Abstract
This study aims to review the epidemiological studies on the association between cadmium (Cd) exposure during pregnancy and neonatal anthropometric measures. Electronic search of PubMed, Scopus, Web of Science, and Cochrane Collaboration was conducted till end of 2018. Pooled estimates were performed using a fixed-effects model or random-effects model. A total of 22 studies included in the meta-analyses. Subgroup analyses on sample type (maternal urine, maternal blood, cord blood, and placenta), meta-regression, and sensitivity analysis were performed to seek the sources of heterogeneity. In the random-effects meta-analysis of included studies, the pooled correlation coefficient between maternal exposure to Cd with birth weight was − 0.04 [95% CI (− 0.07, − 0.01), with birth length and head circumference as − 0.01 [95% CI (− 0.04, 0.02)] and − 0.02 [95% CI (− 0.06, 0.02)]. Our findings showed significant relationship between Cd exposure of pregnant women with low birth weight, but not with birth length and head circumference.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cadmium (Cd) is a type of heavy metal, which is naturally found in soil and is also produced as a result of modern industrial processes. Based on the list of hazardous substances by the US-Environment Protection Agency (US-EPA), it appears as one of the 126 priority contaminants; moreover, the International Agency for Research on Cancer considered it as a human carcinogen [1]. Human exposure to minerals like the Cd occurs through drinking water, food such as cereals, seafood, vegetables, and polluted air and inhalation of tobacco smoke [2, 3]. Due to its long half-life (> 20 years), after ingestion, it can be accumulated in various organs especially in kidneys. In adults, long-term exposure to Cd would lead to undesirable effects on the kidneys and bones, as well as an increased risk of cancer and mortality [2, 4]. Cd also can be considered as an endocrine disruptor compound and thus may affect reproduction and child development [5] and is associated with cardiometabolic risk factors and liver enzymes in adolescents [6]. As among various groups of population, pregnant women and their fetus are at increased risk for adverse effects of environmental contaminants [7], and in recent decades, various epidemiological studies have been conducted to assess the relationship between contaminants and low birth weight and preterm birth [8,9,10]. Meanwhile, several studies have reported significant relationships of Cd exposure with pregnancy and fetal outcomes [11,12,13].
Adverse birth outcomes, including stillbirth, spontaneous abortion, small-for-gestational age (SGA), and low birth size, are significant determinants of infant health and survival. The length of gestation and the size of the fetus are considered as a general factor for evaluating the fetal growth [9]. Typically, the birth weight of less than 2500 g was defined as low birth weight, which may be due to in utero growth retardation, early delivery, and small for gestational age and older maternal age [9]. The infant mortality rate of low-birth-weight infants is approximately 24 times higher than that of normal weight newborns [14]. Similarly, delivery before the full 32 weeks of gestation increases the neonatal mortality rate by 74 times [14].
One of the various factors affecting birth weight is exposure to common environmental pollutants as carbon monoxide, Cd, and other heavy metals [9, 15]. Epidemiological studies have found controversial results on the relationship between maternal exposure to Cd and birth weight and birth length [16,17,18,19,20]. To the best of our knowledge, no recent systematic review or meta-analysis has considered this issue. Therefore, the aims of the present study are to systematically review the most recent epidemiological studies on the association between maternal Cd exposure and neonatal anthropometric measures.
Materials and Methods
The systematic reviews of the observational studies were conducted based on PRISMA guidelines (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) [21].
Eligibility Criteria
Studies were selected based on the following criteria:
- 1.
Participants: women during term pregnancy (however having no chronic disease or long-term medication use) and their infants
- 2.
Outcome: studies examining the association between Cd exposure of pregnant women and adverse birth outcome
- 3.
Study design: studies with cohort, cross-sectional and case–control design
- 4.
Language: studies published in English
Search Strategy
PubMed, Scopus, Web of Science, and Cochrane Collaboration search were conducted to identify related studies published up to end of 2018. The following keywords, as well as MESH terms (Medical Subject Heading) in PubMed, were used to find related articles: “birth weight,” “pregnancy outcome,” and “birth outcome.” These terms are combined with the Boolean operator “OR.” The keyword “Cadmium” was added and combined with the former using Boolean operator “AND.” Finaly, the search was restricted to human studies and English and the final list was checked for further studies.
Study Selection and Data Extraction
All the considered publications were screened for relevance by two independent reviewers, and any disagreement on the title and abstract of studies was dissolved by discussion. The full texts of relevant studies were checked based on inclusion criteria by researchers. The final list of eligible studies was prepared after a consensus between the two researchers. The extracted data from all eligible studies were as follows: year of publication, location, first author’s last name, study design, sample size, and method of Cd exposure assessment, outcome definition, and results expressed as the correlation coefficient (r) or β-coefficient and 95% confidence intervals (CIs), as well as variables used in the adjustment. Qualitative evaluation of individual articles was independently assessed by two researchers and the scoring system based on the STROBE guideline was applied [22]. The objective of the study, the study design, the selection of participants, variables, data sources and measurements, statistical methods, results in data and main results and study limitation were evaluated. Each of 22 questions in the check list were scored as 0 or 1. The correlation between scores obtained from two researchers was 0.70. Means of obtained scores for each study are presented in Table 1. Based on these mean scores, the quality of studies was categorized into three groups of high (mean score 16.5–22), moderate (mean score 12–16), and low quality (mean score < 12). Overall, 68.2% of studies had high quality and other studies were of moderate quality.
Statistical Analysis
The r (correlation) of studies was used. In the case of missed r values, reported β-coefficient, RR, and OR, values were converted to r. Fisher z-transformation of the r is applied for pooled analysis. The potential heterogeneity across studies was evaluated using the Cochran’s Q-test and expressed using the I2 index. The pooled results for Fisher z-transformation were calculated by the fixed-effects model (for I2 < 50%) or the random-effects model (for I2 > 50%). Publication bias was evaluated by the Egger’s and Begg’s tests. Subgroup analyses, meta-regression, and sensitivity analysis were performed to seek the sources of heterogeneity. The sensitivity analyses were performed based on sequential algorithm [23]. All statistical analyses were conducted using STATA software (ver12.0, STATA Corp, College Station, Texas, USA).
Results
Study Selection
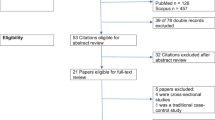
A total of 394 studies were found after the initial search. Of these, 274 studies were excluded after reviewing the title and abstracts (the duplicate studies, non-relevant studies, and studies not compatible with our inclusion criteria such as the animal or in-vitro studies, other heavy metals). Twenty-five studies were identified and reviewed; finally, 22 studies were included in the systematic review and meta-analysis (Fig. 1). Of the included studies, 22 had assessed birth weight, 11 had evaluated the birth length, and 8 had assessed the head circumference at birth.
Study Design and Population
This systematic review includes ten cross-sectional, seven cohorts, and five case–control studies, conducted in ten different countries in four continents of Asia, America, Africa, and Europe. Asian studies were as follows: one in Bangladesh [11], seven in China [24,25,26,27,28,29,30], two in Japan [17, 19], and one in Taiwan [12]. European studies were conducted in the following four countries: one in Spain [31], one in Poland [32], one in Finland [33], and one in Italy [34], respectively. Seven other studies were related to the USA (5 study) [20, 35,36,37,38] and Africa (2 study) [39, 40]. Studies that included the birth length were one in USA, one in Bangladesh, one in Japan, one in South Africa, one in Taiwan, one in Nigeria, and five in China [11, 12, 19, 20, 24, 27,28,29,30, 39, 40].
Of these 11 studies, 8 of them had also assessed the head circumference (one in Bangladesh, one in Japan, one in South Africa, one in Taiwan and two in China, one in Nigeria, and one in the USA). The publication year of studies ranged from 1987 to 2017. Sample sizes ranged from 45 [34] to 5364 [29] representing a total of 11,788 participants for birth weight, 9998 for birth length, and 4143 participants for birth head circumference. The mean age of pregnant women in the selected studies ranged from 25 to 33 years. Eight (45%) studies have used mothers’ urine sample, and others (55%) have used maternal blood or cord blood samples to evaluate the relationship between cadmium exposure and neonatal anthropometric measures. The mean STORBE score and the main characteristics of each 22 studies included in the systematic review are shown in Table 1.
Association Between Cd and Birth Weight
The pooled analysis of Cd exposure was significantly associated with low birth weight (Fisher-z = − 0.07; 95% CI (− 0.11, − 0.02)) using the random-effects model. However, significant heterogeneity was detected for the meta-analysis of association between Cd exposure and birth weight (I2 = 76.3%, p < 0.001), Therefore, the subgroup analysis, meta-regression, and sensitivity analysis was used to explore the potential sources of heterogeneity.
Subgroup Analysis
Results of subgroup analysis based on resource of Cd exposure showed that urinary Cd was negatively associated with birth weight but it was not significant (Fisher-z = − 0.05; 95% CI (− 0.15, 0.04); I2 = 81.9%); the association between cord blood Cd and birth weight was not also significant (Fisher-z = −0.04; 95% CI (− 0.13, 0.06); I2 = 62.6%). Birth weight had significant negative association with maternal blood Cd (Fisher-z = − 0.12; 95% CI (− 0.20, − 0.03); I2 = 83.1%). The placental Cd was not associated with birth weight (Fisher-z = − 0.03; 95% CI (− 0.16, 0.11); I2 = 50.1%) (Fig. 2).
Meta-Regression
Meta-regression identified the mean gestational age as the main source of the heterogeneity and could explain 72.62% of the heterogeneity, but mean maternal age, mean maternal BMI, and sample size of studies were not significant (p > 0.05).
Sensitivity Analysis
In the sensitivity analysis based on sequential algorithm, a single study was excluded from the calculations each time. The study of Huang (2017) had largest decrease in I2 (I2 = 72.5%); therefore, this study was dropped out and then re-analysis reduced sets of studies and performed the same rule one step before. In this step, the study of Tawari (2013) (I2 = 58.7%) was omitted. The studies of Salpietro (2002) for maternal blood Cd and cord blood Cd with I2 = 49.6% and I2 = 37.6%, respectively, were dropped out in the next steps. Finally, with omitting these three studies, the pooled estimate was obtained Fisher-z = − 0.04; 95% CI (− 0.07, − 0.01) with I2 = 37.6% (Fig. 3).
Publication Bias
The p values for Begg’s test was 0.08 and for Egger’s test was 0.349 for all studies and for studies obtained from sensitivity analysis were 0.984 and 0.946, respectively; therefore, it revealed no obvious publication bias among these studies. Figure 4 shows the funnel plot of included all studies (a) and studies obtained from sensitivity analysis (b). The funnel plot for studies obtained from sensitivity analysis suggested stronger asymmetry.
Association Between Cd and Birth Length
The pooled analysis of Cd exposure was not significantly associated with the birth length (Fisher-z = − 0.03; 95% CI (− 0.07, 0.01)) using the random-effects model. The heterogeneity was not significant (I2 = 62.7%; p = 0.001). Figure 5 shows subgroup analysis based on resource of Cd exposure, and the pooled estimates were not significant in subgroups. Begg’s test and Egger’s test revealed publication bias among these studies (p = 0.015 and 0.042, respectively). Figure 4 c shows funnel plot of included studies. The trim-and-fill method for publication bias in meta-analysis did not change the pooled effect size. The univariate meta-regression indicated that none of the factors (including mean maternal age, mean maternal BMI, mean gestational age, and sample size of studies) contributed to the heterogeneity of meta-analysis (all p > 0.05). Result of sensitivity analysis showed with omitting study of Tawari (2013), the pooled estimate obtained − 0.01; 95% CI (−0.04, 0.02) with I2 = 41.1% and p = 0.054. Begg’s test and Egger’s test revealed no obvious publication bias among these studies (p = 0.089 and 0.125, respectively).
Association Between Cd and Birth Head Circumference
The pooled analysis of Cd exposure was not significantly associated with the birth length (Fisher-z = − 0.04; 95% CI (− 0.09, 0.01)) using the random-effects model. The heterogeneity was significant (I2 = 61.3%; p = 0.003). Figure 6 shows subgroup analysis based on resource of Cd exposure; the pooled estimates were not significant in subgroups. The univariate meta-regression indicated that none of the factors (including mean maternal age, mean maternal BMI, mean gestational age, and sample size of studies) contributed to the heterogeneity of meta-analysis (all p values > 0.05). Begg’s test and Egger’s test revealed no obvious publication bias among these studies. The p values for these tests were greater than 0.05 (p = 0.336 and 0.273, respectively). Figure 4 d shows the funnel plot of included studies. Results of the sensitivity analysis showed with dropping study of Zhang (2017): female, the pooled estimated obtained − 0.02; 95% CI (−0.06, 0.02) with I2 = 45.2% (p = 0.051).
Discussion
In this study, we systematically reviewed the current evidence of the association of maternal Cd exposure with neonatal anthropometric measures, i.e., birth weight, length, and head circumference. Our findings revealed significant relationship between Cd exposure of pregnant mother with low birth weight but not with birth length and head circumference.
In general, Cd has higher concentrations in the body of women than in men, and it is suggested that the accumulation of this compound in pregnant women is more than in non-pregnant women [29, 41]. During pregnancy, Cd accumulates in placenta, which can reduce utero-placental blood flow or affect the synthesis and metabolism of placental hormones [30, 42, 43]. On the other hand, the penetration of Cd to the placenta affects the transfer of nutrients to the fetus [39, 44, 45], which in turn may disrupt the fetal growth. Cd can also affect fetal growth through substitution with zinc ions. Zinc is one of the essential elements for the fundamental biological functions of the human body, and it also plays an important role in the growth process and reproductive and immune system [46]. Moreover, zinc can be effective in pregnancy outcome [40, 47], so the deficiency of this element during pregnancy has been associated with the delivery of low birth weight infants [40]. Cd and zinc have the same affinity on sulfur, nitrogen, and oxygen ligands due to the similarity of the electron configuration and valance states. Therefore, in many biological processes, Cd can replace Zn and disrupt their function [40, 48]. In recent years, interest in the study of the effect of cadmium toxicity on birth outcomes has increased. Several studies have reported significant relationship between prenatal Cd exposure and adverse birth outcome [11, 19, 49]. But in some studies, there has been no relationship [50, 51]. Because of differences in the results of various studies, for the first time, the current systematic review and meta-analyses were conducted on the relationship between maternal exposure to Cd and anthropometric measures. The findings of this meta-analysis indicated that exposure to Cd in pregnancy is associated with low birth weight. In spite of the difference between the types of samples (maternal urine, maternal blood, cord blood and placenta), in general, our study articles showed that the mother’s exposure to cadmium was associated with a reduction in birth weight. However, in some studies, different results have been reported. For instance, in the cohort study of Cheng et al. and the case–control study of Huang et al., which examined the maternal urine; there was positive relationship between maternal exposure and birth weight [25, 30]. These findings are consistent with the results of some studies that examined Cd in umbilical cord blood [12, 26, 33, 39]. Maternal blood is another biological sample that has been studied to assess the relationship between the Cd exposure of pregnant women and birth weight. There are controversial results regarding birth weight, while in 12 studies, it often confirms a reduction in birth weight. However, one-third of these studies reported a reverse relationship [20, 26, 29, 38]. Among studies that used the placenta sample, a cross-sectional study linked the levels of Cd in the placenta with reduction of birth weight [38], whereas some other studies did not confirm this association [31, 37].
Evidence of the association between Cd exposure of pregnant women and the decreased birth length is also inconsistent. A cross-sectional study of 209 pregnant women in China found a significant association between Cd exposure and reduction of birth length [27]. This finding was consistent with the results of a case–control study on 125 pregnant and 35 non-pregnant women conducted by Tawari [40]. Likewise, some other studies found a weak but non-significant association between Cd exposure and low birth length [12, 19, 39]. However, three cohort studies on pregnant women with an average age of between 26 and 28 have found a reverse association between Cd exposure and reduction in birth length [11, 24, 29]. Finally, despite the difference between results, the relationship between cadmium exposure of pregnant women and birth length was not significant. Furthermore, findings of this meta-analyses indicated that maternal exposure of Cd was not associated with birth head circumference. These findings are consistent with the results of some previous studies included in this systematic review [11, 12, 19, 20, 24, 39].
Strength and Limitations of the Current Review
Recently, research publications about the probability maternal exposure to heavy metals and adverse outcome are increasing rapidly. However, no systematic review and meta-analysis have ever been reported to investigate the association between maternal Cd exposure and anthropometric measures. The main strength of this study is its novelty and the applied findings that can be useful in preventing intrauterine growth retardation. The findings of this review also have some limitations. First, there are different types of samples for evaluation of Cd exposure (maternal urine, maternal blood, cord blood and placenta), which reduces the sample size in each group. The second limitation of this study was the evidence of hetrogenty in selected studies that performed random-effect model in the analyses. The source of heterogeneity was detected by subgroup analyses, meta-regression, and sensitivity analysis. Third, there was no adjustment of covariates for association between Cd exposure and birth size. The effect size of the included studies was corelation cofficient.
Conclusion
In conclusion, this systematic and meta-analyses revealed a weak but significant association between maternal exposure to Cd and birth weight. However, there was no relationship between cadmium levels during pregnancy and birth length and head circumference. Therefore, future research is highly encouraged to re-evaluate the impact of new data.
Refrences
Waisberg M, Black W, Waisberg C, Hale B (2004) The effect of pH, time and dietary source of cadmium on the bioaccessibility and adsorption of cadmium to/from lettuce (Lactuca sativa L. cv. Ostinata). FCT J 42:835–842. https://doi.org/10.1016/j.fct.2004.01.007
Järup L, Åkesson A (2009) Current status of cadmium as an environmental health problem. TAAP J 238:201–208. https://doi.org/10.1016/j.taap.2009.04.020
Alexander J, Benford D, Cockburn A, Cravedi JP, Dogliotti E et al (2009) Scientific opinion of the panel on contaminants in the food chain. EFSA J 980:1–139
Straif K, Benbrahim-Tallaa L, Baan R, Grosse Y, Secretan B, El Ghissassi F et al (2009) A review of human carcinogens—part C: metals, arsenic, dusts, and fibers. Lancet Oncol J 10:453–454. https://doi.org/10.1016/S1470-2045(09)70134-2
Henson MC, Chedrese PJ (2004) Endocrine disruption by cadmium, a common environmental toxicant with paradoxical effects on reproduction. Exp Biol Med (Maywood) 229:383–392. https://doi.org/10.1177/153537020422900506
Kelishadi R, Askarieh A, Motlagh ME, Tajadini M, Heshmat R, Ardalan G et al (2013) Association of blood cadmium level with cardiometabolic risk factors and liver enzymes in a nationally representative sample of adolescents: the CASPIAN-III study. J Environ Public Health. https://doi.org/10.1155/2013/142856
Sutton P, Giudice LC, Woodruff TJ (2010) Reproductive environmental health. Curr Opin Obstet Gynecol 22:517–524. https://doi.org/10.1097/GCO.0b013e3283404e59
Ferguson KK, O’Neill MS, Meeker JD (2013) Environmental contaminant exposures and preterm birth: a comprehensive review. J Toxicol Environ Health B 16:69–113. https://doi.org/10.1080/10937404.2013.775048
Windham G, Fenster L (2008) Environmental contaminants and pregnancy outcomes. Fertil Steril 89:e111–e116. https://doi.org/10.1016/j.fertnstert.2007.12.041
Diaz-Jimenez J, Arroyo V, Ortiz Burgos C, Carmona Alferez R, Linares Gil C (2016) Effect of environmental factors on low weight in non-premature births: a time series analysis. PLoS One. https://doi.org/10.1371/journal.pone.0164741
Kippler M, Tofail F, Gardner R, Rahman A, Hamadani JD, Bottai M et al (2011) Maternal cadmium exposure during pregnancy and size at birth: a prospective cohort study. Environ Health Perspect 120:284–289. https://doi.org/10.1289/ehp.1103711
Lin CM, Doyle P, Wang D, Hwang YH, Chen PC (2011) Does prenatal cadmium exposure affect fetal and child growth? Environ Int 68:641–646. https://doi.org/10.1136/oem.2010.059758
Kippler M, Wagatsuma Y, Rahman A, Nermell B, Persson LÅ, Raqib R et al (2012) Environmental exposure to arsenic and cadmium during pregnancy and fetal size: a longitudinal study in rural Bangladesh. Reprod Toxicol 34:504–511. https://doi.org/10.1016/j.reprotox.2012.08.002
MacDorman MF, Mathews T (2013) Infant deaths-United States, 2005–2008. MMWR Surveill Summ 62:171–175
Butler AS, Behrman RE (2007) Preterm birth: causes, consequences, and prevention. Institute of Medicine (US) Committee on Understanding Premature Birth and Assuring Healthy Outcomes
Llanos MN, Ronco AM (2009) Fetal growth restriction is related to placental levels of cadmium, lead and arsenic but not with antioxidant activities. Reprod Toxicol 27:88–92. https://doi.org/10.1016/j.reprotox.2008.11.057
Shirai S, Suzuki Y, Yoshinaga J, Mizumoto Y (2010) Maternal exposure to low-level heavy metals during pregnancy and birth size. J Environ Sci Health A 45:1468–1474. https://doi.org/10.1080/10934529.2010.500942
Zhang YL, Zhao YC, Wang JX, Zhu HD, Liu QF, Fan YG et al (2004) Effect of environmental exposure to cadmium on pregnancy outcome and fetal growth: a study on healthy pregnant women in China. J Environ Sci Health A 39:2507–2515. https://doi.org/10.1081/ESE-200026331
Nishijo M, Nakagawa H, Honda R, Tanebe K, Saito S, Teranishi H et al (2002) Effects of maternal exposure to cadmium on pregnancy outcome and breast milk. Occup Environ Med 59:394–397. https://doi.org/10.1136/oem.59.6.394
Bloom MS, Louis GMB, Sundaram R, Maisog JM, Steuerwald AJ, Parsons PJ (2015) Birth outcomes and background exposures to select elements, the Longitudinal Investigation of Fertility and the Environment (LIFE). Environ Res 138:118–129. https://doi.org/10.1016/j.envres.2015.01.008
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med 151:264–269. https://doi.org/10.7326/0003-4819-151-4-200908180-00135
Von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP (2007) The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Ann Intern Med 147:573–577. https://doi.org/10.7326/0003-4819-147-8-200710160-00010
Patsopoulos NA, Evangelou E, Ioannidis JP (2008) Sensitivity of between-study heterogeneity in meta-analysis: proposed metrics and empirical evaluation. Int J Epidemiol 37:1148–1157. https://doi.org/10.1093/ije/dyn065
Guo J, Wu C, Qi X, Jiang S, Liu Q, Zhang J et al (2017) Adverse associations between maternal and neonatal cadmium exposure and birth outcomes. Sci Total Environ 575:581–587. https://doi.org/10.1016/j.scitotenv.2016.09.016
Huang K, Li H, Zhang B, Zheng T, Li Y, Zhou A et al (2017) Prenatal cadmium exposure and preterm low birth weight in China. J Expo Sci Environ Epidemiol. https://doi.org/10.1038/jes.2016.41
Hu X, Zheng T, Cheng Y, Holford T, Lin S, Leaderer B et al (2015) Distributions of heavy metals in maternal and cord blood and the association with infant birth weight in China. J Reprod Med 60:21–29
Sun H, Chen W, Wang D, Jin Y, Chen X, Xu Y (2014) The effects of prenatal exposure to low-level cadmium, lead and selenium on birth outcomes. Chemosphere 108:33–39. https://doi.org/10.1016/j.chemosphere.2014.02.080
Zhang Y, Xu X, Chen A, Davuljigari CB, Zheng X, Kim SS et al (2018) Maternal urinary cadmium levels during pregnancy associated with risk of sex-dependent birth outcomes from an e-waste pollution site in China. Reprod Toxicol 75:49–55. https://doi.org/10.1016/j.reprotox.2017.11.003
Yang J, Huo W, Zhang B, Zheng T, Li Y, Pan X et al (2016) Maternal urinary cadmium concentrations in relation to preterm birth in the Healthy Baby Cohort Study in China. Environ Int 94:300–306. https://doi.org/10.1016/j.envint.2016.06.003
Cheng L, Zhang B, Zheng T, Hu J, Zhou A, Bassig B et al (2017) Critical windows of prenatal exposure to cadmium and size at birth. Int J Environ Res Public Health. https://doi.org/10.3390/ijerph14010058
Falcon M, Vinas P, Perez-Carceles M, Luna A (2003) Placental cadmium and lipid peroxidation in smoking women related to newborn anthropometric measurements. Arch Environ Contam Toxicol 45:278–282. https://doi.org/10.1007/s00244-003-0118-1
Bizoń A, Milnerowicz-Nabzdyk E, Zalewska M, Zimmer M, Milnerowicz H (2011) Changes in pro/antioxidant balance in smoking and non-smoking pregnant women with intrauterine growth restriction. Reprod Toxicol 32(3):360–367. https://doi.org/10.1016/j.reprotox.2011.08.007
Kantola M, Purkunen R, Kröger P, Tooming A, Juravskaja J, Pasanen M et al (2000) Accumulation of cadmium, zinc, and copper in maternal blood and developmental placental tissue: differences between Finland, Estonia, and St. Petersburg. Environ Res 83:54–66. https://doi.org/10.1006/enrs.1999.4043
Salpietro CD, Gangemi S, Minciullo PL, Briuglia S, Merlino MV, Stelitano A et al (2002) Cadmium concentration in maternal and cord blood and infant birth weight: a study on healthy non-smoking women. J Perinat Med 30:395–399. https://doi.org/10.1515/JPM.2002.061
Luo Y, McCullough LE, Tzeng J-Y, Darrah T, Vengosh A, Maguire RL et al (2017) Maternal blood cadmium, lead and arsenic levels, nutrient combinations, and offspring birthweight. BMC Public Health. https://doi.org/10.1186/s12889-017-4225-8
Vidal AC, Semenova V, Darrah T, Vengosh A, Huang Z, King K et al (2015) Maternal cadmium, iron and zinc levels, DNA methylation and birth weight. BMC Pharmacol Toxicol. https://doi.org/10.1186/s40360-015-0020-2
Loiacono NJ, Graziano JH, Kline JK, Popovac D, Ahmedi X, Gashi E et al (1992) Placental cadmium and birthweight in women living near a lead smelter. Arch Environ Health 47:250–255. https://doi.org/10.1080/00039896.1992.9938357
Kuhnert B, Kuhnert P, Debanne S, Williams T The relationship between cadmium zinc and birth weight in pregnant women who smoke. Am J Obstet Gynecol 157:1247–1251. https://doi.org/10.1016/S0002-9378(87)80303-4
Röllin HB, Kootbodien T, Channa K, Odland JØ (2015) Prenatal exposure to cadmium, placental permeability and birth outcomes in coastal populations of South Africa. PLoS One. https://doi.org/10.1371/journal.pone.0142455
Tawari E, Anetor J, Charles-davis M (2013) Cadmium levels in pregnancy, influence on neonatal birth weight and possible Amelioration by some essential trace elements. Toxicol Int 20:108–112. https://doi.org/10.4103/0971-6580.111558
Vilahur N, Vahter M, Broberg K (2015) The epigenetic effects of prenatal cadmium exposure. Curr Environ Health Rep 2:195–203. https://doi.org/10.1007/s40572-015-0049-9
Stasenko S, Bradford EM, Piasek M, Henson MC, Varnai VM, Jurasović J et al (2010) Metals in human placenta: focus on the effects of cadmium on steroid hormones and leptin. J Appl Toxicol 30:242–253. https://doi.org/10.1002/jat.1490
Chertok R, Kullgren B, Burbank D (1984) The effects of CdCl2 on the matemal-to-fetal clearance of 67Cu and placental blood flow. Proc Soc Exp Biol Med 176:138–142. https://doi.org/10.3181/00379727-176-41853
Kippler M, Hoque AW, Raqib R, Öhrvik H, Ekström EC, Vahter M (2010) Accumulation of cadmium in human placenta interacts with the transport of micronutrients to the fetus. Toxicol Lett 192:162–168. https://doi.org/10.1016/j.toxlet.2009.10.018
Kippler M, Lönnerdal B, Goessler W, Ekström EC, El Arifeen S, Vahter M (2009) Cadmium interacts with the transport of essential micronutrients in the mammary gland—a study in rural Bangladeshi women. Toxicology. https://doi.org/10.1016/j.tox.2008.12.009
Roohani N, Hurrell R, Kelishadi R, Schulin R (2013) Zinc and its importance for human health: an integrative review. J Res Med Sci 18:144–157
Singh P, Khushlani K, Veerwal P, Gupta R (1987) Maternal hypozincemia and low-birth-weight infants. Clin Chem 33:1950
Nieboer E, Richardson DH (1980) The replacement of the nondescript term ‘heavy metals’ by a biologically and chemically significant classification of metal ions. Environ Pollut (Series B) 1:3–26. https://doi.org/10.1016/0143-148X(80)90017-8
Johnston JE, Valentiner E, Maxson P, Miranda ML, Fry RC (2014) Maternal cadmium levels during pregnancy associated with lower birth weight in infants in a North Carolina cohort. PLoS One. https://doi.org/10.1371/journal.pone.0109661
Odland J, Nieboer E, Romanova N, Thomassen Y, Lund E (1999) Blood lead and cadmium and birth weight among sub-arctic and arctic populations of Norway and Russia. Acta Obstet Gynecol Scand 78:852–860. https://doi.org/10.1034/j.1600-0412.1999.781004.x
Osman K, Åkesson A, Berglund M, Bremme K, Schütz A, Ask K et al (2000) Toxic and essential elements in placentas of Swedish women. Clin Biochem 33:131–138. https://doi.org/10.1016/S0009-9120(00)00052-7
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khoshhali, M., Rafiei, N., Farajzadegan, Z. et al. Maternal Exposure to Cadmium and Fetal Growth: a Systematic Review and Meta-Analysis. Biol Trace Elem Res 195, 9–19 (2020). https://doi.org/10.1007/s12011-019-01819-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-019-01819-y