Abstract
A study was conducted to determine the effect of dietary selenium (Se) concentration and source for broiler chickens on performance, nutrient digestibility, plasma Se, glutathione peroxidase (GPx) activity, and thiobarbituric acid reactive substances (TBARS). A total of 700 1-day-old broiler chicks were assigned to 7 diets with 20 birds per cage and 5 replicates per treatment. The experimental diets were fed for 32 days in 2 phases (phase 1, day 0 to 14 and phase 2, day 15 to 32). Treatments were as follows: control (without Se supplementation), sodium selenite (SeS; 0.15, 0.30, or 0.45 ppm), and hot-melt extruded sodium selenite (SeHME; 0.15, 0.30, or 0.45 ppm). There were significant linear responses (P < 0.01) for higher plasma Se concentration in SeS and SeHME treatments. Moreover, an increased (P < 0.01) Se concentration of plasma occurred in SeHME treatment compared with that in SeS treatment. The serum GPx analyses revealed that supplemental SeS and SeHME increased significantly the activity of GPx in the plasma in phase 1 (P < 0.05) and phase 2 (P < 0.05). There were significant linear (P < 0.01) responses of SeS and SeHME treatments for the expression of SelW, GPx1, GPx3, and GPx4 in the livers and spleens. In addition, SeHME showed an upregulated expression of GPx-4 in the livers (P < 0.01) and SelW in the spleens (P < 0.05) compared with SeS treatment. SeHME showed a lower TBARS on day 9. Moreover, a decreased (P < 0.01) TBARS occurred in SeS treatment compared with that in control treatment. In conclusion, SeHME can increase antioxidant activity and Se absorption, consequently being a more suitable source of Se than regular sodium selenite.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Selenium (Se) is an essential trace mineral involved in the structural component of many antioxidant enzymes such as glutathione peroxidase (GPx) to protect tissues and cells from damage [1,2,3]. Se is widely used as a dietary supplement for poultry. The most common Se supplement that mainly has been used in poultry diets is the inorganic Se in the form of sodium selenite. However, in recent years, the supplementation of essential minerals at levels above the requirement to ensure the optimum growth has been an area of increasing concern over contamination of the environment through excess mineral excretion. Therefore, the interest is being increased to use alternative options such as organic or nano-supplements, due to their higher bioavailability [4, 5]. Nano-sized particles were shown to have improved bioavailability via higher specific surface area, activity, and catalytic efficiency [6,7,8]. Previous research has shown that nano-Se was more effective than sodium selenite at preventing oxidative damage and enhancing GPx expression in broiler chickens [5, 7, 9, 10] or laying hens [11]. Diet supplemented with 0.20 mg/kg of nano-Se could effectively increase the GPx activity of serum, hepatic GPx, and Se concentration of the livers in chickens [12]. However, the kind of nano-process also affects the bioavailability and efficacy of nano-particles. The new generation of nano-particles may have a higher potential of nano-particle dispersion in the final product.
The hot-melt extrusion (HME) technique is known as one of the top-down and environmentally friendly techniques that are widely used for nano-particle trace element production in the pharmaceutical industry [13]. In addition, the source of incorporated polymer matrix into the HME process may affect the uniformity of Se dispersion. The incorporation of copolymers as the binder of wet or dry granulation and the HME technique increase the solubility and dispersion of poorly water-soluble drugs [13]. This technique has, to our knowledge, not extensively been studied in animal models. The higher bioavailability of trace minerals may reduce environmental pollution through the reduction of excreted trace minerals. We hypothesized that Se nano-particles prepared by the HME process can improve the absorption of Se in poultry and thus be more efficient in comparison to traditional inorganic Se. The aim of the current study was to investigate the effects of nano-Se on growth performance, nutrient digestibility, antioxidant status, and thiobarbituric acid reactive substances (TBARS) on broiler chickens.
Materials and Methods
The Preparation of SeHME
Na2SeO3, Span 80, Tween 80, and polyethylene glycol 6000 were mixed at a 20:12:4:64 ratio prior to the feeding process. The processed mixture was moved to a feed hopper at a 45 g/min speed. The twin-screw system that worked with the hot-melt extruder (STS-25HS, Hankook E.M. Ltd., Pyeongtaek, Korea) connected to a round-shaped die (1-mm diameter) was used for the preparation of extruded materials [13]. The temperatures of barrel and die sections were maintained at 45 °C and 40 °C, respectively. The speed of the screw was 150 rpm during the HME process. By passing through conveying and kneading sections in the barrel, samples were extruded from the die section. Extruded substances were solidified and were milled using the HBL-3500S grinder (Samyang Electronics Co., Gunpo, Korea). Particle properties of Na2SeO3 nano-particles dispersed in distilled water (DW) were elucidated. The hydrodynamic size, polydispersity index, and zeta potential of Na2SeO3 nano-particles were measured by dynamic light scattering (DLS) and laser Doppler methods (ELS-Z1000; Otsuka Electronics, Tokyo, Japan). Powder of Na2SeO3 extrudate was dispersed in DW at 20 mg/ml and their particle properties were tested. The particle shape of Na2SeO3 extrudate was observed by transmission electron microscopy (TEM). The dispersion of Na2SeO3 nano-particles was loaded onto the copper grid with film and dried for 10 min. That sample was then observed by TEM (JEM 1010; JEOL, Tokyo, Japan). The final HME contained sodium selenite content of 20%.
Animals and Experimental Design
A total of 700 1-day-old broiler chicks (Ross 308) were randomly allotted with an average initial body weight (BW) of 44.5 ± 0.3 g on the basis of initial BW according to a completely randomized design (CRD) in the Research Center of Animal Life Sciences at Kangwon National University. There were 5 replicates in each treatment with 20 birds/replicate. The treatment included the following: control (without Se supplementation), inorganic (0.15 ppm, 0.30 ppm, and 0.45 ppm), and HME (0.15 ppm, 0.30 ppm, and 0.45 ppm). The analyzed values for the concentration of Se in the diets were presented in Table 1. The experimental diets were fed for 32 days in 2 phases (phase 1, day 0 to 14 and phase 2, day 15 to 32). The diets were formulated to provide all of the nutrients to meet or exceed the nutrient requirements listed in the Ross 308 nutrient specification [14] with the exception of Se (Table 2).
Blood, Slaughtering, and Tissue Sampling
At days 14 and 32, five birds per replicate were randomly selected and individually weighed for collection of blood. Blood samples were collected via the wing vein after 2-h feed withdrawal, and the ethylenedianminacetic acid (EDTA) tube and serum separate (SST) tube were used. Then, those samples were centrifuged (Union 32R, Bucheon, Korea) at 3000 rpm at 4 °C for 15 min to obtain the serum. The hemolysis-free serum was transferred immediately and frozen at − 20 °C until further biochemical analyses. At day 32 of the feeding trial, 2 birds per replicate were chosen as representative samples and were slaughtered to collect tissue samples. The livers and spleens samples were obtained and frozen directly in liquid nitrogen and stored at − 80 °C until analysis.
RNA Extraction and Real-Time PCR
Total RNA was isolated from the livers (50 mg) and spleens (100 mg) samples via RNAiso plus kit (Takara Biotechnology, Japan) according to the manufacturer’s instruction. The samples were resuspended in DEPC (diethyl pyrocarbonate)–treated water. The quality and quantity of RNA were evaluated through electrophoresis in 1.0% (w/v) formaldehyde denaturing agarose gel. The prepared RNA concentration was evaluated by Nanodrop 1000 (Thermo Fisher Scientific, USA) and OD260/OD280 ratio was approximately 1.8–2.0.
To reverse transcribe with refined RNA into the cDNA, the first strand cDNA synthesis kit (Takara Co. Ltd., Japan) was used following the manufacturer’s instructions. In this process, house-keeping gene, β-actin was introduced to adjust the quantity of input cDNA to maintain the role in internal control [15]. To determine mRNA levels, real-time PCR analysis was conducted with SYBR Premix EX Taq (Takara Co. Ltd., Japan) used in ABI 7500 Real-Time PCR System following the manufacturer’s instructions. The primer sequences (Table 3) for this real-time PCR analysis were prepared by Primer 5.0 software with chicken cDNA sequences from GenBank. A total of 20 μl reaction system included 10 μl SYBR Premix Ex Taq, 0.8 μl of forward and reverse primers (10 μM), 0.4 μl ROX Reference Dye II (50×), 2.0 μl cDNA template, and 6 μl dd H2O. Cycling conditions were as follows: 30 s at 95 °C, 40 cycles of denaturation step at 95 °C for 3 s, 60 °C annealing step for 34 s, and a 72 °C extension step for 15 s.
Thiobarbituric Acid Reactive Substances
The thigh muscles that were divided into 5 equal portions were pooled and minced with a conventional meat grinder (Omega TE22, Lima Food Machinery, Evesham, Worcestershire, UK). For the refrigerated storage experiment, the thigh muscles were packed in sealable plastic bags immediately and stored for 0, 3, 6, and 9 days at 4 °C. A pool of minced thigh muscle samples was submitted for analyses. To examine TBARS, the analytical method was performed according to Shinnhuber [16]. First, 0.5 g of meat (thigh) sample, 3 ml of 1% thiobarbituric acid (TBA) solution (0.02 mM), and 17 ml of 25% trichloroacetic acid (TCA) were blended. This prepared sample was heated in a boiling water bath (BW-20G, Jeio Tech, Korea) for 30 m and cooled in cold water for 10 m. Then, 5 ml of supernatant was collected and mixed with 3 ml chloroform and centrifuged at 3500 rpm in 30 m under room temperature. After this, optical density was operated in 532 nm by a spectrophotometer (UV-mini-1240, Shimadzu Corp., Japan) with samples. The final TBARS value was calculated in milligrams MA (Malonaldehyde) per 1 kg of sample.
The TBARS analysis used this formula as:
Growth Performance
The body weight (BW) and feed intake (FI) of individual birds per pen were recorded weekly, and weight gain (WG) and feed conversion ratio (FCR) were calculated as follows: FCR = total feed consumed by birds / total weight gain.
Statistical Analysis
Statistical analysis of the current experimental data was completed by using the GLM procedure of SAS (SAS Inst. Inc., Cary, NC) in a randomized complete block design. The linear and quadratic contrasts were used to compare the effects of increasing dietary Se source preparation levels (0 ppm, 0.15 ppm, 0.30 ppm, and 0.45 ppm). Orthogonal contrasts were done to compare the SeS treatments with the hot-melt extruded sodium selenite (SeHME) treatments. Pen replicate was the experimental unit for all measurements. Treatment means were separated by Turkey multiple range tests at P < 0.05 statistical level. The linear regression model with the following equation was used to compare the relative bioavailability (RBV) of Se from SeHME and SeS: y = b + c1x1 + c2x2, in which y is the response variable (serum SE and GPx activities), b is the intercept, c1 is the slope of SeS, x1 is the supplemental SeS intake, c2 is the slope of SeHME, and x2 is the supplemental SeHME. The RBV of SeHME as compared with SeS was calculated as the ratio of their linear slopes as described by [17].
Results
Plasma Profiles and Relative Bioavailability of Se
The effects of dietary Se concentration and source on plasma profiles and estimates of the RBV of Se are presented in Table 4. In phase 1, there were significant linear and quadratic responses (P < 0.01) for higher plasma Se concentration in SeS treatment. In addition, the plasma Se concentration increased linearly (P < 0.05) in supplemental SeHME. An increased Se concentration of plasma occurred in SeHME treatment that in SeS (P < 0.05). Compared with SeS (100%), the RBV of SeHME was 115% and 111% based on plasma Se in phases 1 and 2, respectively. The serum GPx activity analyses revealed that supplemental SeS and SeHME significantly increased the activity of GPx in the plasma in phase 1 (P < 0.05) and phase 2 (P < 0.01). Compared with the SeS group, the plasma GPx activity was increased (P < 0.01) with the supplementation of dietary SeHME in phase 1. However, only a tendency for higher (P = 0.06) plasma GPx was observed in SeHME treatment that in SeS in phase 2. Using GPx activity as the criterion, the RBV values of SeHME to SeS were 178 and 209 in phases 1 and 2, respectively. There was a linear increase (P < 0.01) in the white blood cells (WBC). Comparing the SeS and SeHME diets, the WBC increased (P < 0.05) significantly in diets with SeHME. No statistical significance was found for RBC. In phase 2, the plasma Se concentration increased (linear and quadratic, P < 0.01) as supplementation of Se increased in the SeS and SeHME diets. The supplemental SeHME significantly increased (P < 0.05) the plasma Se compared with SeS. However, there no significant difference was observed in the WBC and RBC.
Gene Expression of Selenoproteins
The effects of dietary Se concentration and sources on gene expression are presented in Table 5. There was a significant linear response of SeS and SeHME treatments (P < 0.01) for the expression of SelW, GPx1, GPx2, GPx3, and GPx4 in the livers. In addition, SeHME showed an upregulated (P < 0.01) expression of GPx-4 compared with SeS. In the spleens, the effect of the SeS dose approached significance (P < 0.05) towards the upregulation of the relative expression of SelW, GPx2, GPx3 (linear, P < 0.05), GPx1, and GPx4 (linear and quadratic, P < 0.05). In the spleens, increasing SeHME dose from 0 to 0.45 ppm upregulated the relative expression of SelW, GPx1, GPx3, and GPx4 (linear, P < 0.01) and showed a tendency for higher relative expression of GPx2 (quadratic, P = 0.08). There was a quadratic effect (P < 0.05) for SeHME to upregulate the relative expression of SelW. There was a significantly higher (P < 0.05) expression of SelW in SeHME treatment compared with that in SeS treatment.
TBARS Analysis
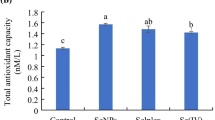
The effects of dietary Se concentration and sources on TBARS of chicken thigh meat are presented in Table 6. No statistical significance was found for TBARS until day 6. However, the SeHME chicks showed a lower (P < 0.01) TBARS on day 9. Moreover, a decreased (P < 0.01) TBARS occurred in SeS treatment compared with that in control treatment.
Growth Performance
The effects of dietary Se concentration and sources on growth performance are presented in Table 7. In phase 1, there was a linear improvement (P < 0.01) in weight gain and a tendency (P = 0.08) for higher feed intake as the dietary SeHME concentration increased. Comparing the SeS and SeHME diets, the relative growth performance was increased (P < 0.05) significantly in diets with SeHME. In phase 2 and also the overall period, Se supplementation had no effects on growth performance, feed intake, and FCR.
Discussion
One of the main focuses of this study was to compare the bioavailability of dietary Se in the forms of selenite and SeHME in broiler chickens. The linear increase in the Se concentration of serum explained that the increasing supplemented doses of Se sources were absorbed properly via the gastro-intestinal tract. The dietary Se level and the absorption rate were reflected in Se concentrations in the plasma [4, 5] or livers [9]. In serum, the Se concentration in SeHME was higher than in SeS. Previous studies have also shown that nano-particles had a higher absorption efficiency than common inorganic trace minerals in broiler chickens [8, 12]. The magnitude of variation between the plasma Se of broilers fed diets supplemented with Se was much less than the difference values in the GPx activity. Nano-size particles have been indicated to increase the efficiency of absorption owing to small particle size and high surface area.
Broilers receiving supplementation of SeHME increased WBC compared to those receiving the SeS at 14 days of age. Moreover, dietary Se supplementation resulted in a linearly higher WBC counts in chickens, and these significant changes may be associated with a predominant antioxidant role of Se and possibly higher blood cells lifespan [18]. The biological effect of Se on farm animals is usually related to selenoprotein production, which participates in the differentiation and proliferation of immunological cells [19]. Conversely, the supplementation of Se had no consistent effect on WBC in the second phase on day 32. Thus, long-term Se supplementation cannot affect WBC.
In addition to serum Se, immune and antioxidant indicators are routinely used to determine the long-term Se nutritional status of broiler chickens [5, 9, 20]. In this study, the plasma GPx was linearly increased in Se-supplemented diets. The results of this study were in agreement with the findings of Bakhshalinejad [20], who reported that the higher dose (0.4 vs 0.1 ppm) of dietary Se increased plasma GPx activity of broiler chickens at 42 days of age. The extent of plasma GPx activity modulation by a different Se source or level was in accordance with that of Hu [5] and Zhou and Wang [9], who observed an increased plasma GPx activity in Se-enrich diets. Se is a key trace mineral to control the antioxidant components by regulating the Se-based antioxidant enzymes and body glutathione pool [1]. The presence of significant differences in serum GPx content between chickens fed Se-supplemented and control diets is an expected result because of Se contents, and Se is strongly connected to the activity of GPx as a determinant antioxidant enzyme in tissues [7]. The linear increase in serum and organs in the GPx activity showed a great response of antioxidant status to dietary Se.
As we mentioned before, Se is a key mineral for optimum antioxidative activity and immune response [2, 21]. This experiment showed that supplementation of Se to the diet linearly increased the activity of selenoproteins in broilers. Selenoproteins can be produced by hydrogen selenide as a metabolite derived from sodium selenite through reduction by thiols [3]. GPx is the primary defense against oxidative metabolites, with an integral role to modulate free radicals [19, 22]. Glutathione is collectively beneficial for intracellular non-enzymatic activity, with the most important role as the main component of endogenous cellular redox buffers [3, 18, 22]. In addition, previous studies reported that the relationship between GPx activity and Se supply is associated with the form of Se as organic and inorganic [7]. Our findings on the linear increase in GPx activity can be supported by the research of Zhou and Wang [9] who indicated an increase in GPx activity in blood and livers of broiler chickens fed increasing levels of Se. Similar results have been reported in rats [23] and red deer [24]. Moreover, supplementation of Se into the diet increased GPx activity in different organs [5, 18, 25]. We did not compare the organic and inorganic sources in this study, but the results showed that GPx activity in the livers and spleens were almost similar between broilers fed inorganic sources as SeS or SeHME, with an exception of the livers GPx4 and spleens SelW that was significantly higher in broilers fed SeHME. It is well established that the antioxidant status is directly proportional to the body Se pool. The protective role of GPx4 on the reversing oxidation of lipid peroxides, particularly in arachidonic acid and linoleic acid, separates it from other GPx family members [26]. We could not rule out that the source of Se affects antioxidant enzyme ratio due to mostly insignificant differences between SeHME and SeS groups. However, according to the data obtained in the present work, the antioxidant status of broiler chickens was linearly improved by increasing Se supplementation to the diet while SeHME was superior to SeS source of Se in increasing the livers GPx4 and spleens SelW. These results indicated that Se consumption exerts antioxidant properties.
To evaluate the influences of Se on antioxidant status of broilers, the levels of TBARS as a key marker of lipid peroxidation were investigated. Oxygen radical formation has been reported to damage cellular compartments by adverse effects on DNA, RNA, and lipids [19, 21, 27]. The hydroxyl radical from accumulated H2O2 readily attaches to polyunsaturated fatty acids and increase the peroxidation of lipids that directly decrease meat quality [25]. The lower TBARS in SeHME treatment may be partially explained by the higher GPx4 activity. The higher activity of GPx can be considered as antioxidative compounds due to the prohibiting effects on hydroperoxide to prevent lipid oxidation in organs [21, 23, 27]. GPx4 plays a different physiological role in several ways compared to the other GPx. GPx4 extensively protects lipids from peroxidation, particularly involving in linoleic acid and arachidonic acid metabolism [28]. Therefore, in this study, Se seems to have a role in lipid oxidation by increasing GPx4. In animal cells, the principal enzymatic antioxidant system for hydrogen peroxide detoxification is associated with GPx activity [2, 23, 29]. The synthesis of selenoproteins is regulated by the concentration of Se in the diet, thus affecting the amount of absorbed Se; as a result, it positively contributes to Se homeostasis. More Se absorbed from SeHME could have greatly affected selenoprotein synthesis in the livers, contributing to increased hepatic selenoproteins mRNA expression. It seems that the differences between the sources of Se were directly correlated to its antioxidative enzyme content. Concomitantly with the increase of plasma Se with Se-supplemented diets, higher levels of GPx presumably improved the antioxidant status of breast meat.
It was noted that supplementation of Se is required for optimal growth [2, 3]. The present results showed a linear improvement of ADG by supplementation of SeHME source in phase 1, which was consistent with previous reports [9, 10]. However, the ADG was not significantly different between Se level and source in phase 2, indicating that SeS and SeHME did not add obvious growth advantage in broiler chickens, presumably because the Se requirement of broiler chickens is lower in older age. Moreover, the results in phase 1 suggest that SeHME can improve growth performance more effectively than SeS for meeting the dietary Se requirement of broiler chickens and can relatively reduce the amount of dietary Se additive. Supplementation of SeHME may efficiently increase the rate of intestinal absorption due to smaller particle size. Same as our study, nano-Se in broiler chickens [5] also showed a greater growth performance than with common inorganic salts due to increased Se absorption. Particle size is an effective parameter, as absorption efficiency enhances with the reduction of particle diameter [3, 6, 29]. Nano-particles are easier to absorb as small particle size (around 200 nm) of Se could be absorbed through the mucus barrier as an intact nano-particle [5, 30]. The relatively higher production cost of organic and nano-particles compared with inorganic sources has limited their application in broiler chickens’ diet. However, environmental issues must take into account regardless of the economic cost of making diets. It is crucial for the poultry industry to use highly absorbable dietary Se to decrease the environmental pollution.
Conclusion
The present results showed that dietary supplementation with SeHME increased plasma Se concentration, plasma GPx activity, and expression of GPx4 in the livers of broiler chickens. Further, the supplemented SeHME had a greater influence in decreasing thigh TBARS, probably via influencing the antioxidant status of broiler chickens.
References
Diplock AT (1992) Selenium, antioxidant nutritions, and human diseases. Biol Trace Elem Res 33:155–156. https://doi.org/10.1007/BF02784005
Suchy P, Strakova E, Herzig I (2014) Selenium in poultry nutrition: a review. Czech J Anim Sci 59:495–503. https://doi.org/10.17221/7730-CJAS
Skalickova S, Milosavljevic V, Cihalova K, Horky P, Richtera L, Adam V (2017) Selenium nanoparticles as a nutritional supplement. Nutr 33:83–90. https://doi.org/10.1016/j.nut.2016.05.001
Payne RL, Southern LL (2005) Comparison of inorganic and organic selenium sources for broilers. Poult Sci 84:898–902. https://doi.org/10.1093/ps/84.6.898
Hu CH, Li YL, Xiong L, Zhang HM, Song J, Xia MS (2012) Comparative effects of nano elemental selenium and sodium selenite on selenium retention in broiler chickens. Anim Feed Sci Technol 177:204–210. https://doi.org/10.1016/j.anifeedsci.2012.08.010
Thiry C, Schneider YJ, Pussemier L, De Temmerman L, Ruttens A (2013) Selenium bioaccessibility and bioavailability in Se-enriched food supplements. Biol Trace Elem Res 152:152–160. https://doi.org/10.1007/s12011-013-9604-0
Boostani A, Sadeghi AA, Mousavi SN, Chamani M, Kashan N (2015) Effects of organic, inorganic, and nano-Se on growth performance, antioxidant capacity, cellular and humoral immune responses in broiler chickens exposed to oxidative stress. Livest Sci 178:330–336. https://doi.org/10.1016/j.livsci.2015.05.004
Mohammadi V, Ghazanfari S, Mohammadi-Sangcheshmeh A, Nazaran MH (2015) Comparative effects of zinc-nano complexes, zinc-sulphate and zinc-methionine on performance in broiler chickens. Br Poult Sci 56:486–493. https://doi.org/10.1080/00071668.2015.1064093
Zhou X, Wang Y (2011) Influence of dietary nano elemental selenium on growth performance, tissue selenium distribution, meat quality, and glutathione peroxidase activity in Guangxi Yellow chicken. Poult Sci 90:680–686. https://doi.org/10.3382/ps.2010-00977
Ahmadi M, Ahmadian A, Seidavi AR (2018) Effect of different levels of nano-selenium on performance, blood parameters, immunity and carcass characteristics of broiler chickens. Poult Sci 6:9–108. https://doi.org/10.22069/PSJ.2018.13815.1276
Lin X, Yang T, Li H, Ji Y, Zhao Y, He J (2019) Interactions between different selenium compounds and essential trace elements involved in the antioxidant system of laying hens. Biol Trace Elem Res (Online published. https://doi.org/10.1007/s12011-019-01701-x
Wang Y (2009) Differential effects of sodium selenite and nano-Se on growth performance, tissue Se distribution, and glutathione peroxidase activity of avian broiler. Biol Trace Elem Res 128(2):184–190. https://doi.org/10.1007/s12011-008-8264-y
Lee SY, Nam S, Choi Y, Kim M, Koo JS, Chae BJ, Kang WS, Cho HJ (2017) Fabrication and characterizations of hot-melt extruded nanocomposites based on zinc sulfate monohydrate and soluplus. Appl Sci 7:902. https://doi.org/10.3390/app7090902
Aviagen (2014) Ross broiler management manual. http://pt.aviagen.com/assets/Tech_Center/Ross_Broiler/Ross_Broiler_Manual. 9:350–364
Jin X, Jia T, Liu R, Xu S (2018) The antagonistic effect of selenium on cadmium-induced apoptosis via PPAR-γ/PI3K/Akt pathway in chicken pancreas. J Hazard Mater 357:355–362. https://doi.org/10.1016/j.jhazmat.2018.06.003
Sinnhuber RO, Yu TC (1977) The 2-thiobarbituric acid reaction, an objective measure of the oxidative deterioration occurring in fats and oils. J Jap Soc Fish Sci 26:259–267. https://doi.org/10.5650/jos1956.26.259
Littell RC, Henry PR, Lewis AJ, Ammerman CB (1997) Estimation of relative bioavailability of nutrients using SAS procedures. J Anim Sci 75:2672–2683. https://doi.org/10.2527/1997.75102672x
Biller-Takahashi JD, Takahashi LS, Mingatto FE, Urbinati EC (2015) The immune system is limited by oxidative stress: dietary selenium promotes optimal antioxidative status and greatest immune defense in pacu Piaractus mesopotamicus. Fish Shellfish Immunol 47:360–367. https://doi.org/10.1016/j.fsi.2015.09.022
Pisoschi AM, Pop A (2015) The role of antioxidants in the chemistry of oxidative stress: a review. Eur J Med Chem 97:55–74. https://doi.org/10.1016/j.ejmech.2015.04.040
Bakhshalinejad R, Akbari Moghaddam Kakhki R, Zoidis E (2018) Effects of different dietary sources and levels of selenium supplements on growth performance, antioxidant status and immune parameters in Ross 308 broiler chickens. Br Poult Sci 59:81–91. https://doi.org/10.1080/00071668.2017.1380296
Gurkan F, Atamer Y, Ece A, Kocyigit Y, Tuzun H, Mete M (2004) Relationship among serum selenium levels, lipid peroxidation, and acute bronchiolitis in infancy. Biol Trace Elem Res 100:97–104. https://doi.org/10.1385/BTER:100:2:097
Sies H (1999) Glutathione and its role in cellular functions. Free Radic Biol Med 27:916–921. https://doi.org/10.1016/S0891-5849(99)00177-X
Markovic SD, Djačić DS, Cvetković DM, Obradović AD, Žižić JB, Ognjanović BI, Štajn AŠ (2011) Effects of acute in vivo cisplatin and selenium treatment on hematological and oxidative stress parameters in red blood cells of rats. Biol Trace Elem Res 142:660–670. https://doi.org/10.1007/s12011-010-8788-9
Pilarczyk B, Drozd R, Pilarczyk R, Tomza-Marciniak A, Jankowiak D, Hendzel D, Kuba J, Kowalska J (2011) Glutathione peroxidase (GSHPx) activity in the liver of red deer in relation to hepatic selenium concentrations, sex, body weight and season of the year. Biol Trace Elem Res 144:560–569. https://doi.org/10.1007/s12011-011-9022-0
Yao H, Zhao W, Zhao X, Fan R, Khoso PA, Zhang Z, Liu W, Xu S (2014) Selenium deficiency mainly influences the gene expressions of antioxidative selenoproteins in chicken muscles. Biol Trace Elem Res 161:318–327. https://doi.org/10.1007/s12011-014-0125-2
Reeves MA, Hoffmann PR (2009) The human selenoproteome: recent insights into functions and regulation. Cell Mol Life Sci 66:2457–2478. https://doi.org/10.1007/s00018-009-0032-4
Zia S, Islam F (2000) Selenium altered the levels of lipids, lipid peroxidation, and sulfhydryl groups in straitum and thalamus of rat. Biol Trace Elem Res 77:251–259. https://doi.org/10.1385/BTER:77:3:251
Chadio SE, Pappas AC, Papanastasatos A, Pantelia D, Dardamani A, Fegeros K, Zervas G (2015) Effects of high selenium and fat supplementation on growth performance and thyroid hormones concentration of broilers. J Trace Elem Med Biol 29:202–207. https://doi.org/10.1016/j.jtemb.2014.09.010
Habibian M, Sadeghi G, Ghazi S, Moeini MM (2015) Selenium as a feed supplement for heat-stressed poultry: a review. Biol Trace Elem Res 165:183–193. https://doi.org/10.1007/s12011-015-0317-4
Kachuee R, Abdi-Benemar H, Mansoori Y, Sánchez-Aparicio P, Seifdavati J, Elghandour MM, Guillén RJ, Salem AZ (2019) Effects of sodium selenite, L-selenomethionine, and selenium nanoparticles during late pregnancy on selenium, zinc, copper, and iron concentrations in Khalkhali goats and their kids. Biol Trace Elem Res 1–14. https://doi.org/10.1007/s12011-018-1618-1
Funding
This work was supported by Korea Institute of Planning and Evaluation for Technology in Food, Agriculture, Forestry and Fisheries (IPET) through Agri-Bio industry Technology Development Program, funded by Ministry of Agriculture, Food and Rural Affairs (MAFRA) (No.116073-3).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The protocol for the present experiment was approved by the Institutional Animal Care and Use Committee of Kangwon National University, Republic of Korea.
Conflict of Interest
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lee, J., Hosseindoust, A., Kim, M. et al. Biological Evaluation of Hot-Melt Extruded Nano-selenium and the Role of Selenium on the Expression Profiles of Selenium-Dependent Antioxidant Enzymes in Chickens. Biol Trace Elem Res 194, 536–544 (2020). https://doi.org/10.1007/s12011-019-01801-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-019-01801-8