113878
Alimentarius C (2015) General standard for contaminants and toxins in food and feed (codex stan 193-1995)
Yi Y-J, Zhang S-HJES, Research P (2012) Heavy metal (Cd, Cr, Cu, Hg, Pb, Zn) concentrations in seven fish species in relation to fish size and location along the Yangtze River 19 (9):3989–3996
Agah H, Leermakers M, Elskens M, Fatemi SMR, Baeyens W (2008) Accumulation of trace metals in the muscle and liver tissues of five fish species from the Persian Gulf. Environ Monit Assess 157(1):499–514. https://doi.org/10.1007/s10661-008-0551-8
Olmedo P, Hernández AF, Pla A, Femia P, Navas-Acien A, Gil F (2013) Determination of essential elements (copper, manganese, selenium and zinc) in fish and shellfish samples. Risk and nutritional assessment and mercury–selenium balance. Food Chem Toxicol 62:299–307. https://doi.org/10.1016/j.fct.2013.08.076
Goldhaber SB (2003) Trace element risk assessment: essentiality vs. toxicity. Regul Toxicol Pharmacol 38(2):232–242. https://doi.org/10.1016/S0273-2300(02)00020-X
Perl DP, Olanow CWJJoN, Neurology E (2007) The neuropathology of manganese-induced Parkinsonism. 66 (8):675–682
Liu M, Xu Y, Nawab J, Rahman Z, Khan S, Idress M, Ud din Z, Ali A, Ahmad R, Khan SA, Khan A, Khan MQ, Tang Y-T, Li G (2020) Contamination features, geo-accumulation, enrichments and human health risks of toxic heavy metal(loids) from fish consumption collected along Swat river, Pakistan. Environ Technol Innov 17:100554. https://doi.org/10.1016/j.eti.2019.100554
Yi Y, Yang Z, Zhang S (2011) Ecological risk assessment of heavy metals in sediment and human health risk assessment of heavy metals in fishes in the middle and lower reaches of the Yangtze River basin. Environ Pollut 159(10):2575–2585. https://doi.org/10.1016/j.envpol.2011.06.011
Bervoets L, Blust R, Verheyen R (2001) Accumulation of metals in the tissues of three spined stickelback (Gasterosteus aculeatus) from natural fresh waters. Ecotoxicol Environ Saf 48(2):117–127. https://doi.org/10.1006/eesa.2000.2010
Tuzen M (2009) Toxic and essential trace elemental contents in fish species from the Black Sea, Turkey. Food Chem Toxicol 47(8):1785–1790. https://doi.org/10.1016/j.fct.2009.04.029
Australia New Zealand Food Authority (1998) Food Standards Code. Standard A12 issue 37
Shah AQ, Kazi TG, Arain MB, Jamali MK, Afridi HI, Jalbani N, Baig JA, Kandhro GA (2009) Accumulation of arsenic in different fresh water fish species – potential contribution to high arsenic intakes. Food Chem 112(2):520–524. https://doi.org/10.1016/j.foodchem.2008.05.095
Cui D, Zhang P, Li H, Zhang Z, Luo W, Yang Z (2020) Biotransformation of dietary inorganic arsenic in a freshwater fish Carassius auratus and the unique association between arsenic dimethylation and oxidative damage. J Hazard Mater 391:122153. https://doi.org/10.1016/j.jhazmat.2020.122153
Salehi H, Ebrahimi AA, Ehrampoush MH, Salmani MH, Fard RF, Jalili M, Gholizadeh A (2020) Integration of photo-oxidation based on UV/Persulfate and adsorption processes for arsenic removal from aqueous solutions. Groundw Sustain Dev 10:100338. https://doi.org/10.1016/j.gsd.2020.100338
Pilehvarian AA, Malekirad AA, Bolandnazar N-S, Rezaei M (2015) Heavy metal bioaccumulation in different fish species in the coast of the Persian Gulf, Iran. Toxin Rev 34(4):215–219
Jinadasa BKKK, Fowler SW (2019) Critical review of mercury contamination in Sri Lankan fish and aquatic products. Mar Pollut Bull 149:110526. https://doi.org/10.1016/j.marpolbul.2019.110526
Radomyski A, Lei K, Giubilato E, Critto A, Lin C, Marcomini AJMpb (2018) Bioaccumulation of trace metals in aquatic food web. A case study, Liaodong Bay, NE China 137:555–565
Driscoll CT, Mason RP, Chan HM, Jacob DJ, Pirrone NJEs, technology (2013) Mercury as a global pollutant: sources, pathways, and effects. 47 (10):4967–4983
EPA (2012) Quantitative Risk Assessment Calculations. Sustainable Futures / P2 Framework Manual 2012 EPA-748-B12–001 13. Quantitative Risk Assessment Calculations 13.:1–11
Gholami Z, Abtahi M, Golbini M, Parseh I, Alinejad A, Avazpour M, Moradi S, Fakhri Y, Mousavi Khaneghah A (2019) The concentration and probabilistic health risk assessment of nitrate in Iranian drinking water: a case study of Ilam city. Toxin Rev:1–10
Pirsaheb M, Fakhri Y, Karami M, Akbarzadeh R, Safaei Z, Fatahi N, Sillanpää M, Asadi A (2019) Measurement of permethrin, deltamethrin and malathion pesticide residues in the wheat flour and breads and probabilistic health risk assessment: a case study in Kermanshah, Iran. International Journal of Environmental Analytical Chemistry:1–12
Rezaei H, Jafari A, Kamarehie B, Fakhri Y, Ghaderpoury A, Karami MA, Ghaderpoori M, Shams M, Bidarpoor F, Salimi M (2019) Health-risk assessment related to the fluoride, nitrate, and nitrite in the drinking water in the Sanandaj, Kurdistan County, Iran. Hum Ecol Risk Assess 25(5):1242–1250
Atamaleki A, Yazdanbakhsh A, Fakhri Y, Mahdipour F, Khodakarim S, Mousavi Khaneghah A (2019) The concentration of potentially toxic elements (PTEs) in the onion and tomato irrigated by wastewater: a systematic review; meta-analysis and health risk assessment. Food Res Int 125:108518. https://doi.org/10.1016/j.foodres.2019.108518
Fakhri Y, Rahmani J, Oliveira CAF, Franco LT, Corassin CH, Saba S, Rafique J, Khaneghah AM (2019) Aflatoxin M1 in human breast milk: a global systematic review, meta-analysis, and risk assessment study (Monte Carlo simulation). Trends Food Sci Technol 88:333–342. https://doi.org/10.1016/j.tifs.2019.03.013
Fakhri Y, Saha N, Miri A, Baghaei, Mehdi, Roomiani L, Ghaderpoori M, Taghavi M, Keramati H, Bahmani Z (2018) Metal concentrations in fillet and gill of parrotfish (Scarus ghobban) from the Persian Gulf and implications for human health. Food Chem Toxicol 115(2018):348–354
Rahmani J, Fakhri Y, Shahsavani A, Bahmani Z, Urbina MA, Chirumbolo S, Keramati H, Moradi B, Bay A, Bjørklund G (2018) A systematic review and meta-analysis of metal concentrations in canned tuna fish in Iran and human health risk assessment. Food Chem Toxicol 118(2018):753–765
Razzaghi N, Ziarati P, Rastegar H, Shoeibi S, Amirahmadi M, Conti GO, Ferrante M, Fakhri Y, Khaneghah AM (2018) The concentration and probabilistic health risk assessment of pesticide residues in commercially available olive oils in Iran. Food Chem Toxicol 120:32–40
Fakhri Y, Sahab N, Ghanbari, Sahebe, Rasouli M, Miri A, Avazpour, Moayed, Rahimizadeh A, Riahih S-M, Ghaderpoori M, Keramati H, Moradi B, Amanidaz N (2018) Carcinogenic and non-carcinogenic health risks of metal (oid) s in tap water from Ilam city, Iran. Food Chem Toxicol 118(2018):204–211
Heshmati A, Ghadimi S, Mousavi Khaneghah A, Barba FJ, Lorenzo JM, Nazemi F, Fakhri Y (2018) Risk assessment of benzene in food samples of Iran's market. Food Chem Toxicol 114(2018):278–284
Rahmani J, Alipour S, Miri A, Fakhri Y, Seyed-Mohammad R, Hassan K, Moradi M, Nazak A, Rokhsane HP, Zohreh B (2018) The prevalence of aflatoxin M1 in milk of Middle East region: A systematic review, meta-analysis and probabilistic health risk assessment. Food Chem Toxicol 118(2018):653–666
Yousefi M, Shemshadi G, Khorshidian N, Ghasemzadeh-Mohammadi V, Fakhri Y, Hosseini H (2018) Mousavi Khaneghah A (2018) Polycyclic aromatic hydrocarbons (PAHs) content of edible vegetable oils in Iran: a risk assessment study. Food Chem Toxicol 118:480–489
Fakhri Y, Mohseni-Bandpei A, Oliveri Conti G, Keramati H, Zandsalimi Y, Amanidaz N, Hosseini Pouya R, Moradi B, Bahmani Z, Rasouli Amirhajeloo L (2017) Health risk assessment induced by chloroform content of the drinking water in Iran: systematic review. Toxin Rev 36(4):322–330
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 38 kb)
Rights and permissions
About this article
Cite this article
Thai, V.N., Dehbandi, R., Fakhri, Y. et al. Potentially Toxic Elements (PTEs) in the Fillet of Narrow-Barred Spanish Mackerel (Scomberomorus commerson): a Global Systematic Review, Meta-analysis and Risk Assessment. Biol Trace Elem Res 199, 3497–3509 (2021). https://doi.org/10.1007/s12011-020-02476-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-020-02476-2