Abstract
Methyl mercury (MeHg) is a developmental neurotoxin that causes irreversible cognitive damage in offspring of gestationally exposed mothers. Currently, no preventive drugs are established against MeHg developmental neurotoxicity. The neuroprotective effect of gestational administration of a flavanoid against in utero toxicity of MeHg is not explored much. Hence, the present study validated the effect of a bioactive flavanoid, fisetin, on MeHg developmental neurotoxicity outcomes in rat offspring at postnatal weaning age. Pregnant Wistar rats were simultaneously given MeHg (1.5 mg/kg b.w.) and two doses of fisetin (10 and 50 mg/kg b.w. in two separate groups) orally from gestational day (GD) 5 till parturition. Accordingly, after parturition, on postnatal day (PND) 24, weaning F1 generation rats were studied for motor and cognitive behavioural changes. Biochemical and histopathological changes were also studied in the cerebral cortex, cerebellum and hippocampus on PND 25. Administration of fisetin during pregnancy prevented behavioural impairment due to transplacental MeHg exposure in weaning rats. Fisetin decreased the levels of oxidative stress markers, increased enzymatic and non-enzymatic antioxidant levels and increased the activity of membrane-bound ATPases and cholinergic function in F1 generation rats. In light microscopic studies, fisetin treatment protected the specific offspring brain regions from significant morphological aberrations. Between the two doses of fisetin studied, 10 mg/kg b.w. was found to be more satisfactory and effective than 50 mg/kg b.w. The present study shows that intake of fisetin during pregnancy in rats ameliorated in utero MeHg exposure-induced neurotoxicity outcomes in postnatal weaning F1 generation rats.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Developmental methyl mercury (MeHg) exposure is a perilous environmental issue, progressing drastically across the planet as a critical threat to the coming generations [1]. The developing fetal brain exposed to MeHg, in utero, via maternal food habits is subjected to irreversible motor and cognitive damages, which is manifested as neurobehavioural complications after birth, in both humans and animals [2]. Mercury released from various natural and anthropogenic sources is bioaccumulated in fish, marine mammals and vegetables cultivated in contaminated sewage water as MeHg, and hence, it reaches the human system via the food chain [3, 4]. The immature developing central nervous system (CNS) is highly vulnerable to exogenous toxins even at very lower exposure levels when compared to the adult brain [5–7]. The F1 generation of mothers exposed to MeHg during gestation showed various neurological and cognitive impairments [8, 9].
The mechanism between MeHg and CNS developmental changes is not well defined till date, but many studies have shown involvement of oxidative imbalance in the brain of pups exposed to MeHg in utero [10–16]. Na+-K+ ATPase is involved in cellular metabolism and also in uptake, storage and metabolism of neurotransmitters [17–20]. Ca2+-ATPase equilibrizes the intracellular calcium levels. Mg2+-ATPase balances the intracellular Mg2+ levels and is involved in the protein synthesis and growth of neurons [21–23]. Our study investigated the effect of prenatal exposure to MeHg on membrane-bound ATPases in weaning rats. The cholinergic system, involving choline esterases, has an important role in maintaining cognition [24–26]. Studies have shown that AchE activity is inhibited by MeHg exposure which finally results in cholinergic hyperactivity and epileptic convulsions [27, 28].
Novel preventive strategies are needed against the MeHg toxicity elicited during the critical stages of brain development. The fetal environment is highly vulnerable to mild imbalance in homeostasis, and hence, preventive approaches with a natural product would be much safer with minimal side effects. There is a considerable body of evidence for the hypothesis that a plant-based diet can be effective against various metal-related toxicity disorders [29, 30]. Considering the limited data available on the neuroprotective role of gestational administration of flavanoids on MeHg developmental toxicity outcomes in the rat F1 generation, we have studied the effect of fisetin (3,7,3′,4′-tetrahydroxyflavone, Fig. 1) on transplacental MeHg neurotoxicity. Fisetin is commonly found in fruits and vegetables like onions, cucumbers, apples, persimmons and strawberries; nuts; wine, etc. Proven neuroprotective effects of fisetin are enhancement of long-term memory, antidepressive action, protection from ischemic reperfusion injury, reduction in behavioural deficits, reduction of lipid peroxide production, neurotrophic activity, activation of neurite outgrowth, antioxidant effects and induction of intracellular levels of glutathione [31, 32]. This wide range of beneficial effects made us investigate the effect of gestational administration of fisetin on in utero MeHg exposure-induced toxic outcomes like motor and cognitive impairment, altered antioxidant status, increased oxidative stress and declined activities of membrane-bound ATPases, impaired cholinergic function and cytoarchitectural aberrations in specific brain regions of postnatal F1 generation weaning rats.
Materials and Methods
Drug, Chemicals and Stains
Methyl mercury (II) chloride [CAS No: 115-09-03] and fisetin [CAS No: 345909-34-4] were purchased from Sigma-Aldrich Company, USA. All the chemicals and reagents used for the estimations were of AR grade.
Dosage Selection
The dosage of methyl mercury and fisetin was determined based on previous studies. We gave 1.5 mg/kg b.w of MeHg orally to the pregnant mother rats (groups II, III and IV) from gestational day (GD) 5 till parturition to create developmental neurotoxicity in the F1 generation. The level of the MeHg dosage was based on the literature showing that at this exposure level, the mercury concentration in newborn rats was comparable to that found in human infants born in populations with high dietary fish consumption [33]. The effective and safe dosages of fisetin were chosen based on previous literatures that are given below. As this is a pioneer study that explored the efficacy of gestational administration of fisetin, we used two different proven effective doses of fisetin (10 and 50 mg/kg b.w.) that authenticated its neuroprotective characteristics in many previous studies [34–36].
Animals
The study subjects were F1 generation weaning rat pups of pregnant Wistar rats (Rattus norvergicus). Female Albino rats of the Wistar strain of 12–14 weeks of age and 180–200 g weight obtained from the Central Animal House, Dr. ALMPGIBMS, University of Madras, Taramani campus, Chennai-113, Tamil Nadu, India, were used for the study. The animals were allowed to acclimatize to the laboratory conditions 7 days prior to the beginning of the experiment.
Breeding
After the acclimatization period (1 week), all the female rats in each group were allowed to mate with fertile male rats (1:2) overnight. The mating and conception was confirmed by observing live sperms in vaginal smears, and the day was designated as gestational day (GD) 0.
Caring
The rats were sheltered separately in polypropylene cages. A standard pellet diet purchased from Hindustan Lever Ltd and sterilized water were given ad libitum. All the individual cages were maintained at 23 ± 3 °C temperature, average humidity and hygienic conditions.
Ethical Approval
The experimental animals were handled according to the university and institutional ethical guidelines (IAEC No. 01/09/2015) after ethical committee approval was obtained.
Experimental Design and Grouping
The mother rats were partitioned into six groups (n = 6 rats in each group) and housed individually in independent cages with a proper label denoting the date of GD 0. The experimental period was from GD 5 (day after fetal implantation, to avoid chance of miscarriage/abortion) until delivery. All the mothers delivered after 21 days of gestation. The offspring were maintained in standard conditions till the weaning period, 21 days after birth. Based on the grouping of the mother rats, the offspring were allotted into six different groups (n = 6 pups in each group) for the present study.
-
Group I:
Normal pregnant rats administered with 0.1% DMSO (vehicle control) orally from GD 5 till parturition
-
Group II:
Pregnant rats administered with 1.5 mg/kg b.w. of MeHg (dissolved in distilled water) orally from GD 5 till parturition
-
Group III:
Pregnant rats administered with 1.5 mg/kg b.w. of MeHg and 10 mg/kg b.w. of fisetin (dissolved in 0.1% DMSO) orally from GD 5 till parturition
-
Group IV:
Pregnant rats administered with 1.5 mg/kg b.w. of MeHg and 50 mg/kg b.w. of fisetin (dissolved in 0.1% DMSO) orally from GD 5 till parturition
-
Group V:
Pregnant rats administered with 10 mg/kg b.w. of fisetin alone (dissolved in 0.1% DMSO) orally from GD 5 till parturition
-
Group VI:
Pregnant rats administered with 50 mg/kg b.w. of fisetin alone (dissolved in 0.1% DMSO) orally from GD 5 till parturition
Sampling
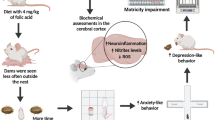
The day of parturition was counted as postnatal day 0 (PND 0). The pups were maintained in standard conditions with mother rats till weaning. Required training for the specific behavioural studies was given from PND 21 to PND 23, and the final evaluations of neurobehavioural studies were taken on PND 24. After 24 h of neurobehavioural studies, on PND 25, six animals from each group were euthanized by cervical dislocation for the study of biochemical parameters (Fig. 2).
Study outline. Pregnant Wistar rats were administered MeHg and fisetin based on the grouping, orally, from GD 5 till parturition. The weaning pups were given training for specific behavioural studies from PND 21 to PND 23, and the final readings were taken on PND 24. On PND 25, the animals were euthanized for the study of biochemical and histopathological changes
Brain Dissection and Homogenization
A midline incision was made on the anesthetized rat to view the skull. Then, a small incision at the posterior aspect of the parietal bone and a rigid but gentle cut in the anterior part of the frontal bone were made to remove the brain easily. The brains were extracted out instantly and washed in cool saline to remove blood. The three brain regions were promptly segregated out from the intact brain carefully on an ice plate in accordance with the method of Glowinski and Iverson [37]. The brain regions were homogenized in ice-cold 0.1 M Tris–HCl buffer, pH 7, using a Potter-Elvehjem homogenizer filled with Teflon pestle and a homogenate (10% w/v) solution was produced. The biochemical parameters were assessed immediately after homogenization of the tissues.
Neurobehavioural Toxicity Study
Two pups from each litter were chosen randomly and gave three days trial from PND 21 to PND 23 for doing behavioural tests. On PND 24, final readings for behavioural tests were taken. Total calmness and absence of external disturbances were maintained throughout the test. Proper rest was given to each rat in between each test and was done in room temperature.
Rotarod Test
Motor activity was evaluated by determining the time spent in seconds by the rat on a rotating rod of 2.5 cm diameter. The speed of the rotation was increased slowly from 0 rpm and the velocity of 10 rpm was kept constant for the experiment. The rats were not allowed to stay on the rotating rod for not more than 60 s. On PND 24, three trials with an interval of 2 min resting time were given. Average latency to fall out of the three trials was noted as a measurement of motor activity [38]. Latency is a time interval between the stimulation and response, or a time delay between the cause and the effect of some physical change in the system being observed.
Morris Water Maze Test
Spatial learning and memory were evaluated using the Morris water maze test. A circular pool of 180 cm diameter × 75 cm height was filled to a depth of 45 cm with black opaque water at 22 °C. A plexiglass (14 × 14 cm), painted in black, was placed 1.5 cm below the water level. A hidden platform was placed in the middle of one of the four quadrants in the maze. On PND 21 to 23, 3-day trials (four trials per day with a 1-min resting time interval) were given to each rat, and the final test was taken on PND 24. The pups from each were released from different positions into one of the four quadrants. Escape latency was calculated within 60 s of the test [32].
Forced-Swim Test
Depressive syndrome was evaluated by keeping the rat in an unexpected situation. The rat was suddenly put in a vertical glass cylinder 25 cm high and 14 cm in diameter filled with water (30 °C) to a depth of 20 cm. The water depth was balanced to ensure that the hind limbs and tail were not touching the base. Each rat was placed in the cylinder for not more than 6 min. After a series of struggles to escape, the rat became immobile and the floating time was determined as the immobility time in seconds [39].
Open-Field Test
General locomotor and explorative activity was evaluated by an open field test. A 60 cm × 90 cm floor covered with an opaque wall of 30 cm height was divided into six equal squares. The pups were placed at the centre of the open field, and the rearing and resting time in seconds and distance travelled were observed for 5 min. A silent and undisturbing environment was maintained to study the locomotor and explorative behaviour of the animals [40].
Nociceptive Testing (Hot Plate Test)
Pain thresholds of the offspring were evaluated by a hot plate test. The rats were kept in a suitable glass beaker, which was kept on a hot plate of 55 °C. The latency period was calculated as the time taken for the rats to respond to the thermal pain, which was shown by licking their paws or jumping from the beaker. The latency time for the pain sensation was kept to a maximum of 45 s to prevent burning of skin [41].
Biochemical Studies
Protein concentrations in the samples were determined by using the standard method of Lowry et al., using bovine serum albumin as the standard [42].
Determination of Markers of Lipid and Protein Oxidation
Lipid peroxidation (LPO) was measured based on the method of Devasagayam and Tarachand [43]. Malondialdehyde formed as a product of lipid peroxidation reacts with thiobarbituric acid (TBA) to form a pink chromogen (TBA 2-malondialdehyde adduct) and was measured at 532 nm absorbance with a spectrophotometer. The malondialdehyde (MDA) content of the sample was expressed as nanomoles of MDA formed/milligram protein. The marker of oxidized proteins, protein carbonyl content, was determined by the method of Levine et al. at 366 nm absorbance [44]. The results were calculated as micromoles of carbonyl group/milligram/minute/milligram protein.
Determination of Non-enzymatic Antioxidants
Glutathione (GSH) level in the selected brain regions was measured based on the method of Moron et al. [45]. The reduced glutathione was expressed as micrograms/milligram protein. NO production was evaluated by determining its metabolites nitrate (NO3 −) and nitrite (NO2 −), by using the method of Miranda et al. [46].
Determination of Enzymatic Antioxidants
Glutathione-S-transferase (GST) activity was measured based on the method of Habig and Jakoby, and the absorbance was measured at 340 nm and expressed as nanomoles of CDNB conjugated/minute/milligram protein [47]. The glutathione reductase (GR) activity was determined using the method of Carlberg and Mannervik [48]. GR reduces (oxidized glutathione) GSSG to GSH; the disappearance of NADPH during this process is assessed at 340 nm. Glutathione peroxide (GPx) activity was determined using the method of Wendel by measuring NADPH utilized at 340 nm [49]. The GPx reduces hydroperoxides into GSSG by utilizing GSH. GSSG is converted into GSH by GR using NADPH as a reducing equivalent donor. The total superoxide dismutase (SOD) activity was measured using the method of Kono in which auto-oxidation of hydroxylamine was calculated at 560 nm for 2 min at 30-s intervals using a UV spectrophotometer [50]. Total thiol and protein-bound thiol were measured using Ellman’s reagent [51]. Catalase (CAT) activity was assessed using the method of Aebi in which formation of a stable complex of H2O2 with ammonium molybdate was used to assay the CAT activity, and the absorbance was measured spectrophotometrically at 405 nm [52, 53].
Determination of Membrane-Bound ATPases
Na+-K+ ATPase (EC 3.6.1.3) activity was assayed by using the method of Bonting [54]. The homogenate (0.2 ml) was mixed in 184 mM Tris–HCl buffer (1 ml) (pH 7.5), 50 mM MgSO4 (0.2 ml), 50 mM KCl (0.2 ml ), 600 mM NaCl (0.2 ml), 1 mM EDTA (0.2 ml) and 10 mM ATP (0.2 ml) and kept for 15 min at 37 °C. One millilitre of ice-cold 10% TCA was used to arrest the reaction. Liberated Pi was estimated in the protein-free supernatant.
Ca2+ ATPase (EC 3.6.1.3) activity was assayed by using the method of Hjerton and Pan [55]. The homogenate (0.1 ml) was mixed in 125 mM Tris–HCl buffer (0.1 ml) (pH 8), 50 mM CaCl2 (0.1 ml) and 10 mM ATP (0.1 ml) and incubated for 15 min at 37 °C. Ice-cold 10% TCA (0.5 ml) was used to arrest the reaction.
Mg2+ ATPase (EC 3.6.1.3) activity was assayed by using the method of Ohnishi et al. [56]. The homogenate (0.1 ml) was mixed in 375 mM Tris–HCl buffer (0.1 ml) (pH 7.6), 25 mM MgCl2 (0.1 ml), 10 mM ATP (0.1 ml) and water (0.1 ml) and incubated for 15 min at 37 °C. Ice-cold 10% TCA (0.5 ml) was used to arrest the reaction.
Liberated inorganic phosphorus (Pi) in protein-free supernatant was estimated using the method of Fiske and Subbarow [57]. ATPase activities were expressed in millimoles of Pi liberated per minute per milligram protein [57].
Determination of Acetyl Cholinesterases
The activity of acetylcholine esterase (AChE) was determined using the method of Ellman in specific brain regions [58]. The method is based on the hydrolyzation of acetylthiocholine iodide (substrate) to thiocholine and butyric acid. 5-Thio-2-nitrobenzoic acid (yellow colour) produced by the reaction between thiocholine and 5,50-dithiobis (2-nitrobenzoic acid) was measured by a spectrophotometer at 412 nm.
Histopathological Studies
The whole brain from three randomly selected pups (PND 25) from each group was used for histopathological examination.
Haematoxylin and Eosin (H&E) Staining
The animals were given ether anesthesia and sacrificed by transcardiac perfusion with 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.4). The brains were excised out and weighed, and postfixation was done using the same fixative. The tissue blocks were further embedded in paraffin wax, and 5-μm-thick serial coronal sections were cut using a microtome. The sections from three specific brain regions were used for haematoxylin and eosin (H&E) [59].
Cresyl Violet (CV) Staining
Nissl body staining was done to assess the extent of neuronal damage in the cerebral cortex, cerebellum and hippocampus in rat offspring [60]. Coronal brain sections were stained with 0.1% (w/v) cresyl violet acetate (Nissl stain) for 5 min, dehydrated through graded concentrations of ethanol and cleared in xylene. The stained sections were visualized and analyzed using a light microscope. The appearance of Nissl-stained dark neurons (NDN) was analyzed in the cerebral cortex, cerebellum and hippocampus of the control and experimental groups. The digital images were captured by using a microscope (BX40; Olympus, Melville, NY) and a digital camera [Nikon D70 DSLR (6.1 megapixels)]. The digital images were analyzed with image analysis software (Optimas version 6.1; Media Cybernetics, Del Mar, CA).
Mercury (Hg) Analysis
The whole brain from six randomly selected pups from each group was used for measurement of Hg accumulation. Isolated whole brain was immediately frozen and stored at −80 °C until analysis using atomic absorption spectroscopy.
Statistical Analysis
Data are represented as mean ± SD. Statistical comparisons were performed by one-way analysis of variance (ANOVA) using SPSS 20 software. If ANOVA indicated significant differences, Tukey’s post hoc test was performed to compare mean values between the study groups and control. A value of p < 0.05 was considered as statistically significant.
Results
Hg Accumulation
Table 1 shows the effect of gestational administration of fisetin against mercury accumulation in the whole brain of control and experimental rats. On PND 25, the brain Hg accumulation was significantly (p < 0.05) high in MeHg-exposed pups, whereas simultaneous gestational administration of fisetin in MeHg-exposed mother rats reduced the Hg accumulation significantly [10 mg/kg b.w. (p < 0.01) and 50 mg/kg b.w. (p < 0.05)]. Groups treated solely with fisetin showed no significant changes in comparison with the control.
Gestational Characteristics
Table 2 shows the protective effect of fisetin administration during pregnancy on gestational characteristics in the control and experimental rats. Oral administration of 1.5 mg/kg b.w. MeHg from GD 5 till parturition caused significant alterations in the maternal and offspring characteristics, whereas simultaneous gestational administration of fisetin in MeHg-exposed mother rats prevented MeHg-induced maternal and fetal toxicity. Groups treated solely with fisetin showed no significant changes in comparison with the control. On PND 21, the body weight of MeHg-exposed pups was significantly (p < 0.05) less in comparison with that of the control, whereas simultaneous gestational administration of fisetin in MeHg-exposed mother rats increased the body weight of pups significantly [10 mg/kg b.w. (p < 0.01) and 50 mg/kg b.w. (p < 0.05)] in comparison to group II. Groups treated solely with fisetin showed no significant changes in comparison with the control.
Oxidative Stress Markers
Table 3 shows the effect of gestational administration of fisetin on LPO and protein carbonyl levels in specific brain regions of pups belonging to MeHg-exposed mother rats. On PND 25, the levels of LPO and protein carbonyl were found to be increased significantly (p < 0.001) in all the brain regions of MeHg-exposed pups, whereas simultaneous gestational administration of fisetin in MeHg-exposed mother rats reduced the levels of LPO and protein carbonyl significantly [10 mg/kg b.w. (p < 0.01) and 50 mg/kg b.w. (p < 0.05)]. Groups treated solely with fisetin showed no significant changes in comparison with the control.
Non-enzymatic Antioxidants
Table 4 shows the effect of fisetin on GSH and NO in specific brain regions of pups belonging to MeHg-exposed mother rats. On PND 25, the levels of GSH and NO were significantly decreased (p < 0.001) in all the brain regions of MeHg-exposed pups, whereas simultaneous gestational administration of fisetin in MeHg-exposed mother rats preserved the levels of GSH and NO significantly [10 mg/kg b.w. (p < 0.01) and 50 mg/kg b.w. (p < 0.05)]. Groups treated solely with fisetin showed no significant changes in comparison with the control.
Enzymatic Antioxidants
Table 5 shows the effect of fisetin on SOD, CAT, GPx, GST and GR activities in specific brain regions of pups belonging to MeHg-exposed mother rats. On PND 25, a significant decrease in the activities of SOD (p < 0.001), CAT (p < 0.001), GPx (p < 0.01) and GST (p < 0.05) and an increase in GR (p < 0.05) were observed in all the brain regions of MeHg-exposed pups, whereas simultaneous gestational administration of fisetin in MeHg-exposed mother rats significantly increased [10 mg/kg b.w. (p < 0.01) and 50 mg/kg b.w. (p < 0.05)] SOD, CAT, GPx and GST activities and decreased GR activity [10 mg/kg b.w. (p < 0.01) and 50 mg/kg b.w. (p < 0.05)] in all the specific brain regions. Groups treated solely with fisetin showed no significant changes in comparison with the control.
Thiol Status
Table 6 shows the effect of fisetin on total thiol (T-SH) and protein-bound thiol (Pr-SH) content in specific brain regions of pups belonging to MeHg-exposed mother rats. On PND 25, protein-bound thiol and total thiol content were significantly decreased (p < 0.001) in all the brain regions of MeHg-exposed pups, whereas simultaneous gestational administration of fisetin in MeHg-exposed mother rats preserved protein thiol and total thiol content of the brain regions significantly [10 mg/kg b.w. (p < 0.01) and 50 mg/kg b.w. (p < 0.05)]. Groups treated solely with fisetin showed no significant changes in comparison with the control.
Neurobehaviour of Pups on PND 24
Figure 3 shows the neuroprotective effect of fisetin on defective motor co-ordination due to transplacental MeHg exposure in rat offspring. On PND 24, pups exposed to MeHg in utero showed significantly less time latency to fall (p < 0.001) in the rotarod test, whereas pups born to fisetin-administered pregnant rats showed significantly increased time latency to fall [10 mg/kg b.w. (p < 0.01) and 50 mg/kg b.w. (p < 0.05)] from the rotating rod. Figure 4 shows the neuroprotective effect of fisetin on learning and memory impairment due to transplacental MeHg exposure in rat offspring. On PND 24, pups exposed to MeHg in utero showed significantly increased escape latency (p < 0.001) in the Morris water maze test, whereas pups born to fisetin-administered pregnant rats showed significantly decreased escape latency [10 mg/kg b.w. (p < 0.01) and 50 mg/kg b.w. (p < 0.05)] in comparison to MeHg-exposed pups. Figure 5 shows the neuroprotective effect of fisetin on depression syndromes due to transplacental MeHg exposure in rat offspring. On PND 24, pups exposed to MeHg in utero showed significantly increased immobility time (p < 0.001) in the forced-swim test, whereas pups born to fisetin-administered pregnant rats showed significantly decreased immobility time [10 mg/kg b.w. (p < 0.01) and 50 mg/kg b.w. (p < 0.05)] in comparison to MeHg-exposed pups.
Neuroprotective effect of gestational administration of fisetin on defective motor co-ordination due to transplacental MeHg exposure in rat offspring. Pups of mother rats from each group were used for the rotarod test. On PND 24, the time taken to fall (latency to fall) in seconds from the rotating rod was noted as a measure of cerebellar motor co-ordination function of the weaning rats. The data represent mean values ± SD (n = 6 animals in each group). Three asterisks p < 0.001, significantly different from the control; one number sign p < 0.05 and two number signs p < 0.01, significantly different from the MeHg-exposed group (group II); one-way ANOVA with Tukey’s post hoc test was used for statistical analysis
Neuroprotective effect of gestational administration of fisetin on learning and memory impairment due to transplacental MeHg exposure in rat offspring. Pups of mother rats from each group were used for the Morris water maze test. After 3 days of training (from PND 21 to PND 23), on PND 24, the time taken by the rat to find the hidden platform (escape latency) was taken as a measure of memory function in the weaning rats. The data represent mean values ± SD (n = 6 animals in each group). Three asterisks p < 0.001, significantly different from the control; one number sign p < 0.05 and two number signs p < 0.01, significantly different from the MeHg-exposed group (group II); one-way ANOVA with Tukey’s post hoc test was used for statistical analysis
Neuroprotective effect of gestational administration of Fisetin on depression syndromes due to transplacental MeHg exposure in rat offspring. Pups of mother rats from each group were used for Forced swim test. On PND 24, the rats were kept on an unanticipated adverse situation by putting the rat in a water-filled beaker. Floating time taken by the rat above water without any mobility (immobility time) was considered as a measure of depression behaviour. The data represent mean values ± SD (n = 6 animals in each group). Three asterisks p < 0.001, significantly different from the control; one number sign p < 0.05 and two number signs p < 0.01, significantly different from the MeHg-exposed group (group II). One-way ANOVA with Tukey’s post hoc test was used for statistical analysis
Figure 6a–c shows the neuroprotective effect of fisetin on defective locomotor activity and emotionality behaviour due to transplacental MeHg exposure in rat offspring. On PND 24, pups exposed to MeHg in utero showed significantly decreased distance travelled (p < 0.01, Fig. 6a), decreased resting time (p < 0.01, Fig. 6b) and increased number of rears (p < 0.001, Fig. 6c) in the open field test, whereas pups born to fisetin-administered pregnant rats showed significantly increased resting time [10 mg/kg b.w. (p < 0.01) and 50 mg/kg b.w. (p < 0.05)], increased distance travelled [10 mg/kg b.w. (p < 0.05) and 50 mg/kg b.w. (p < 0.05)] and decreased number of rears [10 mg/kg b.w. (p < 0.001) and 50 mg/kg b.w. (p < 0.01)] in comparison to MeHg-exposed pups. Figure 7 shows the neuroprotective effect of fisetin on impaired nociception signals due to transplacental MeHg exposure in rat offspring. On PND 24, pups exposed to MeHg in utero showed significantly increased time for pain sensation (p < 0.05) in the hot plate test, whereas pups born to fisetin-administered pregnant rats showed significantly decreased time for pain sensation [10 mg/kg b.w. (p < 0.01) and 50 mg/kg b.w. (p < 0.05)] in comparison to MeHg-exposed pups. Groups treated solely with fisetin showed no significant changes in comparison with the control in all the behavioural tests.
a–c Neuroprotective effect of gestational administration of fisetin on defective locomotor activity and emotionality behaviour due to transplacental MeHg exposure in rat offspring. Pups of mother rats from each group were used for the open-field test on PND 24. The pups were placed at the centre of the open field (60 cm × 90 cm) and were observed for 5 min. The distance travelled in centimetres (a), the resting time in seconds (b) and the number of rears (c) were noted as a measure of locomotor activity and emotionality behaviour of the animals. The data represent mean values ± SD (n = 6 animals in each group). Two asterisks p < 0.01 and three asterisks p < 0.001, significantly different from the control. One number sign p < 0.05, two number signs p < 0.01 and three number signs p < 0.001, significantly different from the MeHg-exposed group (group II). One-way ANOVA with Tukey’s post hoc test was used for statistical analysis
Neuroprotective effect of gestational administration of fisetin on impaired nociception signals due to transplacental MeHg exposure in rat offspring. Pups of mother rats from each group were used for the hot plate test on PND 24. The latency to pain sensation was calculated as the time taken for the rats (within 45 s) to respond to the thermal pain, which was shown by licking their paws or jumping from the beaker. The data represent mean values ± SD (n = 6 animals in each group). One asterisk p < 0.05, significantly different from the control. One number sign p < 0.05 and two number signs p < 0.01, significantly different from the MeHg-exposed group (group II). One-way ANOVA with Tukey’s post hoc test was used for statistical analysis
Membrane-Bound ATPases
Figure 8a–c shows the neuroprotective effect of gestational administration of fisetin on the activities of Na+-K+ ATPase, Ca2+ ATPase and Mg2+ ATPase in specific brain regions of control and experimental rats. On PND 25, the specific activity of membrane-bound ATPases was found to be decreased significantly [Na+-K+ ATPase (p < 0.001), Ca2+ ATPase (p < 0.01) and Mg2+ ATPase (p < 0.05)] in all the three brain regions of MeHg-exposed pups, whereas simultaneous gestational administration of fisetin in MeHg-exposed mother rats significantly preserved the activities of Na+-K+ ATPase [10 mg/kg b.w. (p < 0.001) and 50 mg/kg b.w. (p < 0.01)], Ca2+ ATPase [10 mg/kg b.w. (p < 0.01) and 50 mg/kg b.w. (p < 0.05)] and Mg2+ ATPase [10 mg/kg b.w. (p < 0.05) and 50 mg/kg b.w. (p < 0.05)]. Groups treated solely with fisetin showed no significant changes in comparison with the control.
a–c Neuroprotective effect of gestational administration of fisetin on the altered activities of Na+-K+ ATPase, Ca2+ ATPase and Mg2+ ATPase due to transplacental MeHg exposure in the rat offspring brain. Pups of mother rats from each group were euthanized on PND 25, and the activity of Na+-K+ ATPase (a), Ca2+ ATPase (b) and Mg2+ ATPase (c) was evaluated in the cerebral cortex, cerebellum and hippocampus of the rat offspring brain. The data represent mean values ± SD (n = 6 animals in each group). One asterisk p < 0.05, two asterisks p < 0.01 and three asterisks p < 0.001, significantly different from the control. One number sign p < 0.05, two number signs p < 0.01 and three number signs p < 0.001, significantly different from the MeHg-exposed group (group II). One-way ANOVA with Tukey’s post hoc test was used for statistical analysis
AchE Activity
Figure 9 shows the neuroprotective effect of fisetin on the activities of AchE in specific brain regions of control and experimental rats. On PND 25, the specific activity of AchE was found to be decreased significantly (p < 0.001) in all the brain regions of MeHg-exposed pups, whereas simultaneous gestational administration of fisetin in MeHg-exposed mother rats preserved the AchE activity significantly [10 mg/kg b.w. (p < 0.001) and 50 mg/kg b.w. (p < 0.01)]. Groups treated solely with fisetin showed no significant changes in comparison with the control.
Neuroprotective effect of gestational administration of fisetin on the altered activities of AchE due to transplacental MeHg exposure in the rat offspring brain. AchE activity was assessed in pups of mother rats from each group on PND 25 in the cerebral cortex, cerebellum and hippocampus of the rat brain. The data represent mean values ± SD (n = 6 animals in each group). Three asterisks p < 0.001, significantly different from the control. Two number signs p < 0.01 and three number signs p < 0.001, significantly different from the MeHg-exposed group (group II). One-way ANOVA with Tukey’s post hoc test was used for statistical analysis
Histopathological Studies
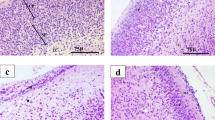
Figure 10a–c represents the neuroprotective effect of gestational administration of fisetin on the altered cellular morphology due to transplacental MeHg exposure in specific brain regions of pups belonging to MeHg-exposed mother rats (H&E staining). On PND 25, light microscopic studies showed widespread neurodegeneration in specific motor and cognitive regions of offspring brain due to transplacental exposure to MeHg. Neurons in the Purkinje layer of the cerebellum were diminished, and a large number of pyknotic nuclei (CA1 and CA2 regions) in the hippocampus and appearance of gliosis in the cerebral cortex were observed in pups of MeHg-exposed mother rats. Fisetin administration protected the brain of the developing fetus from much cellular morphological alterations in the three brain regions. Groups treated solely with fisetin showed no significant changes in comparison with the control.
a–c Neuroprotective effect of gestational administration of fisetin on the altered cellular morphology due to transplacental MeHg exposure in rat offspring. Pups of mother rats from each group were used for the histopathological study on PND 25. Haematoxylin and eosin staining of the cerebral cortex, cerebellum and hippocampus of rat offspring is shown in a, b and c respectively. A Control with normal morphology (H&E, ×400). B MeHg (1.5 mg/kg b.w.) showing neuronal damage (H&E, ×400). In the cerebral cortex (a), MeHg exposure caused extensive neurodegeneration; appearance of astrocyte gliosis is denoted with arrows. In the cerebellum (b), MeHg exposure caused decreased neurons in the Purkinje layer (PL) of the cerebellum and is denoted with arrows. The granular layer (GL) and molecular layer (ML) are also shown. In the hippocampus (c), MeHg exposure caused increased appearance of pyknotic nuclei in the CA1 and CA2 regions as shown by arrows. C MeHg + fisetin (10 mg/kg b.w.) protected neurons from degeneration (H&E, ×400). D MeHg + fisetin (50 mg/kg b.w.) protected neurons from degeneration (H&E, ×400). E Fisetin (10 mg/kg b.w.) showing near-normal morphology (H&E, ×400). F Fisetin (50 mg/kg b.w.) showing near-normal morphology (H&E, ×400)
Figure 11a–c represents the neuroprotective effect of gestational administration of fisetin on changes in Nissl body characteristics in specific brain regions of pups belonging to MeHg-exposed mother rats (Cresyl violet staining). On PND 25, Nissl-stained dark neurons (NDN), a feature of extensive damage with neuronal cell loss with atrophic neurons, shrunken cytoplasm and damaged nuclei in the cerebral cortex, cerebellum and cortical and pyramidal cells of the hippocampus (CA1 and CA2 regions) were observed. Fisetin administration protected the brain of the developing fetus from morphological changes; a decrease in NDN was observed in the three regions on PND 25. Groups treated solely with fisetin showed no significant changes in comparison with the control.
a–c Neuroprotective effect of gestational administration of fisetin on changes in Nissl body characteristics in specific brain regions of pups belonging to MeHg-exposed mother rats. Pups of mother rats from each group were used for the histopathological study on PND 25. Cresyl violet staining of the cerebral cortex, cerebellum and hippocampus of rat offspring is shown in a, b and c respectively. The appearance of Nissl-stained dark neurons (NDN) was analyzed in the cerebral cortex, cerebellum and hippocampus of control and experimental groups. A control with normal morphology (CV, ×400), B MeHg showing increased NDN (CV, ×400), C MeHg + fisetin (10 mg/kg b.w.) showing decreased Nissl-stained dark neurons (NDNs) (CV, ×400), D MeHg + fisetin (50 mg/kg b.w.) showing decreased NDNs (CV, ×400), E fisetin (10 mg/kg b.w.) showing near-normal morphology (CV, ×400), F fisetin (50 mg/kg b.w.) showing near-normal morphology (CV, ×400)
Discussion
The current study is the first that exemplified a novel protective effect of the flavanoid fisetin against the neurotoxicity caused by MeHg during the most susceptible stage of life in rats. The experiment was designed to represent the current scenario of gestational exposure to MeHg in pregnant mothers via contaminated seafood consumption habits which result in motor and cognitive disorders in the developing fetus. MeHg is easily absorbable from the gastrointestinal (GI) tract to the circulation (90–95%) and can easily cross the placental and blood-brain barriers and cause oxidative stress in the brain [61–64].
The half-life of MeHg is about 52–93 days [65] in adult rats, and in the current study, the exposure protocol followed resulted in significant accumulation of mercury in F1 generation rat brains even 25 days after the halt of MeHg exposure. Results from a number of studies on gestational exposure to MeHg have also shown significant accumulation of inorganic mercury in the offspring brain of both humans and rats [65]. The fisetin-administered group showed comparatively less Hg concentration in the current study. MeHg binds to the –SH group of cysteine, and the MeHg/L-cysteine (MeHg-Cys) complex closely resembles L-methionine which is the substrate for L-type neutral amino acid carrier transport (LAT) that transports MeHg across the placenta and blood-brain barrier [63]. Further studies have to be done to explore the mechanism of inhibition of Hg accumulation in the fetal brain by fisetin, whether it is at the level of the LAT system or at the placental barrier, or else by any direct interaction with the MeHg structure. Throughout the study, there was no change in food and water consumption habits of pregnant mother rats. Moreover, the fisetin-administered group showed no symptoms of overdosage or any other toxicity signs. The present study found extended gestational days and decreased maternal body weight gain in MeHg-exposed mother rats whereas co-administration with fisetin showed near-normal maternal characteristics.
Impairment of motor co-ordination is one of the toxic behavioural outcomes of prenatal MeHg exposure in the F1 generation of humans and laboratory rodents [66, 67]. In the present study, rat offspring exposed to MeHg during gestation were unable to spend enough time on the rotating rod, whereas concurrent administration of fisetin protected the offspring from severe motor impairment. The present results are reinforcing the alleviating effects of fisetin on impaired motor neurons which were reported in many pioneering studies [68]. In the open-field test, MeHg-exposed rat offspring showed less resting time, travelled less distance, but showed increased rearing time, indicating the reduction in arousal or increase in the level of emotionality of the rats which was reported in an earlier similar study [40]. The concurrent developmental exposure to fisetin ameliorated the impairment in spontaneous locomotor activity of rat offspring during postnatal weaning days.
In the forced-swim test, rat offspring showed depression symptoms. Our findings are in accordance with the previous studies in which rat offspring exposed to MeHg in utero showed behavioural despair to an aversive situation, in the forced-swim test [39]. The concurrent administration of fisetin prevented the development of depressive behaviour. The anti-depressant effect of fisetin has been validated previously in a mouse model which showed the role of non-adrenergic and serotonergic neurons. The Morris water maze test showed evidence for impairment of learning and memory due to transplacental exposure to MeHg. Our results are supporting the earlier studies [69]. Simultaneous administration of fisetin during gestation protected from learning and memory impairment. Previous studies show that fisetin promotes ERK-dependent improvement of memory and the learning process [68]. Nociception signals were also found to be damaged due to in utero MeHg exposure which was evident from the hot plate test, which showed decreased sensitivity to pain in rat pups of the present study. Concurrent administration of fisetin during gestation showed an improvement in nociception signals of the postnatal weaning pups.
In the present study, high brain levels of LPO and protein carbonyl indicate an oxidative imbalance in the system. MeHg generates reactive oxygen species (ROS) which cause peroxidation of lipid membranes and cause oxidative modifications of proteins in the brain [15]. Protein carbonyls are novel markers of oxidized proteins, and carbonylation of proteins disrupts many functions of receptors, signalling molecules, transporters and enzymes [44]. Reduction of LPO and protein carbonyl levels in the fisetin-administered group can be explained based on its high free radical scavenging property [70]. In the present study, prenatal MeHg exposure caused reduced thiol content in rat offspring brain regions, which could be due to covalent binding of MeHg to reactive thiols of cellular proteins, called S-mercuration, that results in formation of protein adducts [71]. GSH is the most abundant intracellular thiol compound in the CNS, which was highly reduced in all the three regions of the MeHg-exposed rat offspring brain in the present study.
Transplacental exposure to MeHg impaired the antioxidant status significantly which was seen as reduced activity of enzymatic (GPx, GST, SOD and CAT) and non-enzymatic (NO and GSH) antioxidants in weaning rat pups. A previous study on an animal model showed similar results due to MeHg exposure [16]. GR is responsible for the formation of GSH from GSSG. MeHg reduces the GSH level by binding to it and hence increases GR activity [16]. Simultaneous gestational administration of fisetin preserved the levels of antioxidants in F1 generation rat brains, which can be explained, based on the previous report that showed inhibition of lipid peroxidation by fisetin and reduction of oxidative stress leading to improvement of memory and learning. Concurrent administration of fisetin in pregnant rats preserved the developmental profile of the endogenous GSH antioxidant system, which is in agreement with the study that showed fisetin retains endogenous GSH levels and preserves mitochondrial function during oxidative stress [71, 72].
In the present study, the activities of membrane-bound enzymes Na+-K+ ATPase, Ca2+ ATPase and Mg2+ ATPase were decreased markedly due to prenatal MeHg exposure in the rat offspring brain. The decreased activity of ATPases could be due to increased S-mercuration of these membrane-bound proteins by MeHg. Increased intracellular Ca2+ concentration is found to be an important factor in the onset of MeHg neurotoxicity. Ca2+ ATPase expels Ca2+ ions and maintains a low Ca2+ level in the cytosol which is essential for maintaining ionic balance in the neurons [21, 22, 73, 74]. Na+-K+ ATPase and Mg2+ ATP are involved in calcium homeostasis by maintaining cellular levels of Na+ and Mg+ ions respectively. Inhibition of these ATPases might finally affect the electrochemical balance in the neurons which might finally affect the action potential and neuronal survival [75, 76]. Concurrent gestational administration of fisetin prevented the dwindling of ATPase activity during weaning periods of F1 generation rats in all the three regions.
The cholinergic system is an important therapeutic target for many neuropsychological disorders. AChE catalyzes hydrolysis of the important neurotransmitter acetylcholine to its inactive metabolites, choline and acetate. Prompt removal of free acetylcholine from the synapse is crucial for absolute function of cognition, learning and the memory process [20, 26, 27]. In the present study, MeHg exposure in utero caused decreased activity of the specific cholinergic marker protein, AchE. Our results are in accordance with those of previous studies [27, 77] . Decreased AchE activity was manifested as inefficiency to execute well in the Morris water maze test by postnatal weaning rats. Synchronous administration of fisetin in mother rats during pregnancy substantially prevented the declining of AchE activity due to MeHg in the rat offspring brain during its postnatal life, which is in agreement with many earlier documented studies that have improved the impairment of the cholinergic system by fisetin in several disorders. Earlier investigations showed involvement of AchE in induction of long-term potentiation in hippocampal pyramidal neurons which helps in enhancement of cognitive function; simultaneously, previous studies showed the ability of fisetin to promote long-term potentiation in the hippocampus and enhancement of memory [31, 32, 36]. Further studies are required to identify the exact mechanism of the protective effect of fisetin against impairment of the cholinergic system due to developmental MeHg exposure.
In light microscopic studies, transplacental exposure to MeHg caused significant changes in the microstructure of the cerebral cortex, cerebellum and hippocampus. Vast neuronal degeneration in the Purkinje layer of the cerebellum, presence of increased pyknotic nuclei in the hippocampus and appearance of gliosis in the cerebral cortex were observed in the pups which was reported previously [78–80]. In Nissl staining analysis, developmental MeHg exposure caused increased presence of Nissl-stained dark neurons (NDN), which was similar to a previous report [81], whereas fisetin-exposed offspring rats caused reduced NDN levels due to its antioxidant activity. Supplementation of fisetin in pregnant mother rats significantly mitigated the morphological damages due to in utero MeHg exposure in specific motor and cognitive centers of the rat offspring brain and consequently improved its physiological outcome, which was evident in neurobehavioural studies.
Conclusion
As the developing nervous system is susceptible to MeHg-induced toxicity, our study demonstrates that fisetin restrains the development of oxidative stress due to MeHg and maintains the antioxidant status, membrane-bound ATPases and AChE activities in the developing brain. Fisetin also mitigated MeHg-induced cytoarchitectural damages in specific motor and cognitive centers of F1 generation rats. Additionally, fisetin hampered neurobehavioural toxicity manifestations of MeHg in weaning rat pups. Taken together, gestational intake of fisetin protects the developing rat brain from transplacental MeHg toxicity. Further investigation on the neuroprotective effect of this flavanoid is warranted.
References
Sheehan MC, Burke TA, Navas-Acien A, Breysse PN, McGready J, Fox MA (2014) Global methylmercury exposure from seafood consumption and risk of developmental neurotoxicity: a systematic review. Bull World Health Organ 92:254–269
Gary J, Myers PW, Davidson WGE, Van EW, Thurston SW, Strain JJ, Shamlaye CF, Bovetd P (2014) Methylmercury exposure and developmental neurotoxicity. Bull World Health Organ 93(2):132
Boening DW (2000) Ecological effects, transport, and fate of mercury: a general review. Chemosphere 40:1335–1351
Johnson CL (2004) Mercury in the environment: sources, toxicities, and prevention of exposure. Pediatr Ann 33:437–442
Matsumoto H, Koya G, Takeuchi T (1965) Fetal Minamata disease. A neuropathological study of two cases of intrauterine intoxication by a methyl mercury compound. J Neuropathol Exp Neurol 24:563–574
Choi BH, Lapham LW, Amin-Zaki L, Saleem T (1978) Abnormal neuronal migration, deranged cerebral cortical organization, and diffuse white matter astrocytosis of human fetal brain: a major effect of methylmercury poisoning in utero. J Neuropathol Exp Neurol 37:719–733
Takeuchi T (1982) Pathology of Minamata disease. With special reference to its pathogenesis. Acta Pathol Jpn 32(Suppl 1):73–99
Myers GJ, Marsh DO, Davidson PW, Cox C, Shamlaye CF, Tanner M, Choi A, Cernichiari E, Choisy O, Clarkson TW (1995) Main neurodevelopmental study of Seychellois children following in utero exposure to methylmercury from a maternal fish diet: outcome at six months. Neurotoxicology 16:653–664
Van WE, Thurston SW, Myers GJ, Strain JJ, Weiss B, Zarcone T, Watson GE, Zareba G, McSorley EM, Mulhern MS, Yeates AJ, Henderson J, Gedeon J, Shamlaye CF, Davidson PW (2013) Prenatal methyl mercury exposure in relation to neurodevelopment and behavior at 19 years of age in the Seychelles Child Development Study. Neurotoxicol Teratol 39:19–25
Aschner M, Syversen T, Souza DO, Rocha JB, Farina M (2007) Involvement of glutamate and reactive oxygen species in methylmercury neurotoxicity. Braz J Med Biol Res 40:285–291
Halliwell B (1992) Oxygen radicals as key mediators in neurological disease: fact or fiction? Ann Neurol 32:10–15
Mori N, Yasutake A, Hirayama K (2007) Comparative study of activities in reactive oxygen species production/defense system in mitochondria of rat brain and liver, and their susceptibility to methylmercury toxicity. Arch Toxicol 81:769–776
Franco JL, Braga HC, Stringari J, Missau FC, Posser T, Mendes BG, Leal RB, Santos AR, Dafre AL, Pizzolatti MG, Farina M (2007) Mercurial-induced hydrogen peroxide generation in mouse brain mitochondria: protective effects of quercetin. Chem Res Toxicol 20:1919–1926
Mari M, Morales M, Colell A, García-Ruiz C, Jose C, Fernández-Checa (2009) Mitochondrial glutathione, a key survival antioxidant. Antioxid Redox Signal 11:2685–2700
Farina M, Rocha JBT, Aschner M (2011) Mechanisms of methylmercury-induced neurotoxicity: evidence from experimental studies. Life Sci 89:555–563
Stringari J, Nunes AK, Franco JL, Bohrer D, Garcia SC, Dafre AL, Milatovic D, Souza DO, Rocha JB, Aschner M, Farina M (2008) Prenatal methylmercury exposure hampers glutathione antioxidant system ontogenesis and causes long-lasting oxidative stress in the mouse brain. Toxicol Appl Pharmacol 227:147–154
Mynett-Johnson L, Murphy V, McCormack J, Shields DC, Claffey E, Manley P, McKeon P (1998) Evidence for an allelic association between bipolar disorder and Na+, K+ adenosine triphosphatase alpha subunit gene (ATP1A3). Biol Psychiatry 44:47–51
Wood AJ, Elphick M, Grahame-Smith DG (1989) Effect of lithium and of other drugs used in the treatment of manic illness on the cation-transporting properties of Na+, K+-ATPase in mouse brain synaptosomes. J Neurochem 52:1042–1049
Erecinska M, Silver IA (1994) Ions and energy in mammalian brain. Prog Neurobiol 43:37–71
Carageorgiou H, Pantos C, Zarros A, Stolakis V, Mourouzis I, Cokkinos D, Tsakiris S (2007) Changes in acetylcholinesterase, Na+K+ ATPase, and Mg2+ ATPase activities in the frontal cortex and the hippocampus of hyper and hypothyroid adult rats. Metab Clin Exp 56:1104–1110
Chiu VC, Mouring D, Haynes DH (1983) Action of mercurials on the active and passive transport properties of sarcoplasmic reticulum. J Bioenerg Biomembr 15:13–25
Freitas AJ, Rocha JB, Wolosker H, Souza DO (1996) Effects of Hg2+ and CH3Hg+ on Ca2+ fluxes in rat brain microsomes. Brain Res 738:257–264
Haynes DH (1983) Mechanism of Ca2+ transport by Ca2+-Mg2+-ATPase pump: analysis of major states and pathways. Am J Phys 244:G3–12
Everitt BJ, Robbins TW (1997) Central cholinergic systems and cognition. Annu Rev Psychol 48:649–684
Carageorgiou H, Tzotzes V, Sideris A, Zarros A, Tsakiris S (2005) Cadmium effects on brain acetylcholinesterase activity and antioxidant status of adult rats: modulation by zinc, calcium and L-cysteine co-administration. Basic Clin Pharmacol Toxicol 97:320–324
Appleyard ME (1995) Acetylcholinesterase induced long-term potentiation in CA1 pyramidal cells by a mechanism dependent on metabotropic glutamate receptors. Neurosci Lett 190:25–28
Petruccioli L, Turillazzi P (1991) Effect of methylmercury on acetylcholinesterase and serum cholinesterase activity in monkeys, Macaca fascicularis. Bull Environ Contam Toxicol 46:769–773
Wootten V, Brown DR, Callahan BG, Vetrano K, Wadman P, Melia J, Mulligan T, Schatz RA (1985) Behavioral and biochemical alterations following in utero exposure to methylmercury. Neurobehav Toxicol Teratol 7:767–773
Flora SJ (2009) Structural, chemical and biological aspects of antioxidants for strategies against metal and metalloid exposure. Oxidative Med Cell Longev 2:191–206
Ross JA, Kasum CM (2002) Dietary flavonoids: bioavailability, metabolic effects, and safety. Annu Rev Nutr 22:19–34
Deeba N, Syed Vaqar M, Adhami M, Khan I, Mukhtar H (2013) Inhibition of Akt/mTOR signaling by the dietary flavonoid fisetin. Anti Cancer Agents Med Chem 13:995–1001
Zhen L, Zhu J, Zhao X, Wu H, An Y, Li S, Du X, Lin M, Wang Q, Xu Y, Pan J (2012) The antidepressant like effect of fisetin involves the serotonergic and noradrenergic system. Behav Brain Res 228:359–366
Amin-Zaki L, Majeed MA, Elhassani SB, Clarkson TW, Greenwood MR, Doherty RA (1979) Prenatal methylmercury poisoning. Clinical observations over five years. Am J Dis Child 133:172–177
Khan N, Deeba N, Syed MH (2013) Fisetin: a dietary antioxidant for health promotion. Antioxid Redox Signal 19:151–162
Chuang JY, Chang PC, Shen YC et al (2014) Regulatory effects of fisetin on microglial activation. Molecules 15:8820–8839
Prakash D, Gopinath K, Sudhandiran G (2013) Fisetin enhances behavioral performances and attenuates reactive gliosis and inflammation during aluminum chloride-induced neurotoxicity. NeuroMolecular Med 15:192–208
Glowinski J, Iversen LL (1996) Regional studies of catecholamines in the rat brain. J Neurochem 13:655–669
Sakamoto M, Nakano A, Kajiwara Y, Naruse I, Fujisaki T (1993) Effects of methyl mercury in postnatal developing rats. Environ Res 61:43–50
Onishchenko N, Tamm C, Vahter M, Tomas H, Johnson JA, DA J, Sandra C (2007) Developmental exposure to methylmercury alters learning and induces depression-like behavior in male mice. Toxicological Sci 97:428–437
Yoshida M, Suzuki M, Satoh M, Yasutake A, Watanabe C (2011) Neurobehavioral effects of combined prenatal exposure to low-level mercury vapor and methylmercury. J Toxicol Sci 36:73–80
Beyrouty P, Stamler CJ, Liu JN, Loua KM, Kubow S, Chan HM (2006) Effects of prenatal methylmercury exposure on brain monoamine oxidase activity and neurobehaviour of rats. Neurotoxicol Teratol 28:251–259
Lowry OH, Risebrough NJ, Farr AL, Randal RJ (1951) Protein measurement with folin phenol reagent. J Biol Chem 193:265–270
Devasagayam TP, Tarachand U (1987) Decreased lipid peroxidation in rat kidneys during gestation. Biochem Biophys Res Commun 145:134–138
Levine RL, Williams JA, Stadtman ER, Shater E (1994) Carbonyl, assays for determination of oxidatively modified proteins. Methods Enzymol 233:346–357
Moron MS, Depierre JW, Mannervik B (1979) Levels of glutathione, glutathione reductase and glutathione S-transferase activities in rat lung and liver. Biochim Biophys Acta 582:67–78
Miranda KM, Espey MG, Wink DA (2001) A rapid, simple spectrophotometric method for simultaneous detection of nitrate and nitrite. Nitric Oxide 5:62–71
Habig WH, Jakoby WB (1981) Assays for differentiation of glutathione S-transferases. Methods Enzymol 77:398–405
Carlberg I, Mannervik B (1985) Glutathione reductase. Methods Enzymol 113:484–490
Wendel A (1981) Glutathione peroxidase. Methods Enzymol 77:325–333
Kono Y (1978) Generation of superoxide radical during autoxidation of hydroxylamine and an assay for superoxide dismutase. Arch Biochem Biophys 186:189–195
Sedlak J, Lindsay RH (1968) Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal Biochem 25:192–205
Goth L (1991) A simple method for determination of serum catalase activity and revision of reference range. Clin Chim Acta 196:143–152
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Bonting SL (1970) Sodium-potassium activated adenosinetriphosphatase and cation transport. In Bittar EE (ed) Membranes and ion transport. Interscience Publishers, Ltd., London, 1:257–263
Hjerton S, Pan H (1983) Purification and characterization of two forms of a low affinity Ca2+-ATPase from erythrocyte membranes. Biochim Biophys Acta 728:281–288
Ohnishi T, Suzuki T, Suzuki Y, Ozawa K (1982) A comparative study of plasma membrane Mg2+-ATPase activities in normal, regenerating and malignant cells. Biochim Biophys Acta 684:67–74
Fiske CK, Subbarow Y (1925) The colorimetric determination of phosphorus. J Biol Chem 66:376–406
Worek F, Eyer P, Thiermann H (2012) Determination of acetylcholinesterase activity by the Ellman assay: a versatile tool for in vitro research on medical countermeasures against organophosphate poisoning. Drug Test Anal 4:282–291
Feldman AT, Wolfe D (2014) Tissue processing and hematoxylin and eosin staining. Methods Mol Biol 1180:31–43
Furuta Y, Kobori O, Shimazu H, Morioka Y, Okuyama Y (1985) A new in vivo staining method, cresyl violet staining, for fiberoptic magnified observation of carcinoma of the gastric mucosa. Gastroenterol Jpn 20:120–124
Nielsen JB, Andersen O (1992) The toxicokinetics of mercury in mice offspring after maternal exposure to methylmercury—effect of selenomethionine. Toxicology 74:233–241
Mansour MM, Dyer NC, Hoffman LH, Schulert AR, Brill AB (1973) Maternal-fetal transfer of organic and inorganic mercury via placenta and milk. Environ Res 6:479–484
Kerper LE, Ballatori N, Clarkson TW (1992) Methylmercury transport across the blood-brain barrier by an amino acid carrier. Am J Phys 26:761–765
Floyd RA (1999) Antioxidants, oxidative stress, and degenerative neurological disorders. Exp Biol Med 222:236–245
Yaginuma-Sakurai K, Murata K, Iwai-Shimada M, Nakai K, Kurokawa N, Tatsuta N, Satoh H (2012) Hair-to-blood ratio and biological half-life of mercury: experimental study of methylmercury exposure through fish consumption in humans. J Toxicol Sci 37:123–130
Franco JL, Teixeira A, Meotti FC, Ribas CM, Stringari J, Garcia PSC, Moro AM, Bohrer D, Bairros AV, Dafre AL, Santos AR, Farina M (2006) Cerebellar thiol status and motor deficit after lactational exposure to methylmercury. Environ Res 102:22–28
Gimenez-Llort L, Ahlbom E, Dare E, Vahter M, Ogren S, Ceccatelli S (2001) Prenatal exposure to methylmercury changes dopamine-modulated motor activity during early ontogeny: age and gender-dependent effects. Environ Toxicol Pharmacol 9:61–70
Maher P, Dargusch R, Bodai L, Paul E, Gerard PJM, Marsh JL (2011) ERK activation by the polyphenols fisetin and resveratrol provides neuroprotection in multiple models of Huntington’s disease. Hum Mol Genet 20:261–270
Wu J, Cheng G, Lu Z, Wang M, Tian J, Bi Y (2016) Effects of methyl mercury chloride on rat hippocampus structure. Biol Trace Elem Res 171:124–130
Ravichandran N, Suresh G, Ramesh B, Siva GV (2011) Fisetin, a novel flavonol attenuates benzo(a)pyrene-induced lung carcinogenesis in Swiss albino mice. Food Chem Toxicol 49(5)
Kanda H, Shinkai Y, Kumagai Y (2014) S-Mercuration of cellular proteins by methylmercury and its toxicological implications. J Toxicol Sci 39:687–700
Chiruta C, Schubert D, Dargusch R, Maher P (2012) Chemical modification of the multitarget neuroprotective compound fisetin. J MedChem 55:378–389
Magour S (1986) Studies on the inhibition of brain synaptosomal Na+/K+-ATPase by mercury chloride and methyl mercury chloride. Arch Toxicol 9:393–396
Chuu JJ, Liu SH, Lin-Shiau SY (2001) Effects of methyl mercury, mercuric sulfide and cinnabar on active avoidance responses, Na+/K+-ATPase activities and tissue mercury contents in rats. Proc. Natl. Sci. Counc. Repub. China B 25:128–136
Mekahli D, Bultynck G, Parys JB, De Smedt H, Missiaen L (2011) Endoplasmic-reticulum calcium depletion and disease. Cold Spring Harb 3:1–30
Zhang Y, Lu R, Liu W, Wu Y, Qian H, Zhao X, Wang S, Xing G, Yu F, Aschner M (2013) Hormetic effects of acute methylmercury exposure on grp78 expression in rat brain cortex. Dose Response 11:109–120
Cheng JP, Yang YC, Hu WX, Yang L, Wang WH, Jia JP, Lin XY (2005) Effect of methylmercury on some neurotransmitters and oxidative damage of rats. J Environ Sci 17:469–473
Gao Y, Yan CH, Yu XD, Wu SH (2006) Effects of perinatal exposure to methylmercury on the structure of hippocampus and cerebellum in young rats. Wei Sheng Yan Jiu 35:402–405
Falluel-More A, Sokolowski K, Sisti HM, Zhou X, Tracey J, Shors TJ, DiCicco-Bloom E (2007) Developmental mercury exposure elicits acute hippocampal cell death, reductions in neurogenesis, and severe learning deficits during puberty. J Neurochem 103:1968–1981
Burbacher TM, Rodier PM, Weiss B (1990) Methylmercury developmental neurotoxicity: a comparison of effects in humans and animals. Neurotoxicol Teratol 12:191–202
Carvalho MC, Nazari EM, Farina M, Muller YMR (2008) Behavioral, morphological, and biochemical changes after in ovo exposure to methylmercury in chicks. Toxicol Sci 106:180–185
Acknowledgments
The financial assistance and award of Junior Research Fellowship (JRF) to the first author under the Department of Science and Technology - Promotion of University Research and Scientific Excellence (DST - PURSE) Phase II programme is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Jacob, S., Thangarajan, S. Effect of Gestational Intake of Fisetin (3,3′,4′,7-Tetrahydroxyflavone) on Developmental Methyl Mercury Neurotoxicity in F1 Generation Rats. Biol Trace Elem Res 177, 297–315 (2017). https://doi.org/10.1007/s12011-016-0886-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-016-0886-x