Abstract
Selenium (Se) is recognized as a necessary trace mineral in animal diets, including those of birds. Lead (Pb) is a toxic heavy metal and can damage organs in humans and animals. Complex antagonistic interactions between Se and heavy metals have been reported in previous studies. However, little is known regarding the effects of Se on Pb-induced toxicity and the expression of inflammatory factors and heat shock proteins (HSPs) in the cartilage of chickens. In this present study, we fed chickens either with Se or Pb or both Se and Pb supplement and later analyzed the mRNA expressions of inflammatory factors (inducible nitric oxide synthase (iNOS), nuclear factor-kappa B (NF-κB), tumor necrosis factor-α (TNF-α), and cyclooxygenase-2 (COX-2)) and HSPs (Hsp27, Hsp40, Hsp60, Hsp70, and Hsp90). The results showed that Se and Pb influenced the expression of inflammatory factors and HSP genes in the chicken cartilage tissues. Additionally, we also found that antagonistic interaction existed between Se and Pb supplementation. Our findings suggested that Se could exert a antagonistic effect on Pb in chicken cartilage tissues.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Lead (Pb) is a metal element that can be toxic to living organisms, and expose it to nature could induce a wide range of biochemical and physiological dysfunctions in humans and laboratory animals [1, 2], such as negative effects on the birds’ general health, reproduction, and behavior, potentially leading to death [3]. Most previous studies documented that Pb could circulate in the body via blood and accumulate in bones and other vital organs (liver, kidneys, etc.) [4]. Clinical study indicated that exposure to Pb can lead to osteopenia, osteoporosis, impaired bone healing, and even osteoarthritis [5]. Pb toxicity also could cause inhibition of growth plate development in rats, increased cartilage-lead levels in ducks (higher than that of bone), and increased cartilage formation with delayed maturation and calcification and increased formation of fibrous tissue during bone repair in mouse [6, 7]. It had also been reported that Pb could induce oxidative stress by generating free radicals and reactive oxygen species (ROS) [8]. However, the toxic role of Pb is unclear, and the exact mechanism of Pb toxicity still needs to be further studied.
Inflammation was an important indicator of organism damage due to excess heavy metals. Inducible nitric oxide synthase (iNOS) could prevent autoimmunity to a certain degree and could function as an intracellular and intercellular signaling molecule shaping the immune response [9]. Nuclear factor-kappa B (NF-κB) was one of most important transcription factors in terms of inflammatory responses and could control the expression of numerous genes, such as iNOS, cyclooxygenase-2 (COX-2), and tumor necrosis factor-a (TNF-α) [10]. TNF-α was a proinflammatory cytokine that was produced by macrophages [11] and activated NF-κB. COX was the key enzyme in the biosynthesis of prostaglandins. COX-2 expression was usually induced by various cytokines, mitogens, and stresses [12]. Some of the interactions between heavy metals and inflammatory factors have already been reported in chickens. Arsenic exposure increased the messenger RNA (mRNA) level of TNF-α in mice liver [13] and NF-κB, TNF-α, COX-2, and iNOS in chicken gastrointestinal tract [14]. Excess Mn exposure resulted in inflammatory injury to chicken testis tissue through NF-κB/iNOS-COX-2 signaling pathway [15].
Heat shock proteins (HSPs) were major molecular chaperones that perform important functions in the folding/unfolding and translocation of proteins as well as in the assembly/disassembly of protein complexes [16], and HSPs possessed the ability to modulate the cellular antistress responses and played key roles in protecting organisms from metal stress [17]. Some previous studies indicated that heavy metal stresses could cause an increase in the mRNA level of HSPs in different tissues of western painted turtles [18] and chromium could upregulate the expressions of Hsp27 and Hsp70 in mice liver [19].
Selenium (Se) is an essential micronutrient for living organisms and is a well-established antioxidant [20], which can prevent or decrease the harmful effects of metal on the antioxidant system in different tissues [21–23]. The protective effect of Se against the different heavy metal toxicity in biological systems appears to have been studied [8, 24]. Se could protect the effect against cadmium-induced cytotoxicity in chicken splenic lymphocytes [25]. Se also had protective effects on the impairments of spatial learning, memory, and synaptic structural plasticity induced by Pb exposure in weaned rats [26]. Se deficiency could cause exudative diathesis and skeletal myodegeneration in birds which would be rapidly recovered by Se supplementation [27].
In summary, there was some research on Pb toxicity and Se on the antagonism against Pb. However, too little is known about the effect of Pb on the mRNA level of inflammatory injury and HSPs and the antagonistic effect of Se on Pb toxicity in chicken cartilage tissues. Herein, we established a model of Se and Pb interactions in chicken meniscus cartilage and sword cartilage and detected the effect of Se and Pb on the gene expression of inflammatory cytokines and HSPs.
Materials and Methods
Birds and Diets
All procedures used in this study were approved by the Institutional Animal Care and Use Committee of Northeast Agricultural University. A total of 360 male broiler chickens (1 day old; Weiwei Co. Ltd., Harbin, China) were randomly divided into four groups (90 chickens per group). Each group was separated into six pens with 15 chickens per pen. Throughout the entire experimental period, chickens were allowed ad libitum consumption of food and water. In this present study, we fed chickens a supplemental diet of Pb, Se, and Pb + Se to control Pb-induced toxicity and the antagonistic functions of Se in chicken muscle tissues. The Se group was fed a Se-adequate (sodium selenite) diet that contained 1 mg/kg Se and 0.5 mg/kg Pb. The Pb group was fed a Pb-supplemented (Pb acetate) diet containing 0.2 mg/kg Se and 350 mg/kg Pb. The Se + Pb group was fed a Se and Pb compound diet containing 1 mg/kg Se and 350 mg/kg Pb. Finally, the control group was fed a basic diet containing 0.2 mg/kg Se and 0.5 mg/kg Pb. Chickens were euthanized when they were 90 days old. The meniscus cartilage and sword cartilage tissues were quickly removed and rinsed with ice-cold sterile deionized water, frozen immediately in liquid nitrogen, and stored at −80 °C until required for subsequent experiments.
Quantification of Inflammatory Factor and HSP Gene mRNA
The extraction of total RNA (30-mg tissue; n = 6/diet group) was performed using TRIzol reagent according to the manufacturer’s instructions (Invitrogen, China). Then, the dried RNA pellets were resuspended in 50 μl of diethyl-pyrocarbonate-treated water. After determining the concentration and purity of the total RNA at 260/280 nm, first-strand complementary DNA (cDNA) was synthesized from 5 μg of total RNA using oligo dT primers and Superscript II reverse transcriptase according to the manufacturer’s instructions (Invitrogen, China). Finally, synthesized cDNA was diluted five times with sterile water and stored at −80 °C before use.
After quantification, the expression levels of HSPs and inflammatory genes were determined by real-time quantitative reverse transcription PCR with SYBR Premix ExTaq TM (Takara, China), and the real-time PCR was performed with ABI PRISM 7500 (Applied Biosystems). The PCR primers (Table 1) were designed using Oligo Primer Analysis software (version 6.0) and synthesized by Invitrogen (Shanghai, China).
Reactions consisted of the following: 10 μl of 2× SYBR Green I PCR Master Mix (TaKaRa, China), 0.4 μl of 50× ROX reference Dye II, 0.4 μl of each primer (10 μM), 2 μl of diluted cDNA, and 6.8 μl of PCR-grade water. The PCR procedure for HSPs, inflammatory genes, and GAPDH consisted of 95 °C for 30 s followed by 40 cycles at 95 °C for 15 s and 60 °C for 30 s. Results (fold changes) were expressed as 2−ΔΔCt in which ΔΔCt = (CtHSPs − CtGADPH)B/C/D − (CtHSPs − CtGADPH)A. A is the control group, B is the +Pb group, C is the +Se group, and D is the Se + Pb group. Inflammatory genes use the same formula to calculate the 2−ΔΔCt.
Western Blot Analysis
Protein extracts were subjected to SDS-polyacrylamide gel electrophoresis under reducing conditions on 12 % gels. Separated proteins were then transferred to nitrocellulose membranes using a tank transfer for 2 h at 200 mA in Tris glycine buffer containing 20 % methanol. Membranes were blocked with 5 % skim milk for 16–24 h and incubated overnight with diluted primary chicken antibody Hsp70 (1:500) and Hsp90 (1:500; Hsp90 and Hsp70 production of polyclonal antibody by our lab) followed by a horseradish peroxidase (HRP)-conjugated secondary antibody against rabbit IgG (1:1500, Santa Cruz, CA, USA). To verify equal loading of samples, the membrane was incubated with monoclonal β-actin antibody (1:1000, Santa Cruz, CA, USA), followed by a HRP-conjugated goat antimouse IgG (1:1000). The signal was detected by X-ray films (Trans Gen Biotech Co., China). The optical density (OD) of each band was determined by Image VCD gel imaging system, and the Hsp70 and Hsp90 expressions were detected as the ratio of OD of Hsp70 and Hsp90 and OD of β-actin, respectively.
Statistical Analyses
Statistical analyses of all the data were performed using the SPSS for Windows software (version 13; SPSS Inc., Chicago, IL, USA). The measurements were considered significantly different when P < 0.05 was obtained by a one-way analysis of variance. All the data displayed a normal distribution and passed equal variance testing. Differences between means were assessed using Tukey’s honest significant difference test for post hoc multiple comparisons. The data were expressed as the mean ± standard deviation. In addition, a principal component analysis (PCA) was used to define the most important parameters, which could be used as key factors for individual variations using the Statistics 6.0 program.
Results
Effects of Se, Pb, and Se + Pb on mRNA Levels of iNOS, NF-κB, TNF-α, and COX-2 in Chicken Meniscus Cartilage and Sword Cartilage
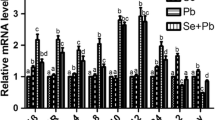
The mRNA levels of inflammatory cytokine genes in broiler chickens’ meniscus cartilage were detected. As shown in Fig. 1, Pb increased the mRNA levels of iNOS, NF-κB, TNF-α, and COX-2 in the +Pb group of the meniscus cartilage tissue (P < 0.05), and there was no significant difference between the +Se group and control group in the mRNA levels of iNOS, NF-κB, and COX-2 (P > 0.05), except the mRNA level of TNF-α. In the Se + Pb group, the mRNA level of NF-κB was increased, but the increased degree was less than that in the +Pb group compared with the control group (P < 0.05). Other three genes were not influenced (P > 0.05).
Likewise in Fig. 2, Pb increased the mRNA levels of iNOS, NF-κB, TNF-α, and COX-2 in the +Pb group of the sword cartilage tissue (P < 0.05), and there was no significant difference between the +Se group and control group in the mRNA levels of iNOS, NF-κB, TNF-α, and COX-2 (P > 0.05). In the Se + Pb group, the mRNA levels of iNOS, TNF-α, and COX-2 were increased but the increased degree was less than that in the +Pb group compared with the control group (P < 0.05), except NF-κB (P > 0.05).
Effects of Se, Pb, and Se + Pb on mRNA Levels of Hsp27, Hsp40, Hsp60, Hsp70, and Hsp90 in Chicken Meniscus Cartilage and Sword Cartilage
The mRNA levels of HSP genes in broiler chickens’ meniscus cartilage were detected. As shown in Fig. 3, Pb increased the mRNA levels of Hsp27, Hsp40, Hsp60, Hsp70, and Hsp90 in the +Pb group of the meniscus cartilage tissue (P < 0.05), and there was no significant difference between the +Se group and control group in the mRNA levels of Hsp27, Hsp40, Hsp60, Hsp70, and Hsp90 (P > 0.05). In the Se + Pb group, the mRNA levels of Hsp27, Hsp40, Hsp60, Hsp70, and Hsp90 were not influenced compared with the control group (P > 0.05).
Likewise in Fig. 4, Pb increased the mRNA levels of Hsp27, Hsp40, Hsp60, Hsp70, and Hsp90 in the +Pb group of the sword cartilage tissue (P < 0.05), and there was no significant difference between the +Se group and control group in the mRNA levels of Hsp27, Hsp40, Hsp60, Hsp70, and Hsp90 (P > 0.05). In the Se + Pb group, the mRNA levels of Hsp27, Hsp60, Hsp70, and Hsp90 were increased but the increased degree was less than that in the +Pb group compared with the control group (P < 0.05), except Hsp40 (P > 0.05).
Effects of Se, Pb, and Se + Pb on Western Bolt Analysis of Hsp70 and Hsp90 Levels in Chicken Meniscus Cartilage and Sword Cartilage
The protein expression levels of Hsp70 and Hsp90 were examined by western blots. The results (Figs. 5 and 6) revealed that the Hsp70 and Hsp90 protein expressions in +Pb group were gradually increased in meniscus cartilage and sword cartilage of chickens compared with control group. Also, western blots of Hsp70 and Hsp90 results were consistent with Hsp70 and Hsp90 mRNA responses to Pb-induced toxicity.
Principal Component Analysis
Using PCA, all the parameters that were measured in the present study were distinguished on the ordination plots that corresponded to the first and second principal components (95.109 and 4.891 %, respectively) in chicken’s meniscus cartilage (Fig. 7a) and (95.602 and 4.398 %, respectively; Fig. 7b) in sword cartilage. Furthermore, the observed correlations among the parameters were confirmed and quantified according to Spearman’s test (Tables 2 and 3). These results indicated the correlation of all the biomarkers suggesting a possibly positive or negative relationship. The results revealed that Hsp40, Hsp60, Hsp70, and Hsp90 had a close positive correlation with each other in meniscus cartilage. Similar tendency had been observed in iNOS and COX-2 as well. In addition, Hsp40 and Hsp60, TNF-α, and COX-2 presented a high correlation in sword cartilage of chicken. Similar phenomenon has been tested in the sword cartilage of chicken as well.
Discussion
A previous study revealed that a complex antagonistic interaction existed between Se and other heavy metals. Se can reduce the content of certain heavy metals, such as Pb, Cd, and Hg [25, 28, 29]. Wang et al. reported that Se alleviated Pb-induced increase of inflammation factor and HSP expression in chicken livers [30]. The chicken cartilage and bone was the main accumulation tissue. Gao et al. reported that Se and Pb influenced the expression of selenoprotein genes in chicken cartilage tissue [31]. However, the effects of Se against Pb toxicity on mRNA levels of inflammatory cytokines and HSP genes have been little reported in chickens. In this current study, we detected the effects of dietary Se against Pb toxicity on mRNA levels of inflammatory cytokine (iNOS, NF-κB, TNF-α, and COX-2) and HSP (Hsp27, Hsp40, Hsp60, Hsp70, and Hsp90) genes in the cartilage tissue of broiler chickens. The results showed that the treatment of Se and Pb influenced the gene expression of inflammatory cytokines and HSPs in chicken cartilage tissues.
Pb is one of high toxic heavy metals and has been known that it is not essential in living organisms. Pb exposure also could cause rat liver inflammatory injury [26]. NF-κB was a heterodimeric transcription factor that translocates to the nucleus and mediated the transcription of a vast array of proteins involved in inflammatory response [32]. NF-κB was involved in regulation of COX-2 and iNOS expressions, which were important enzymes that mediate inflammatory processes [33]. Several previous studies indicated that Pb exposure increased the levels of TNF-α and COX-2 proinflammatory enzymes in mouse microglial cell line [34]. Pb was responsible for a significant morphological alteration and a decline in cell function and TNF-α release which increased inflammation in mice testicular macrophages [35]. In the current study, we found that in the Pb group, the mRNA levels of NF-κB, TNF-α, COX-2, and iNOS were significantly higher than that in the control group. In accordance with the above research results, our results suggested that excess Pb could cause inflammation of chicken cartilage tissues.
As molecular chaperones, HSPs are highly conserved cellular stress proteins present in every organism from bacteria to humans. HSPs could protect organisms from a number of stress conditions including heavy metal stress [36]. Several previous studies presented that the mRNA levels of HSPs (Hsp27, Hsp40, Hsp60, Hsp70, and Hsp90) in chicken livers were increased when induced by dietary Se deficiency [37]. LcHsp27 expression showed a dramatic upregulation after a combined stress of temperature and cadmium [38]. The levels of HSP mRNA (Hsp90, Hsp70, Hsp60, Hsp40, and Hsp27) and protein (Hsp70 and Hsp60) were significantly increased (P < 0.05 or P < 0.01) with the As2O3 treatment [39]. Continuous Pb treatment elevated Hsp70 level in oribatid mite [40]. In this study, we found that Hsp27, Hsp40, Hsp60, Hsp70, and Hsp90 mRNA levels of chicken cartilage tissues were significantly increased (P < 0.05) in the +Pb group. It implied that HSPs may be the biomarkers of Pb poisoning.
Se is one of the essential micronutrients for living organisms, closely related to human and animal health. One of its main functions is an antioxidant action involved in protection against damage caused by free radicals and oxidative stress [41]. Some previous studies demonstrated that the antagonistic effect of Se on other heavy metals, such as Se, could protect the nitric oxide and the gene expression of inflammatory cytokines induced by cadmium in chicken splenic lymphocytes [42]. Se could ameliorate cadmium (Cd)-induced brain damage in chickens [43] and provide protective effects on the chronic poisoning of arsenic and decreased Hsp70 mRNA level which arsenic induced in rat livers [44]. Se also could provide certain protective actions against Pb-induced toxicity in chicken livers [45]. In our study, most of the mRNA levels of NF-κB, TNF-α, iNOS, COX-2, Hsp27, Hsp40, Hsp60, Hsp70, and Hsp90 decreased in the Se + Pb group compared to those in the Pb group. This finding is consistent with previous studies that indicated that Pb accumulation could be alleviated by Se treatment. Therefore, we hypothesized that Se may have a negative effect on Pb and could alleviate Pb-induced toxicity in chicken cartilage tissues.
In conclusion, we demonstrated that Se can alleviate Pb-dependent toxicity in chicken cartilage tissues. Moreover, changes in the mRNA expression of inflammatory cytokines and HSPs are indeed associated with the antagonistic function of Se that protects against Pb-induced toxicity.
References
Adonaylo VN, Oteiza PI (1999) Lead intoxication: antioxidant defenses and oxidative damage in rat brain. Toxicology 135:77–85
Flora SJ, Pande M, Mehta A (2003) Beneficial effect of combined administration of some naturally occurring antioxidants (vitamins) and thiol chelators in the treatment of chronic lead intoxication. Chem Biol Interact 145:267–280
Fisher IJ, Pain DJ, Thomas VG (2006) A review of lead poisoning from ammunition sources in terrestrial birds. Biol Conserv 131:421–432
Hartup BK (1996) Rehabilitation of native reptiles and amphibians in DuPage County, Illinois. J Wildl Dis 32:109–112
Roschger JG, Hofstaetter B, Pemmer Z et al (2013) Differential accumulation of lead and zinc in double-tidemarks of articular cartilage. Osteoarthr Cartil 21:1707–1715
Yıldızgören M T., Baki A E, Kara M, et al. (2014) Ultrasonographic measurement of the femoral cartilage thickness in patients with occupational lead exposure, J Expo Sci Environ Epidemiol. 1-3
Zoeger N, Roschger P, Hofstaetter JG et al (2006) Lead accumulation in tidemark of articular cartilage. Osteoarthr Cartil 14:906–913
Ates B, Orun I, Talas ZS, Durmaz G, Yilmaz I (2008) Effects of sodium selenite on some biochemical and hematological parameters of rainbow trout (Oncorhynchus mykiss Walbaum, 1792) exposed to Pb2+ and Cu2+. Fish Physiol Biochem 34:53–59
Bogdan C (2001) Nitric oxide and the immune response. Nat Immunol 2:907–916
Cogswell JP, Godlevski MM, Wisely GB et al (1994) NF-kappa B regulates IL-1 beta transcription through a consensus NF-kappa B binding site and a nonconsensus CRE-like site. J Immunol 153:712–723
Raabe T, Bukrinsky M, Currie RA (1998) Relative contribution of transcription and translation to the induction of tumor necrosis factor-alpha by lipopolysaccharide. J Biol Chem 273:974–980
Yuan CJ, Mandal AK, Zhang Z, Mukherjee AB (2000) Transcriptional regulation of cyclooxygenase-2 gene expression: novel effects of nonsteroidal anti-inflammatory drugs. Cancer Res 60:1084–1091
Das S, Santra A, Lahiri S, Guha Mazumder DN (2005) Implications of oxidative stress and hepatic cytokine (TNF-alpha and IL-6) response in the pathogenesis of hepatic collagenesis in chronic arsenic toxicity. Toxicol Appl Pharmacol 204:18–26
Xing M, Zhao P, Guo G et al (2015) Inflammatory factor alterations in the gastrointestinal tract of cocks overexposed to arsenic trioxide. Biol Trace Elem Res 167:288–299
Du Y, Zhu Y, Teng X et al (2015) Toxicological effect of manganese on NF-kappaB/iNOS-COX-2 signaling pathway in chicken testes. Biol Trace Elem Res 168:227–234
Bernabo P, Rebecchi L, Jousson O et al (2011) Thermotolerance and Hsp70 heat shock response in the cold-stenothermal chironomid Pseudodiamesa branickii (NE Italy). Cell Stress Chaperones 16:403–410
Chen H, Xu XL, Li YP, Wu JX (2014) Characterization of heat shock protein 90, 70 and their transcriptional expression patterns on high temperature in adult of Grapholita molesta (Busck). Insect Sci 21:439–448
Ramaglia V, Harapa GM, White N, Buck LT (2004) Bacterial infection and tissue-specific Hsp72, -73 and -90 expression in western painted turtles. Comp Biochem Physiol Toxicol Pharmacol : CBP 138:139–148
Lee J, Lim KT (2012) Inhibitory effect of SJSZ glycoprotein (38 kDa) on expression of heat shock protein 27 and 70 in chromium (VI)-treated hepatocytes. Mol Cell Biochem 359:45–57
Klotz LO, Kroncke KD, Buchczyk DP, Sies H (2003) Role of copper, zinc, selenium and tellurium in the cellular defense against oxidative and nitrosative stress. J Nutr 133:1448S–1451S
Yao H, Liu W, Zhao W et al (2014) Different responses of selenoproteins to the altered expression of selenoprotein W in chicken myoblasts. RSC Adv 4:64032–64042
Liu C, Fu J, Liu C, Li S (2015) The role of nitric oxide and autophagy in liver injuries induced by selenium deficiency in chickens. RSC Adv 5:50549–50556
Jiang ZH, Khoso PA, Yao HD et al (2015) SelW regulates inflammation-related cytokines in response to H2O2 in Se-deficient chicken liver. RSC Adv 5:37896–37905
Orun I, Talas ZS, Ozdemir I, Alkan A, Erdogan K (2008) Antioxidative role of selenium on some tissues of (Cd2+), Cr3+)-induced rainbow trout. Ecotoxicol Environ Saf 71:71–75
Chen X, Zhu YH, Cheng XY, Zhang ZW, Xu SW (2012) The protection of selenium against cadmium-induced cytotoxicity via the heat shock protein pathway in chicken splenic lymphocytes. Molecules 17:14565–14572
Han XJ, Xiao YM, Ai BM et al (2014) Effects of organic selenium on lead-induced impairments of spatial learning and memory as well as synaptic structural plasticity in rats. Biol Pharm Bull 37:466–474
Zhang JL, Zhang ZW, Shan AS et al (2014) Effects of dietary selenium deficiency or excess on gene expression of selenoprotein N in chicken muscle tissues. Biol Trace Elem Res 157:234–241
Bjerregaard P, Fjordside S, Hansen MG, Petrova MB (2011) Dietary selenium reduces retention of methyl mercury in freshwater fish. Environ Sci Technol 45:9793–9798
Shao CC, Li N, Zhang ZW et al (2014) Cadmium supplement triggers endoplasmic reticulum stress response and cytotoxicity in primary chicken hepatocytes. Ecotoxicol Environ Saf 106:109–114
Wang H, Li S, Teng X (2015) The antagonistic effect of selenium on lead-induced inflammatory factors and heat shock proteins mRNA expression in chicken livers. Biol Trace Elem Res. doi:10.1007/s12011-015-0532-z
Gao H, Liu CP, Song SQ et al (2015) Effects of dietary selenium against lead toxicity on mRNA levels of twenty-five selenoprotein genes in the cartilage tissue of broiler chicken. Biol Trace Elem Res. doi:10.1007/s12011-015-0579-x
Karin M, Yamamoto Y, Wang QM (2004) The IKK NF-kappa B system: a treasure trove for drug development. Nat Rev Drug Discov 3:17–26
Surh YJ, Chun KS, Cha HH et al (2001) Molecular mechanisms underlying chemopreventive activities of anti-inflammatory phytochemicals: down-regulation of COX-2 and iNOS through suppression of NF-kappa B activation. Mutat Res 480–481:243–268
Kumawat KL, Kaushik DK, Goswami P, Basu A (2014) Acute exposure to lead acetate activates microglia and induces subsequent bystander neuronal death via caspase-3 activation. Neurotoxicology 41:143–153
Barbhuiya SASK, Chakraborty S, Sengupta M (2013) Studies of lead toxicity on inflammatory damage and innate immune functions in testicular macrophages of male Swiss albino mice. Mod Res Inflamm 2:75–81
Lindquist S, Craig EA (1988) The heat-shock proteins. Annu Rev Genet 22:631–677
Liu CP, Fu J, Xu FP, Wang XS, Li S (2015) The role of heat shock proteins in oxidative stress damage induced by Se deficiency in chicken livers. Biometals 28:163–173
Yang QL, Yao CL, Wang ZY (2012) Acute temperature and cadmium stress response characterization of small heat shock protein 27 in large yellow croaker, Larimichthys crocea. Comp Biochem Physiol Toxicol Pharmacol : CBP 155:190–197
Guo Y, Zhao P, Guo G et al (2015) Effects of arsenic trioxide exposure on heat shock protein response in the immune organs of chickens. Biol Trace Elem Res Biol. doi:10.1007/s12011-015-0389-1
Kohler HR, Alberti G, Seniczak S, Seniczak A (2005) Lead-induced Hsp70 and Hsp60 pattern transformation and leg malformation during postembryonic development in the oribatid mite Archegozetes longisetosus Aoki. Comp Biochem Physiol Toxicol Pharmacol : CBP 141:398–405
Rayman MP (2000) The importance of selenium to human health. Lancet 356:233–241
Liu S, Xu F, Fu J, Li S (2015) Protective roles of selenium on nitric oxide and the gene expression of inflammatory cytokines induced by cadmium in chicken splenic lymphocytes. Biol Trace Elem Res 168:252–260
Liu LL, Zhang JL, Zhang ZW et al (2014) Protective roles of selenium on nitric oxide-mediated apoptosis of immune organs induced by cadmium in chickens. Biol Trace Elem Res 159:199–209
Xu Z, Wang Z, Li JJ et al (2013) Protective effects of selenium on oxidative damage and oxidative stress related gene expression in rat liver under chronic poisoning of arsenic. Food Chem Toxicol 58:1–7
Xu T, Gao X, Liu G (2015) The antagonistic effect of selenium on lead toxicity is related to the ion profile in chicken liver. Biol Trace Elem Res. doi:10.1007/s12011-015-0422-4
Acknowledgments
This work was supported by China Postdoctoral Science Foundation (No. 2012 M520702); the National Natural Science Foundation of China (31402267); the Start-up Foundation for Doctors of Northeast Agricultural University, China (No. 2012RCB92); Heilongjiang Province Science Foundation for Youths (QC2014C015); Heilongjiang Provincial Department of Education Science and Technology research project (No. 12541024); Foundation for Young Talents in Higher Education of Heilongjiang, China (UNPYSCT-2015009); the Young Talents Project of Northeast Agricultural University (No. 14QC18, No. 14QC20); the International Postdoctoral Exchange Fellowship Program (No. 20130006); and the Students Innovative Training (SIPT) program of Northeast Agricultural University (No. 201510224109, 201510224110).
Author information
Authors and Affiliations
Corresponding authors
Additional information
All other authors have read the manuscript and have agreed to submit it in its current form for consideration for publication in the journal.
Shu Fang Zheng and HuanYu Song contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zheng, S., Song, H., Gao, H. et al. The Antagonistic Effect of Selenium on Lead-Induced Inflammatory Factors and Heat Shock Protein mRNA Level in Chicken Cartilage Tissue. Biol Trace Elem Res 173, 177–184 (2016). https://doi.org/10.1007/s12011-016-0630-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-016-0630-6