Abstract
Selenium and zinc are well-known essential trace elements with potent biological functions. However, the possible health benefits of the combined administration of dietary selenium and zinc have not been studied extensively. In this study, we prepared selenium- and zinc-enriched mushrooms (SZMs) containing increased levels of selenium and zinc. The effects of SZMs on antioxidant and antitumor activities were evaluated. Mice were fed with either a control diet or a diet supplemented with SZMs or sodium selenite and zinc sulfate for 6 weeks. Antioxidant capacity was investigated by measuring the activities of antioxidant enzymes and the levels of lipid peroxide products. Results showed that treatment with SZMs significantly increased the activities of glutathione peroxidase (GPx) and superoxide dismutase and decreased the levels of malondialdehyde and lipofuscin. Furthermore, using a mouse model of lung tumors, we found that SZMs significantly decreased the number of tumor nodes with an increase in the activity of GPx. SZMs had a greater effect on the increase in both antioxidant and antitumor activities than did sodium selenite and zinc sulfate. These findings suggest that SZMs may be effective for improving antioxidant capacity and preventing tumors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Reactive oxygen species (ROS) are constantly generated in the human body during cell metabolism and diseases. Overproduction of ROS can damage cellular components and compromise cell viability [1]. To protect against the potential harmful effects of ROS, cells possess an antioxidant defense system that includes superoxide dismutase (SOD), catalase, and glutathione peroxidase (GPx) [2]. However, once the antioxidant system is overwhelmed by an increased production of ROS, oxidative stress will occur. Oxidative stress has been implicated in the pathogenesis of various diseases including cardiovascular disease, liver injury, and cancer [3]. Therefore, strategies to enhance antioxidant protection may be effective at preventing and treating oxidative stress-induced diseases.
Selenium and zinc are well-known essential trace elements with potent antioxidant effects. Selenium exerts its antioxidant effects through selenoproteins, such as GPx and thioredoxin reductases [4]. Zinc acts as an antioxidant because it is an important cofactor of SOD [5]. Accumulating evidence has indicated that a deficiency in selenium or zinc results in decreased antioxidant capacity [6], whereas supplementation with selenium or zinc improves antioxidant status [7, 8].
Recently, there has been an increased interest in evaluating the combined effects of antioxidants. Given that many antioxidants work synergistically, it seems more appropriate to administer treatment using a combination of antioxidants rather than with a single antioxidant. In fact, the combined administration of vitamin C and E has been shown to be synergistically protective against oxidative damage [9, 10]. In addition, selenium has been found to enhance the antioxidant effect of vitamin E [11]. Beneficial effects of combined selenium and zinc treatment in decreasing cadmium-induced oxidative stress have also been reported [12, 13]. However, these two studies used sodium selenite and zinc chloride, the chemical forms of selenium and zinc. Evidence regarding the combinative effects of dietary-sourced selenium and zinc remains limited, and more research on their possible antioxidant and antitumor properties is needed.
In the present study, selenium- and zinc-enriched mushrooms (SZMs) that are enriched in both selenium and zinc were produced by using the oyster mushroom (Pleurotus ostreatus), which is a common edible mushroom, as a carrier. Oyster mushrooms are capable of accumulating large amounts of trace elements and incorporating them into organic compounds. Moreover, the mushroom has many beneficial health effects, such as reducing blood cholesterol concentration, modulating immune function, preventing oxidative damage, and inhibiting tumor growth and metastasis [14–16]. Therefore, SZMs would be expected to be more effective in triggering antioxidant defense. Here, we evaluated the potential antioxidant activity of SZMs and also assessed their antitumor effects in a mouse tumor model.
Materials and Methods
Preparation of SZMs
Mushrooms were grown on a basal substrate containing corncobs and cottonseed hulls according to standard farming methods. SZMs were produced by supplementing sodium selenite and zinc sulfate in the composts. Mushrooms collected on the peak harvest day of the two flush were air-dried and then pulverized with a hammering grinder. The products were passed through a 60-mesh sieve for further use. The SZMs contained 39.8 mg of selenium and 201.2 mg of zinc per kg of powder.
Animals
Kunming mice (10–14 weeks old) were purchased from the Experimental Animal Center of Hebei Medical University (Shijiazhuang, Hebei, China). They were handled under standard laboratory conditions and had free access to food and water. The mice were allowed to acclimatize to our facility for at least 1 week before being used in the experiments. All animal experiments were performed in accordance with to the National Institutes of Health Guidelines on the Care and Use of Laboratory Animals.
Experimental Protocols
Two experiments were carried out to assess the effects of SZMs as detailed below. In the experiment evaluating antioxidant activity, 28 mice were randomized equally into four groups and received the following diets: the control group received a basal powder diet; the low-SZM and high-SZM groups received the basal powder diet supplemented with 1 and 5 % SZM, respectively; the inorganic selenium and zinc (ISZ) group received the basal powder diet supplemented with sodium selenite and zinc sulfate (which contained the equivalent amount of selenium and zinc as 5 % SZM). The mice in high-SZM group received about 300 μg of selenium and 1.5 mg of zinc per kg body weight per day. These doses were chosen on the basis of previous studies [17, 18]. At the end of the study, whole blood was collected from the tail vein. The livers and hearts were quickly removed, weighed, and homogenized in the appropriate buffers. The activities of antioxidant enzymes and the levels of lipid peroxide products were investigated.
The experiment evaluating antitumor activity included four groups of 12 mice each: the untreated and control groups were fed a basal powder diet; the SZM group was fed the basal powder diet containing 2 % SZM; and the ISZ group was fed the basal powder diet containing sodium selenite and zinc sulfate (which contained the equivalent amount of selenium and zinc as 2 % SZM). During administration, the untreated group was maintained on tap water, whereas the other three groups were given urethane to induce lung tumor.
Mouse Lung Tumor Model
Mice were given drinking water containing urethane at a concentration of 1,000 ppm for 12 weeks as previously described [19, 20]. Daily water consumption was measured and was found to be similar among all experimental groups. At the end of the experiments, the mice were killed, and the samples of whole blood and liver were collected for the determination of GPx activity and malondialdehyde (MDA) level, respectively. The lungs were removed, and the number of tumor nodules was counted under a dissecting microscope. For histopathological examination, lung tissue samples were fixed in 10 % neutral buffered formalin, embedded in paraffin, and stained with hematoxylin and eosin.
Activities of Antioxidant Enzymes
The activity of GPx was measured using the coupled enzymatic method [21], while the activity of SOD was assayed using pyrogallol method [22], as previously described. Results were expressed as U/ml or U/mg protein. The concentration of total protein was determined using the Bradford method [23] with bovine serum albumin as the standard.
Levels of Lipid Peroxide Products
The level of MDA was measured using the thiobarbituric acid method [24]. The results were expressed as nmol/mg protein. Lipofuscin (LPF) was isolated from heart homogenates [25], and autofluorescence was measured using a spectrofluorometer. The fluorescence intensity was calibrated with quinine sulfate.
Statistical Analysis
Results are expressed as mean ± SD. The statistical significance of differences was calculated by one-way analysis of variance followed by the Bonferroni post hoc test. A P value of less than 0.05 was considered statistically significant.
Results
SZMs Upregulate the Activities of GPx and SOD
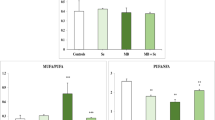
The mice fed with 1 % SZM had significantly higher whole blood GPx activity, whereas mice fed with 5 % SZM exhibited increased whole blood GPx, erythrocyte, and liver SOD activities, compared with that exhibited by the control mice (Fig. 1). The oral administration of sodium selenite and zinc sulfate only increased whole blood GPx activity. No significant differences were observed in liver GPx activities among all groups. Notably, the activities of whole blood GPx and liver SOD in mice fed with 5 % SZM were significantly higher than those in mice fed with sodium selenite and zinc sulfate.
The effects of SZMs on the activities of GPx and SOD. Mice were fed the indicated diet for 6 weeks. The activities of GPx in the whole blood (a) or liver (b) and SOD in erythrocytes (c) or the liver (d) were measured. Values are shown as mean ± SD; n = 7 mice in each group. The asterisks indicate differences from the control group; the number signs indicate differences from the ISZ group; P < 0.05
SZMs Reduce the Levels of MDA and LPF
To assess whether the increased activities of the antioxidant enzymes induced by SZMs were associated with the decreased production of lipid peroxide, the levels of MDA in the liver and LPF in the heart were measured (Fig. 2). Compared with mice fed with control diets, mice fed with 5 % SZM or sodium selenite and zinc sulfate exhibited significant decreases in the levels of both MDA and LPF. No significant differences were observed between mice fed with 1 % SZM and those fed the control diet. The level of LPF was significantly lower in mice fed with 5 % SZM than those in mice fed with sodium selenite and zinc sulfate, whereas the level of MDA was similar in the two groups.
The effects of SZMs on the levels of MDA and LPF. Mice were fed the indicated diet for 6 weeks. The levels of MDA in the liver (a) and LPF in the heart (b) were measured. Values are shown as mean ± SD; n = 7 mice in each group. The asterisks indicate differences from the control group; the number sign indicates difference from the ISZ group; P < 0.05
SZMs Inhibit the Development of Tumors Induced by Urethane in Mice
Numerous studies have suggested that antioxidants inhibit tumor development [26, 27]. Therefore, we investigated whether SZMs could protect mice against tumor growth. In response to treatment with urethane, all of the mice developed macroscopically visible lung tumors. However, the number of tumor nodules on the lung surface was significantly lower in mice fed with SZM or sodium selenite and zinc sulfate than in mice fed with control diet (Fig. 3a). The most significant tumor inhibition was observed in mice fed with SZM.
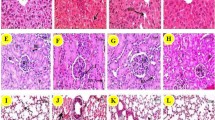
SZMs inhibit the development of urethane-induced tumors in mice. Mice were fed the indicated diet and were treated or not treated with urethane in their drinking water for 12 weeks. a The number of tumor nodules on the lung surface was measured. Values are shown as mean ± SD; n = 12 mice in each group. The asterisks indicate differences from the control group, the number sign indicates difference from the ISZ group, P < 0.05. b Representative histological sections of lung tumors stained with hematoxylin and eosin. Original magnification ×200
Histopathological examination revealed that the tumor was sharply demarcated from the surrounding tissue. Slight compression of adjacent tissues was occasionally observed. The tumor was composed of well-differentiated cells with round to oval nuclei and moderately abundant eosinophilic cytoplasm (Fig. 3b). There were no significant differences in histological findings among the three groups treated with urethane.
The Protective Effect of SZMs is Associated with Increased Activity of GPx
The effect of SZMs on GPx activity and MDA level under tumor conditions was examined (Fig. 4). GPx activity was significantly inhibited in mice exposed to urethane compared with untreated mice. Oral administration of SZM or sodium selenite and zinc sulfate restored the decrease in GPx activity. However, treatment with urethane failed to alter the level of MDA. Mice fed with SZM or sodium selenite and zinc sulfate had a slight but not significant decrease in the MDA level compared to mice fed with control diet. There was no significant difference in the GPx activity or MDA level between the two experimental diet groups.
The effects of SZMs on the activity of GPx and the level of MDA in urethane-treated mice. Mice were fed with the indicated diet and were treated or not treated with urethane in their drinking water for 12 weeks. The activity of GPx in the whole blood (a) and the level of MDA in the liver (b) were measured. Values are shown as mean ± SD; n = 8 mice in each group. The asterisks indicate differences from the control diet, P < 0.05
Discussion
The combined administration of appropriate antioxidants through diet and supplements may be more effective at enhancing antioxidant capacity and providing health benefits. Studies have demonstrated the synergistic effects of several antioxidants such as vitamins C, E, and selenium [9–11]. The combined administration of selenium and zinc has also been found to reverse cadmium-induced oxidative stress [12, 13]. In agreement with these observations, we found that SZMs, which contain elevated levels of selenium and zinc, possess antioxidant properties, including enhancing the activities of antioxidant enzymes such as SOD and GPx and reducing lipid peroxide products such as MDA and LPF.
Although the antitumor roles of selenium and zinc alone have been recognized for many years, little is known about the antitumor activity of combined treatment with selenium and zinc. Evidence from epidemiological and animal studies has indicated that low selenium and zinc status is inversely correlated with an increased risk of tumors [28, 29]. Therefore, combined treatment with selenium and zinc is necessary for more effectively reducing the risk of tumor formation. In the present study, we found that both SZM and inorganic selenium and zinc significantly inhibited the development of lung tumors in an animal model. This result suggests that combined treatment with selenium and zinc is beneficial for tumor control. Notably, the antitumor effect of SZM was greater than that of inorganic selenium and zinc. Thus, SZMs seem to be a highly effective source of selenium and zinc for tumor prevention.
Whether the antitumor effect of SZMs is mediated by increased antioxidant activity is of considerable interest. The activity of GPx was significantly lower in urethane-induced lung tumor mice than the activity observed in the normal mice, demonstrating the involvement of this antioxidant enzyme. In contrast, the antitumor effect of SZMs was associated with an increased activity of GPx. Studies have found that GPx plays an important role in preventing tumors; for example, deficiency in GPx was shown to augment the development of colonic tumors in susceptible mice [30], whereas the overexpression of GPx was found to decrease the rate of tumor cell growth [31] and protect cells against DNA damage [32], which could be important in reducing the risk of tumors. Taken together, these results indicate that the increased activity of GPx mediated by SZMs may, at least in part, be responsible for preventing the development of tumors. In contrast, little change was observed in the level of MDA after 12 weeks of treatment with urethane. This again could be explained by the fact that MDA is unstable and can be further oxidized in vivo [33]. Thus, it is possible that the level of MDA did not completely reveal the magnitude of lipid peroxidation that occurred. Further studies are needed to investigate the contribution of antioxidant defenses in the SZM-induced antitumor effect.
The effects of antioxidants on cancer are controversial, with some studies showing benefit and others showing no benefit and even possible harm [34]. Our results showed that SZM had a potential beneficial effect on cancer prevention. We cannot exclude the possibility that SZM may also exert its effects through other mechanisms such as enhancing immune function. The immune system plays an important role in the prevention of tumors by recognizing and destroying tumor cells. Treatment with selenium or zinc has been shown to be associated with multiple immunomodulatory effects, such as enhancing NK cell activity and affecting T cell proliferation and differentiation [35–37]. In addition to selenium and zinc, some constituents of SZMs, particularly β-glucans, may be associated with beneficial immunomodulatory and antitumor effects. Indeed, β-glucans or β-glucan-containing extracts have been reported to exhibit significant immunomodulatory effects [38, 39]. Therefore, SZMs would likely be effective in modulating immune function. Further analysis of the effect of SZM on immune function may potentially extend its applications.
In summary, this study demonstrates that SZMs are beneficial in improving antioxidant activity and protecting mice against the development of lung cancer. Supplementation with SZMs might be an inexpensive, safe, and effective approach for increasing antioxidant capacity and preventing tumors.
References
Wellen KE, Thompson CB (2010) Cellular metabolic stress: considering how cells respond to nutrient excess. Mol Cell 40:323–332
Finkel T, Holbrook NJ (2000) Oxidants, oxidative stress and the biology of aging. Nature 408:239–247
Qadri I, Iwahashi M, Capasso JM, Hopken MW, Flores S, Schaack J, Simon FR (2004) Induced oxidative stress and activated expression of manganese superoxide dismutase during hepatitis C virus replication: role of JNK, p38 MAPK and AP-1. Biochem J 378:919–928
Rotruck JT, Pope AL, Ganther HE, Swanson AB, Hafeman DG, Hoekstra WG (1973) Selenium: biochemical role as a component of glutathione peroxidase. Science 179:588–590
Greenough MA, Volitakis I, Li QX, Laughton K, Evin G, Ho M, Dalziel AH, Camakaris J, Bush AI (2011) Presenilins promote the cellular uptake of copper and zinc and maintain copper chaperone of SOD1-dependent copper/zinc superoxide dismutase activity. J Biol Chem 286:9776–9786
Oteiza PI, Olin KL, Fraga CG, Keen CL (1995) Zinc deficiency causes oxidative damage to proteins, lipids and DNA in rat testes. J Nutr 125:823–829
Sahin K, Kucuk O (2003) Zinc supplementation alleviates heat stress in laying Japanese quail. J Nutr 133:2808–2811
Hu Y, Mclntosh GH, Le Leu RK, Young GP (2010) Selenium-enriched milk proteins and selenium yeast affect selenoprotein activity and expression differently in mouse colon. Br J Nutr 104:17–23
Moison RM, Beijersbergen van Henegouwen GM (2002) Topical antioxidant vitamins C and E prevent UVB-radiation-induced peroxidation of eicosapentaenoic acid in pig skin. Radiat Res 157:402–409
Honarbakhsh S, Schachter M (2009) Vitamins and cardiovascular disease. Br J Nutr 101:1113–1131
Beck MA (2007) Selenium and vitamin E status: impact on viral pathogenicity. J Nutr 137:1338–1340
el Jihen H, Imed M, Fatima H, Abdelhamid K (2008) Protective effects of selenium (Se) and zinc (Zn) on cadmium (Cd) toxicity in the liver and kidney of the rat: histology and Cd accumulation. Food Chem Toxicol 11:3522–3527
Messaoudi I, EI Heni J, Hammouda F, Said K, Kerkeni A (2009) Protective effects of selenium, zinc, or their combination on cadmium-induced oxidative stress in rat kidney. Biol Trace Elem Res 130:152–161
Puttaraju NG, Venkateshaiah SU, Dharmesh SM, Urs SM, Somasundaram R (2006) Antioxidant activity of indigenous edible mushrooms. J Agric Food Chem 54:9764–9772
Keenan JM, Goulson M, Shamliyan T, Knutson N, Kolberg L, Curry L (2007) The effects of concentrated barley beta-glucan on blood lipids in a population of hypercholesterolaemic men and women. Br J Nutr 97:1162–1168
Hetland G, Johnson E, Lyberg T, Bernardshaw S, Tryggestad AM, Grinde B (2008) Effects of the medicinal mushroom Agaricus Murill on immunity, infection and cancer. Scand J Immunol 68:363–370
Abdo KM (1994) NTP toxicity studies of sodium selenate and sodium selenite (CAS Nos. 13410-01-0 and 10102-18-8) administered in drinking water to F344/N rats and B6C3F1 mice. Toxic Rep Ser 38:1–E5
Batra N, Nehru B, Bansal MP (1998) The effect of zinc supplementation on the effects of lead on the rat testis. Reprod Toxicol 12:535–540
Kristiansen E, Clemmensen S, Meyer O (1990) Chronic ethanol intake and reduction of lung tumours from urethane in strain A mice. Food Chem Toxicol 28:35–38
Chan PC (1996) NTP technical report on toxicity studies of urethane in drinking water and urethane in 5 % ethanol adiministered to F344/N rats and B6C3F1 mice. Toxic Rep Ser 52:1–91
Yamamoto Y, Takahashi Y (1993) Glutathione peroxidase isolated from plasma reduces phospholipid hydroperoxides. Arch Biochem Biophys 305:541–545
Prohaska JR (1983) Changes in tissue growth, concentrations of copper, iron, cytochrome oxidase and superoxide dismutase subsequent to dietary or genetic copper deficiency in mice. J Nutr 113:2048–2058
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Buege JA, Aust SD (1978) Microsomal lipid peroxidation. Methods Enzymol 52:302–310
Siakotos BL, Strehler AN (1974) Procedures for the isolation of lipofuscin (age pigment) from normal heart and liver. Methods Enzymol 31(Pt A):425–432
Li Y, Schellhorn HE (2007) New developments and novel therapeutic perspectives for vitamin C. J Nutr 137:2172–2184
Khandrika L, Kumar B, Koul S, Maroni P, Koul HK (2009) Oxidative stress in prostate cancer. Cancer Lett 282:125–136
Federico A, Iodice P, Federico P, Del Rio A, Melloine MC, Catalano G, Federico P (2001) Effects of selenium and zinc supplementation on nutritional status in patients with cancer of digestive tract. Eur J Clin Nutr 55:293–297
Cavdar AO, Gozdasoglu S, Babacan E, Mengubas K, Unal E, Yavuz G, Tacyildiz N (2009) Zinc and selenium status in pediatric malignant lymphomas. Nutr Cancer 61:888–890
Chu FF, Esworthy RS, Chu PG, Longmate JA, Huycke MM, Wilczynski S, Doroshow JH (2004) Bacteria-induced intestinal cancer in mice with disrupted Gpx1 and Gpx2 genes. Cancer Res 64:962–968
Liu J, Hinkhouse MM, Sun W, Weydert CJ, Ritchie JM, Oberley LW, Cullen JJ (2004) Redox regulation of pancreatic cancer cell growth: role of glutathione peroxidase in the suppression of the malignant phenotype. Hum Gene Ther 15:239–250
Baliga MS, Wang H, Zhuo P, Schwartz JL, Diamond AM (2007) Selenium and GPx-1 overexpression protect mammalian cells against UV-induced DNA damage. Biol Trace Elem Res 115:227–242
Loiseaux-Meunier MN, Bedu M, Gentou C, Pepin D, Coudert J, Caillaud D (2001) Oxygen toxicity: simultaneous measure of pentane and malondialdehyde in humans exposed to hyperoxia. Biomed Pharmacother 55:163–169
Bjelakovic G, Nikolova D, Simonetti RG, Gluud C (2004) Antioxidant supplements for prevention of gastrointestinal cancers: a systematic review and meta-analysis. Lancet 364:1219–1228
Bromme CS, McArdle F, Kyle JA, Andrews F, Lowe NM, Hart CA, Arthur JR, Jackson MJ (2004) An increase in selenium intake improves immune function and poliovirus handling in adults with marginal selenium status. Am J Clin Nutr 80:154–162
Hoffmann FW, Hashimoto AC, Shafer LA, Dow S, Berry MJ, Hoffmann PR (2010) Dietary selenium modulates activation and differentiation of CD4+ T cells in mice through a mechanism involving cellular free thiols. J Nutr 140:1155–1161
Murakami M, Hirano T (2008) Intracellular zinc homeostasis and zinc signaling. Cancer Sci 99:1515–1522
Brown GD, Gordon S (2003) Fungal beta-glucans and mammalian immunity. Immunity 19:311–315
Yan H, Kakuta S, Nishihara M, Sugi M, Adachi Y, Ohno N, Iwakura Y, Tsuji NM (2011) Kjellmaniella crassifolia Miyabe (Gagome) extract modulates intestinal and systemic immune responses. Biosci Biotechnol Biochem 75:2178–2183
Acknowledgments
This study was supported in part by funds from the Fifth Hospital of Shijiazhuang.
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yan, H., Chang, H. Antioxidant and Antitumor Activities of Selenium- and Zinc-Enriched Oyster Mushroom in Mice. Biol Trace Elem Res 150, 236–241 (2012). https://doi.org/10.1007/s12011-012-9454-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-012-9454-1